Abstract
Purpose:
Long-term vascular access (VA) is critical for patients with kidney disease or hyperlipoproteinemia, yet stenotic lesions often compromise its patency, necessitating frequent interventions. This study compares the efficacy of three endovascular treatment modalities—plain old balloon angioplasty with drug-coated balloon (POBA+DCB), scoring balloon with drug-coated balloon (SB+DCB), and FLEX vessel preparation with drug-coated balloon (VP+DCB)—in the treatment of VA stenoses.
Materials and methods:
A retrospective analysis was conducted on 175 VA interventions performed between June 2022 and October 2024 at a dedicated VA center. Patients were divided into three groups based on treatment modality: POBA+DCB (n = 57), SB+DCB (n = 47), and VP+DCB (n = 71). Technical success was achieved in all cases, and complete balloon effacement was highest in the POBA and VP groups (100%) compared to SB (91.5%, p = 0.0046).
Results:
Reinterventions per patient-year were significantly lower in the VP group (0.372) compared to POBA (0.695) and SB (0.917) (p < 0.0001). Patency outcomes favored VP+DCB, with target lesion primary patency (TLPP) rates at 6 and 12 months of 86.0% and 73.6%, respectively, compared to 79.1% and 40.1% for POBA and 67.7% and 41.2% for SB. Access circuit primary patency (ACPP) at 6 and 12 months was 84.7% and 57.0% for VP, 78.5% and 37.9% for POBA, and 65.4% and 35.6% for SB. In multivariate analysis, prior interventions and reintervention frequency significantly impacted patency loss.
Conclusions:
These findings suggest that VP+DCB may provide superior patency and reduced reintervention rates compared to POBA+DCB and SB+DCB, offering a promising alternative for the management of VA-stenoses in real-world clinical settings.
Keywords
Introduction
Long-term vascular access (VA) is crucial for patients with chronic and end-stage kidney disease or hyperlipoproteinemia. Despite the advancements of recent decades, both arteriovenous fistulae (AVF) and grafts (AVG) face challenges, particularly regarding the development of stenotic lesions that can impede maturation or lead to VA-dysfunction and failure. These complications necessitate secondary interventions, associated with additional costs and affecting quality of life. Hence, KDOQI-Guidelines recommend a goal of ⩽3 interventions per-year to maintain VA-patency before considering another options. 1
Percutaneous transluminal angioplasty (PTA) has been the mainstay of endovascular treatment for VA-stenosis. However, the vessel wall trauma associated with PTA often leads to recurrent stenoses and recoil, which diminishes target lesion primary patency (TLPP). While “plain-old balloon angioplasty” (POBA) has been the most common type of treatment for many years, various additional technologies (cutting-, scoring-balloons (SB) or vessel preparation (VP)) have been developed and shown promise in improving interventional outcomes. One such balloon technology, that of SB, has been introduced to potentially improve the treatment of resistant stenotic lesions. 2 However, their effectiveness compared to POBA in VA-stenoses has not been extensively studied. Additionally, VP has been described in both peripheral arterial disease and VA-stenosis.2,3 VP may reduce PTA-associated diffuse barotrauma by facilitating a more controlled vessel expansion along the lines of microincisions. The additional application of drug-coated balloons (DCB) in VA has been increased with however mixed results.4–8 Uptake of drug application from DCBs might be improved after the use of either SB or VP, yet comparative data for these different treatment approaches in VA are lacking.
The purpose of this study was to compare the outcomes of POBA+DCB, SB+DCB and VP+DCB in the treatment of VA-stenoses and evaluate up to 12-month outcomes regarding the efficacy, patency and reintervention rates.
Materials and methods
Study design
A prospectively collected anonymized database of patients that underwent angioplasty VA-procedures (surgically created AVFs, AVGs and percutaneous AVFs (pAVF)) at a dedicated VA-center between June 2022 and October 2024 was retrospectively analyzed. Starting in June 2022, TriWedge® SB (BrosMed medical, Dongguan, China) has been introduced alongside the standard high- or ultra high-pressure PTA-balloon. This SB is characterized by 3 triangular scoring elements positioned at 4, 8 and 12 o’clock axially to the balloon. Starting in May 2023, FLEX Vessel-Prep® device (VentureMed, Minneapolis, USA) was introduced to our practice as a treatment option for VA-lesions. This device is designed to create longitudinal, controlled-depth, circumferential micro-incisions along the length of a lesion by a retrograde pullback approach of a self-sizing treatment element under low atmospheric pressure (maximal expansion up to 10 mm) with 0.25 mm microsurgical blades placed on the back side of three struts spaced at 180 degree intervals[3]. Three datasets were retrospectively reviewed, matched and compared. Only procedures using DCB were included in the analysis. This study complied with Ethics Committee and Institutional Review Board requirements and was conducted in accordance with the Declaration of Helsinki.
Procedures
All procedures were performed due to hemodynamically significant stenosis (>50%) in the VA-circuit, including patients with thrombosed VA after a thrombectomy to treat the culprit lesion. The choice of the treatment type was left to the surgeon’s discretion. For POBA, Mustang, Athletis (Boston Scientific, Marlborough, USA) or Conquest40 (BD and company, Franklin Lakes, USA) balloons were used. Procedures were performed in similar fashion under ultrasound guidance. In cases with difficult anatomy or the inability to visualize the area of interest (e.g. cephalic arch, axillary vein, etc.), fluoroscopy with a portable c-arm was additionally utilized. When POBA or SB-angioplasty were performed, the balloon was inflated between nominal and rated burst pressure (RBP) for ⩾3 min, with repeated inflation times and increasing balloon sizes, when necessary (incomplete balloon effacement, >30% residual stenosis, and/or <50% access flow (Qa) increase from pre-procedural baseline). Attention was paid to slow inflation and deflation times to reduce the risk of rupture and/or recoil. After PTA, a paclitaxel-coated DCB of either the same or 1mm larger diameter (Lutonix (BD); INPACT (Medtronic, Minneapolis, USA) or Aperto (Cardionovum, Bonn, Germany)) was applied for ⩾3 min. When VP was used, the procedure was performed first with 4-8 passes through the stenotic lesion(s) using the VP-device and identifying the changes in the lesion under ultrasound, when feasible. After both SB and VP, if there was no significant lumen increase, a POBA was used to additionally treat the lesion, followed by DCB for ⩾3 min. If there was an adequate lumen increase (complete balloon effacement, <30% residual stenosis, and >50% Qa after SB or ⩾50% lumen increase after VP), a DCB was applied for ⩾3 min. Adverse events were reported according to the Society of Interventional Radiology (SIR) guidelines. 9
Definitions
Patency rates were defined according to the postinterventional patency rates per SIR-recommendations 10 and Gray 11 : TLPP was the interval from the intervention until the next reintervention at/adjacent to the original location or until access abandonment. Access circuit primary patency (ACPP) was the interval from the intervention until the next reintervention on the access circuit or access abandonment. Secondary patency (SP) was the interval from the intervention until access abandonment. Technical success was the successful restoration of the lumen of the lesion with <30% residual stenosis. Reinterventions including access abandonment were counted as number of interventions per-patient-year. Juxtaanastomotic stenosis (JAS) was defined as 2–2.5 cm from the arteriovenous anastomosis extending into the AVF vein. Outflow vein stenosis was the area outside JAS and cephalic arch/axillary vein (typically cannulation site).
Demographics
Two-hundred fourteen interventions on VA (excluding stentgraft placements) were performed between June 2022 and October 2024. Of those, 175 patients were treated with DCB-angioplasty after POBA (n = 57), SB (n = 47), and VP (n = 71) and were included in the analysis. Patients’ demographics and VA-related history (Figure 1) are described in Table 1. Significantly more patients in the VP-group had previous interventions on their VA-circuit (53/71 (74.65%)) as compared to both POBA (32/57 (56.14%) and SB (25/47 (53.19%)) (p = 0.025). Additionally, significant difference was seen in the number of prior interventions on VA-circuit per group, with more previous interventions (mean 2.3, 95%CI 2.73, SD 2.22) in the VP-group as compared to both POBA (mean 1.00, 95%CI 1.46, SD 1.25) and SB (mean 1.04, 95%CI 1.55, SD 1.44) (p<0.0001). In total, 62.9% of patients had 1 or more previous interventions on their VA circuit. The distribution of different types of VA per each group was similar (p = 0.72) with majority of treated accesses being the RCAVFs.
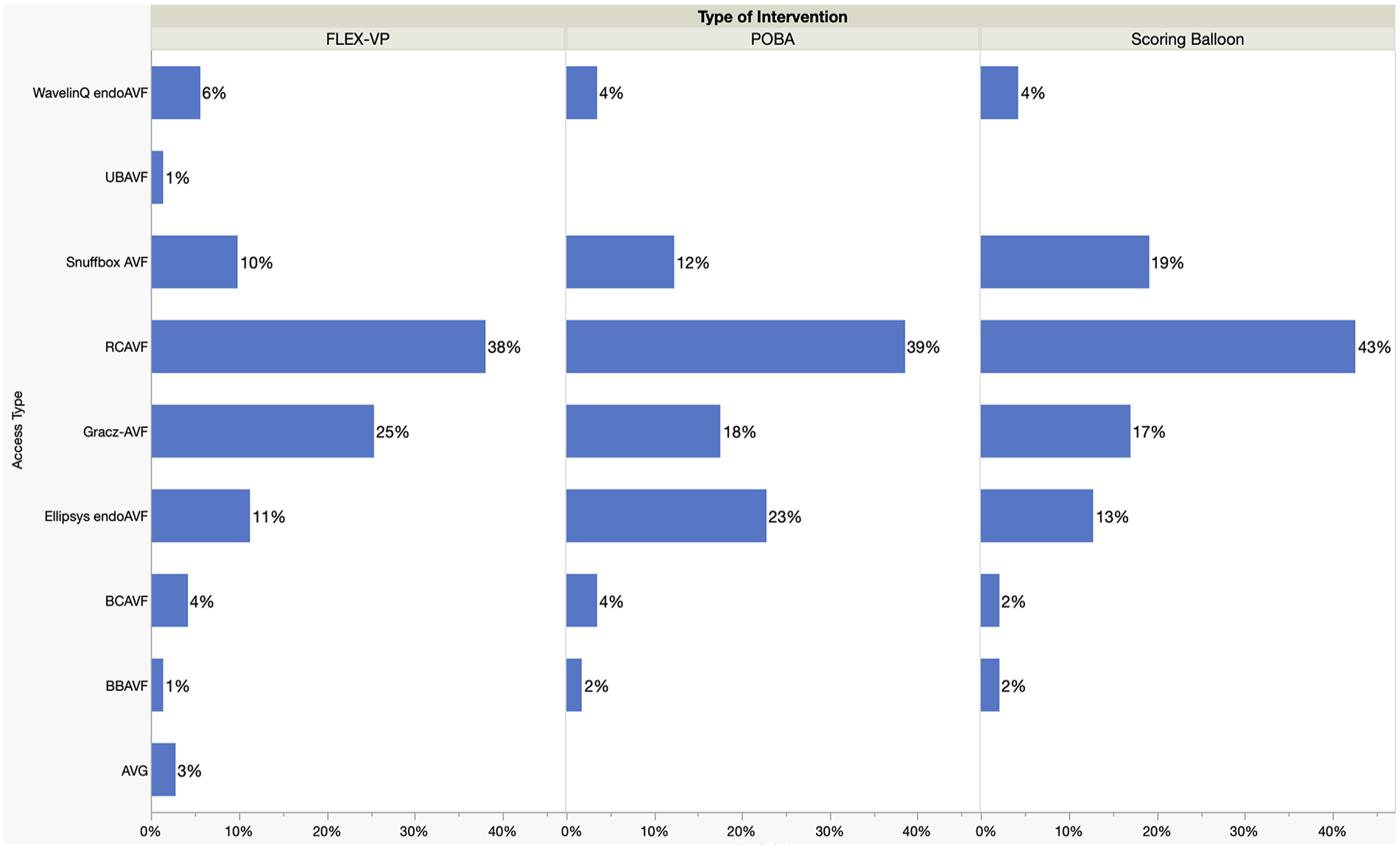
Distribution of different vascular access types per intervention-group.
Baseline patient and vascular access related demographics.
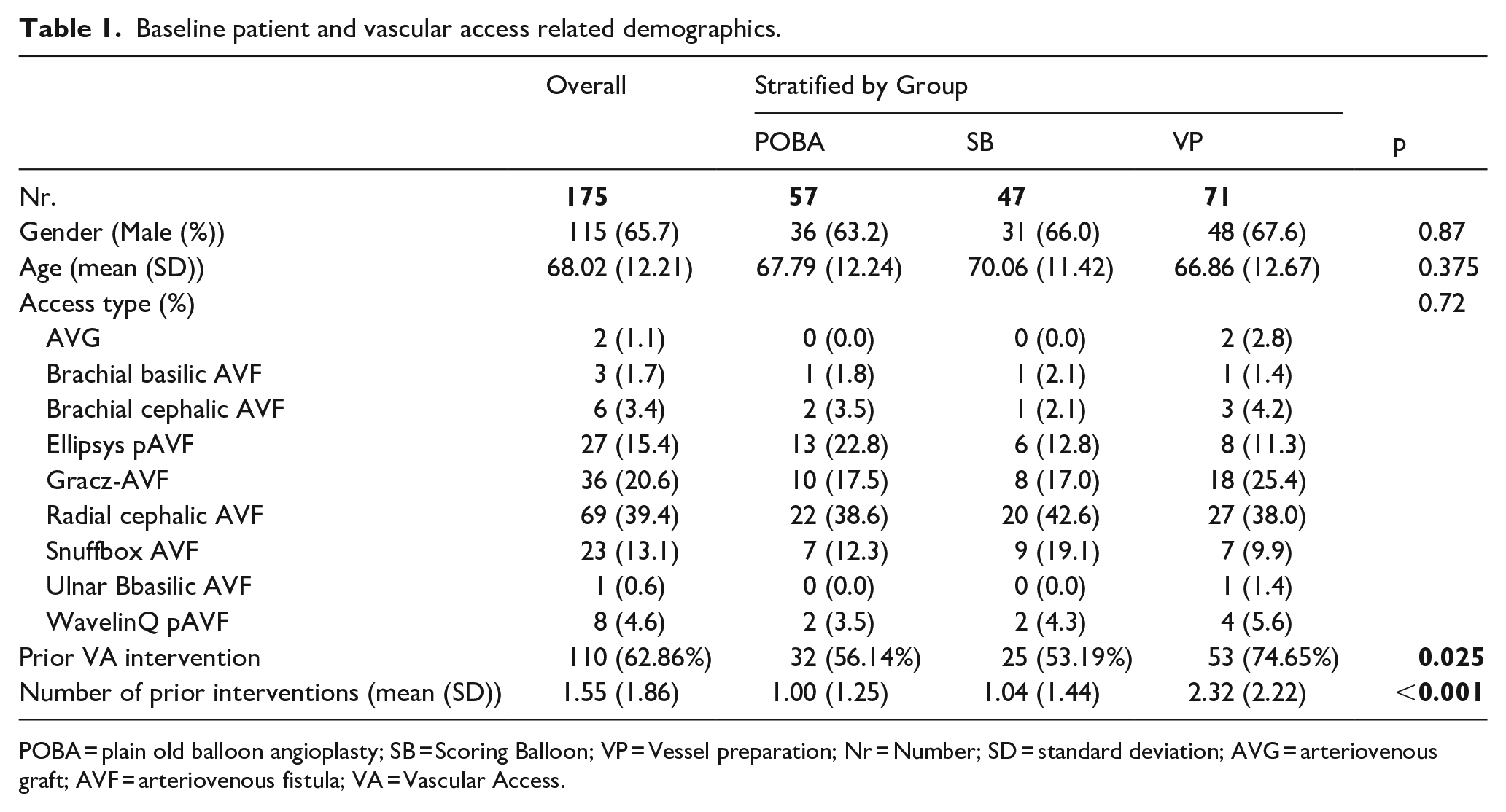
POBA = plain old balloon angioplasty; SB = Scoring Balloon; VP = Vessel preparation; Nr = Number; SD = standard deviation; AVG = arteriovenous graft; AVF = arteriovenous fistula; VA = Vascular Access.
Statistical analysis
Basic exploratory analysis was conducted to evaluate all study variables using descriptive statistics. Frequencies and percentages were calculated for categorical variables, while continuous variables were summarized as means with SD or medians with ranges, as appropriate. Normality of data distribution was assessed using the Shapiro-Wilk and Kolmogorov-Smirnov tests. For initial comparisons of baseline demographics, analysis of variance (ANOVA) was used for continuous variables across groups; where significant differences were detected, pairwise t-tests with Bonferroni correction were performed. Non-normally distributed continuous variables were compared using Mann-Whitney U tests. Associations between categorical variables and study groups were assessed using chi-squared or Fisher’s exact tests, as appropriate.
Patency loss was analyzed using Kaplan-Meier survival curves and Cox proportional hazard ratios (HRs), with HRs and their corresponding 95% confidence intervals (95% CI) reported. Multivariate Cox regression analyses were performed to further evaluate patency loss. Variables with a p-value <0.20 in univariate Cox regression were included alongside balloon type in the multivariate models. The hazard proportionality assumption was tested using Schoenfeld’s residuals. Censoring events in survival analyses included loss to follow-up, patient death, or the patient reaching the study endpoint without a patency loss event.
All statistical analyses were conducted using JMP version 18 (Cary, NC, USA), and R version 4.2.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
From 175 patients, technical success was achieved in 100% of patients across all three groups. Complete balloon effacement was achieved in 100% of POBA- and VP cases, and 91.5% in SB-group (p = 0.0046). 38% of the patients (27/71) in the VP-group were directly treated using a DCB without the need for POBA. The most common treatment segment was the outflow vein stenosis in 118/175 (67.4%), followed by JAS in 88/175 patients (50.2%). 22.9% of patients had combined JAS and outflow vein stenosis, 26.9% isolated JAS and 40.6% isolated outflow vein stenosis. In addition to that, cephalic arch stenosis was treated in 8 patients (4.6%), axillary vein, graft-venous anastomosis (2 AVGs and one RCAVF with interposition graft) and AV-anastomosis were treated in 3 patients (1.7%) each, respectively. Significantly more patients with JAS were in the POBA-group (70.2%) as compared to the SB- (48.9%) and VP-groups (35.2%) (p = 0.0004) (Table 2). No adverse events were reported.
periprocedural outcomes.
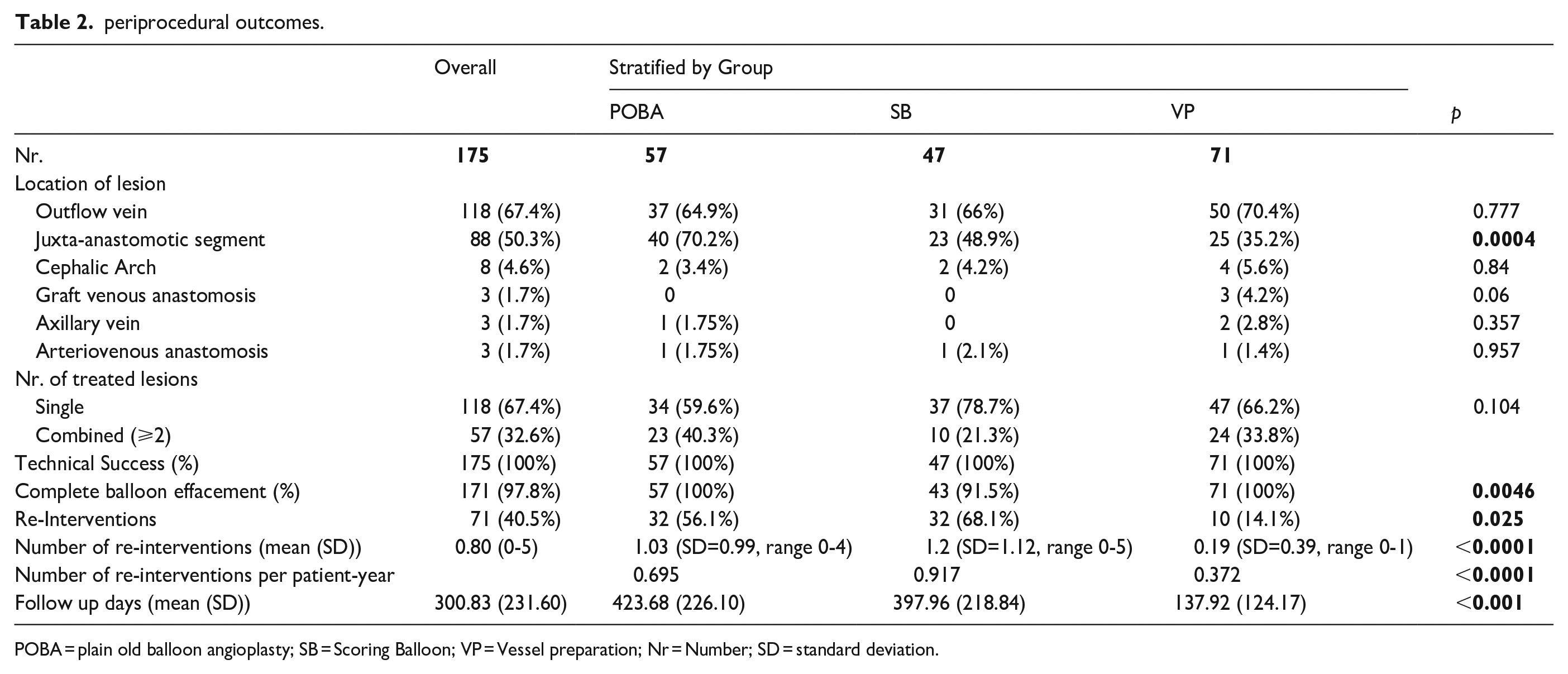
POBA = plain old balloon angioplasty; SB = Scoring Balloon; VP = Vessel preparation; Nr = Number; SD = standard deviation.
Reinterventions
71/175 (40.5%) patients underwent secondary reinterventions (Table 2). There was significant difference in reinterventions between groups with 14.1% of patients in the VP-group (10/71), 56.1% in the POBA-group (32/57) and 68.1% in the SB-group (32/47) during the study period. Mean number of reinterventions was 0.19 (SD = 0.39, range 0–1) for VP-, 1.03 (SD=0.99, range 0-4) for POBA- and 1.2 (SD = 1.12, range 0-5) for SB-group (p < 0.0001). Due to significantly different (p < 0.0001) mean follow-up period per group (424 days for POBA (SD = 226.1, range 27-850 days), 398 days for SB (SD = 218.8, range 2-809 days) and 138 days for VP (SD=124.2, range 2-496 days)), the number of reinterventions per-patient-year was analyzed. This resulted in a total number of reinterventions per-patient-year of 0.695, 0.917 and 0.372 for POBA-, SB- and VP-groups, respectively, with statistical significance for the VP-group comparing to both POBA and SB-groups (p < 0.0001). Interestingly, from 8 interventions involving cephalic arch (4 in VP-group and 2 each in POBA- and SB-groups, respectively), only 1 VP-patient required 1 reintervention on target lesion, whereas all POBA- and SB-patients lost the TLPP.
Patency analysis
The TLPP at 6 and 12 months was 79.1% and 40.1% for POBA, 67.7% and 41.2% for SB and 86.0% and 73.6% for VP, respectively (Figure 2). Median days to TLPP loss were 314 and 301 in the POBA and SB-groups, respectively. Median days to TLPP loss was not reached for VP-group (HRSB vs. POBA = 1.375, 95%CI = 0.787–2.403, p = 0.264; HRVP vs. POBA = 0.491, 95%CI = 0.228–1.060, p = 0.070). However, there was no statistical significance in TLPP difference between the POBA, SB and VP-groups. The HR for TLPP loss in the SB-group relative to POBA was 1.277 (95%CI = 0.783–2.082, p = 0.327) and in the VP group relative to POBA was 0.570 (95%CI = 0.269–1.207, p = 0.142, Table 3). The logrank test statistic was 4.97 (p = 0.08). In multivariate analysis, history of prior intervention and number of reinterventions were included in the model (logrank test p-values = 0.02 and <0.001, respectively). The overall model was significant (logrank test p < 0.001). The HR of the number of reinterventions was 2.103 (95%CI = 1.721-2.570, p < 0.001, Table 4).
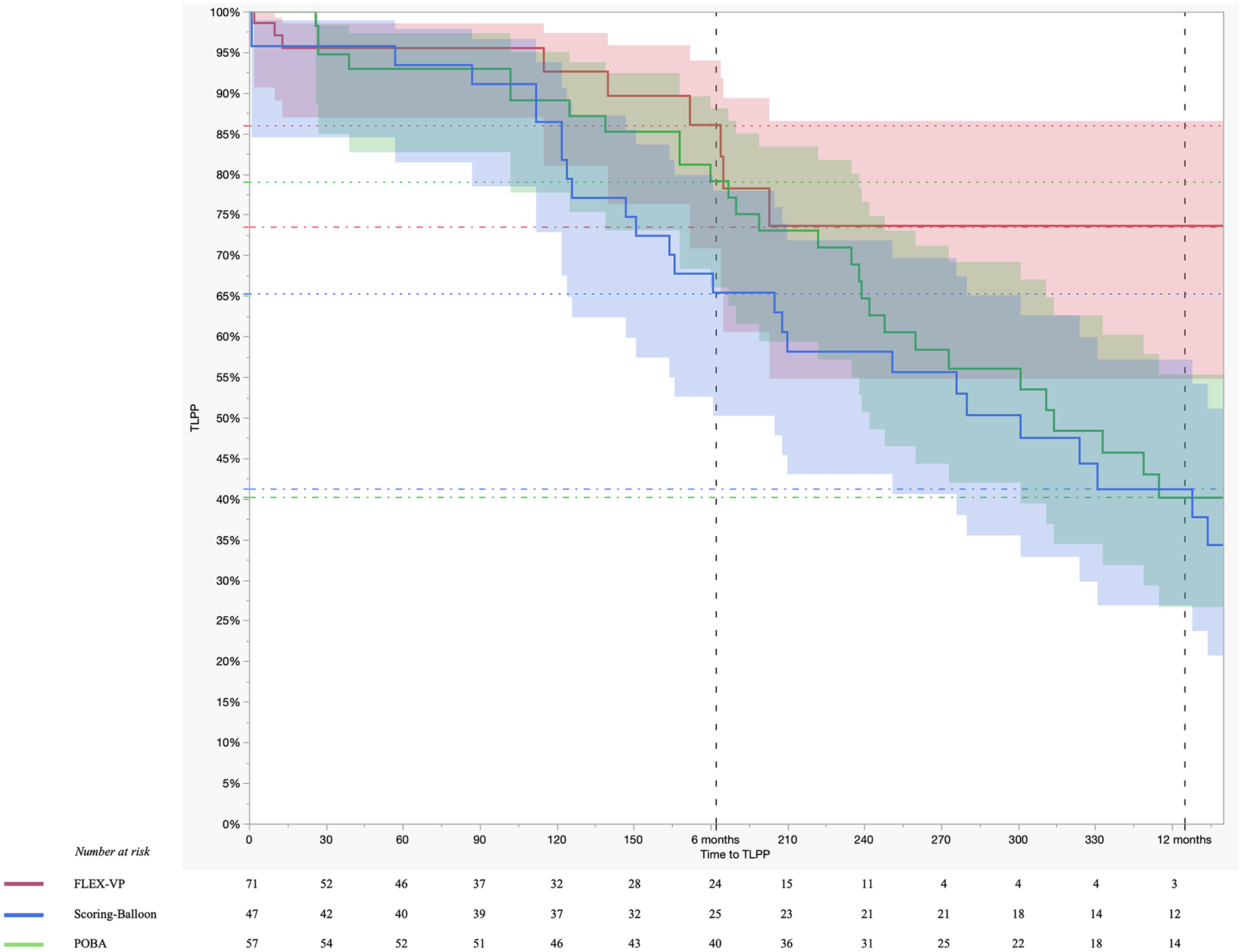
Kaplan-Meier analysis of target lesion primary patency rates.
Univariate Cox regression analyses of access circuit primary patency, secondary patency and target lesion primary patency.
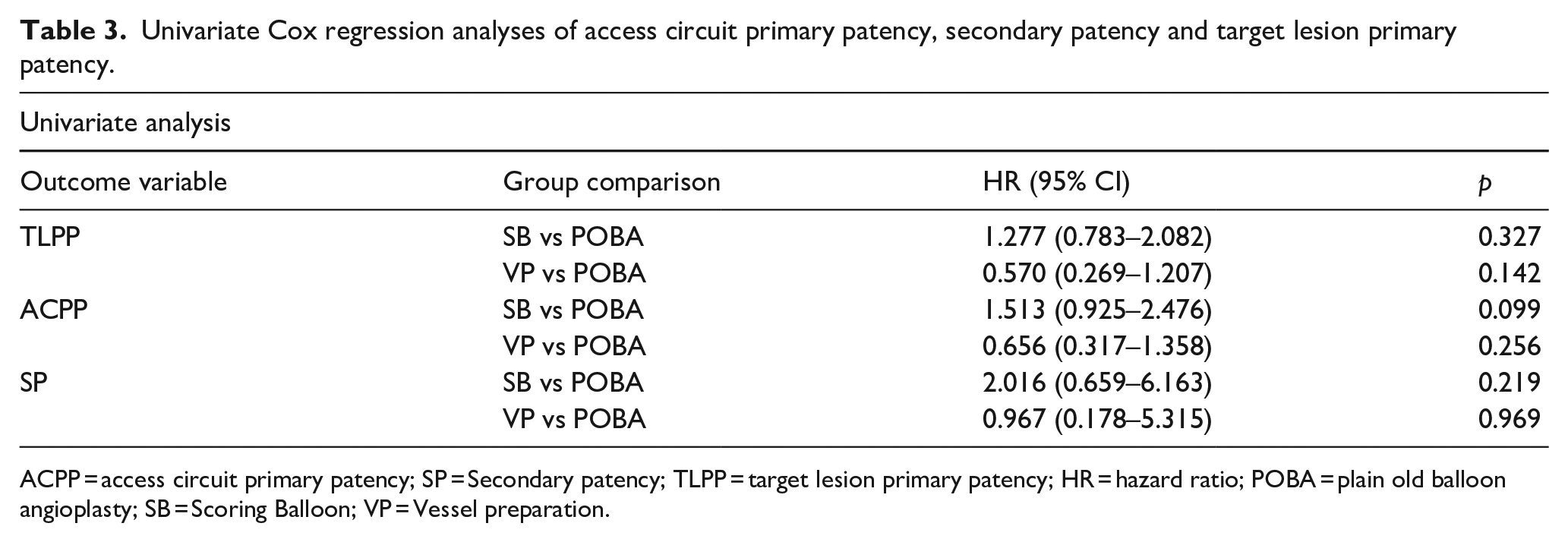
ACPP = access circuit primary patency; SP = Secondary patency; TLPP = target lesion primary patency; HR = hazard ratio; POBA = plain old balloon angioplasty; SB = Scoring Balloon; VP = Vessel preparation.
Multivariate Cox regression analyses of access circuit primary patency, secondary patency and target lesion primary patency.
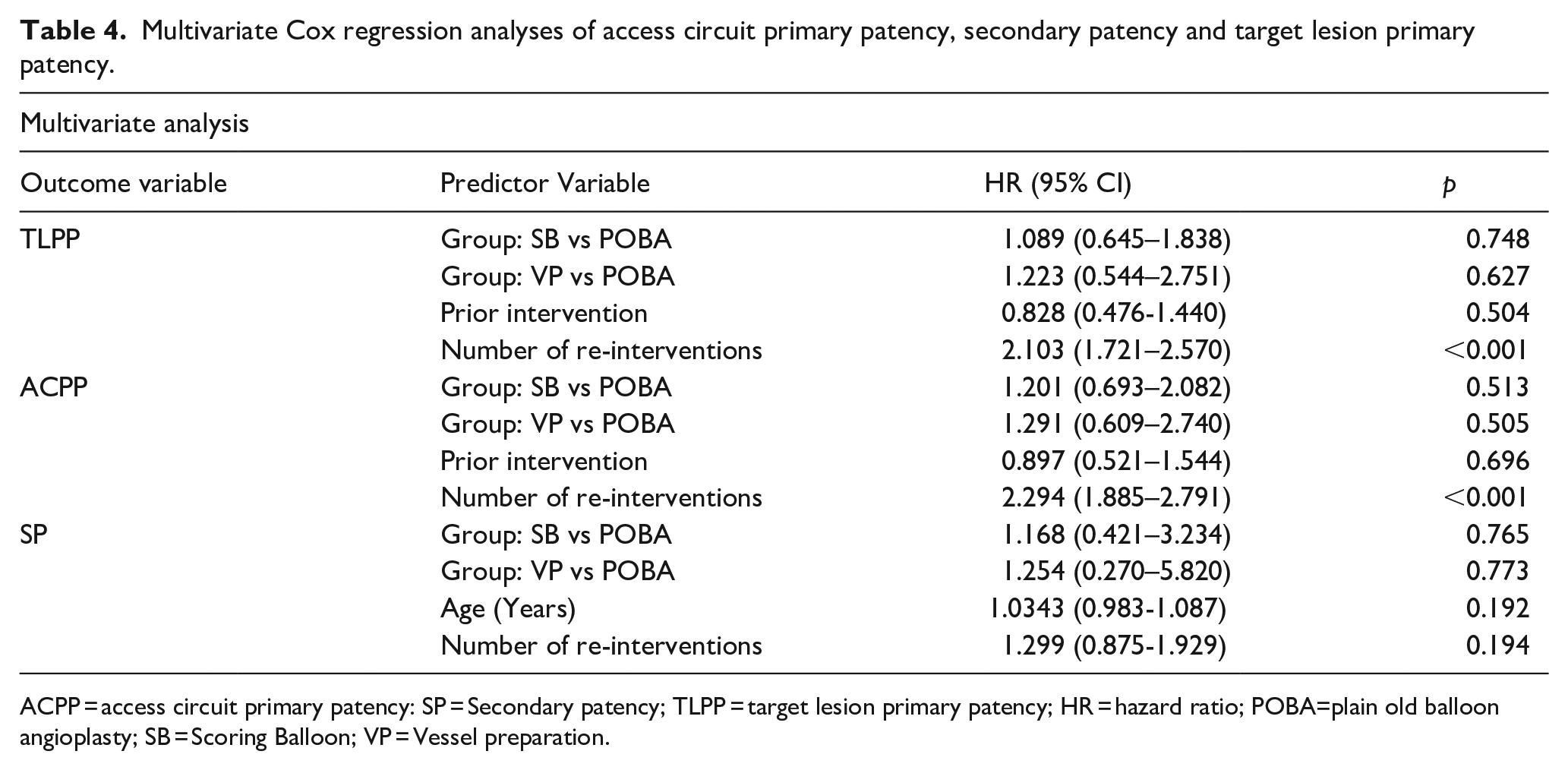
ACPP = access circuit primary patency: SP = Secondary patency; TLPP = target lesion primary patency; HR = hazard ratio; POBA=plain old balloon angioplasty; SB = Scoring Balloon; VP = Vessel preparation.
ACPP was higher in the VP-group without statistical significance between all groups. The HR for ACPP-loss in the SB-group relative to POBA was 1.513 (95%CI = 0.925–2.476, p = 0.099) and in the VP-group relative to POBA was 0.656 (95%CI = 0.317–1.358, p = 0.256) (Table 3). The logrank test statistic was 6.31 (p = 0.04). The ACPP at 6 and 12 months was 78.5% and 37.9% for POBA, 65.4% and 35.6% for SB and 84.7% and 57.0% for VP, respectively (Figure 3). Median days to ACPP loss were 311 among POBA, 280 among SB and were not reached for VP. No significant difference was seen when the analysis was limited to the first year after the intervention (HRSB vs. POBA = 1.491, 95%CI = 0.860–2.584, p = 0.155; HRVP vs. POBA = 0.614, 95%CI = 0.290–1.301, p = 0.203). In multivariate analysis, history of prior intervention and number of reinterventions were included in the model (logrank test p-values = 0.2 and 2 x 10-15 respectively). The overall model was significant (logrank test p < 0.001), but the only significant variable was the number of reinterventions for the VP-group (HR = 2.294, 95%CI = 1.885–2.791, p < 0.001) (Table 4).
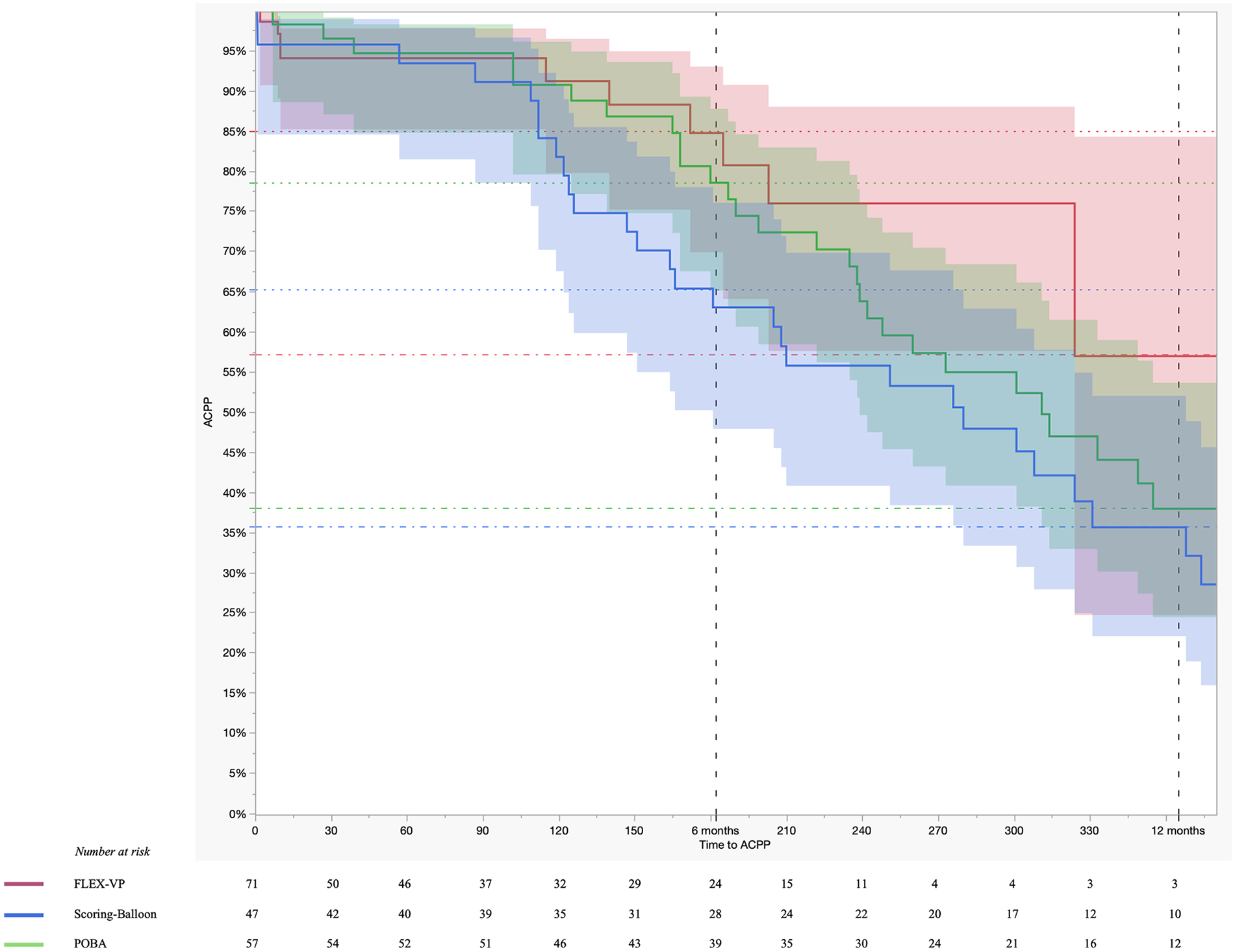
Kaplan-Meier analysis of access circuit primary patency rates.
There was no significant difference regarding SP, with high SP-rates at both 6 and 12 months. The SP rate at 6 and 12 months was 98.0% and 93.7% for POBA, 90.7% and 87.9% for SB and 97.1% and 97.1% for VP, respectively (Figure 4). Median days to SP loss was not reached for any of the groups. The HR for SP loss in the SB-group relative to POBA was 2.016 (95%CI = 0.659–6.163, p = 0.219) and in the VP-group relative to POBA was 0.967 (95%CI = 0.178–5.315, p = 0.969, Table 3). The logrank test statistic was 1.85 (p = 0.40). No significant difference was seen when the analysis was limited to the first year after access creation (HRSB vs. POBA = 2.323, 95%CI = 0.555–9.729, p = 0.249; HRVP vs. POBA = 0.525, 95%CI = 0.083–3.311, p = 0.496) (Table 3). In multivariate analysis, age and number of reinterventions were included in the model (logrank test p-values = 0.1 and 0.07, respectively). The overall model was borderline significant (logrank test p = 0.05), but no individual variables reached the level of significance (Table 4).
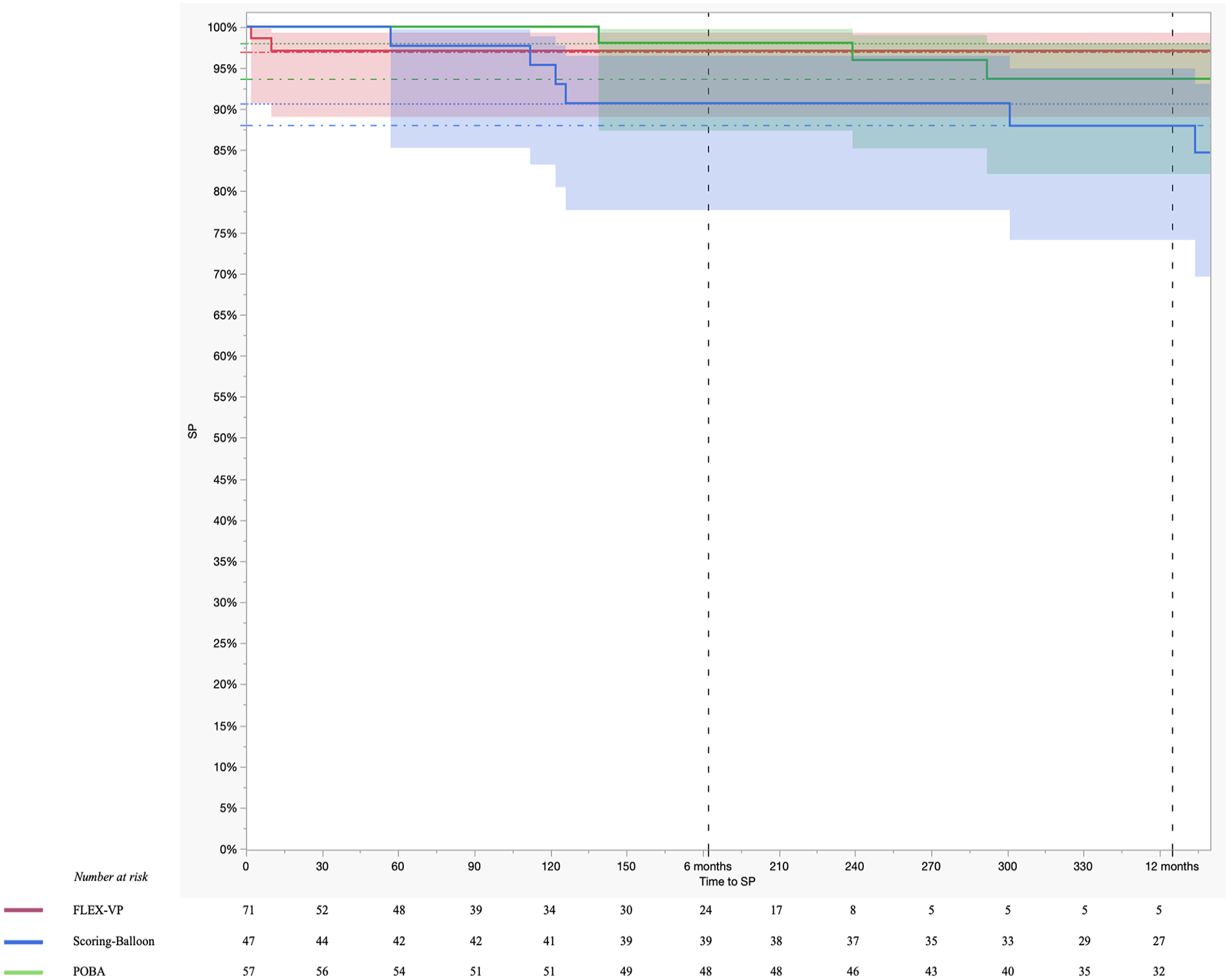
Kaplan-Meier analysis of secondary patency rates.
Discussion
Though PTA remains the preferred method for the treatment of VA stenosis, reported TLPP rates at 6 months range widely between 0-85%: and are very low at 12 months4,6,12,13 Many of these studies have been randomized controlled trials (RCT) with specific patient populations and strict inclusion/exclusion criteria,5,7,14 limiting applicability to a general patient population. Real-world outcomes in diverse patient populations may be different than those reported in RCTs. Additionally, DCBs were recently introduced into the VA space, and the advent of vessel wall scoring technology by using a SB or a VP-device has generated the question if combination of vessel wall scoring and use of a DCB lead to longer inter-procedural intervals through longer TLPP and/or ACPP. This study compares the efficacy of three different endovascular treatment modalities for dysfunctional VA: POBA, SB and VP, each combined with DCBs. Our patient population included surgically created AVFs, pAVFs, and some AVGs in VP-group.
In this series, no significant differences in the hazard of patency loss between the POBA, SB and VP-groups were found in univariate analysis. Across various multivariate analyses, a greater number of prior reinterventions was consistently associated with increased risk of patency loss. The VP-group had the patients with the most prior interventions, and despite that, at 6 and 12 months both TLPP and ACPP were higher than for POBA and SB (86.0% and 73.6% for TLPP and 84.7% and 57.0% for ACPP, respectively). Our results are similar to those reported in a study by Aruny et al., 3 where 148 lesions were treated using the VP prior to POBA-PTA. At 6 months, the TLPP was 62.2%, with average time to revascularization of 202.7 days. For patients with AVFs, TLPP was 67.5%, whereas for AVGs it was 52.4%. Interestingly, among the cases involving cephalic arch stenosis, the 6-months TLPP rate was 70.6%: higher than reported in the Rajan and Falk 15 , Beathard et al. 16 Both, the study by Aruny and our study found no device related adverse events.
Notably, the results from current study regarding the TLPP and ACPP are comparable with those from RCTs and some using stentgrafts for the treatment of VA-stenoses.17–19 Furthermore, the outcomes of VP+DCB from our study demonstrate even higher TLPP and ACPP rates than some studies using stentgrafts.17–19 Knowing that stentgrafts have better outcomes as compared with POBA, this underlines that performing good VP and (possibly prolonged and slow) angioplasty in combination with DCB is at least comparable (and even possibly better) to stentgrafts yet does not leave a stentgraft in the VA-circuit. Dedicated studies comparing VP+DCB versus stentgraft may inform treatment choices in light of the higher cost for stentgrafts, the known development of edge stenosis, and the consequences of stentgrafts in the cannulation segment.20–22 The recent availability of cell-impermeable endoprosthesis (WrapsodyTM, Merit Medical, Salt Lake City, USA) add to the treatment options to be explored in the future. 23
The role of DCBs in the treatment of VA still remains to be defined. Studies with different DCBs (including RCTs) have demonstrated variable outcomes after POBA plus paclitaxel-coated DCBs versus POBA alone5,24,25 – notably without increased mortality in the DCB-groups.8,14 In pAVFs, use of DCBs vs POBA may be associated with a decrease in procedure frequency without a difference in ACPP. 26 A potential benefit of vessel wall scoring prior to use of a DCB was first suggested by a cutting ballon study, where TLPP at 6 months after using a cutting-balloon with DCB was 74.1%, whereas in the group treated with cutting-balloon alone it was 65.9%, and only 40.4% using POBA+DCB. 27 In comparison, our POBA+DCB-group demonstrated 79% TLPP at 6 months, while TLPP was 86% for VP+DCB and the SB+DCB was lower at 67.7%. Of the three different pre-treatment groups (VP, SB, and POBA), POBA is the technique with the greatest variability of opening the vessel wall to drug exposure: PTA is a “controlled” rupture of the vein wall, which is modified by the degree of wall elasticity, scar tissue, etc. We did not collect data in the POBA group on US or fluoroscopic signs of extravasation which would indicate a large possible exposure to drug delivered by the DCB. Similarly, there is no objective measure of barotrauma associated with the use of an ultra-high pressure versus a normal pressure angioplasty balloon, which is thought to be relevant in stenotic recoil and more aggressive myointimal hyperplasia. One can only speculate how a high-pressure DCB, such as the Aperto, affects the need for high pressure ballons after VP or SB. 25 Interestingly, an economic evaluation has demonstrated that DCBs can be cost-effective in VA-treatment 28 and when combined with VP might even further reduce the follow-up costs for healthcare systems by significantly reducing the number of reinterventions.
Annualized re-intervention rates were smallest in the VP-group compared to POBA- and SB-groups (p < 0.0001), despite the significantly higher rate of prior interventions per year in the VP-group. Overall shorter follow-up in the VP-group (mean follow-up 138 days, vs 398 and 424 days in SB and POBA, respectively) may have introduced a bias in the other direction.
Use of Sirolimus-coated DCBs in the MATILDA trial demonstrated promise in treating VA-stenosis in an initial single arm trial. 29 RCTs comparing POBA versus sirolimus-coated DCB (IMPRESSION) and POBA, paclitaxel-, and sirolimus-coated DCBs (PAVE-2) are under way in Singapore and the UK, respectively. These trials will inform studies on what drug to combine with any form of vein wall preparation to improve drug delivery30,31 If the sirolimus-balloon demonstrates even better outcomes in the future, theoretically this might additionally improve of the outcomes in combination with the VP-device.
Limitations
One limitation is the potential difference in lesion characteristics between the groups. We observed a significant difference in lesion location, with a higher proportion of JAS in the POBA-group. This disparity could have influenced our results and warrants further investigation with more balanced lesion distributions, when more patients are being treated. Moreover, there was a significantly different follow-up between groups. Additionally, the retrospective and non-randomized character of the study remain the main known limitation of a comparison analysis.
Conclusion
In conclusion, both TLPP and ACPP were trending higher after VP as compared to POBA and SB-angioplasty at up to 12 months. The number of prior interventions was identified as a significant factor affecting patency rates, yet the number of previous interventions was significantly higher and follow-up reinterventions lower in VP-group as compared to both other groups. Future research should focus on evaluating the cost-effectiveness of VP and DCB in a RCT and identifying potential benefit in different lesion locations and investigating factors that influence the long-term success after endovascular treatment of VA-stenoses.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RS: Consultant for Becton Dickinson and Company/BARD, Laminate medical Inc., Medtronic Inc., XELTIS BV, VentureMed; received speaker fees from BrosMed Medical and Cardionovum; Scientific Advisory Board member for VENOVA Medical.
TIM: none
DMH: Consultant for Becton Dickinson and Company/BARD, Laminate medical Inc., Medtronic Inc., Merit Medical, Venostent; Scientific Advisory Board member for Nephrodite, VENOVA Medical
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
