Abstract
Background:
Epicutaneo-caval catheters (ECC) are vital in neonatal intensive care units (NICU) in enabling prolonged venous access. Despite their benefits, central line-associated bloodstream infections (CLABSI) pose a potential risk. The ECC removal procedure may contribute to an increased risk of post removal sepsis through biofilm release. Antimicrobial-impregnated ECCs have been proposed as a potential solution to prevent this complication, but research on their effectiveness in neonates is limited.
Objective:
To compare post-ECC or neonatal peripherally inserted central catheter (n-PICCs) removal sepsis rates when using conventional or antimicrobial-impregnated catheters in neonates.
Methods:
A retrospective, single center cohort analysis using 2019 data from 421 neonates after successful ECC insertion at the Women’s Wellness and Research Center, a large tertiary level NICU in Qatar. The study systematically collected data on demographics, insertion, and removal as well as microbiology, and infection data. Statistical analyses compared conventional and antimicrobial ECCs, with a focus on the incidence of sepsis within 72 hr post-removal.
Results:
After excluding non-eligible, 349 cases were included in the analysis (138 conventional, 211 antimicrobial-impregnated). There were no significant demographic differences between the two groups. The CLABSI incidence was higher amongst the antimicrobial ECC group (4% vs 0.6%,
Conclusion:
The study noted a higher, but statistically insignificant, incidence of post removal sepsis when using antimicrobial-impregnated ECCs. This finding raises questions about the effectiveness of antimicrobial ECCs in preventing post-removal sepsis in this patient population. Further randomized trials are needed to assess the role of antimicrobial ECCs amongst neonates and to refine neonatal ECC care strategies.
Keywords
Introduction
Epicutaneo-caval catheters (ECCs) or neonatal peripherally inserted central catheter (n-PICCs) are frequently used in neonatal intensive care units (NICUs) to establish prolonged venous access for the administration of total parenteral nutrition (TPN) and medications, a common practice for neonates, particularly those with very low birth weight (VLBW). The use of ECCs presents notable benefits, including the avoidance of recurrent, uncomfortable, and painful venous cannulations, streamlined delivery of concentrated essential nutrition to support optimal growth, and mitigation of constraints associated with peripheral access.1,2
Notwithstanding the infrequent incidence of complications relative to other vascular access devices, central line-associated bloodstream infections (CLABSIs) is a substantial contributor to morbidity and mortality in the neonatal period.3,4 According to findings from the PREVAIL randomized controlled trial, bloodstream infections (BSIs) that occur during the presence of ECCs constitute less than half of all BSIs in NICU and high-dependency care settings. 5 The association between ECC removal, the potential role of biofilm stripping and the occurrence of bloodstream infections is noteworthy. 6
Biofilms attached to the interior and exterior surfaces of an ECC harbor microorganisms that might be dislodged or stripped away during the removal process.7,8 The process of removal has the potential to release a bolus of microorganisms in the circulation, increasing the risk of bacteremia and subsequent BSI.8,9 Biofilms, characterized as stable microbial communities enveloped in extracellular polymeric substances, form on catheter surfaces within 24 hr of insertion. 9 The extent and location of biofilm formation is influenced by the duration of catheterization. Short-term catheters (<10 days) exhibit greater biofilm formation on their external surface (extraluminal), while long-term catheters (⩾30 days) have increased biofilm formation within the catheter inner lumen (intraluminal).9,10
In response to the concern of BSIs, antimicrobial-impregnated central venous catheters have been recommended by manufacturers for use in adults and children.11,12 The most recent 2023 recommendations from the Society for Healthcare Epidemiology of America (SHEA) concerning antimicrobial-impregnated central venous catheters recommends that they should be used under specific conditions. 13 These conditions include hospital units or patient populations with a CLABSI rate above institutional goals despite compliance with essential CLABSI prevention practices, patients with limited venous access and a history of recurrent CLABSI, and patients at heightened risk of severe sequelae from a CLABSI, such as those with recently implanted intravascular devices like a prosthetic heart valve or aortic graft. This updated guidance underscores the importance of targeted utilization of antimicrobial catheters based on clinical indications.
Previous clinical incidents and studies have reported varying incidences of sepsis following ECC removal. Incidences ranging from 2.1% to 13% have been documented in different populations.7,8,14–17 Given the lack of data regarding BSIs related to ECC removal in the NICU, various practice measures have been implemented to mitigate the risk of BSIs following ECC removal in neonates. These measures include the use of prophylactic antimicrobial agents,6,14 implementation of aseptic techniques during catheter removal, and adherence to bundled infection prevention strategies. However, there is limited data about the use of antimicrobial-impregnated ECCs in neonates, as well as on complications following their removal. This study aims to evaluate the incidence of sepsis following ECC removal in a NICU and to compare outcomes between conventional and antimicrobial-impregnated ECCs to clarify sepsis risks associated with ECC removal.
Method
Design and setting
In this retrospective, single-center cohort investigation, de-identified data collected during routine clinical practice from 1st January 2019 till 31st December 2019 was analyzed. The primary aim was to assess the occurrence of sepsis subsequent to the removal of ECCs and compare outcomes between conventional central venous catheters and antimicrobial-impregnated central venous catheters.
The study was conducted in the tertiary level NICU of the Women’s Wellness and Research Center (WWRC), Hamad Medical Corporation, Doha, Qatar. The WWRC accommodates a tertiary NICU with 112 beds, offering comprehensive care for newborns starting from 24 weeks gestational age. The NICU, managing an annual caseload exceeding 4000 newborns, operates with a dedicated neonatal vascular access team (NeoVAT) comprising 30 specialized nursing, and 12 medical staff, possessing the requisite expertise to perform and assist in ECC insertions.
Participants and sample size
The study encompassed all term and preterm neonates admitted to the NICU requiring intravenous therapy through successful insertion of an ECC. The patient study enrollment and reasons for exclusion were summarized in Figure 1.
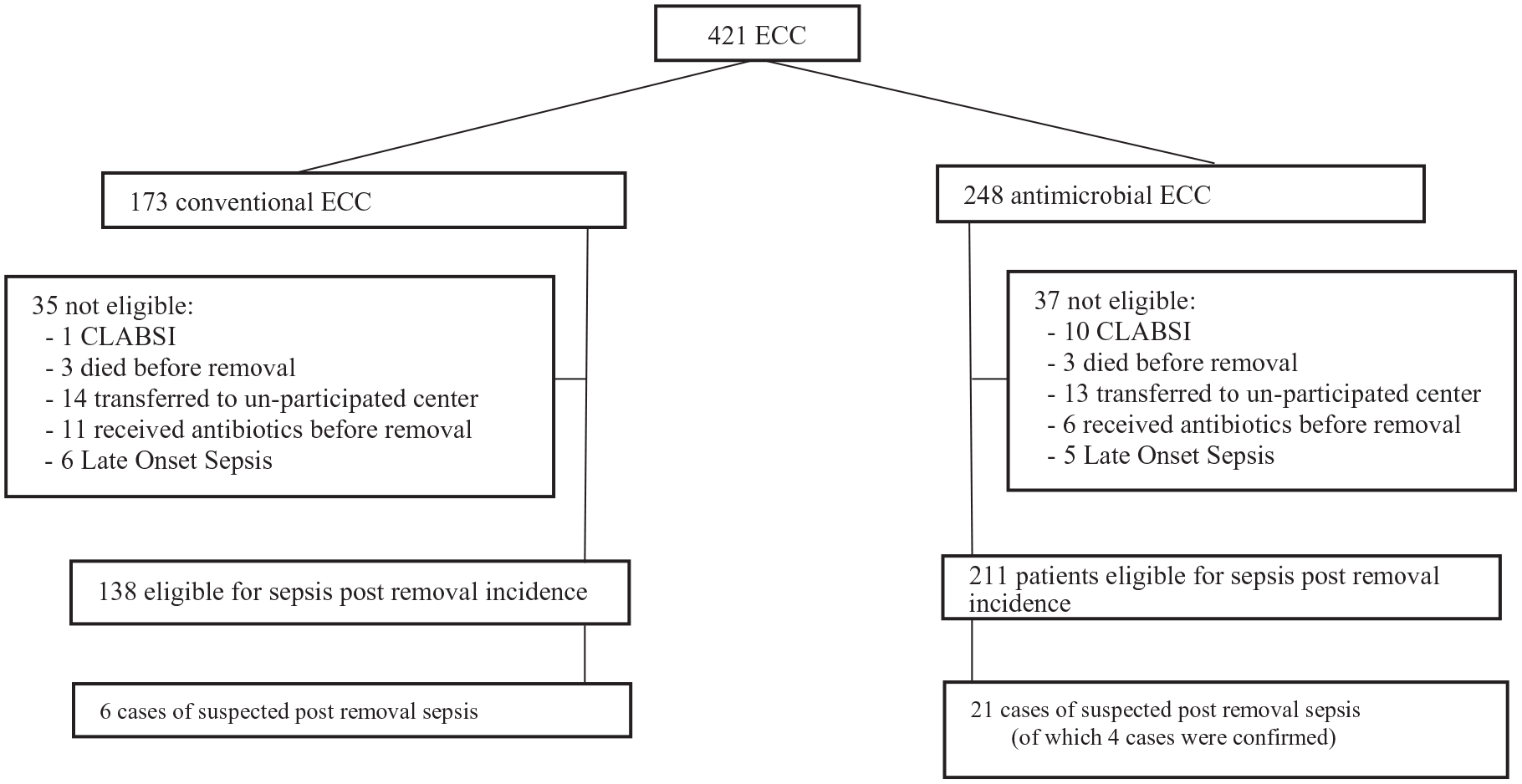
Study flow chart.
Procedure
ECC cannulation in our unit is strictly performed following local hospital policy and current international standards of practice.2,18 In the study setting, ECC insertions were routinely conducted by the NICU’s NeoVAT. 19 We use both the conventional ECC, PremiCath® 1Fr, and the antimicrobial-impregnated ECC, PremiStar® 1Fr, both manufactured by Vygon and made of radiopaque polyurethane. The antimicrobial-impregnated catheter contains Rifampicin and Miconazole, which provide local antimicrobial action on the catheter surface.
Our catheter maintenance and care bundles to prevent CLABSI include a fully sterile insertion bundle, a daily vascular access device assessment bundle, a line dressing change bundle, and a catheter access bundle. Key elements of these bundles include the use of maximal personal protective equipment, adherence to proper hand hygiene techniques, optimal selection of catheter site and device, daily assessment of central line necessity, and the use of a specialized central line trolley (Mayo Stand). Additional procedures involve a pre-procedure timeout, timing the procedure, using a sterile central line insertion kit, cleaning the catheter hub with chlorhexidine, applying a transparent semipermeable dressing, employing a needleless connector, using an alcohol-impregnated port protector, and consistently using a two-person technique. Further measures include the application of cyanoacrylate glue for sealing the insertion site, catheter fixation, and stabilization, as well as for bleeding and infection prevention. 20 We also use diluted lipid emulsion as a lubricant for guidewire removal, 21 the vein viewer imaging system for vascular assessment, and the microsite and Modified Seldinger Technique (MST) for catheter insertion. 22 Our approach emphasizes early ECC insertion and removal, the use of antimicrobial-impregnated catheters, speak-up campaigns to address infection control breaches, and maintaining a closed IV tubing system at all times. 23
Additionally, we have developed the mnemonic “5 Rights for Vascular Access” for neonatal vascular access pre-briefing: choosing the Right vascular access device for the Right patient, administering the Right therapy in the Right vein for the Right duration. Catheter removal is performed by the same team, following the established hospital protocols.
Measurements and data collection
The analysis of patient data includes a comprehensive set of demographic and baseline information, covering factors such as sex, gestational age at birth (expressed in weeks and days), mode of delivery, presence of chorioamnionitis, and birth weight in grams. Specific data pertinent to the ECC insertion procedure encompasses details such as the date and time of insertion, date and time of removal, the number of successful insertion attempts, reasons for catheter removal, catheter type (conventional or antimicrobial), duration of ECC indwelling, date of CLABSI diagnosis, results of blood cultures, administration of antibiotics before removal, presence of sepsis signs and symptoms within 72 hr of ECC removal, and information regarding patient transfer or mortality.
All instances of CLABSI were diagnosed in accordance with infection control committee guidelines. The diagnostic procedures for CLABSI adhered to the guidelines delineated in the National Healthcare Safety Network (NHSN) Patient Safety Component Manual, as published by the Centers for Disease Control and Prevention (CDC) in the USA. 24 In the context of this study, sepsis post ECC removal was specifically defined as the presence of signs and symptoms of sepsis occurring within 72 hr after ECC removal. This definition also involved the requisites of collecting blood cultures and initiating antibiotic therapy.
Statistical analyses
The unit of analysis in this study was each ECC insertion. Data distribution was summarized using numbers and percentages, mean, and standard deviation as applicable. Differences in prenatal and newborn characteristics, as well as outcomes between conventional ECC and antimicrobial ECC, were assessed using Chi-square, Fisher’s exact, and
Patient and public involvement were not part of the conception, design, or development of the study protocol. Additionally, neither patients nor the public were involved in data acquisition, analyses, interpretation, or the development of this manuscript. Outcome measurements were based on recent literature and were determined through a collaborative effort among the researchers.
Results
This study included a total of 421 successful ECC insertions in neonates, comprising 173 conventional ECCs (41.1%) and 248 antimicrobial ECCs (58.9%). No significant differences in demographic characteristics were observed between the two groups. The mean gestational age was 29.9 weeks in the conventional ECC group and 29.7 weeks in the antimicrobial ECC group (
Regarding clinical outcomes, late-onset sepsis unrelated to the central catheter occurred in six patients (3.5%) in the conventional group and five patients (2%) in the antimicrobial group (
The duration of ECC indwelling was less than 7 days in 43 patients (24.9%) in the conventional group and between 7 and 14 days in 98 patients (56.6%). In the antimicrobial group, the duration was 53 patients (21.4%) less than 7 days, 144 patients (58.1%) between 7 and 14 days, and 51 patients (20.6%) beyond 14 days (
Demographic and clinical characteristics.

CLABSI: central line-associated bloodstream infections; ECC: epicutaneo-caval catheters.
Specific complications associated with the removal of conventional and antimicrobial catheters are represented in Table 2. The percentages indicate the proportion of each complication category relative to the total number of ECC removals for each catheter type. Notably, the most common reason for ECC removal in both types was elective removal due to it no longer being required. While Table 1 refers to all cases, Table 2 presents data on the ECCs that were removed. The additional three ECCs (one conventional and two antimicrobial) that constitute a discrepancy were initially left in place after clinical risk assessment determined that the potential lack of vascular access due to the unavailability of alternative vascular access outweighed the risks worsening infection due to no removal of the catheter.
Complications and reasons for ECC removal.

CLABSI: central line-associated bloodstream infections; ECC: epicutaneo-caval catheters.
In the eight cases above, the ECC was removed due to CLABSI. The discrepancy arises from three ECCs (one conventional and two antimicrobial) out of the 11 CLABSI cases in Table 1 that were initially left in place after a clinical risk assessment.
A total of 349 patients (138 conventional and 211 antimicrobial impregnated ECC) met the eligibility criteria for inclusion in calculating the incidence of sepsis post ECC removal. Patient enrollment and reasons for exclusion are delineated in Figure 1.
Table 3 outlines the demographic details of the included patients. No significant differences in demographic characteristics were identified between the two groups. The mean gestational age was 29.9 ± 3.5 weeks in the conventional ECC group and 29.7 ± 3.6 weeks in the antimicrobial ECC group (
Patient characteristics and outcomes post ECC removal.

SD: standard deviation; ECC: epicutaneo-caval catheters.
Regarding the incidence of sepsis post removal, the conventional group exhibited an incidence of 4.3% (6/138), while the antimicrobial group had an incidence of 10% (21/211), yielding a
In the conventional group, all instances of post-epicutaneo-caval catheters (ECC) removal were categorized as “suspected sepsis,” and no positive cultures were obtained. Nevertheless, all patients in this group experienced successful recovery. In contrast, within the antimicrobial group, four out of 21 cases had positive blood cultures, confirming the presence of infection. Additionally, 17 cases in the antimicrobial group were classified as “suspected sepsis” despite negative blood cultures. Unfortunately, among these 17 cases, one patient died. Table 4 provides a detailed overview of the microbiological aspects, including the number of cases, identified organisms, and outcomes associated with ECC-related infections.
Summary of catheter-associated infections and outcomes by catheter type.

Discussion
ECCs are commonly utilized in the NICU for long-term intravenous drug administration and TPN, especially in VLBW infants. However, ECCs can also serve as a potential vector of BSIs or CLABSIs. 25 Several confounding variables are known including, contamination of the catheter hub or the catheter itself can result in bacterial colonization and subsequent infection. 26 ECC can develop micro-punctures from repeated insertion and removal or from cannulation of the catheter itself, these micro-punctures can be used as entry sites for bacteria. 27 A microfilm can be formed on the catheter, which can lead to CLABSIs when the microfilm ruptures and causes leakage of the bacteria into the blood stream.27–29 This biofilm may become dislodged upon its removal, releasing bacteria into the bloodstream. To mitigate the risk of BSI following catheter removal, several studies have investigated the use of prophylactic antibiotic doses. However, the findings from these studies have been inconclusive, with some demonstrating a reduction in BSI incidence, while others finding no significant differences.6–8,16,30,31
Extensive research has been conducted to better understand biofilm formation and its clinical implications.27,28,31,32 Biofilm formation poses a challenge as it makes infections resistant to antibiotic treatment. In response, various techniques and strategies known as “biofilm prevention strategies” have been developed to target different stages of biofilm maturation.28,32 One such strategy involves the use of antimicrobial-impregnated catheters, which have shown promise in significantly reducing the incidence of CLABSIs in neonatal and pediatric patients. 33 While it is important to note that antimicrobial-impregnated catheters and central line protection devices may not prevent all CLABSIs, they have demonstrated a reduction in incidence when compared to standard catheters in some studies.5,34–36 Additionally, other studies have indicated a significant decrease in ECC colonization, signs of sepsis, or catheter-related BSIs with the use of antimicrobial-impregnated central venous catheters.37–40
Recent studies, however, have challenged the previously reported advantages of antimicrobial-impregnated catheters. The study conducted by Klemme et al. 41 failed to find any statistical significance regarding the efficacy of such catheters. Similarly, the PREVAIL trial found no evidence of benefit or harm associated with rifampicin- and miconazole-impregnated ECCs in neonatal care. 5 Additionally, in a meta-analysis conducted by Lai and Yue, 42 antimicrobial-impregnated catheters were not found to be effective in reducing the incidence of BSIs in children and neonates compared to conventional catheters. 42 This meta-analysis included a limited number of heterogeneous studies, suggesting the need for further research to determine the effectiveness of antimicrobial-impregnated catheters in reducing the incidence of BSIs in neonates and children.
Lai and Yue, 42 considered the use of various bundle interventions in the trials, which might have contributed to a reduction in baseline infection rates during the study period. Additionally, the duration of neonatal catheterization was relatively short, and improved catheter aseptic practices were implemented in this high-risk cohort, which could have influenced the outcomes in both groups. 42
Furthermore, two recently published studies conducted in our NICU focused on neonatal catheter care. The first study emphasized the significant benefits of an octyl-based tissue adhesive for catheter securement, reducing complications like phlebitis, suspected sepsis, and premature removal of ECCs. These findings align with global literature, affirming octyl-based tissue adhesive safety and efficacy in neonatal catheter care. 20 In contrast, the second NICU study investigated over 2000 infants, evaluating the impact of rifampicin-miconazole-impregnated ECCs on CLABSI rates. Surprisingly, the study found no significant difference in CLABSI rates compared to standard ECCs over a 4-year period. 43 This study suggested that the use of rifampicin-miconazole-impregnated catheters did not result in a reduction in the incidence of CLABSIs when compared to standard catheters.
In the NICU setting, the incidence of sepsis following the removal of ECCs is relatively low due to the implementation of protocols aimed at preventing skin infection around the ECC line. However, there is limited research available on the effectiveness of rifampicin-miconazole impregnated central venous catheters or ECCs in reducing the occurrence of sepsis after catheter removal in neonates. Existing studies on antimicrobial-impregnated central venous catheters or ECCs have primarily focused on the 48–72 h immediately following catheter removal, and data beyond this time period is lacking.
The findings of our study indicate that there is no significant difference in the incidence of sepsis post-catheter removal between antimicrobial impregnated ECCs and conventional ECCs in a predominantly premature infant population. These results align with the existing evidence that miconazole and rifampicin may not consistently prevent BSIs in neonates.
The discrepancy between our findings and those of other studies on central venous catheter impregnation with minocycline and rifampicin in children and adults,12,39,44 can be attributed to the potentially weaker effect of miconazole and rifampicin impregnation compared to minocycline and rifampicin impregnation. Also, because it primarily targets invasive fungal infections, which are less common in the neonatal population, miconazole may not play a significant role in this impregnation, especially given the current practice of using fluconazole prophylaxis in VLBW infants in our NICU during the study period. Furthermore, rifampicin may be less effective as an antibacterial agent when used alone or in combination with miconazole.
There could be several other factors contributing to the findings of ineffectiveness of rifampicin-miconazole-impregnated ECCs in reducing BSIs in neonates, including limitations in the studies themselves. Some studies may have had a small sample size, which could have limited their ability to detect a significant difference in the incidence of BSI. Variations in study design, such as different definitions of BSI or variations in catheter insertion techniques, could have also influenced the results. Patient populations may have differed between studies, including variations in the types of neonatal populations (e.g. premature vs term infants) or differences in the underlying conditions of the neonates (e.g. presence of other infections or conditions). Moreover, microorganisms may develop resistance to rifampicin and/or miconazole, which could reduce the effectiveness of the antimicrobial coating. Finally, even with an antimicrobial-coated catheter, proper catheter care is crucial to reduce the risk of infection. Inadequate catheter care could increase the risk of BSIs regardless of the type of catheter used.
The higher CLABSI rate in the antimicrobial group compared to the conventional group, warrants further exploration. While insertion and maintenance bundles were similar, speculatively additional factors such as the nature of the antimicrobials used, development of drug resistance, and patient demographics could contribute to this disparity. Moreover, the higher, although statistically not significant, rates of post-removal sepsis in antimicrobial-impregnated catheters may be attributed to factors such as organism resistance in the biofilm. These findings, however, require validation through randomized or larger cohort studies.
A significant challenge might be encountered in diagnosing CLABSI following the removal of the ECC. The potential for blood cultures to yield positive results due to skin contaminants rather than true pathogens complicates the accuracy of CLABSI diagnoses post-ECC removal, raising concerns about the possible overestimation of CLABSI incidence. Mermel, 45 in his benchmark study, discusses the risk factors for catheter-related infections and emphasizes the critical need to distinguish between true infections and contaminants. Similarly, O’Grady et al. 12 provide guidelines for the prevention of intravascular catheter-related infections, highlighting the challenges in accurately diagnosing these infections due to potential contamination. These studies underscore the importance of implementing stringent contamination control measures and developing additional diagnostic criteria to better differentiate between true infections and contaminant-positive cultures in future research.12,45
Strength and limitations
All neonates meeting the eligibility criteria were systematically enrolled, and the substantial sample size that was both inclusive and representative of the NICU population, and increasing the statistical power of the study. This statistical power was instrumental in minimizing the potential impact of selection bias and elevating the generalizability of the study’s outcomes to comparable contexts. However, notwithstanding these methodological strengths, it is imperative to acknowledge limitations of this research.
The study was conducted within a single NICU and collected data for only 1 year which may limit the generalizability of the findings. Future studies with larger sample sizes and multi-center designs are needed to further validate the results. Additionally, while the study focused on comparing the incidence of sepsis following ECC removal between conventional and antimicrobial-impregnated catheters, it did not include data on the cost-effectiveness of antimicrobial-impregnated catheters in the NICU setting. Assessing the economic implications of using these catheters could provide valuable insights for healthcare decision-making. Furthermore, long-term outcomes of neonates who have received antimicrobial-impregnated catheters, including potential effects on antibiotic resistance, should be investigated to better understand the implications of their use in clinical practice.
Conclusion
Our study indicates a potential trend toward a higher incidence of sepsis following ECC removal in neonates with antimicrobial ECCs compared to conventional ECCs, although this difference did not reach statistical significance. This highlights the complexity of post-removal complications and the need for further investigation.
These findings underscore the importance of careful consideration when selecting ECC types in neonatal care. While antimicrobial ECCs may help prevent CLABSIs in other patient settings, this protection was not observed in our study. The findings suggest that antimicrobial ECCs failed to prevent post-removal sepsis.
The observed trend prompts a call for more research to refine our understanding of catheter-related complications in neonates and guide evidence-based clinical decision-making. Additional randomized trials are necessary to comprehensively evaluate the effectiveness of antimicrobial ECCs in neonatal care.
Until conclusive evidence is available, clinicians should remain informed about the latest recommendations for preventing catheter-related BSIs in NICUs and incorporate best practices in catheter care.
Footnotes
Acknowledgements
Special thanks to the entire ECC team doctors and nurses, and to the infection control team in WWRC. Furthermore, the authors would like to thank Dr Kevin Hugill for his invaluable insights and contributions to the final revisions of this article.
Author contributions
M.A. and A.G. were main investigators, undertaking the conceptualization and design of the study, overseeing data collection, initiating the manuscript’s initial draft, and participating in subsequent revisions. M.M., F.A., J.A.R., M.A., and M.v.R. undertook responsibilities for data collection, clearing, and anonymization. The data analysis was performed by M.A. and A.G. M.v.R. contributed to the editorial content review and provided final revisions to the manuscript. All authors actively participated in the critical review and approval of the ultimate manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Research Center (MRC), Hamad Medical Corporation.
Ethics approval
The study protocol (MRC-01-21-426) was approved by the local institution review body (IRB). As the data source was anonymized, the local IRB deemed that participant consent was not feasible nor required as they determined the study a “chart review.” Participants and their parents were not involved in the design, conduct, or reporting of this study
