Abstract
Objective:
To compare relative risk (RR) of central line-associated bloodstream infection (CLABSI) between hospitals using Clave needleless connector technologies (NCT) and comparator hospitals using non-Clave NCT. And, to estimate avoided CLABSIs, mortality, and cost savings.
Background:
Needleless connectors, while protective against needlestick injury, have long been implicated as a potential risk for CLABSI. Significant RR reduction of CLABSI among the many NCT has not been clinically demonstrated.
Methods:
The U.S. Healthcare-Associated Infections database was accessed for CLABSI data for calendar year 2019 via the Centers for Medicare and Medicaid Services website. This dataset was merged with the Clave NCT manufacturer’s 2019 database to identify hospitals purchasing Clave NCT (MicroClave™, NanoClave™, Clave Neutron™, Clave™, Microclave™ Clear) and non-Clave NCT comparator hospitals. Sub-analysis of Clave NCT hospitals included: (1) Clave NCT mixed-use and (2) Clave high-volume use hospitals. The standardized infection ratio (SIR) was generated to estimate CLABSI RR after adjusting for intern/resident-to-bed-ratio (IRB), care location, and hospital demographics. Using the RR, avoided CLABSIs, mortality, and cost savings were calculated.
Results:
A total of 2987 eligible hospitals (1288 Clave NCT, 1699 non-Clave NCT) with 17,452,575 central line-days were evaluated. All three Clave NCT hospital groups showed a statistically significant reduction in RR compared to the non-Clave NCT hospitals. The RR in Clave NCT hospitals was 0.93, a 7% decrease in CLABSI risk (
Conclusion:
The use of the Clave NCT, in and of itself, is an effective risk reduction strategy for CLABSI prevention, reduced mortality, and substantial cost savings.
Keywords
Introduction
Catheter related bloodstream infections (CRBSI)s, estimated by central line associated bloodstream infections (CLABSI) surveillance criteria, are coupled with serious adverse outcomes including excess mortality and healthcare costs. 1 A 2017 U.S. (United States) Agency for Healthcare Research and Quality (AHRQ) pre-pandemic meta-analysis reported an estimate of 150 excess deaths for every 1000 in-hospital CLABSI events (RR = 2.72). 2 In the same report, the additional cost for these infections was estimated to be $48,108 (95% CI $27,232–$68,983).
The progress achieved in reducing CLABSI by 2019 in the U.S. was dismantled by the unprecedented challenges to healthcare systems during the global COVID-19 pandemic. U.S. acute care hospitals experienced a 7% increase in CLABSIs between the 2020 and 2021 pandemic years with the largest increase (10%) in intensive care units (ICU). 3 By the end of 2022, the average CLABSI standardized infection ratio (SIR) dramatically increased by 60% since 2019. 4
The impact of COVID-19 on international CLABSI rates was severe. During the pandemic years CLABSI rates in ICUs in Saudi Arabia and the Middle East increased approximately 16% compared to 20195; 76% in 21 Brazilian hospitals5,6; 86% hospitals in seven hospitals in seven International Nosocomial Infection Control Consortium (INICC) developing countries5,7; and 47% in >3400 U.S. National Healthcare Safety Networks (NHSN) hospitals. 8
With the conviction that CLABSI is preventable, the 2022 U.S. Department of Health and Human Services’ National Action Plan set a goal to reduce CLABSI by 40% in ICUs and general wards by 2028. 9 Clinicians will depend on evidence-based guidelines to drive practice change and outcome improvement efforts. These guidelines rely heavily on medical device technologies such as antimicrobial dressings, securement devices, disinfection caps, lock flush solutions, etc. for the prevention of intraluminal and extraluminal sources of CRBSI.10–12 Needleless connectors (NC), originally developed for healthcare worker needlestick prevention, have long been challenged regarding their potential role in the risk of CRBSI. Over 30 NC technologies (NCT) with varying device designs are currently marketed globally with no evidenced-based recommendation for choosing one device over another for maximum protection against microbial ingress, infection, and occlusion.10,11
A recent prospective, clinically simulated in vitro experimental study compared 20 different NCs to quantify differences in bacterial transfer and biofilm formation.
13
This study was controlled by utilizing two daily
The purpose of this secondary data analysis was to compare the risk of CLABSI between hospitals using Clave NCT and comparator hospitals using non-Clave NCT. The data was further analyzed to estimate avoided CLABSI, mortality, and cost of CLABSI.
Methods
Study design
This study is a secondary data analysis of prospective clinical data obtained from the U.S. Center for Medicare and Medicaid Services (CMS) and the Clave NCT manufacturer (ICU Medical, Inc., San Clemente, CA) for the calendar year (CY) January 1–December 31, 2019. The data was used to calculate the SIR and relative risk (RR) of CLABSI among hospitals that utilized the study Clave NCT compared with hospitals where patients received any other type of NC. Clave NCT included the Clave™, MicroClave™, MicroClave Clear™, NanoClave™, and Clave Neutron™ connectors. Subset analyses estimated avoided CLABSIs, mortality, and cost savings.
Data sources
The healthcare-associated infection (HAI) national dataset for CY 2019 was accessed and downloaded via the CMS Healthcare Associated Infections website. 14 From this database, NHSN central line-days (CLD), CLABSI observed events, and predicted CLABSI events were extracted and aggregated for the analysis. The CMS hospital characteristics file was also downloaded. Data from this file including intern/resident-to-bed ratio (IRB), rural or urban status, bed size, and geographic regional categories were merged with the HAI data for one CMS dataset. 15 Inclusion criteria were all hospitals with one or more central line-days that reported both CLABSI and hospital characteristics data.
The Clave NCT customer database for CY 2019 was provided by the manufacturer. The two databases were merged categorizing hospitals using Clave NCT (study hospitals) versus those not using the study NCs (comparator hospitals) for a final comprehensive dataset.
Study groups
Hospitals were sorted into four groups to compare the effect of Clave NCT versus non-Clave NCT usage on CLABSI. Group 1 included comparator hospitals that could not be linked to Clave NCT purchases (non-Clave NCT). Group 2 included the study hospitals that purchased Clave NCT. Group 2a and 2b were subsets of Group 2: Group 2a were mixed-use hospitals that utilized Clave NCT but with non-exclusive use within the facility; Group 2b included high-volume hospitals within the top quartile of order volume intensity defined as the total number of Clave NCT purchased during the study period divided by the total number of hospital beds. A process flow chart of data acquisition and management is shown in Figure 1.
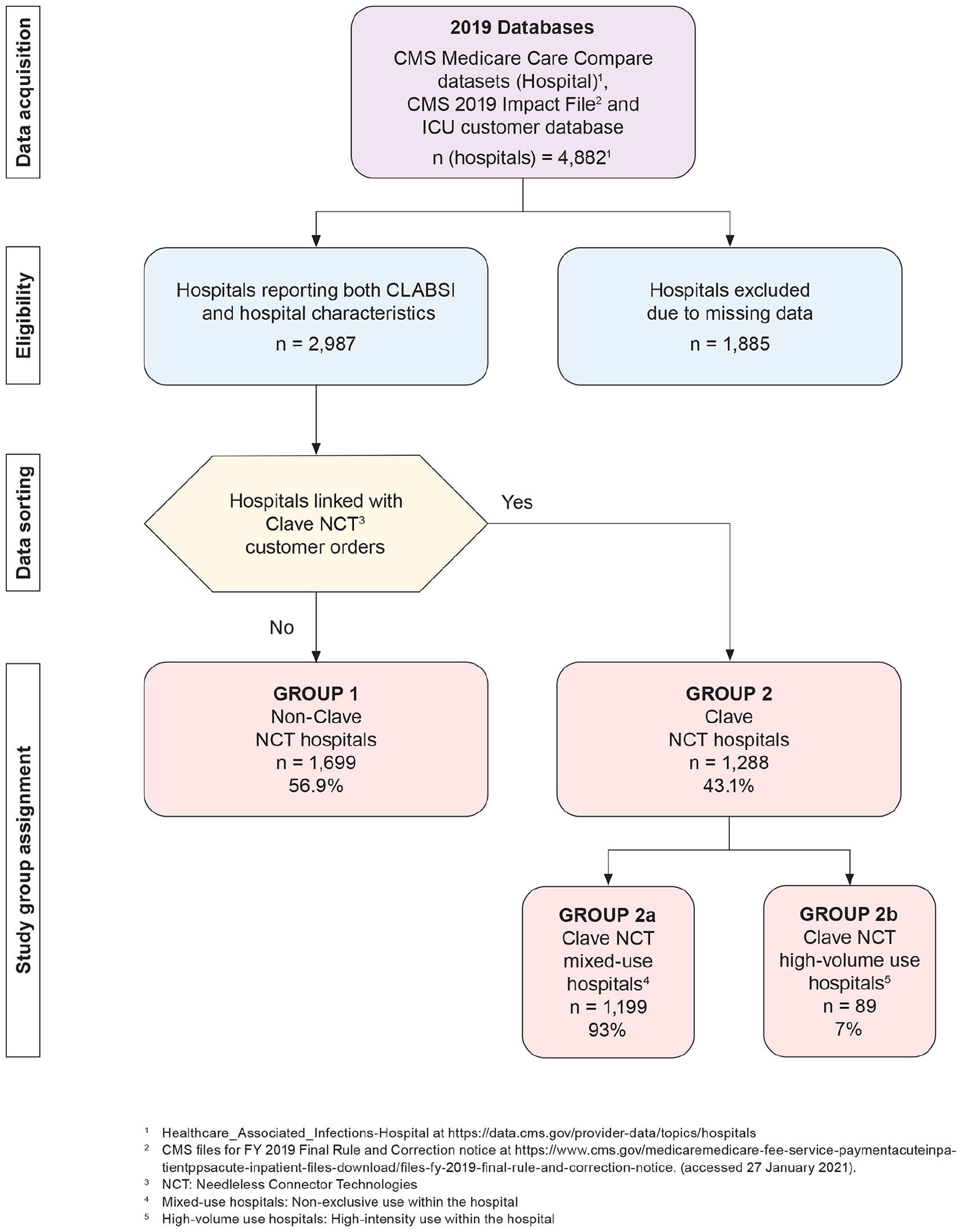
Process flow diagram.
Definitions
Clave needleless connector technologies (NCT)
Clave NCT are NCs characterized by luer-activation, split septum, internal cannula, straight flow path, low internal fluid volume, and minimal (neutral) or zero displacement.
Hospital (as defined this study)
A hospital reporting a unique facility ID within the CMS dataset.
Center for Medicare and Medicaid Services (CMS)
CMS is part of the U.S. Department of Health and Human Services that oversees many federal healthcare programs, including Medicare, Medicaid, and the Children’s Health Insurance Program (CMS.gov). CMS pays acute care hospitals for inpatient stays of Medicare beneficiaries.
Intern/resident-to-bed-ratio (IRB)
The IRB is computed as a ratio of teaching intensity, where the numerator is the number of interns and residents working at a hospital and the denominator the number of beds in the facility. 16
Standardized infection ratio (SIR)
The SIR is a summary measure used by the NHSN to track HAIs, including CLABSI, over time. 3 The SIR compares number of CLABSIs in a facility to the number “predicted” to have occurred, based on a previous year’s reported data. The SIRs are adjusted for infection risk factors such as patient care location, hospital bed size, patient age, teaching hospital, etc. 3 An SIR <1.0 indicates that less CLABSIs were observed than predicted. An SIR >1.0 indicates higher risk of CLABSI than predicted.
Relative risk (RR)
RR compares the risk of a health event that is, CLABSI, among one group with the risk in another group. The ratio of two SIRs measures the RR of CLABSI of corresponding facilities. An RR of <1.0 indicates a decreased risk for the event, in this case, one group protective against CLABSI compared to another group(s). 17 If the RR is >1.0, CLABSI is more likely to occur.
Avoidable mortality
Avoidable mortality is the number of avoided deaths attributable to a given HAI that is, deaths that are preventable. 18
Avoidable cost
Avoidable cost is the expense that would be averted if a given HAI that is, CLABSI had not occurred.
Statistical analysis
Statistical analyses were performed using the merged CMS and manufacturer databases. Number of CLD, observed and predicted CLABSI cases, and unadjusted CLABSI rates were aggregated by Clave NCT study hospitals and non-Clave NCT comparator hospitals as well as the distribution of hospital characteristics for the designated groups.
Standardized infection ratio
The NHSN SIR was calculated as the observed CLABSI cases reported to NHSN during 2019, divided by the predicted number of infections based on the 2015 referent period.
Poisson model
Random Intercept Poisson Regression model19,20 was applied with the observed number of CLABSI events as the dependent variable, and each of the three study Clave NCT groups versus non-Clave NCT as independent variables, while controlling for hospital IRB (0, >0 and ⩽0.25, >0.25 and ⩽0.6, and >0.6), rural/urban status, bed size (<100, 100–300, and >300), and geographic region. Random intercepts accounted for the over-dispersion of observed cases, and logarithm of predicted cases as the offset to the model. Adjusted RR for CLABSI was estimated as the exponential of coefficient of the Clave NCT hospitals. All confidence intervals calculated at 95%.
Avoidance of CLABSI, mortality, and cost savings
The avoided number of CLABSI cases associated with Clave NCT, mixed-use Clave NCT, and Clave high-volume NCT hospitals were estimated as observed number of CLABSI multiplied by (1–1/RR), which is the attributable fraction used in epidemiological studies. 19
Avoided mortality was the multiplication of avoided number of CLABSI by 0.15. 2 The estimate of avoided deaths resulting from the use of Clave NCT was calculated by using the reduction in the RR and the estimated range of 12%–25% mortality rate for CLABSI. 21
The cost savings calculation for Clave NCT, Clave NCT mixed-use, and Clave NCT high-volume hospitals was the multiplication of avoided number of CLABSI by the generally accepted assessment of the cost burden of $48,108 per CLABSI event. 2
Results
Study versus comparator hospitals descriptive statistics
In total, 4882 hospitals were included in the 2019 CMS Hospital Compare database. Among these, 2987 hospitals met eligibility criteria reporting central line-days >1, observed CLABSI rate per 1000 CLD, observed and predicted CLABSIs, and hospital characteristic data. A total of 1885 hospitals did not meet eligibility criteria. (Figure 1). The Clave NCT group included 1288 hospitals (43.1%), and non-Clave NCT group 1699 (56.9%) comparator hospitals. The Clave NCT group had significantly more hospitals in urban locations, major teaching hospitals, increased IRB ratios, and a larger bed size than the comparator hospitals (
Characteristics of hospitals using non-Clave NCT versus Clave NCT in 2019.
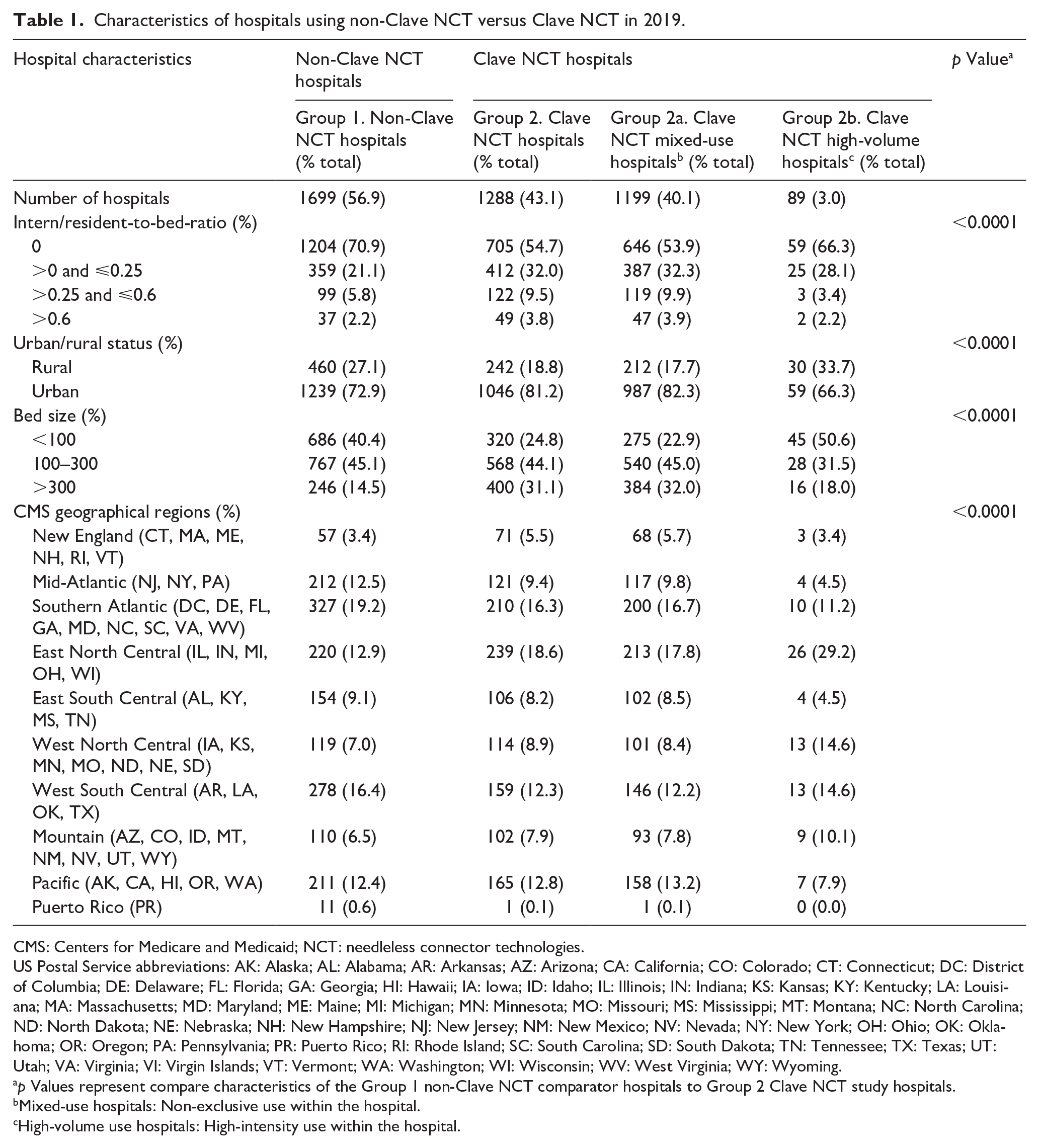
CMS: Centers for Medicare and Medicaid; NCT: needleless connector technologies.
US Postal Service abbreviations: AK: Alaska; AL: Alabama; AR: Arkansas; AZ: Arizona; CA: California; CO: Colorado; CT: Connecticut; DC: District of Columbia; DE: Delaware; FL: Florida; GA: Georgia; HI: Hawaii; IA: Iowa; ID: Idaho; IL: Illinois; IN: Indiana; KS: Kansas; KY: Kentucky; LA: Louisiana; MA: Massachusetts; MD: Maryland; ME: Maine; MI: Michigan; MN: Minnesota; MO: Missouri; MS: Mississippi; MT: Montana; NC: North Carolina; ND: North Dakota; NE: Nebraska; NH: New Hampshire; NJ: New Jersey; NM: New Mexico; NV: Nevada; NY: New York; OH: Ohio; OK: Oklahoma; OR: Oregon; PA: Pennsylvania; PR: Puerto Rico; RI: Rhode Island; SC: South Carolina; SD: South Dakota; TN: Tennessee; TX: Texas; UT: Utah; VA: Virginia; VI: Virgin Islands; VT: Vermont; WA: Washington; WI: Wisconsin; WV: West Virginia; WY: Wyoming.
Mixed-use hospitals: Non-exclusive use within the hospital.
High-volume use hospitals: High-intensity use within the hospital.
A total of 17,452,575 CLD were included in the analysis, of which the study Clave NCT hospitals accounted for 60.7% (10,593,913) and non-Clave NCT comparator hospitals 39.3% (6,858,662).
The non-Clave NCT hospitals experienced 38.4% (4537) of the total CLABSI events and 61.6% (6983) occurred in the Clave NCT hospitals (Table 2).
Descriptive statistics for non-Clave NCT and Clave NCT hospitals CLABSI data.
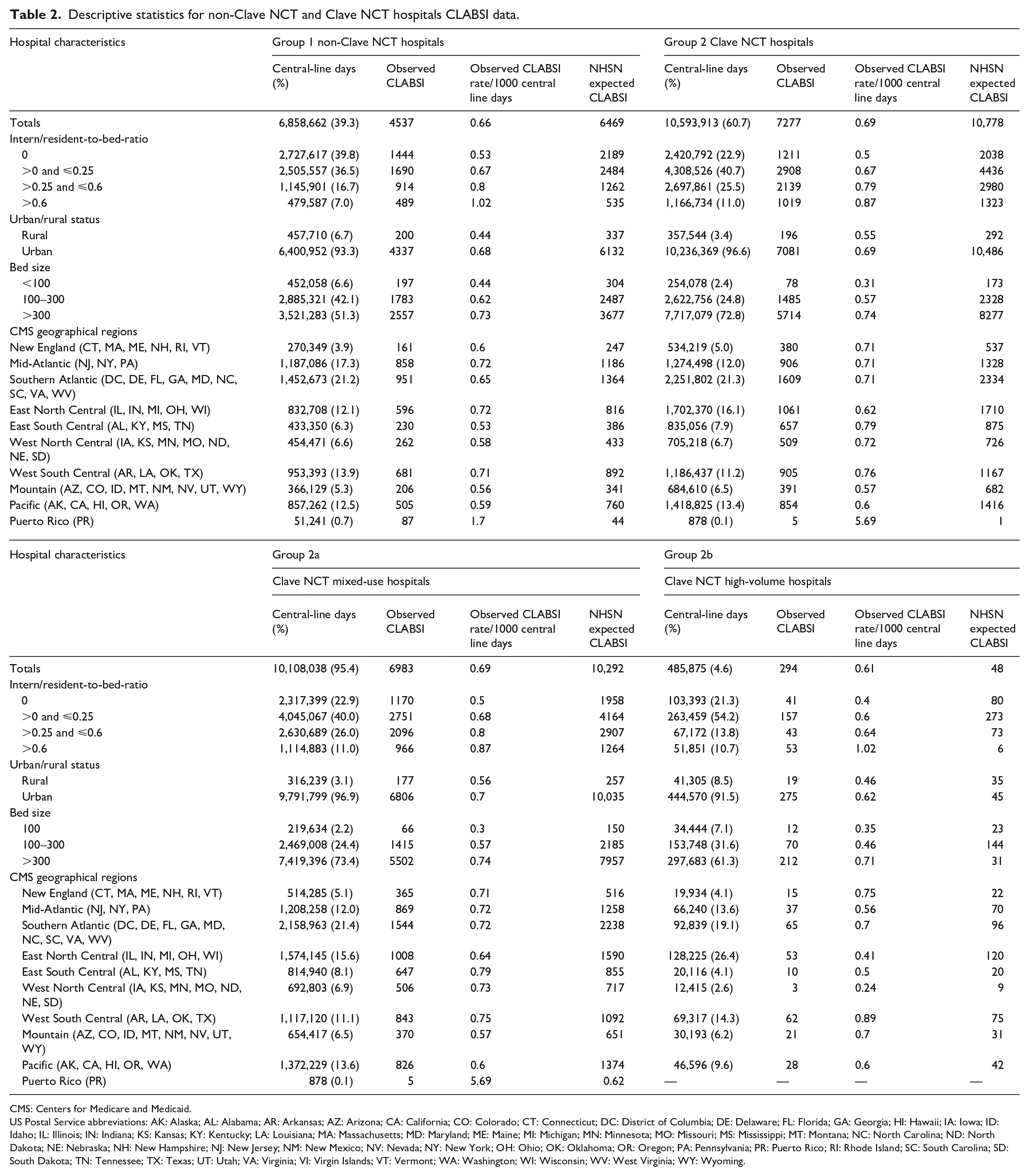
CMS: Centers for Medicare and Medicaid.
US Postal Service abbreviations: AK: Alaska; AL: Alabama; AR: Arkansas; AZ: Arizona; CA: California; CO: Colorado; CT: Connecticut; DC: District of Columbia; DE: Delaware; FL: Florida; GA: Georgia; HI: Hawaii; IA: Iowa; ID: Idaho; IL: Illinois; IN: Indiana; KS: Kansas; KY: Kentucky; LA: Louisiana; MA: Massachusetts; MD: Maryland; ME: Maine; MI: Michigan; MN: Minnesota; MO: Missouri; MS: Mississippi; MT: Montana; NC: North Carolina; ND: North Dakota; NE: Nebraska; NH: New Hampshire; NJ: New Jersey; NM: New Mexico; NV: Nevada; NY: New York; OH: Ohio; OK: Oklahoma; OR: Oregon; PA: Pennsylvania; PR: Puerto Rico; RI: Rhode Island; SC: South Carolina; SD: South Dakota; TN: Tennessee; TX: Texas; UT: Utah; VA: Virginia; VI: Virgin Islands; VT: Vermont; WA: Washington; WI: Wisconsin; WV: West Virginia; WY: Wyoming.
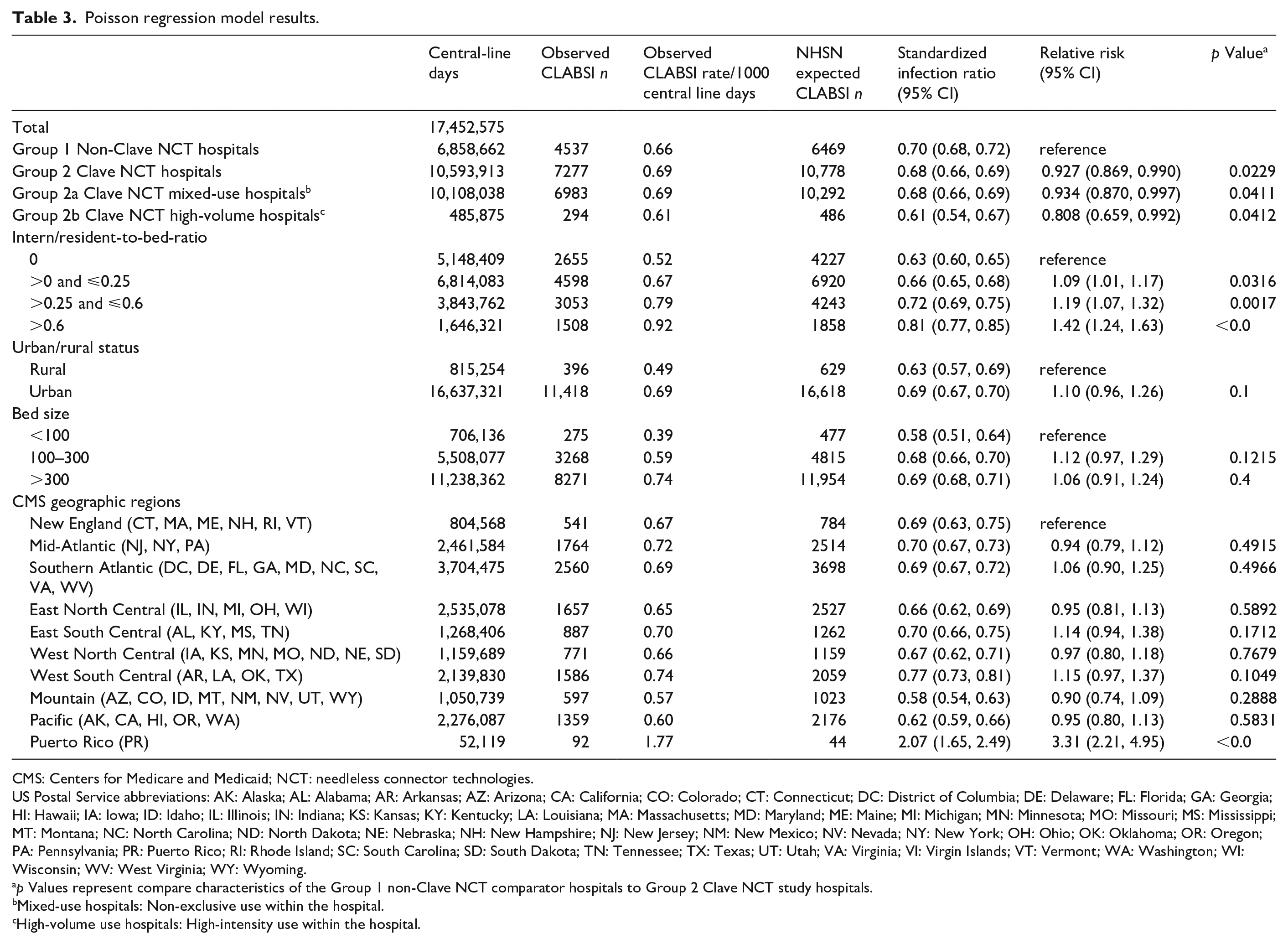
The unadjusted CLABSI rate per 1000 CLD was 0.66 for non-Clave NCT hospitals and 0.69 for Clave NCT hospitals. CLABSI rates in both groups were increased in hospitals with larger IRB ratios, urban setting, and bed size >300. Rates varied among the geographical regions. The SIR for the non-Clave hospitals was a computed 0.70 (95% confidence interval (CI) 0.68, 0.72) and 0.68 (95% CI 0.66, 0.69) for the Clave NCT (Table 3, Figure 2).
Poisson regression model results.
CMS: Centers for Medicare and Medicaid; NCT: needleless connector technologies.
US Postal Service abbreviations: AK: Alaska; AL: Alabama; AR: Arkansas; AZ: Arizona; CA: California; CO: Colorado; CT: Connecticut; DC: District of Columbia; DE: Delaware; FL: Florida; GA: Georgia; HI: Hawaii; IA: Iowa; ID: Idaho; IL: Illinois; IN: Indiana; KS: Kansas; KY: Kentucky; LA: Louisiana; MA: Massachusetts; MD: Maryland; ME: Maine; MI: Michigan; MN: Minnesota; MO: Missouri; MS: Mississippi; MT: Montana; NC: North Carolina; ND: North Dakota; NE: Nebraska; NH: New Hampshire; NJ: New Jersey; NM: New Mexico; NV: Nevada; NY: New York; OH: Ohio; OK: Oklahoma; OR: Oregon; PA: Pennsylvania; PR: Puerto Rico; RI: Rhode Island; SC: South Carolina; SD: South Dakota; TN: Tennessee; TX: Texas; UT: Utah; VA: Virginia; VI: Virgin Islands; VT: Vermont; WA: Washington; WI: Wisconsin; WV: West Virginia; WY: Wyoming.
Mixed-use hospitals: Non-exclusive use within the hospital.
High-volume use hospitals: High-intensity use within the hospital.
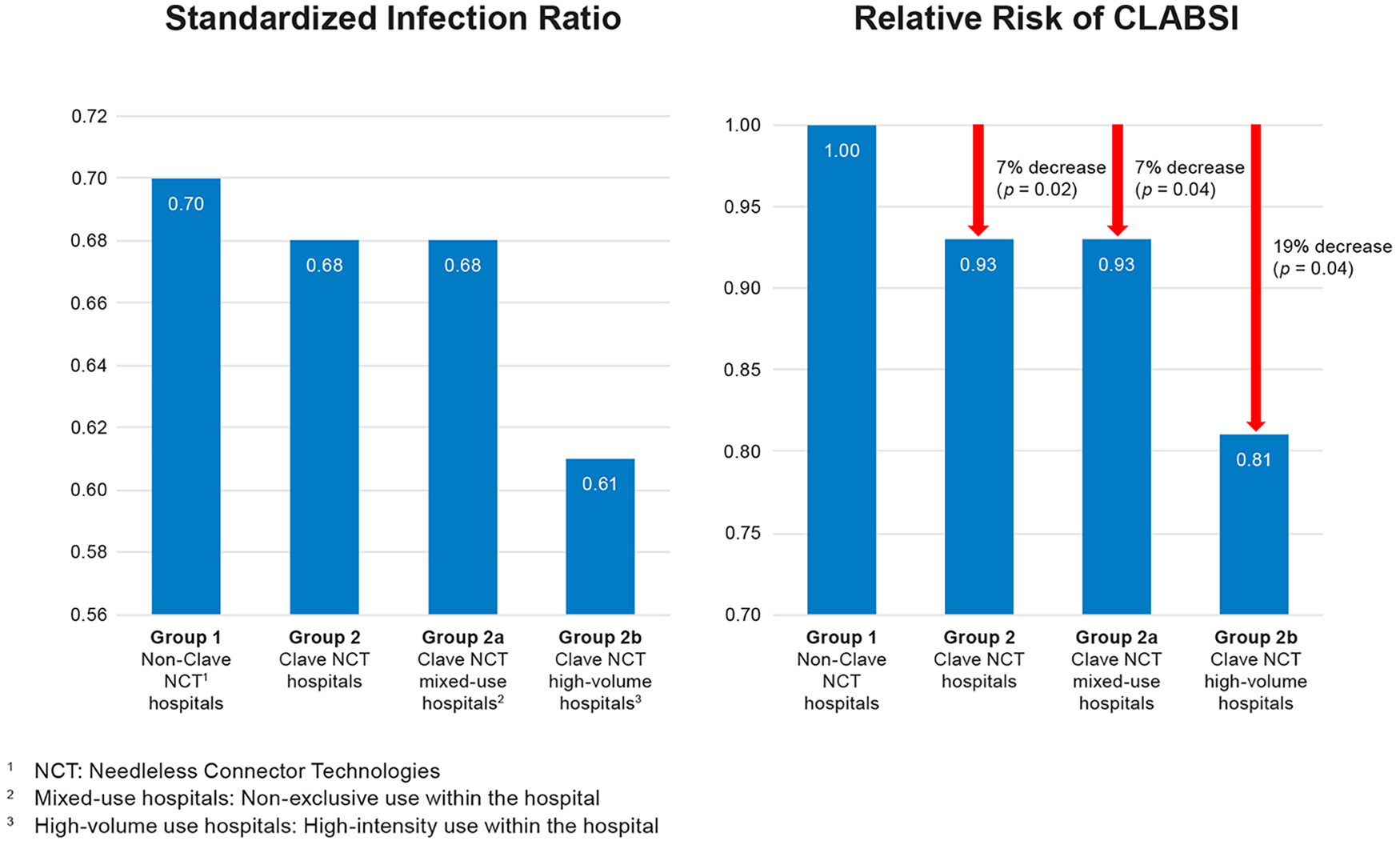
Comparison of standardized infection ratio and relative risk of CLABSI in non-Clave NCT and Clave NCT hospitals.
Relative risk for study versus comparator hospitals
The RR was significantly lower for the Clave NCT hospitals when using the comparator hospitals as reference and adjusting for hospital characteristics, 0.93 (95% CI 0.869, 0.990;
Clave NCT mixed-use and high-volume hospital descriptive statistics
The Clave NCT study group included 1199 (93%) Clave NCT mixed-use hospitals and 89 (7%) Clave NCT high-volume hospitals (Table 2). Clave NCT mixed-use hospitals had 95.4% of Clave NCT hospitals CLD and Clave NCT high-volume hospitals 4.6%. The Clave NCT mixed-use hospitals accounted for 96% (6983) of 7287 CLABSI events and high-volume hospitals 4% (294). The unadjusted CLABSI rate for the Clave mixed-use hospitals was 0.69/1000 CLD and SIR 0.68 (95% CI 0.66, 0.69). The Clave high-volume hospitals experienced the lowest CLABSI rate of 0.61/1000 CLD and SIR 0.61 (95% CI 0.54–0.67) of all hospital groups (Tables 2 and 3 and Figure 2).
Relative risk for Clave NCT mixed-use and high-volume hospitals
The RR of CLABSI was significantly lower in both Clave NCT hospital subgroups: Clave NCT mixed-use hospitals 0.93 (95% CI 0.870–0.997;
Avoidance of CLABSI, mortality, and cost savings
Applying the 7% reduction in RR, an estimated 563 CLABSIs were avoided in the Clave NCT hospitals and 493 in the Clave NCT mixed-use hospitals (Table 4). The Clave NCT high-volume use hospitals experienced a 19% reduction in RR, avoiding approximately 70 CLABSI cases. An estimated 68–141 deaths were prevented by use of the Clave NCT for the time-period evaluated. The realized cost savings of the Clave NCT hospitals was estimated to be $27,095,233. In Clave NCT mixed-use and Clave NCT high-volume hospitals the cost savings was approximated at $23,738,671 and $3,356,560 respectively.
Estimated avoidance of CLABSI, mortality, and cost savings for 2019.
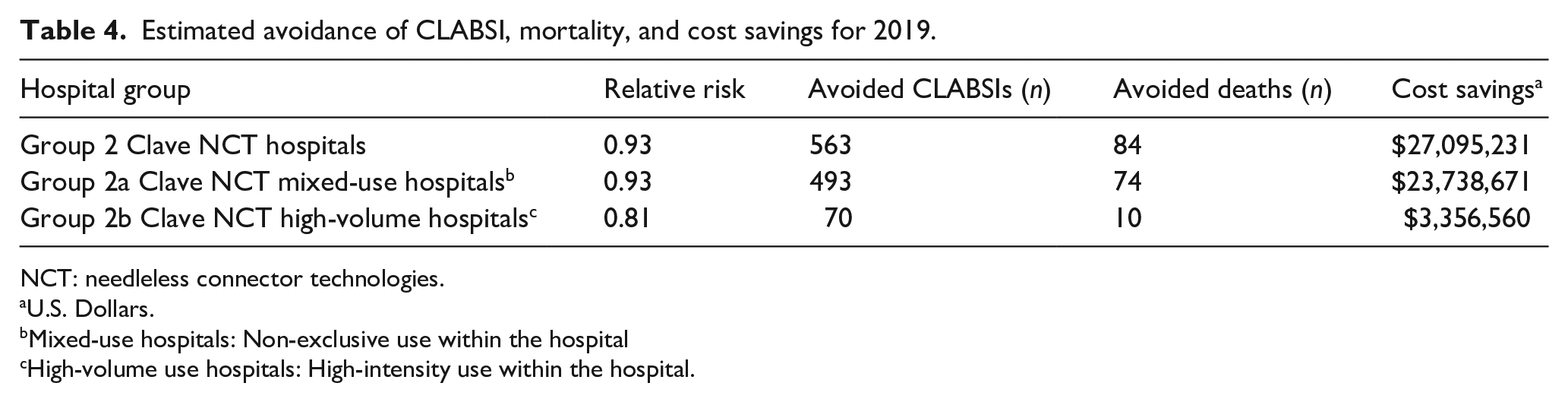
NCT: needleless connector technologies.
U.S. Dollars.
Mixed-use hospitals: Non-exclusive use within the hospital
High-volume use hospitals: High-intensity use within the hospital.
Discussion
To date, this study including 2987 hospitals and over 17.5 million CLD is the largest known secondary public/private database analysis of the relative risk of CLABSI, avoidable CLABSI and mortality, and cost savings associated with the use of an exclusive (Clave) NC technology compared to all other technologies. The results found hospitals using Clave NCT experienced a lower SIR, a statistically significant reduction of RR, an estimated 84 avoided deaths, and over $27 million in cost savings.
Since the introduction of NCT in the 1990s, experts have written or theorized about optimal NC design and selection criteria without rigorous evidence of the comparative risk of infection among the numerous marketed devices.22–25 A 2020 systematic review examining the clinical impact of NC design concluded that more studies analyzing microbial ingress, antiseptic application, and intraluminal biofilm in varying NC designs were necessary to make confident choices.
7
Tabak et al. first introduced the innovative methodology for secondary analysis of existing public (CMS) and private data sources (CareFusion, San Diego, CA) to examine CLABSI risk associated with NCs.
26
The study compared the use of a specific NC design, a positive displacement mechanical valve, in 758 U.S. hospitals with 2316 comparator hospitals using all other NCs for CY 2013. They found that the RR in the study hospitals was not significantly different than the comparator hospitals when adjusting for hospital characteristics (
The expansive experimental in vitro study previously described by Ryder et al. found the two Clave NCs (MicroClave and Neutron) were within the statistically significant group of four NCs with the least bacterial transfer and intraluminal biofilm formation compared to 16 other NCs studied.
13
Further analysis of eight design factors found that NC design features associated with the least bacterial transfer and biofilm formation were those incorporating a split septum, minimal seal length, internal cannula, low surface area and volume, neutral displacement, and simple hydrodynamics of the flow path; all of which are consistent with the Clave technology. The results corroborate the findings of the present study where the use of the Clave NCT had a statistically significant RR reduction of CLABSI in the clinical setting. This was especially noteworthy in the Clave NCT high-volume use hospitals where the CLABSI rate (0.61/1000 CLD), IRB (equal to non-Clave hospitals >0.6), urban settings, and bed size >100 beds for these hospitals was significantly lower than all other NCT groups. The high-volume hospitals also had the lowest SIR (0.61) and a significant 19% reduction in RR (0.81,
CLABSI Is a preventable adverse event associated with enormous morbidity, mortality, and cost burden. 1 CLABSI rates dramatically increased during the Covid-19 pandemic years and currently remain well above the pre-pandemic rate. 4 The encumbrance will likely be intensified when the pending U.S. requirement for Hospital Onset Bacteremia and Fungemia surveillance to include BSI related to all vascular access devices (VAD) will uncover a dramatic increase in CRBSI.29,30 It is now important to re-evaluate infection prevention practices and implement effective medical device technologies to reduce CRBSI risk with all VADs. While CRBSI prevention requires a combination of evidenced-based strategies, this study supports the choice of NCT as an important component of infection prevention bundles.
Strengths and limitations
A major strength of this secondary analysis was the use of the large-scale database of CMS/NHSN publicly reported HAI outcome data. Prospective, longitudinal data collected by practicing infection preventionists using a standardized NHSN CLABSI definition reflects reliable nationwide data and eliminates bias, and conflict of interest issues related to the study objective.
However, the study design is not without limitation. First, while a randomized controlled trial inclusive of all on-market (22+) needleless connectors would be the most robust to test the hypothesis, it would be highly unrealistic and virtually impossible due to the required number of clinical sites and subjects, duration of the study, inability to control for confounding variables, extensive staff education, etc., not to speak of the exorbitant cost.
As an alternative, a secondary data analysis of the large, reliable, and non-biased CDC’s NHSN database was chosen for the evaluation. This data was collected prospectively by infection control practitioners in a majority of U.S. hospitals and reported to NHSN which provides facilities, states, regions, and the nation with data needed to identify problem areas, measure progress of prevention efforts, and ultimately eliminate healthcare-associated infections. This data, which is used for national comparison of CLABSI rates and SIR, is not controlled for specific variables that in part contributes to infection risk for example, choice of catheter, vein puncture technique, skin antisepsis with maximum sterile barrier precautions, exit site, type of fixation and dressing, frequency of dressing change, etc. However, readily available, and widely adopted guidelines, recommendations, and standards addressing infection prevention of CLABSI allows for an assumption of similarity between institutions compared within the study.
To address potential bias of the confounding variables two approaches were employed. Firstly, the variations in prevalence rates due to differences in infection prevention practices can partly be accounted for by the random intercepts. This model assumes the response variable has a Poisson distribution and assumes the logarithm of its expected value can be modeled by a linear combination of unknown parameters. This accounts for the effects of other variables such as the ones mentioned above. Secondly, standardized hospital quality indicators (bed size, intern/resident-to-bed-ratio, rural/urban status, and geographic region) were used as controls for intervening variables. The random intercept Poisson regression model used in this analysis, in part accounts for the combination of unknown variables that may be different among hospitals. In addition, the factors of bed size, intern/resident-to-bed-ratio, rural/urban status, and geographic region that influence quality outcomes3,31,32 were statistically controlled. The random intercept Poisson model is an appropriate choice for analysis when the CLABSI rates are low.
Nearly 2000 hospitals were excluded from the data analysis due to missing CLABSI or hospital characteristic data required for a RR regression model. It is unknown whether the inclusion of these hospitals would have any effect upon the results. We also utilized 2019 pre-Covid 19 data for the analysis. Due to the disruption of standardized care, it was presumed that data from the pandemic years may not reflect actual differences.
Lastly, the percentage of Clave NCT used within mixed-use hospitals was not discernable since the actual number and location of use within a hospital was not obtainable beyond purchase. The impact of variable product usage in Clave NCT mixed-use hospitals was undetermined, nonetheless the RR of was significantly less than the non-Clave NCT hospitals.
Conclusion
The use of NCT is designated as an “unresolved issue” in current standards and recommended CLABSI reduction strategies.10,11 The more recent awaited research to evaluate microbial ingress and intraluminal biofilm formation found statistically significant differences among the 20 NCs studied, therefore some NCT are likely to be of greater risk for CLABSI than others. This precludes an all-inclusive recommendation for all NCT as a risk or as a preventive measure and points to the need for critical evaluation of design features in the selection process.
While CLABSI prevention requires a combination of evidenced-based strategies, we found that the cautious choice of an NC is a critical component of infection prevention bundles. The present study supports the incorporation of Clave NCT as an evidence-based strategy to significantly reduce the risk of CLABSI and mortality with substantial cost savings, especially when used as the primary NC throughout the facility.
Footnotes
Acknowledgements
The authors would like to acknowledge the support provided by Dr Xiaowu Sun, PhD. Dr Sun provided statistical design, analysis, and supported interpretation of the results.
Author contributions
All authors contributed to the study conception and design of the study, analysis and interpretation of the results with support from Dr Xiaowu Sun, draft manuscript preparation and all authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author JB is an employee of ICU Medical. Author MR has received consultancy fees from ICU Medical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by ICU Medical.
Ethical approval
This was a secondary data analysis with no human subject enrollment. Ethical Approval not applicable.
