Abstract
Effective hemodialysis (HD) depends on efficient, durable, and safe vascular access. Despite incessant efforts to reduce the use of tunneled central venous catheter (tCVC), late referrals, poor planning, improper creation of arteriovenous fistula, and lack of appropriate blood vessels are responsible for high prevalence of tCVC in the dialysis population. Specific mechanical problems, such as cracked hubs, broken clamps, leaky connectors, and bloodline rupture, can jeopardize the tCVC integrity. In case of tCVC damage at the distal part of the extracutaneous portion, most catheters are usually fixed using specific repair kits. However, when the bloodline is cracked close to the exit-site, the catheter exchange over a guide wire or the placement at a new site are considered the only viable options. However, the preservation of patient vascular assets should be prioritized, especially in patients with limited or exhausted access possibilities. Thus, in order to rescue the catheter and preserve the patient’s vascular asset, we describe an innovative technique to fix tCVC bloodline perforations located in the proximity of the exit-site. The procedure consists in creating a new exit-site through the shortening of the tunnel tract. The intervention of exit-site relocation by external splicing represents an attractive option to treat unfixable tCVC damage or tunnel erosion without the need for catheter removal or exchange. This mini-invasive procedure is rapid, safe, and optimally tolerated by the patient. However, the prerequisites for this procedure are an exit site-cuff distance of at least 5 cm and the availability of an adequate repair kit. Exposition of the device along the subcutaneous tract due to chronic erosion of the tunnel can be simultaneously resolved by this intervention.
Introduction
Effective hemodialysis (HD) depends on efficient, durable, and safe vascular access. When consistent with patient’s end-stage kidney disease (ESKD) life-plan, arteriovenous fistula (AVF) is recognized as the preferred vascular access for chronic maintenance hemodialysis therapy due to its good patency and lower complication rate.
Despite incessant efforts to reduce the use of tunneled central venous catheter (tCVC), the prevalence of patients affected by ESKD being dialyzed via a tCVC in the United States is approximately 25%, while this value draws near 35% in Italy.1,2 In addition, more than 84% of incident ESRD patients in the United States are initiated on hemodialysis with tunneled catheters. 1
Late referrals, poor planning, and improper creation of AVF, as well as lack of appropriate blood vessels represent the main culprits of the high existence of tCVC in the dialysis population.
The common tCVC complications such as catheter dysfunction, infection, thrombosis, fibrin sheath formation, central vein stenosis, and increased morbidity/mortality related to catheter adoption are well recognized and widely described. 3
However, specific mechanical problems, which jeopardize the catheter integrity, such as cracked hubs, broken clamps, leaky connectors, and bloodline rupture are seldom reported, even though these events may determine severe consequences.3,4
In case of tCVC damage at the distal part of the extracutaneous portion, most catheters are usually fixed using specific repair kits.4,5 However, when the bloodline is cracked close to the exit-site, either catheter exchange over a guide wire or the placement at new site are considered the only viable options. 4 However, especially in patients with limited or exhausted access possibilities, the preservation of patient vascular assets should be prioritized.
In this case report, we describe how we managed to solve a case of Tesio bloodline perforation with simultaneous exposition of the device along the subcutaneous tract due to chronic erosion of the tunnel.
Case report: Rescue of damaged tunneled central venous catheter and restoration of the existing tunnel by exit-site relocation
A 68-year-old woman suffering from ESRD due to unknown cause started HD treatment from the age of 25 years old. She underwent kidney transplantation from deceased-donor at age 27 therefore, the organ was explanted after 20 days due to sepsis from unknown causes. Following this complication, the patient decided to not be transplanted again, and underwent HD treatment ever since. Her medical history is notable for total parotidectomy, intradialytic hemolytic crisis, arterial hypertension, hyper-polymenorrhagia, and left femoral neck chondroma.
To permit a long-term survival and the best quality of life of the patient, initially a distal autologous AVF at the right arm was created having a good functioning and permitting a great dialysis efficiency for almost 20 years. Subsequently, due to AVF failure, various attempts to create a distal AVF at the same arm first, and subsequently a proximal AVF at the left arm were carried out. Both fistulas had a shorter longevity due to recurrent stenosis treated numerous times by percutaneous transluminal angioplasty. Eventually, in December 2019, a long-term cuffed Tesio catheter was placed in the right internal jugular vein. Since then, both arterial and venous branches of the CVC have always showed a good functioning.
In January 2024, during a HD session, an anomalous leakage of blood drops from the venous branch of the CVC was noticed. Hemodialysis treatment was disrupted, and blood was returned to the patient from the arterial line of the CVC. Washes with physiological solution were performed to recognize the site of the CVC fissure, which was adequately identified (Figure 1A and B), and a plastic clamp was placed proximally at the perforation. Moreover, it was noted that the CVC was partially exposed along the subcutaneous tract due to a cutaneous ulceration (Figure 1C and D). However, at the physical exam neither purulent discharge at the catheter epidermal interfaces, nor the presence of clinical inflammation (erythema, swelling, tenderness, or induration) along the catheter tunnel was observed. The blood test results were negative for signs of inflammation (white blood cells 4.85 cells/ul3, C reactive protein 0.92 mg/dl [nv <1]).
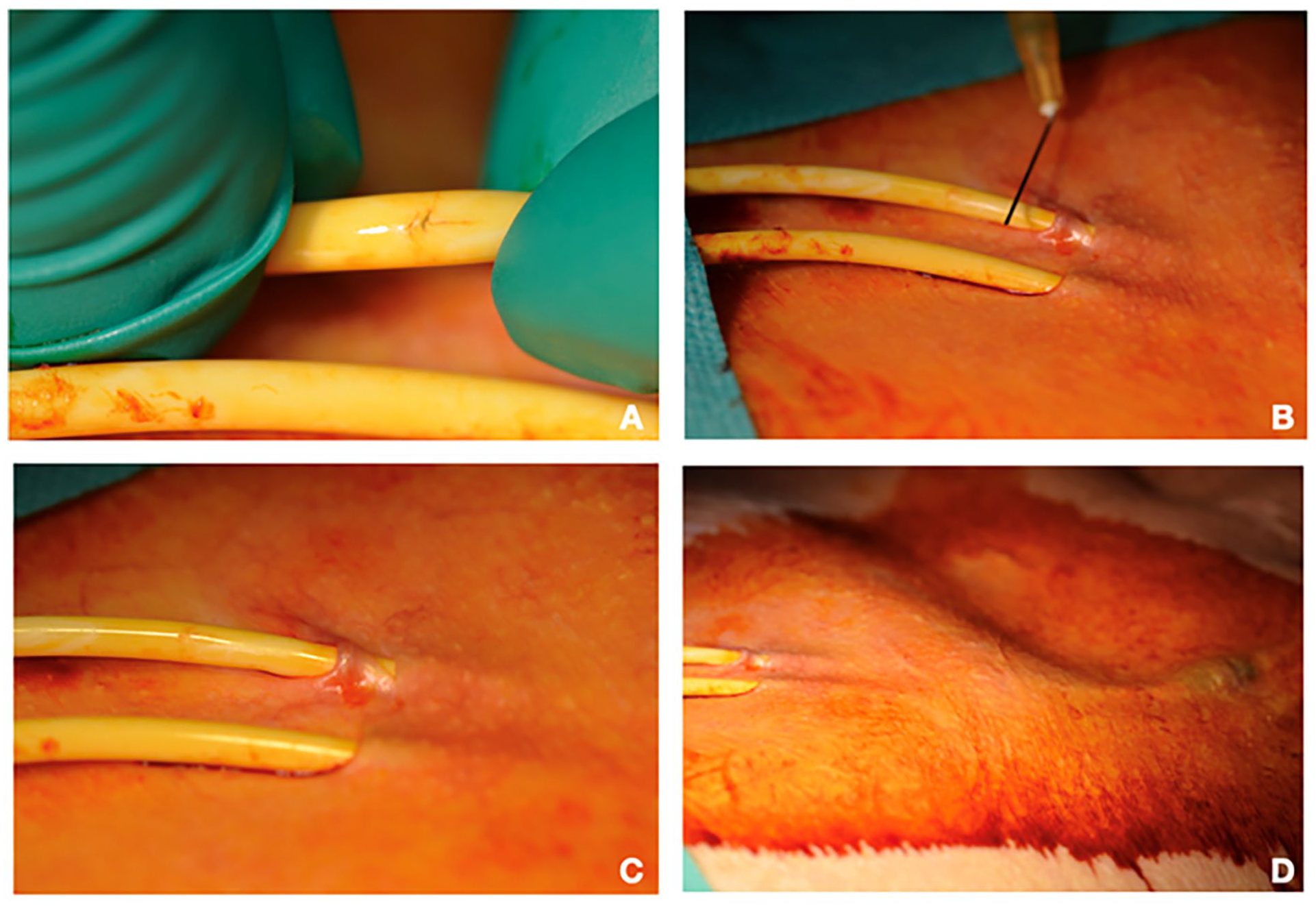
(A–D) Damage of the venous branch of the Tesio catheter with simultaneous tunnel erosion: (A) identification of the tiny perforation on the venous branch of the Tesio catheter; (B) the tip of the needle pinpoints the precise position of the damage on catheter; (C) presence of tunnel erosion approximately 1 cm from the exit-site; (D) it is possible to observe the generous distance between the exit-site and the subcutaneous cuff.
Since the patient carried a long history of HD treatment and the use of the tCVC was the last option as permanent dialysis access due to consecutive AVF failures, we decided to perform a mini-invasive surgical procedure to shorten the tunnel tract and create a new exit-site.
The exit-site and skin were disinfected with 1% povidone iodine, while the extensions and Luer connectors of Tesio catheter were excluded from the surgical field by sterile drapes. Under local anesthesia (lidocaine 1%, Figure 2A), the exit-site relocation by external splicing consisted in creating a skin incision of 3–4 mm in a point located halfway between the previous exit-site and the Dacron cuff of the venous branch of the Tesio catheter (Figure 2B). Hence, by this tiny opening, the tube was retrieved by a blunt dissection (Figure 2C–E). Subsequently, the lumen of the venous branch was cut at approximately 5 mm distance from the perforated area of the tube and lifted out from the skin through the previous incision (Figures 2F and G and 3A and B). Then, the catheter was clamped and accurately disinfected by wrapping all around sterile gauze soaked with 50% sodium hypochlorite solution for 5 min (Figure 3C).
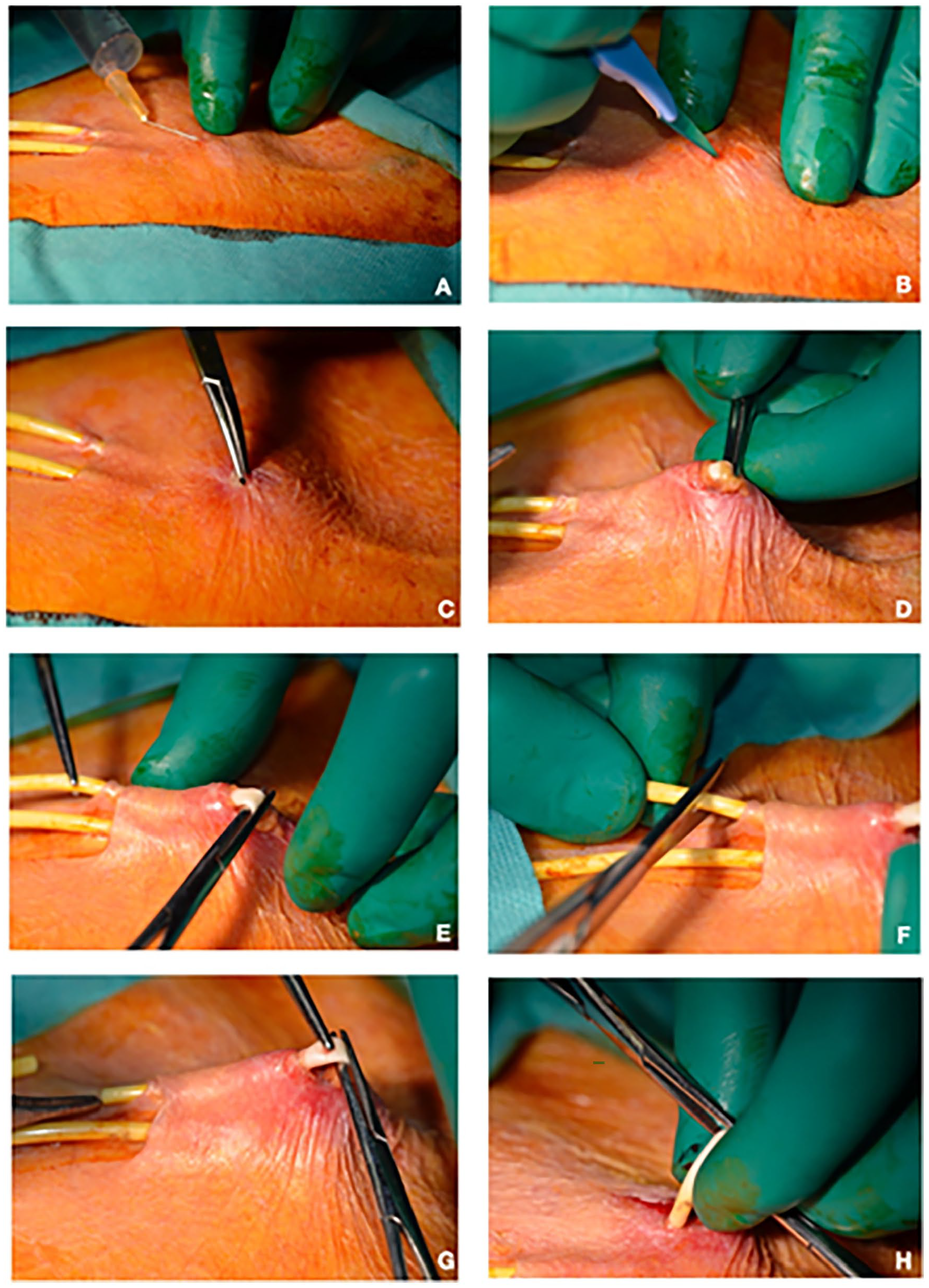
(A–H) Isolation and retrieval of the venous branch of the Tesio catheter: (A) subcutaneous injection of 1% lidocaine solution; (B) 3–5 mm cutaneous incision by scalpel at midway between the subcutaneous cuff and exit-site of the venous branch of the Tesio catheter; (C–E) by the tiny opening the catheter is isolated and retrieved by blunt dissection; (F) the extra-cutaneous portion of the catheter is excised beyond the perforation point; (G, H) the remaining portion of the catheter is lifted out from the subcutis through the previous skin incision.
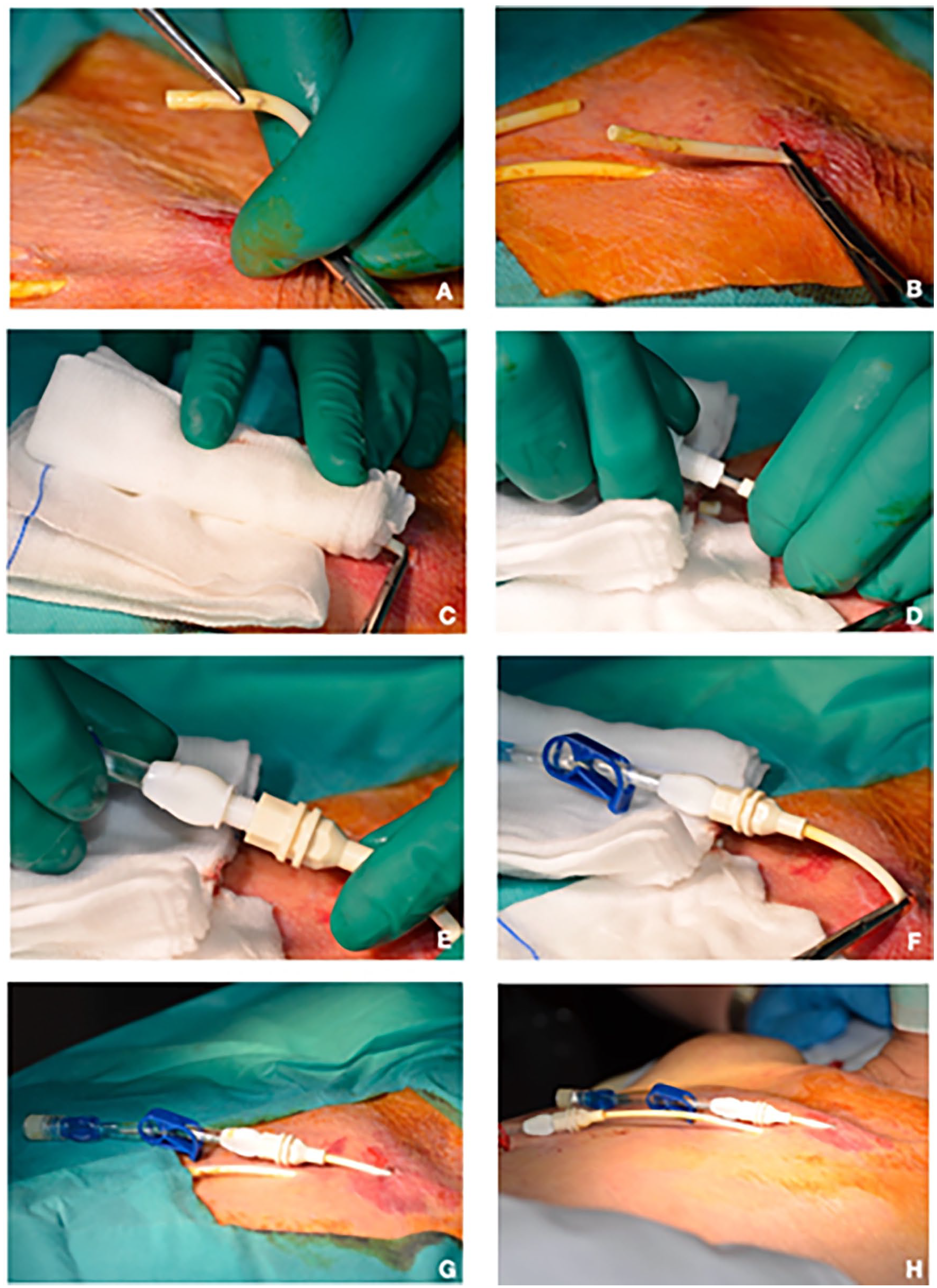
(A–H) Connection of the new extension the venous branch of the Tesio catheter: (A, B) once the catheter is retrieved from the subcutis, it is immediately clamped in proximity of the new exit-site; (C) the new extra-cutaneous portion of the catheter is accurately disinfected by wrapping all around sterile gauze soaked with 50% sodium hypochlorite solution for 5 min; (D–F) the venous branch of the catheter is externally lengthened by connecting a new hub extension; (G, H) final result of the procedure: the tunnel of the venous branch of the catheter has been shortened and the new exit-site is now located 3–4 cm above the previous one.
After dragging out the free end of the catheter branch from the skin, the tube was externally lengthened by connecting a new hub extension (Figure 3D–F). After removing the clamp, the catheter was flushed with 0.9% sodium chloride solution and the lumen was filled with heparin after discarding the volume of the previous anticoagulant lock. As a result, the initial tiny incision became the new exit-site of Tesio venous branch, which was now located 3–4 cm upwards from the old exit-site (Figure 3G and H). Excellent catheter performance was assessed by recording effective blood flow (>300 ml/min) during the following 12 months.
Discussion
Complications related to the wear of tCVC are seldom described in the medical Literature, even though these events are surely more common than reported and might lead to the removal of the catheter. 3 It was observed that almost 20% of premature tCVC removal is caused by mechanical damage, 6 while catheter breakage percentage was estimated to be included between 3% and 9%. In the study by Letachowicz et al. was reported an incidence rate of mechanical failure equal to 0.36/1000 catheters-days. 4 Nearly 67% of the events were related due to luer connector breakage, while line rupture and extension damage occurred in almost 22% and 11% of the cases, respectively.
In case of retrograde tCVC when connector damage, cracked clamps and extension breakage occur, is usually possible to fix the device using specific repair kits. However, if the tCVC damage is located in the proximity of the exit-site, the repairing is unfeasible, because the remaining intact part of the catheter is too short. In such cases, catheter exchange over a guide wire or its implant at new site are deemed the only possible alternatives.
Thus, in order to rescue the catheter and preserve the patient’s vascular asset, we decided to relocate the exit-site shortening the tunnel tract. The intervention enabled to obtain a sufficient catheter length to apply a new extension. These procedures are not unknown for interventionalists who possess experience with peritoneal dialysis (PD) catheter revision. In fact, either in case of PD catheter damage or tunnel infection, exit-site relocations are performed with excellent outcomes.7–9
However, the difference between the two contexts is usually represented by the length of the tunnel and both the distance and the numbers of the cuffs.10,11 In fact, in PD catheters the tunnel length is usually longer, being the deep cuff positioned intramuscularly or at the abdominal fascia (hence, at least approximately 8–10 cm distant from the skin exit-site) and in case of double-cuff PD catheters the subcutaneous superficial cuff can be removed, if necessary. On the other hand, the tCVC Dacron cuff cannot be excised due to the subsequent high risk of catheter extrusion. Additionally, the subcutaneous length of the tCVC tunnel is usually crafted shorter, especially in case of anterograde-tunneling models.
Nevertheless, the experience derived from peritoneal catheter showed that the subcutaneous cuff migrated approximately 1.5 cm toward the exit-site just 1 year after the placement of the device. 12 Furthermore, tunneling the subcutaneous cuff at least 4 cm internal to the exit-site yielded to a decrease of exit-site infection and catheter-related peritonitis as well as the reduction of exit-site infection refractory to medical therapy. 13 In fact, tunnel infections with cuff involvement are unlikely to respond to antibiotic therapy and in most cases require surgical intervention.7,10,14–16
Thus, creating a short tunnel from the exit site to the subcutaneous cuff may predispose to cuff extrusion and bacterial colonization resulting in higher incidence of catheter dislocation and infection-related complications.
In this regard, being the extravascular portion of the Tesio catheter significantly longer than the counterparts, it enables to create a longer and more personalized subcutaneous path. This feature makes feasible tunnel revision either in cases of tCVC unfixable damage or tunnel ulceration. The presented case was emblematic, because the two complications (tCVC perforation and cutaneous decubitus) were simultaneously present, and we were able to resolve both at the same time without the need of catheter replacement via the intervention of exit-site relocation.
Notably, this procedure can be equally performed in case of monotube bi-lumen tCVC with “Y junction” provided that a new suitable Y hub kit is available, and an adequate exit site-cuff distance is present. In fact, it is necessary to create a skin incision slightly larger (5–8 mm) halfway between the previous exit-site and the Dacron cuff of the catheter to retrieve the single catheter tube by the small opening. Hence, after cutting the tube and dragging out the free end of the catheter from the skin as previously described, the tube could be externally lengthened by connecting a new Y hub extension to obtain a very similar result.
Learning points
The weighty learning points of this case report are summarized in Table 1. Additionally, there are few other aspects and helpful tips regarding the technique that may deserve to be deliberated upon further.
Summary of important teaching points.

IV: intravenous; tCVC: tunneled central venous catheter.
(1) The creation of longer tunnel (>5 cm) during tCVC placement increases the future possibility to perform “rescue procedures” in case of mechanical or infectious complications. Furthermore, the creation of a more conspicuous distance between the blood stream and the outside as well between the cuff and the exit-site might be able to minimize, at the same time, the rate of extraluminal catheter-related blood stream infection, the incidence of tunnel infection unresponsive to the antibiotic therapy and the events of cuff extrusion. Finally, the chance of crafting a more personalized tunnel path allows to obtain the most acceptable aesthetic result according to patient’s need and wish.
(2) The cutaneous incision that is carried out at the point located halfway between the previous exit-site and the Dacron cuff of the tCVC should be as tiny as the desired new exit-site. A larger opening should be discouraged to avoid the friction of the catheter around the surrounding tissue and the delay of the sinus epithelization process.
(3) The procedure can be also performed on monotube bi-lumen tCVC with “Y junction” provided that a suitable repair kit is available, and an adequate exit site-cuff distance is present.
(4) In case the subcutaneous tCVC route runs deep, ultrasonographic examination might help to precisely locate the device and assist in the precise choice of the skin incision point.
Conclusion
The intervention of exit-site relocation represents an attractive option to treat unfixable tCVC damage or tunnel erosion without the need for catheter removal or exchange. This mini-invasive procedure showed to be rapid, safe, and optimally tolerated by the patient. However, the prerequisites for this intervention are an exit site-cuff distance of at least 5 cm and the availability of an adequate repair kit.
Footnotes
Author contributions
Research idea and study design: LN, AS; data acquisition: LN, AH; data analysis/interpretation: LN, AS; supervision or mentorship: CA, AS, GC. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
No required (retrospective case report undertaken through a case note review).
Informed consent to participate
Informed consent obtained by the patient.
Informed consent to publish
No doubt that anonymity can be maintained.
