Abstract
Purpose:
To assess 6-month safety and efficacy of a self-expanding stent graft placement to treat stenotic or thrombosed synthetic arteriovenous access graft at the venous anastomoses.
Material and methods:
This prospective, multicenter, post-market surveillance study assessed the GORE® VIABAHN® Endoprosthesis (stent graft) in Japanese patients with stenotic or thrombosed vascular access grafts. Key efficacy and safety outcomes were primary patency (PP) and secondary patency (SP) of the target lesion and the vascular access circuit, repeat interventions, technical and clinical success, incidence of adverse and serious adverse events, and device deficiencies through 6 months.
Results:
The 103 enrolled patients (mean age 71.8 years; 57.3% female) had stent grafts placed in the venous anastomosis of arteriovenous access grafts (on-label). The majority of lesions were stenotic (77.1%) and located in the upper arm (91.4%). Mean lesion length was 42.9 ± 23.6 mm and diameter was 6.7 ± 1.9 mm; 28 patients (27.2%) had elephant trunk placements. Technical and clinical success rates were 100%. At 6 months, Kaplan-Meier estimates of target lesion and circuit PP were 75.1% (95% CI: 65.2, 82.6) and 66.8% (95% CI: 56.4, 75.3), and SP rates were 93.8% (95% CI: 86.7, 97.2) and 93.8% (95% CI: 86.7, 97.2), respectively. Mean cumulative number of reinterventions (0.36; 95% CI: 0.20, 0.51) and incidence of device- or procedure-related adverse events were low (⩽3%). No device deficiencies were reported.
Conclusions:
The 6-month outcomes of the self-expanding stent graft to treat thrombosed venous anastomoses of synthetic hemodialysis vascular access grafts with stenosis demonstrated safety and effectiveness.
Keywords
Introduction
Hemodialysis access is becoming increasingly important as the prevalence of chronic kidney disease and end-stage renal disease continues to rise.1,2 Arteriovenous fistulas (AVFs) remain the most frequently used vascular access for hemodialysis. 3
Arteriovenous grafts (AVG) are one of the options for vascular access for hemodialysis, particularly for patients with end-stage kidney disease that are not suitable candidates for AVF as AVGs are associated with lower risks of vascular access-related events (e.g. infection) compared to a central venous catheter.3,4 The AVG is critical as an access site for dialysis. However, AVGs often fail to function within 18 months after implantation, primarily due to intimal hyperplasia at the site of the venous anastomosis.5,6
A common treatment of a stenotic or occluded anastomosis of the AVG is percutaneous transluminal angioplasty (PTA), but this treatment has reported poor primary patency of 16%–35.5% at 6 months.7,8 A retrospective study in a population of Japanese patients undergoing treatment for vascular access stenosis included a group of 300 cases of angioplasty for AVG and further supported poorer vascular access primary patency rates (20.4%) at 6 months. 8
In the past, several attempts have been made to prolong AVG function using antiplatelet or antithrombotic agents, as well as surveillance with early intervention. One such intervention, bare metal stents (BMS), can maintain full dilation of the vessel lumen and reduce turbulence through the graft. However, BMS do not prevent in-stent cell proliferation, which ultimately leads to in-stent restenosis. In the 1990s, properly designed studies failed to show the superiority of BMS over PTA alone.9,10 Stent grafts, on the other hand, consist of an intraluminal support, that resists muscular contraction of the vessel wall, and a biocompatible barrier to prevent cellular migration and in-stent restenosis. These features of the stent graft may provide an effective and durable treatment for neointimal hyperplastic stenosis, since it addresses the disadvantages of BMS.
The GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface (hereafter, self-expanding stent graft) became commercially available for the use in arteriovenous revision in the United States (US) in 2013, in Europe in 2014, and in Japan in 2016. This study presented here evaluated efficacy and safety outcomes for patients with stenosis or occlusion at the venous anastomosis of arteriovenous access grafts treated with the self-expanding stent graft, following commercial availability in Japan. The focus of this report is the 6-month outcomes for use of the self-expanding stent graft.
Methods
Study design
This prospective, multicenter, post-market surveillance study was conducted between July 2020 and February 2021 across 17 centers in Japan. Study sites with clinical departments that included nephrology, surgery, and/or dialysis were selected. The target patient population were patients with stenosis or occlusion (thrombosed) at the venous anastomosis of synthetic arteriovenous access graft (on-label with Instructions for Use) that were naïve to the use of the self-expanding stent graft. Reported here are the 6-month outcomes in patient cases of on-label use of the self-expanding stent graft.
This study was sponsored by W. L. Gore & Associates (Flagstaff, AZ) and is registered with ClinTrials.gov (CT Registration #NCT04429243). This surveillance study was regulated by the Japanese Ministry of Health, Labour, and Welfare; complies with the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices and Welfare; and was conducted in accordance with Japanese Good Post-Market Surveillance Practice Regulations (GPSP). Per the Japanese Ministry of Health, Labour, and Welfare and Japanese GPSP Regulations, informed consent is determined by each institution’s ethical committee policy to specify whether informed consent was necessary or outcome data could be abstracted while protecting patient’s rights without requiring individual patient consent. Informed consent was not required.
Device and procedures
All patients were treated with the GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface (W. L. Gore & Associates, Flagstaff, AZ). The self-expanding stent graft is a flexible, self-expanding endoluminal endoprosthesis consisting of an expanded polytetrafluorethylene (ePTFE) lining with an external nitinol support extending along its entire length. The devices were available in diameters of 6, 7, and 8 mm and lengths sizes of 2.5, 5.0, and 10.0 cm. In Japan, the device is indicated for placement in the venous anastomosis of synthetic arteriovenous access grafts (device Instruction for Use).
Patient demographic characteristics, medical history, lesion characteristics, and procedural information including vascular access and devices used, dialysis treatment continuation/discontinuation, failure, adverse events, reintervention, and concomitant medications were collected using an electronic data capture system. Information was collected at the recommended follow-up visits 1-, 3-, 6-, 12-, and 24-months post-procedure.
In general, outflow tract vein stenosis was suspected when there was a decrease in graft sound, a decrease in thrill, a decrease in dialysis efficiency, a marked increase in venous pressure during dialysis, venous hypertension possibly due to the graft, prolonged hemostasis time, and a decrease in brachial artery blood flow (generally below 600 mL/min) on ultrasound examination, angiography was performed in these cases. Intervention was indicated in those cases in which more than 50% stenosis was found in the outflow tract vein. Stenotic or occluded lesions were treated with PTA balloon prior to the placement of self-expanding stent grafts. In addition to PTA, patients with occluded lesions may have been treated with either surgical and/or percutaneous thrombectomy to declot occlusions.
Pre-dilation with a balloon catheter was performed in all cases. The balloon for pre-dilation was adjusted to the standard venous diameter, but there were slight differences from facility to facility. The dilation was performed with a balloon size equivalent to the size of the self-expanding stent graft used.
Outcomes and definitions
Efficacy endpoints included primary and secondary patency of the target lesion and of the vascular access circuit. Primary patency of the target lesion was defined as the period during which the patency was maintained from initial treatment until either total occlusion of target lesion or re-treatment due to occlusion or stenosis of the target lesion. For primary patency of the target lesion evaluation, the occlusion or stenosis needed to occur within the stent or within 5 mm from the periphery of the stent graft. Primary patency of the vascular access circuit was defined as the period during which the patency was maintained from initial treatment until occlusion in vascular access or re-treatment of the vascular access circuit. Secondary patency of the target lesion was defined as the period during which the patency was maintained from index procedure including the period of patency after re-treatment. Secondary patency of the vascular access circuit was defined as the period during which the patency was maintained from index procedure until discontinuation of the use (abandonment) of the vascular access circuit. Primary and secondary patency of the target lesions and of the vascular access circuit were estimated by Kaplan-Meier (KM) method.
Additional efficacy endpoints included repeat interventions in the target lesion (number performed after initial treatment) and technical and clinical success. Technical success was defined as <30% residual stenosis after initial treatment. Clinical success was defined as the resumption of normal dialysis for at least one session after the initial treatment.
Subgroup analyses were performed to estimate primary and secondary patency on variables of lesion characteristics (i.e. stenosis vs occlusion), stent graft size, stent graft crossing the elbow, placement and location of stent, use of “elephant trunk” technique (defined here as stent grafts that are under sized to the outflow vein and lie in the outflow vein without vessel wall apposition), sex, age of circuit, previous interventions, and select medical history characteristics.
The safety endpoints included the incidence of device- and procedure-related adverse events at treatment and up to 30 days after treatment. Additionally, the incidence of adverse events, serious adverse events, and device defect in each follow-up period was evaluated.
Statistical method
Continuous parameters are presented as mean ± standard deviation (SD) or 95% confidence interval (95% CI). Categorical variables are presented as a number (percentage). Kaplan-Meier estimates were analyzed by log-rank test. No standard errors for KM estimates of primary patency and secondary patency exceeded 10.0%. p-Values of <0.05 were considered statistically significant. All computations were performed using SAS version 9.3 and JMP version 16.1 (SAS Institute, Inc, Cary, NC).
Results
Patient population
A total of 103 patients had self-expanding stent grafts placed in the venous anastomosis of synthetic arteriovenous access grafts. The baseline patient demographics, medical characteristics, and lesion and procedural details for cases at 6-months are summarized in Table 1. The patients were a mean ± SD age of 71.8 ± 12.0 years, had a mean ± SD body mass index of 23.2 ± 5.1 kg/m2, and the 57.3% were female. Comorbidities in this population included diabetic nephropathy (39.8%), chronic glomerulonephritis (16.5%), and nephrosclerosis (12.6%). Clinical indications for the procedure were primarily for reasons of rising venous pressure (51.5%), low blood flow (30.1%), graft occlusion with thrombus (21.4%), and decreased thrill (16.5%). A total of 105 lesions in 103 patients were treated. Two patients had two lesions originating from the venous anastomosis and within 30 mm of the venous anastomosis. In both cases, the two lesions were covered with one 10 cm long device. Lesion location comprised of 91.4% that were in the upper arm and 66.7% that were located in the basilic vein; 22.9% (n = 24) of the lesions were occluded. Of the 24 patients with occluded lesions, 12 were treated to declot with either surgical and/or percutaneous thrombectomy in addition to PTA treatment. The mean ± SD lesion length was 42.9 ± 23.6 mm and the mean ± SD diameter of the outflow vein landing zone on the venous side was 6.7 ± 1.9 mm. The mean ± SD blood flow of the vascular access site pre-procedure was 361.5 ± 196.8 mL/min and post-procedure was 884.5 ± 271.8 mL/min. The most frequent self-expanding stent graft placements (n = 103) were 6 mm × 10 cm (42.7%) or 6 mm × 5 cm (26.2%) in diameter and length. Use of the elephant trunk technique occurred in 27.2% of the placements. Pre-dilation with a balloon catheter was performed in all 105 lesions treated of which 82 (78.1%) achieved full pre-dilation, 22 (20.9%) did not achieve full pre-dilation, and 1 (1.0%) was unknown.
Patient demographics, medical characteristics, and lesion and procedural details.

Max: maximum; min: minimum; SD: standard deviation.
Patient population is derived of only cases of venous anastomosis of synthetic arteriovenous access grafts the using self-expanding stent graft.
In two cases, one device covered two lesions.
Data were available for 45 (43.7%) patients through the 1-month follow-up window, 91 (88.4%) through 3-month follow-up, and 72 (69.9%) through 6-month follow-up. One patient discontinued from the study at the 6-month interval due to death from gastric cancer.
Technical success, defined as a residual stenosis rate <30% after initial treatment, was achieved in 89 of the 89 patients (100%) with a technical success evaluation; residual stenosis data was not available in 14 cases. Clinical success, defined as resumption of ⩾1 normal dialysis after initial treatment with the self-expanding stent grafts, was achieved in all patients with 30-day data available (n = 55).
Primary and secondary patency
The primary end points of KM-estimated primary and secondary patency of the target lesion and the vascular access circuit are shown in Figures 1 and 2, respectively. At 30 days, the target lesion primary patency was 91.9% (95% CI: 84.4, 95.9) and secondary patency was 98.0% (95% CI: 92.1, 99.5). At 6 months, target lesion primary patency was 75.1% (95% CI: 65.2, 82.6) and secondary patency was 93.8% (95% CI: 86.7, 97.2).
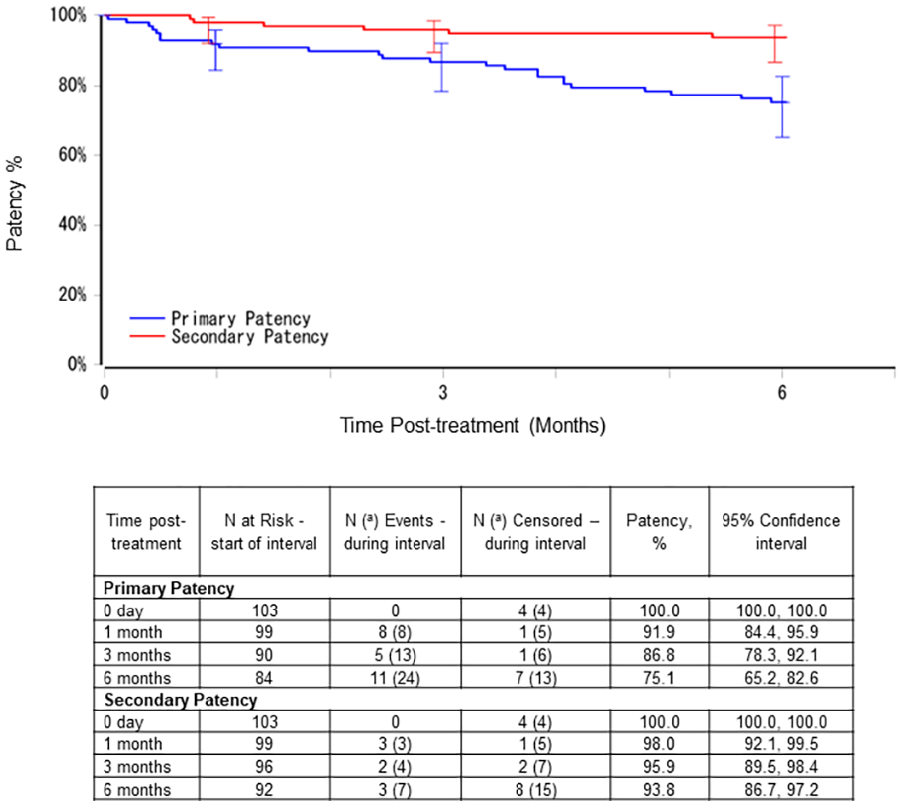
Kaplan Meier estimates of target lesion primary and secondary patency.
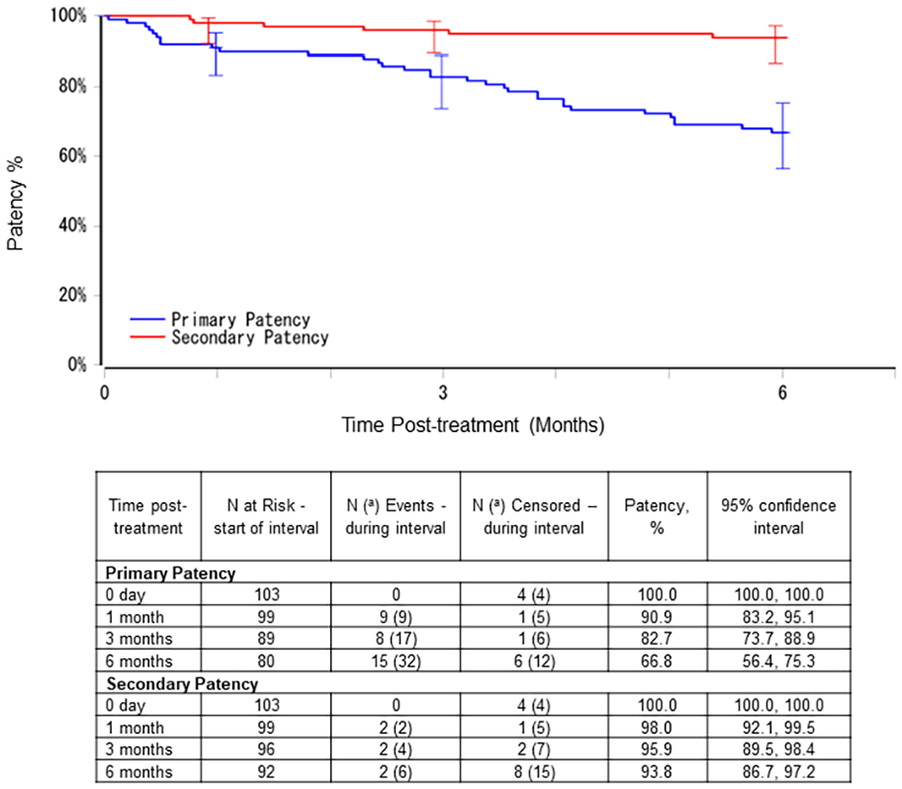
Kaplan Meier estimates of vascular access circuit primary and secondary patency.
The vascular access circuit primary patency was 90.9% (95% CI: 83.2, 95.1) and secondary patency was 98.0% (95% CI: 92.1, 99.5) at 30 days. At 6 months, vascular access circuit primary patency was 66.8% (95% CI: 56.4, 75.3) and secondary patency was 93.8% (95% CI: 86.7, 97.2).
Additional efficacy outcomes
In the 103 patients with data (representing 105 lesions), the mean cumulative number of reinterventions of the target lesion per patient was 0.36 (95% CI: 0.03, 0.17) at 30 days and 0.10 (95% CI: 0.20, 0.51) at 6 months.
The subgroup analyses for select variables assessed for target lesion and vascular access circuit are shown in Table 2. The variable comparisons for lesion characteristics (stenosis vs occlusion), stent graft size, stent graft crossing the elbow, placement and location of stent, use of “elephant trunk” technique, sex, previous interventions, and select medical history characteristics were not statistically significant. Patency estimates for the vascular access circuit variables of number of past interventions and age of circuit were evaluated and were not statistically significant.
Primary and secondary patency outcomes of the target lesion and the vascular access circuit at 6 months by subgroups of VIABAHN implantation.
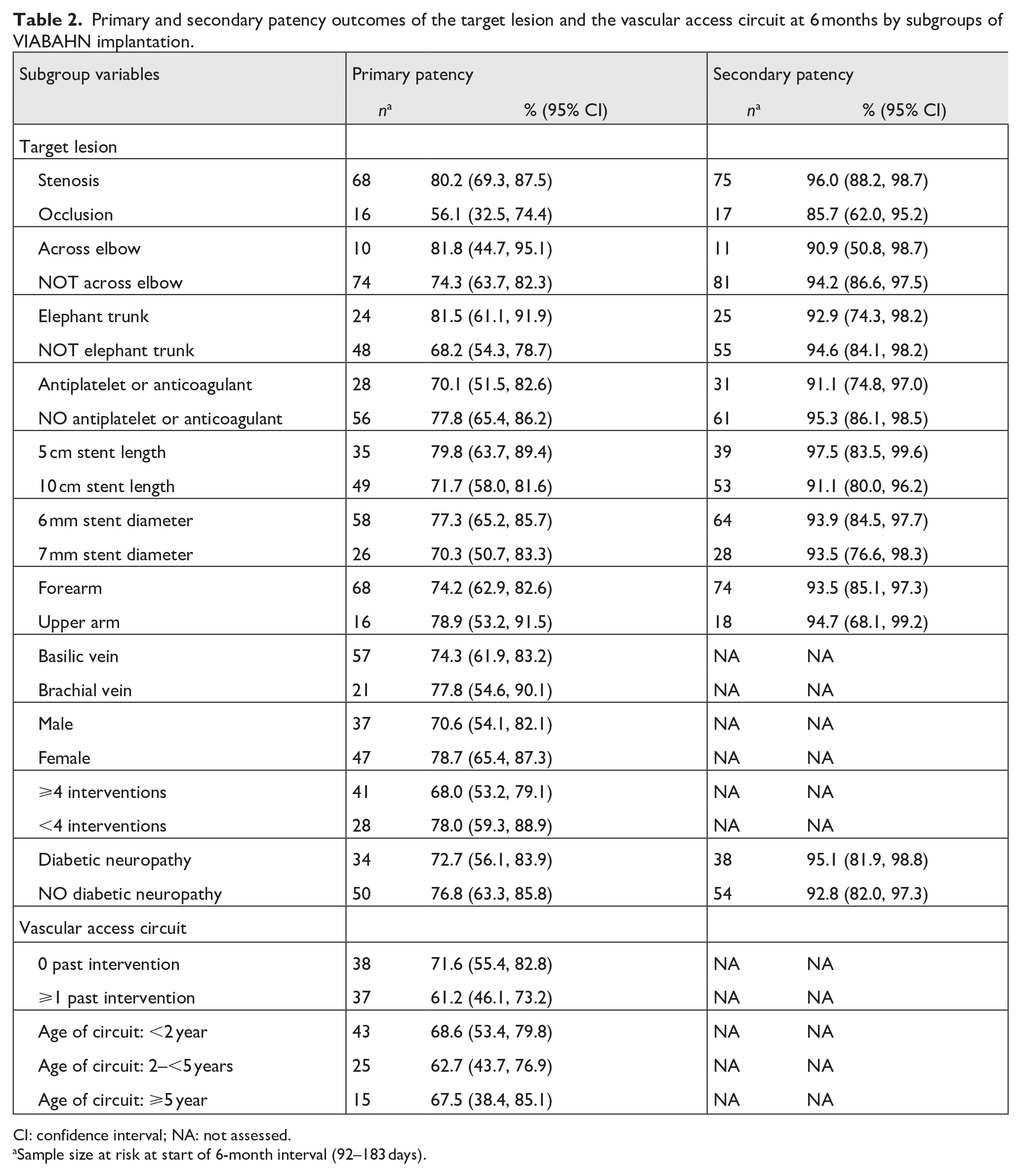
CI: confidence interval; NA: not assessed.
Sample size at risk at start of 6-month interval (92–183 days).
Safety outcomes
Two (1.9%) procedure-related adverse events of vascular injury and venous stenosis occurred on the day of the procedure. The vascular injury occurred during pre-dilation prior to device implant and the stenosis occurred in the brachial ulnar vein approximately 1 cm downstream after insertion of the device. Through 30 days, there were three (3.0%) events of vascular stent occlusion (occlusion of the device). There were no reported serious adverse events at time of procedure, through 30 days, or through 6 months. There were no reports of device defects through 6 months.
Discussion
This prospective, post-marketing surveillance study of Japanese patients with stenosis or occlusion at the venous anastomosis of arteriovenous access graft demonstrated 100% effectiveness of technical and clinical success in patients treated with the self-expanding stent graft. At 6 months, acceptable primary patency for the target lesion (75.1%) and vascular access circuits (66.8%) were achieved and secondary patency for both was 93.8% through 6 months. Safety was demonstrated by the low number of reinterventions and a low incidence of adverse events. The self-expanding stent graft proved durable through 6 months as no device deficiencies were reported.
Comparison of stent grafts and PTA
In one of the first trials to demonstrate better outcomes with stent grafts, outcomes from 190 patients in the US assigned to either balloon angioplasty alone or in addition with a stent graft showed 6-month primary patency of both the target lesion and vascular access circuit to be significantly greater in the stent graft group (51% and 38%, respectively) compared to that observed in the PTA group (23% and 20%, respectively). 7 Performance of the self-expanding stent graft evaluated in this report has been assessed in meta-analysis and strategic literature reviews and include data from studies that compare the self-expanding stent graft and PTA. In a prospective multicenter trial (REVISE) of 293 patients, the 6-month primary patency of the self-expanding stent graft was superior to balloon angioplasty in the target lesion (52.9% vs 35.5%) and the vascular access circuit (43.4% vs 29.4%). 11 An additional prospective study of the self-expanding stent graft (covered) in 110 patients also demonstrated respectable 6-month primary patency of the target lesion (70.3%) and the vascular access circuit (39.2%). 12
Haskal et al. reported on a multicenter, prospective, controlled trial of 270 patients with ⩾50% AVG anastomotic stenosis, with patency results for 191 patients who completed the study and treated with the FLAIR® Stent Graft (FSG; n = 97) or PTA (n = 94). 13 Through 24 months, the treatment area primary patency was 26.9% for FSG versus 13.5% for PTA (p < 0.001), access circuit primary patency was 9.5% for FSG versus 5.5% for PTA (p = 0.01), and index patency function was 7.1 ± 7.0 months/intervention for FSG versus 5.3 ± 5.2 months intervention for PTA. The estimated number of reinterventions before graft abandonment was 3.4 in FSG patients versus 4.3 in PTA patients. Even at 2 years, the use of FSG demonstrated a more than 2-fold sustained advantage over PTA in terms of treatment area and overall access patency. The authors concluded that the time to next intervention was longer in the FSG group.
The 6-month primary patency rates for the target lesion and the access circuit from these previous reports,7,11,12 in addition to this current study, are summarized in Table 3. The 6-month target lesion primary patency rate for stent grafts from prospective studies generally ranges between 50% and 60%. The target lesion primary patency in this current study was 75.1% and may be reflective of potentially contributing factors. In the analysis population, 23% of patients had occluded AVGs at baseline. At 6 months, the occluded AVGs had lower primary patency in the target lesion than AVGs that were stenosed but not occluded. Previously occluded grafts seem more likely to occlude again.11,12 This likelihood may be attributed to the notion that treating occluded grafts is more technically challenging and, as a result, has the potential for increased vascular damage. Stenosed AVG are observed to have better outcomes, likely owing to the simpler interventional procedures, and in turn, lower risk of complications. For example, the REVISE study 11 population included 41% of thrombosed grafts whereas this study includes 23% and this difference may have contributed to better patency outcomes. Another potentially contributing factor is that the diameter of the prosthetic graft sizes used in this study were either 5 mm (53%) or 6 mm (39%), which is a relatively large number of patients having small-diameter prosthetic grafts. This may have resulted in lower access blood flow and reduced vascular injury. In fact, the mean vascular access site blood flow rate after stent graft placement increased from 361.5 mL/min pre-procedure to 884.5 mL/min post-procedure, which is considered high blood flow. 14 The authors opine that another contributing factor may be that the number of elephant trunk cases (n = 28) representing 27.2% of cases in this study. The elephant trunk technique involves using a stent graft that lies within the outflow vein without full apposition to the vessel wall. This approach is used to bypass stenosed or thrombosed segments, maintain blood flow dynamics, and reduce the risk of vessel injury. The diameter of the outflow tract vein was large relative to the diameter of the stent graft used. Patency outcomes of the cases with the elephant trunk procedure are likely attributed to the distal end of the stent graft does not contact a vein, so pannus in the vein in contact does not spill over to the stent graft. Additionally, because the jet flow from the graft does not directly contact the outflow tract vein wall, the shear stress on the outflow tract vein wall is reduced, which may inhibit the formation of intimal thickening in the outflow area. Another study 15 supports that under sizing in this manner observed a statistically significant increase in target lesion primary patency compared to stent that were well apposed. Lastly, in this study, patients were in the lower end of the body mass index range (average 23.2 kg/m2) and this could have been a factor for patency outcome, though this is speculation and would require additional studies to clarify this and other postulated contributing factors to the higher primary patency rate observed in this study.
Summary of 6-month primary patency of stent grafts and percutaneous transluminal angioplasty.

PTA: percutaneous transluminal angioplasty; VSX: GORE® VIABAHN® Endoprosthesis.
Percentages as reported by the reference.
Comparison of stent graft and BMS
There are few comparative studies between BMS and stent grafts at the venous anastomosis of AVG. Kim et al. compared the results of BMS and stent grafts at the venous anastomosis of AVG. 16 Both the primary access and secondary patency rates of the BMS and the covered stent at 3, 6, and 12 months were not significantly different. However, at 3, 6, and 12 months, the covered stent showed higher primary stent patency rates of 100%, 85%, and 70%, respectively than rates for BMS of 75%, 67%, and 49%, respectively (p < 0.01). Further studies are needed to determine if stent grafts have any significance over BMS at the venous anastomosis of the AVG.
Meta-analyses
Several studies and meta-analyses have established the lower losses in 6-month primary patency with stent grafts for AVG venous anastomoses over PTA.17-20 The most recent of these analyses, that involved 1147 patients of which 867 patients were AVGs, showed stent graft implantation had significantly lower loss of primary patency at 6 months for AVG (odds ratio (OR) 4.48, 95% confidence intervals (CI) 1.98, 10.14; p < 0.001) and access circuits compared to PTA (OR 1.91, 95% CI: 1.31, 2.80; p < 0.001). 20 Outcomes from this 6-month analyses adds to the body of evidence supporting the use of stent grafts over PTA.
Limitations
This study was limited by the single-arm design and lack of direct comparison to different approaches. This surveillance study provides real-world data but inherently introduces variability. One such variability is that while pre-dilation with a balloon catheter was performed in all lesions treated, data on the balloon sizes used or maximum balloon dilation obtained were not collected. Further, the lack of this data along with variability from facility to facility, and adjustments to the standard venous diameter precluded factoring this into the sub analyses. Another limitation was that the incomplete or lack of follow-up visits, particularly after 30 days, subsequently limited the analyses sample size. The analysis in this study was confined to on-label use of the device. Therefore, patients designated as off-label use were excluded, which is not reflective the clinical setting and, as a result, lowered the overall number of patients in the analysis.
Conclusions
The 6-month interim outcomes from the study of the self-expanding stent graft placement to treat stenosis at the vein-to-graft of stenotic or thrombosed arteriovenous grafts demonstrates safety and effectiveness. These results were comparable to other similar studies and superior to prior reports using angioplasty alone. Pending longer-term data will provide evidence of durability of the device through 2 years.
Footnotes
Acknowledgements
Medical writing support was provided by Millie Hollandbeck (Phoenix, Arizona) and was funded by W. L. Gore & Associates, Inc. (Flagstaff, AZ). The authors thank Chrystal Redding and Yasunori Arida (W. L. Gore & Associates, Inc.) for their technical review that was limited to assurance of data accuracy.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H. Haruguchi, M. Fukasawa, K. Ikeda, K. Suemitsu, T. Sato, J. Nakamura, T. Noguchi, D. Eguchi, T. Kamimura, and K. Aikawa have no industry relationships to disclose. M. Miyamoto declares: Honorarium from W. L. Gore & Associates, G.K., Medtronic Japan, Boston Scientific Japan, KANAKA Medical Products, Medicon Inc., and Filips Japan. M. Murakami declares: Honoraria for presentations from Boston Scientific, Kaneka Medix, W. L. Gore & Associates, and Medtronic.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: W. L. Gore & Associates, Inc., sponsored this study and the medical writing support that was provided by Millie Hollandbeck (Phoenix, Arizona).
