Abstract
Purpose:
To quantify the rates and identify risk factors for the complications of central venous access devices (CVADs) removal in children.
Method:
Retrospective (2018–2023) review of children undergoing CVADs removal at a single institution. Data are reported as frequency, percentages and median. Logistic regression analysis was used to identify risk factors associated with difficult removal. Receiver Operating Characteristic Curve (ROC) analysis was conducted to identify the age cut-off and positive likelihood ratio (+LH) for the indwelling time associated with complicated removal. p-Value <0.05 were considered statistically significant.
Results:
We identified 208 CVAD removals with a median age of 7.2 (0.2–18.4) years including 116 (55.8%) males. The median CVAD placement duration was 1.26 years (0.4–5.7) years. Indications for insertion included acute lymphoblastic leukaemia (ALL; 78/208, 37.5%), lymphomas (31/208, 14.9%), other malignancies (58/208, 27.9%). Removal indications included completion of treatment (144/208, 69.2%), infection (22/208, 10.6%), malfunction (7/208, 3.4%) and other reasons (35/208, 16.8%). There were 20 (9.6%) complications characterised by difficulty removing the CVAD. Complicated removals were more likely to occur in children with ALL as the primary diagnosis (p = 0.001); independently of the indication for insertion, longer indwelling time was associated with higher risk of complicated removal (p < 0.001). Indwelling time >2.09 years was associated with a 2.87 increased risk of difficult removal.
Conclusion:
In our experience, almost 10% of CVAD removals in children result in complications. These findings are associated with an indwelling time >2 years; strategies to prevent complicated removals should be considered in children requiring long-term central venous access.
Introduction
Central venous access devices (CVADs) are catheters placed into the central venous circulation, through either a thoracic central vein or the iliocaval venous system; these devices allow for long-term venous access to deliver treatments such as antibiotics, chemotherapy, dialysis and total parenteral nutrition. 1 In particular, they are indicated in patients requiring long-term venous access, the infusion of peripherally incompatible solutions (such as those with a high osmolarity), the delivery of multiple or high-volume products, and in patients with difficult intravenous access (DIVA). In the paediatric population, main indications include chemotherapy for malignancy, parental nutrition for intestinal failure and infusion of blood products in haematological conditions.
The choice of device should be guided by duration of use, peripheral vein availability, treatment requirements and patients/carer preference. 2 Non-tunnelled CVADs are placed percutaneously and exit the skin in proximity to the cannulation site. They are intended for short-term venous access. In contrast, tunnelled CVADs are placed subcutaneously and exit the skin at various distance from the cannulation site. Tunnelled catheters may be cuffed in order to reduce dislodgment and bacterial entry; these include Hickman™ and Broviac™ devices.3,4 Totally implantable venous devices are characterised by a catheter connected to a subcutaneous port such as the Port-a-Cath™ and more recently the peripherally inserted central catheters (PICC) port. Such devices are protected by the overlying skin, allowing for a longer duration of use, improved comfort, cosmetic benefit and reduced infection risk.
CVAD insertion, maintenance and removal is subject to complications.5,6 Complications during insertion are reported in 7%–18% of cases and include pneumothorax, haemorrhage, arterial puncture and cardiac rhythm dysfunction.7–9 In the post-operative period, patients may be susceptible to a variety of well-documented complications, including local infection or central line associated blood stream infection (CLABSI), catheter dislodgment and/or malfunction. 10 CVAD removal may be indicated due to the end of treatment or in the presence these complications. Incidents (such as device fragmentation and retention) during removal might occur, and have been reported in 1.8%–16% of cases.11,12 Therefore, surgeons and other health professionals encountering a CVAD that is difficult to remove may need to apply significant traction, extend the original incision or even create a new incision. 12 Previous studies have demonstrated that catheter damage, associated with a ‘roughened’ surface from the reaction to the foreign body and presence of a calcified fibrin sheath, may account for these difficult removals.11–13 Documented risk factors include prolonged device use, smaller lumen size and CVADs inserted in patients with haematological or oncological conditions.12,14
With limited large-scale studies in children, there is an overall paucity of data on the complications related to CVADs removal, which may underestimate the true impact of this specific problem in the paediatric population.15,16
The aim of our study was to report the incidence of complicated CVADs removal and identify risk factors in children.
Methods
Participants
We conducted a retrospective study of all paediatric patients (up to 18 years of age) undergoing an elective or emergency CVAD removal at Monash Children’s Hospital, a tertiary paediatric hospital in Melbourne, Australia, between 2018 and 2023. This study was approved by the Human Research Ethics Committee at Monash Health (HREC RES-23-0000-002Q).
Insertion bundle
All children underwent an USS-guided insertion of the CVAD, according to standardised institutional guidelines, via a modified Seldinger technique (catheter inserted over peel-away sheath) using the access kit provided with the device or a micro-puncture access kit (Micropuncture® Access Set – Cook Medical LLC, Bloomington, USA). The right internal jugular vein (IJV) was the preferred site. However, if it was not suitable, due to thrombosis from previous cannulations or other anatomical abnormalities, the left IJV, and then the subclavian veins, were utilised. Vein patency was assessed with USS in all children before attempting venepuncture. The external diameter of the catheter was determined in order to maintain a catheter-to-vessel ratio less than 45%. 17 In all patient’s silicone lines were utilised; Bard implanted port devices and Hickman™ lines (Bard Access Systems, Inc., USA) and SmartPort® implanted port devices (AngioDynamics, Inc., USA) were used. Implanted port devices were positioned over the chest wall and secured to the fascia using sutures including polypropylene, polyglactin 910, polydioxanone or nylon. Tunnelled CVAD were secured to the skin of the chest wall using a nonabsorbable (polypropylene or nylon) purse string suture or a subcutaneous anchoring device (SecurAcath®, Interrad Medical, USA) in the last 50 patients. At the end of the procedure, tunnelled CVADs and implanted port devices with the needle in situ, were dressed with 3M™ Tegaderm™ I.V. Advanced Securement Dressing (3M Australia Pty Limited). Tunnelled CVADs were also dressed with chlorhexidine gluconate impregnated dressing (BioPatch® – Ethicon, Inc., Somerville, NJ).
Maintenance bundle
The CVADs dressing was changed every 7 days or when soiled, no longer occlusive/adherent or if there was evidence of local inflammation. Tunnelled CVADs, and implanted port devices with the needle in situ, were regularly dressed with 3M™ Tegaderm™ I.V. Advanced Securement Dressing (3M Australia Pty Limited); tunnelled CVADs were also dressed with chlorhexidine gluconate impregnated dressing (BioPatch® – Ethicon, Inc., Somerville, NJ). Implanted port devices accessed for greater than 24 h also had a chlorhexidine gluconate impregnated dressing applied.
When the CVAD was in use, all lumens were flushed with 0.9% saline following the completion of the medication and/or fluid administration, in-between medication administrations, following blood products and parenteral nutrition administration, and after blood sampling.
When the CVAD was not in regular use in a hospital setting, all lumens that did not have a continuous infusion running through were aspirated and flushed with 0.9% saline at least once a day. In the outpatient setting, CVADs were flushed with 0.9% saline and locked with TauroLock™ (TauroPharm GmbH, Waldbüttelbrunn, Germany) after use, or at minimum once every 6 weeks if not in use.
Complicated removals
We followed a prior definition of complicated CVAD removal which included deterioration or fragmentation of the device requiring additional intervention, such as formal neck dissection or other second procedures such as radiologically guided removal of fragments. 11
Data collection
Data were collected following review of each case’s operation report and medical records and recorded on an Excel spreadsheet (Microsoft® Excel® 2016). Demographic information included patient’s age at removal, sex, patient’s primary diagnosis. CVAD-related factors investigated included indwelling time (measured as time period between CVAD insertion and removal) and type of medical condition. We graded the difficulty of CVAD removal using a revised grading tool previously reported. 12
Statistical analysis
Baseline demographic and CVADs data were recorded and reported as frequency, percentages and median (range). Group comparisons were performed using GraphPad Prism 10.2.0 (GraphPad Software, Boston, Massachusetts USA, www.graphpad.com). The Shapiro-Wilk test was used to assess the normality of the demographic data. The Mann-Whitney test was used to compare significant differences between medians for non-normally distributed data. Logistic regression analysis was used to test for risk factors associated with difficult removal. Receiver Operating Characteristic Curve (ROC) analysis was conducted and the Area Under the Curve (AUC) was calculated to identify a criterion and positive likelihood ratio (+LH) for the indwelling time associated with difficult removal. A p-value of <0.05 was considered statistically significant.
Results
A total of 208 patients underwent CVAD removal during the study period and were included in the statistical analysis. Patients’ characteristics are outlined in Table 1. The proportion of males was 55.8% and the median age at CVAD insertion and removal was 4.5 years (0.2–18) and 7.2 (0.6–18.4) years respectively. The median indwelling time was 1.26 years (0.4–5.7).
Patients’ characteristics.
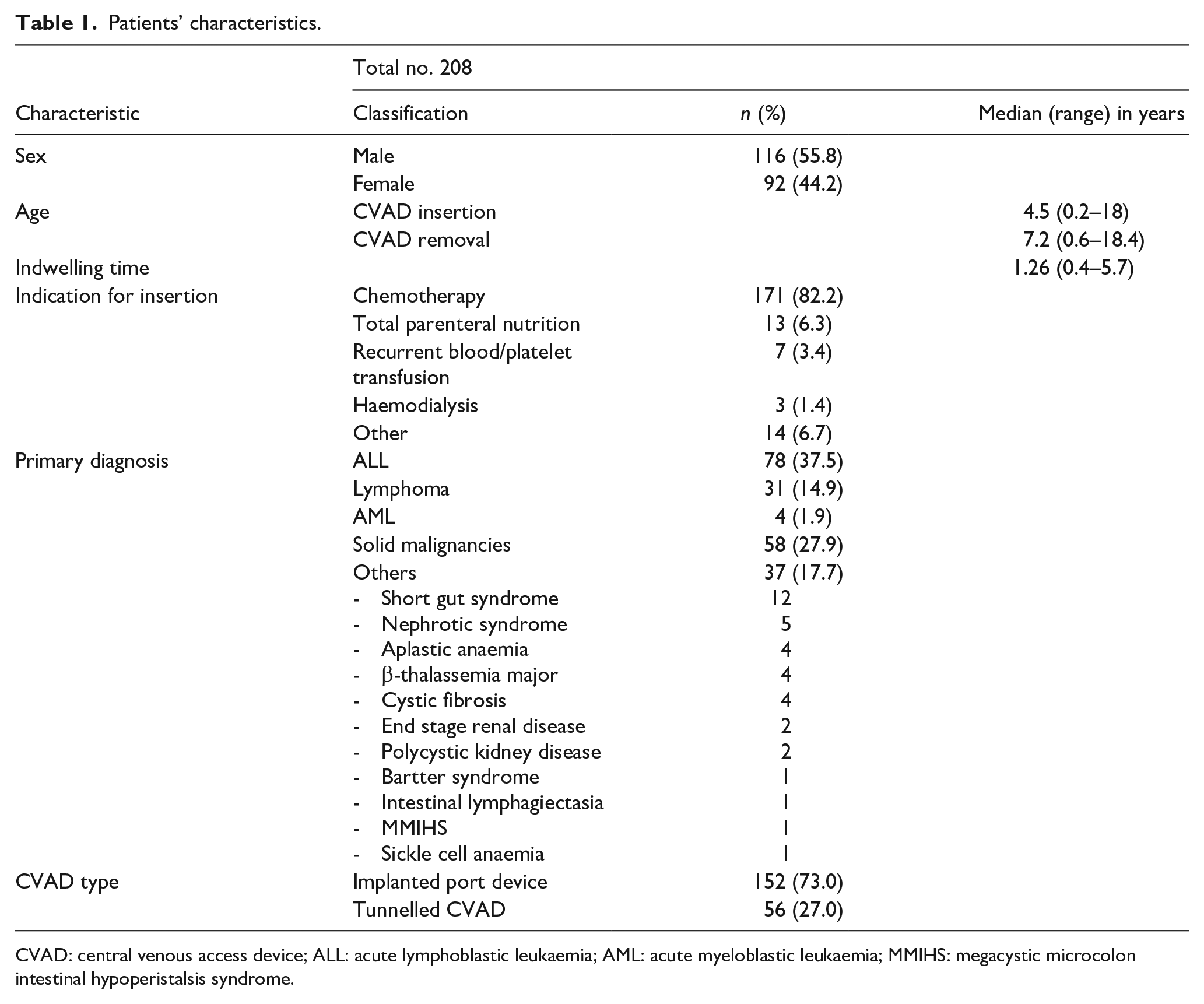
CVAD: central venous access device; ALL: acute lymphoblastic leukaemia; AML: acute myeloblastic leukaemia; MMIHS: megacystic microcolon intestinal hypoperistalsis syndrome.
The majority of insertions were performed to deliver chemotherapy in children with malignant tumours (Table 1).
Indications for removal included completion of treatment (144/208, 69.2%), CLABSI (22/208, 10.6%), for a CLABSI rate of 0.18 × 1000-line days, malfunction (7/208, 3.4%), and other reasons (35/208, 16.8%).
Complicated removals
Twenty CVAD removals (9.6%) were complicated by requiring further intervention. The demographic and intraoperative findings of this cohort are presented in Table 2.
Demographic and intraoperative findings of patients with complicated CVAD removal.
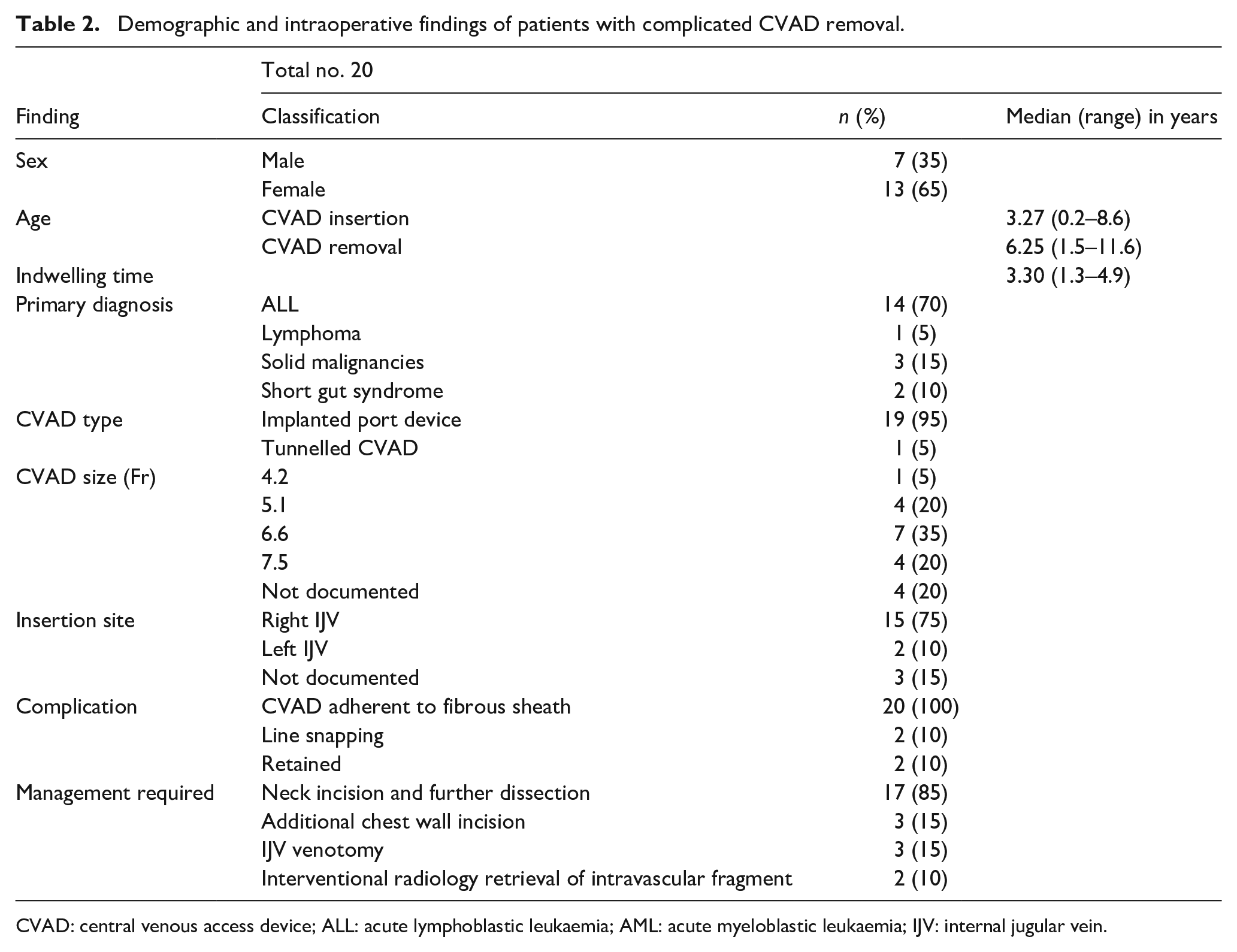
CVAD: central venous access device; ALL: acute lymphoblastic leukaemia; AML: acute myeloblastic leukaemia; IJV: internal jugular vein.
Identified risk factors
Indwelling time for implanted port devices was significantly longer compared to tunnelled CVADs: 2.3 (0.4–5.7) versus 0.5 (0.8–3.3) years, p < 0.0001. The child’s age at removal was overall significantly higher in non-complicated removals; 7.48 years (0.6–18.4) versus 6.25 (1.5–11.6), p < 0.0001. However, indwelling time prior to removal was overall significantly longer in complicated removals compared to uncomplicated removals; 3.3 years (1.3–4.9) versus 1.1 (0.4–5.7), p < 0.0001.
The incidence of ALL was significantly higher in complicated removals; 14/20 versus 78/188 p = 0.01. However, the indwelling time for patients with ALL was significant longer compared to patients with other pathologies: 2.5 (0.4–4.2) versus 0.8 (0.5–5.7) years, p < 0.0001. Logistic regression analysis documented a significant association between the indwelling time and the risk of difficult removal, independently of the primary diagnosis and the type of CVAD (OR 2.9 [95% CI 1.08–7.9], p = 0.001).
ROC curve analysis documented AUC of 0.84 (95% CI 0.77–0.88), p < 0.0001 with a criterion of >2.09 years (94.7% sensitivity, 67.0% specificity) and +LR of 2.87 (95% CI 2.27–3.64).
The degree of difficulty of all included complicated and uncomplicated cases is graded in Table 3. Two cases fell into grade 5, where catheter retention occurred and fragments were unable to be removed during the procedure: these fragements were subsequently removed by the interventional radiology team with endovascular retrieval.
Grading of degree of difficulty at removal of CVAD.

Revised from Wilson et al. 12
Discussion
We report a single-centre study investigating the incidence of complicated CVADs removal in a large series of children. We identified an overall 9.6% incidence of complicated removal in 208 patients; in the literature, the incidence varies from 2% to 16%, which may be attributed to the unique characteristics of each series including patients’ demographics, medical conditions, insertion technique, device use and indwelling time.11,12,14,18 Our study has the second-largest cohort in the existing literature and provides a perspective into the clinical factors for difficult removal. CVAD insertion leads to the formation of a fibrin sheath as part of the reaction to the foreign body, which adheres to the vein wall resulting in resistance during removal.18,19 Risk factors that have previously been identified to contribute to difficult removal are haematological malignancy, chemotherapy, infection, small lumen size and prolonged device use.11,12,14,18 Our study contributes to these findings, confirming that ALL and prolonged device use of more than 2 years are associated with complicated removal. The association with ALL and difficult CVAD removal is well documented in the literature; Wilson et al. reported that patients receiving chemotherapy for ALL were more likely to experience difficult removal (74/98, 76%). 12 Similarly, Teague et al. and Jones and Giacomantonio reported more complicated removal in patients with ALL.11,20 It is possible that particular chemotherapeutic agents used in patients with ALL contribute to the process, causing catheter fixation, although no study has clearly investigated the effects of different chemotherapeutic agents in this context. Nonetheless, it must be noted that chemotherapy administration is very often the main indications for CVAD insertion in children. In our study we found that indwelling time >2 years was independently associated with difficult CVAD removal; we also found that the catheter indwelling time in patients with ALL was significant longer than in patients with other conditions (p < 0.0001). Therefore, we argue that the association between ALL and difficult removal might be simply due to the prolonged indwelling time, rather than the chemotherapic agents. Unfortunately, due to the retrospective nature of this study we have not been able to obtain reliable data regarding the chemotherapic agents used in our population of children; prospective large studies are needed to ascertain the relationship between the type of chemotherapic agent and difficult CVAD removal in children.
CVAD material, specifically silicone, has been hypothesised to contribute to an increased rate of complications.11,14 The link between the type of material and difficult removal remains controversial; Chatani et al., documented the use of silicone catheters being associated with increased risk of difficult removal in adults. 21 However, Wilson et al. reported polyurethane catheters to be at higher risk of difficult removal in children. 12 Other studies have found no difference in the complication rates of CVADs made of silicone or polyurethane. 20 Our series include only silicone catheters, but we have recently moved to polyurethane CVADs in our institution and we are prospectively monitoring their outcomes.
Regarding the types of complications, CVAD removal may be characterised by the need for firm traction, the re-opening of the initial venous access incision, further dissection, venotomy or new incision. The majority of our complicated removals were in grade 3 (55%), requiring re-opening of the initial incision and new neck or chest wall incision. This is of a higher degree of difficulty compared to the majority of patients (60%) in Wilson et al. and all patients in Teague et al. which only required further dissection following the re-opening of the venous access incision.11,12 These discrepancies might be due to patients’ characteristics, insertion technique, type of material and indwelling time.
Whether retained fragments necessitate further retrieval is uncertain. Although retained cuff material from tunnelled CVADs in the subcutaneous can be managed conservatively, it can lead to a foreign body reaction and/or infection; therefore, several authors advocate removal of the cuff at the time of CVAD removal to avoid additional procedure.22,23
Concerns remain of the risk of thrombosis and catheter migration with fragments retained within the central venous system. 20 This has not been extensively studied in children due to limited case numbers, but ongoing follow-up of 5–6 years in Wilson et al. and Milbrandt et al. shows that patients remain asymptomatic.12,18 In our study, we had two cases of intravascular retained fragments; these were retained in the internal jugular vein and later successfully retrieved by interventional radiology; another fragment was retained in the chest wall subcutaneous tissue of a teenage girl and it was decided not to undergo further intervention due to cosmetic concerns of additional incision in the breast region. We acknowledge that our study is limited by its retrospective nature and by being based in a single tertiary paediatric centre, limiting its generalisability to the adult settings. Due to the retrospective study design, we were not able to document the exact insertion technique (e.g. the use of micro-puncture kit) that might be important to reduce endothelial trauma. In addition, the small number of non-oncological patients, of which only two had a difficult removal, did not allow identification of risks associated with different medical conditions.
In conclusion, we have reported on a large population of children with complicated CVADs removal over a 5-year period. In our experience complicated removals are not rare, and important contributing risk factors have been identified which need to be investigated further.
Indwelling time >2 years is a stronger predictor of complicated removal; strategies to elective replace CVADs after this period might need to be considered in children requiring long-term central venous access. Particularly, research should be directed to understand the link between the specific medical conditions, type of infusates, type of CVAD material and difficult removal in children. In addition, it will be important to analyse the role of insertion techniques that might reduce endothelial trauma (e.g. micro-introduction kits), the specific vein used for insertion and the insertion technique (e.g. use of dilators, tunnelling etc.). It is crucial to develop larger-scale prospective studies with longer follow-up periods; improved CVAD surveillance will provide insight and guidance on reducing CVADs removal complications.
Footnotes
Acknowledgements
We would like to thank Dr Guineva JP Wilson, Dr Jessica J Zhang, Dr Richard Barnes and Miss Anne Spranklin for their support in providing us access to their case databases to identify eligible patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
