Abstract
Outcomes after out-of-hospital cardiac arrest (OHCA) remain poor in the UK. In order to increase the chances of successful resuscitation, international society guidelines on cardiopulmonary resuscitation quality have recommended titration of chest compression parameters and vasopressor administration to arterial diastolic blood pressure if invasive catheters are in situ at the time of cardiac arrest. However, prehospital initiation of arterial and central venous catheterisation is seldom undertaken due to the risks and significant technical challenges in the context of ongoing resuscitation in this environment. In 2019, a dedicated programme was started at East Anglian Air Ambulance (EAAA) to enable the safe introduction of contemporary emergency vascular access devices, in order to improve physiological monitoring intra-arrest and deliver nuanced, goal-directed resuscitation in OHCA patients. This programme was entitled Specialist Percutaneous Emergency Aortic Resuscitation (SPEAR). This article details the EAAA SPEAR technique; and the development, implementation and governance of this novel endovascular strategy in our UK physician-paramedic staffed helicopter emergency medical service.
Keywords
Introduction
Resuscitation is attempted by prehospital emergency medical services in approximately 30,000 out-of-hospital cardiac arrest (OHCA) patients per year in the UK. However, fewer than one in ten patients (9%) survive to hospital discharge. 1 Return of spontaneous circulation (ROSC) is the first step to long-term survival, but is only achieved in 40% of OHCA patients. 2 Hence, in order to improve outcomes in patients with refractory OHCA, strategies that increase the likelihood of ROSC in the prehospital phase should be investigated.
Coronary perfusion occurs primarily during diastole, or during the decompression phase of external chest compressions during cardiac arrest.3,4 This blood flow is driven by the diastolic pressure gradient between the proximal aorta and right atrium, developing a resultant coronary perfusion pressure (CPP). 5 Multiple animal and human studies, which measured CPP via invasive central venous and aortic catheters during cardiac arrest, have demonstrated that increased CPP is associated with ROSC and subsequent survival.6 –9 Therefore, a fundamental goal of cardiopulmonary resuscitation is to maximise CPP in order to achieve ROSC. 10
Various strategies have been investigated to augment CPP during cardiac arrest. Animal studies have been conducted where CPR depth and vasopressor administration was titrated in a goal-directed manner to achieve a CPP >20 mmHg. This demonstrated improved rates of ROSC and short-term survival compared to standard chest compression depth with non-titrated bolus dosing of vasopressors.9,11 Accordingly, American society guidelines on CPR quality have recommended titration of resuscitative efforts and use of CPP as a physiological target if invasive arterial/central venous catheters are in situ at the time of cardiac arrest. 4 However, this is not the case in OHCA. Prehospital initiation of central venous catheterisation and arterial pressure monitoring is seldom undertaken; it presents significant technical challenges in the context of ongoing resuscitation and is not without risk. Furthermore, most monitors used by prehospital teams cannot subtract arterial and central venous waveforms in order to display CPP in real-time. Therefore, intra-arrest measurement of CPP is generally not practical in the context of OHCA.
Despite this, direct measurement of the arterial component of CPP is possible during OHCA. Prehospital invasive arterial pressure monitoring has been shown to be feasible, safe and does not adversely affect scene times. 12 Furthermore, recent studies have demonstrated that femoral arterial access is feasible during ongoing resuscitation for cardiac arrest. 13 Femoral diastolic blood pressure (DBP) is highly congruent with aortic DBP (AoDBP) and generation of a higher femoral DBP has also been associated with an increased likelihood of ROSC.13 –17 Although the optimal DBP target is yet to be determined, studies have suggested that a DBP of at least 40 mmHg is required for successful resuscitation to occur.5,6,13
Femoral arterial access allows real time display of invasive blood pressure (IBP) and can also provide feedback on chest compression effectiveness via measurement of systolic blood pressure (SBP) and measure the effect of adrenaline administration on DBP – primarily achieved through peripheral vasoconstriction.5,18 –20 Other benefits include the early detection of ROSC (including low-flow states) with minimal interruption of CPR, swift recognition of re-arrest, access for arterial blood gas (ABG) analysis, and access for primary percutaneous intervention (PPCI) or mechanical circulatory support (MCS) following arrival at hospital.
A dedicated over-the-needle arterial cannula with flow-switch blood control (Becton, Dickinson and Company, New Jersey, USA) had been used for radial artery access for many years at East Anglian Air Ambulance (EAAA). However, these devices were not designed for intra-arrest use, and are not suitable for femoral arterial access, primarily due to their short length. Furthermore, it was recognised that, whilst gaining access to the femoral artery, femoral venous access was also easily achievable, and desirable in order to deliver a reliable route for venous administration of vasoactive medications.
In 2019, a dedicated programme was started at EAAA to enable the safe introduction of contemporary emergency vascular access devices in order to improve physiological monitoring intra-arrest and deliver nuanced, goal-directed resuscitation in OHCA patients. This programme was entitled Specialist Percutaneous Emergency Aortic Resuscitation (SPEAR). This article details the EAAA SPEAR technique; and the development, implementation and governance of this novel endovascular strategy in our UK physician-paramedic staffed helicopter emergency medical service (HEMS).
Setting
East Anglian Air Ambulance (EAAA) is a UK HEMS service that provides prehospital critical care to the statutory ambulance service in the East of England (East of England Ambulance Service NHS Trust (EEAST)). The East of England is a geographic area of 20,000 km2 and has approximately 6.4 million inhabitants. The HEMS system team consists of two teams each composed of a physician and critical care paramedic. They are dispatched from one of two bases (Cambridge and Norwich) by either helicopter or ground response vehicle, depending on patient location, weather and time of day. Each year EAAA is tasked to approximately 3000 primary missions and attends 2000 patients, of which approximately 25% are OHCA.
EAAA SPEAR technique
Rapid assessment phase
After arriving at an OHCA, a rapid assessment phase is undertaken by the HEMS team to understand the timing and circumstances of the arrest; and to ensure that essential treatments such as high-quality CPR (qCPR) and standard advanced life support (ALS) are in progress. Management of ALS is usually continued by or delegated to an appropriately trained EEAST team member. This allows the HEMS team to conduct their rapid assessment; and if required, assist with critical interventions such as intubation. Mechanical CPR (mCPR) is frequently initiated to provide consistent, optimal qCPR and to free-up team members. Intra-arrest insertion of SPEAR catheters is only considered if the OHCA remains refractory despite qCPR/ALS; and if the HEMS team feel that patient factors, OHCA circumstances and trajectory are favourable.
SPEAR phase
Preparation
Once the decision to undertake SPEAR has been made, the procedure must be delivered swiftly, in order to capitalise on its intended benefits and minimise scene time. SPEAR procedure packs are numbered in order and organised in a logical and sequential manner to minimise procedural time and to promote sterility (Table 1 and Figure 1).
SPEAR equipment list.


SPEAR bag and modules.
Despite the time-pressure, diligent preparatory steps are undertaken to reduce the risk of infection. These steps are summarised in Table 2. SPEAR catheters may remain indwelling in hospital, and sheaths can be accessed for interventional cardiology procedures or extracorporeal therapies. Responders on scene should be briefed on what is about to happen. This is essential to ensure the continuous delivery of qCPR/ALS whilst the HEMS team are task-focused on SPEAR; and protects the procedural area from accidental desterilisation by ambulance staff who may be unfamiliar with sterile procedures.
Preparation steps prior to SPEAR catheter insertion.

Ultrasound-guided femoral vascular access
A percutaneous, ultrasound-guided method for femoral arterial and venous access was favoured over blind/landmark and surgical cutdown techniques. However, in order to conduct ultrasound-guided procedures in the prehospital environment; a durable, portable, battery-operated ultrasound device was required. To aid in the delivery of SPEAR and other advanced resuscitative capabilities, a new point-of-care ultrasound (POCUS) device (Butterfly iQ™ Butterfly Network, Massachusetts, USA) was introduced. This has several advantages that help facilitate a prehospital ad-vanced vascular access programme. Firstly, it utilises a novel silicone-chip based technology in a single, portable, battery-operated probe; obviating the need for multiple transducers. In addition, the device’s software has several functions to aid vascular access beyond standard B-mode imaging. This includes simultaneous in-plane and out-of-plane imaging of the region of interest (Biplane Imaging™) and an overlay that enhances in-plane views of needles (Needle Vis™). Lastly, POCUS images and short video clips (obtained during vascular access) are recorded and stored in a secure cloud-based server, and retrospectively reviewed by prehospital consultants who provide feedback on image acquisition and optimisation. This allows for robust clinical governance, quality assurance of images, and continuous individual and institutional learning with POCUS and vascular access.
For SPEAR procedures, the sterile Butterfly iQ™ device is used to obtain a short axis view of the common femoral artery (CFA) and common femoral vein (CFV). Arterial access should be obtained via the CFA. A vessel entry point slightly cephalad of the femoral artery bifurcation is targeted to minimise the risk of vascular trauma, downstream ischaemia or thrombosis from cannulating the superficial femoral artery or a tributary vessel. This entry point may offer enhanced distal limb-flow, and possibly reduce thrombus formation due to stagnation around the sheath in a smaller vessel. Furthermore, to reduce the risk of non-compressible bleeding and potential retroperitoneal haematoma, a high puncture of the external iliac artery above the inguinal ligament must be avoided. The steps for ultrasound-guided femoral arterial access are summarised in Table 3. The same steps can be repeated to access the femoral vein if the administration of vasoactive medications is anticipated, or if reliable venous access is required. 5Fr sheaths (MERIT Prelude®, Merit Medical, Utah, USA) are used for both femoral arterial and venous access.
Steps for ultrasound-guided femoral arterial and venous catheter insertion.

ROSC phase
Once continuous arterial IBP monitoring is established, the effectiveness of qCPR can be assessed and compression position optimised to aim for an AoDBP of >40 mmHg. IBP can also be used to objectively assess for ROSC, including low-flow states, with minimal interruption to CPR. If the patient remains in cardiac arrest, 1 mg adrenaline boluses every 3–5 min are continued as per standard ALS guidelines. For patients who remain in ventricular fibrillation (VF) or pulseless ventricular tachycardia (pVT) despite three failed conventional defibrillation attempts, vector-change defibrillation may be attempted. An intravenous bolus of magnesium sulphate may also be administered in refractory VF/pVT as an adjunct to standard ALS doses of amiodarone.
However, if there is organised cardiac activity on POCUS and/or the presence of an arterial waveform on IABP monitoring identifies a low-flow state, then small adrenaline boluses (10–50 µg) or adrenaline infusion as per Figure 2 can be administered to aim for an AoDBP of >40 mmHg. Infusions are delivered using a compact, battery-operated Micropump (Micrel Medical Devices, Greece). The device is 10 times lighter than standard infusion pumps, which makes it highly advantageous for use in the prehospital arena.
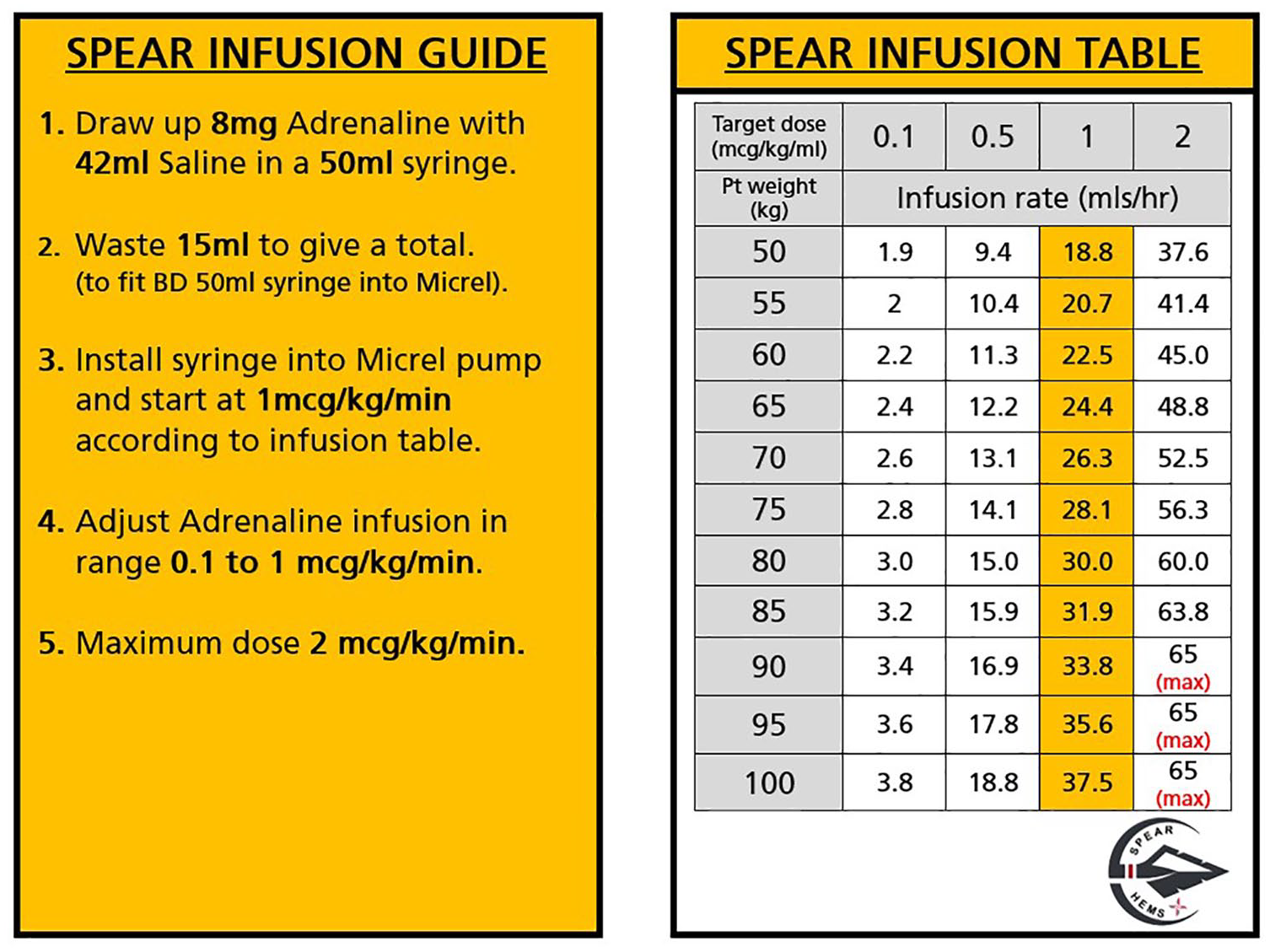
SPEAR infusion guide.
Once ROSC is achieved, adrenaline via bolus or infusion is titrated to individualised mean arterial pressure targets alongside standard neuroprotective post-resuscitation care. Interventions performed on scene are minimised to expedite transfer to definitive care. SPEAR devices can be accessed either on scene or en route to hospital for point-of-care arterial blood gas analysis (i-STAT 1, Abbott, Illinois, USA). These results can be used to guide ventilation strategy and identify potential electrolyte abnormalities. Resuscitation may be terminated if the patient remains in cardiac arrest, considering the clinical context, patient factors, POCUS findings and blood gas analysis if available.
When handing the patient over to in-hospital teams, attention is drawn to SPEAR devices, including highlighting which vessel was accessed and failed punctures. The latter will need to be monitored carefully if the patient receives antiplatelet, anticoagulant or thrombolytic medications. The SPEAR operator will also handover whether ‘full asepsis’ was achieved during insertion. Full asepsis is defined as an insertion following all the preparatory steps (Table 2) without desterilisation at any point during the procedure. This is important as SPEAR catheters may be left in situ or accessed for interventional procedures. If full asepsis could not be achieved on scene, this will be highlighted so the in-hospital teams can replace any intravascular devices at the earliest opportunity. Where immediate angiography or extracorporeal cardiopulmonary resuscitation (eCPR) is being performed, the 5Fr arterial and venous sheaths can be rewired with an 0.035-inch guidewire and upsized, as necessary.
Training and governance
The implementation of SPEAR was delivered in a cautious and step-wise manner. There was a recognition that SPEAR was novel, technically-difficult and required carefully considered application. Furthermore, a high-degree of prehospital experience and situational awareness was required to prevent distraction from standard resuscitative measures, task-fixation or unnecessary prolongation of scene times. An initial pilot phase was conducted, where SPEAR was only delivered by prehospital consultants with extensive experience of OHCA management and expertise in ultrasound-guided percuta-neous vascular access. Initial cases were meticulously reviewed to refine the technique. Following the successful pilot, a structured training programme was designed to expand SPEAR to the rest of the service.
SPEAR course
A 1-day training course, accredited by the Endovascular Resuscitation and Trauma Management Society (EVTM), was developed to incorporate theoretical and practical elements of SPEAR. Internal candidates from EAAA were enrolled onto the course provided they had suitable experience with vascular access techniques and after completing a minimum of 6-months of HEMS. The course was also opened to external candidates from both UK and international advanced resuscitation teams, who were interested in growing their emergency vascular access skills or building capability within their services. This allowed for sharing of experience and inter-service collaboration.
The course consists of three components. First, didactic lectures covering OHCA physiology and pharmacology; the recent literature on endovascular resuscitation; detailed vascular anatomy; and the full technical aspects of delivering SPEAR. Lectures are delivered by EAAA faculty and external experts in the field of endovascular resuscitation, both from the UK and other centres worldwide. Theory is then consolidated in a series of workshops on various practical aspects of SPEAR, including familiarisation with the SPEAR kit, POCUS/sonoan-atomy, and a masterclass in percutaneous ultrasound-guided Seldinger vascular access. The latter is taught using vascular access phantoms and a FemoraLineMan (Simulab Corporation, Seattle, Washington, USA) manikin for ultrasound-guided femoral arterial access. This device allows for visualisation and puncture of the femoral vessels using both short-axis or long-axis approaches. It also provides a good teaching platform for coaching on ‘micro-skills’ such as dilation and wire handling. Lastly, candidates undertake a series of moulages in simulated prehospital environments, including medical and trauma cases that require advanced vascular access. At the time of writing, EAAA have conducted three SPEAR courses training 45 clinicians. A post-course feedback survey has demonstrated that delegates found the course enjoyable, educational and the vast majority would recommend the SPEAR course to colleagues.
Following completion of the SPEAR course, EAAA clinicians undertake a series of supervised SPEAR cases with a SPEAR consultant, prior to being signed off as competent to independently perform femoral arterial and venous catheterisation. The exact number of cases required vary depending on the clinical background and prior experience with the technique. For example, physicians from in-hospital specialities who routinely conduct ultrasound-guided vascular access will require fewer supervised cases than clinicians who do not. Both paramedics and physicians can perform SPEAR at EAAA as part of an interchangeable operator model. A ‘finishing school’ for EAAA clinicians who have conducted SPEAR cases is currently in development.
Competency and credentialling
POCUS images and clips from vascular access cases are saved on the cloud-based imaging archiving system. These saved cases are used as a portfolio of evidence to ensure ongoing practice and competency. Clinical competency is recorded for clinicians on a secure database hosted on the EAAA intranet. Competency for SPEAR is confirmed every 3 months when clinicians must demonstrate knowledge of SPEAR indications and kit familiarity followed by a practical demonstration of ultrasound-guided SPEAR insertion using a vascular access phantom, the FemoraLineMan and simulated cases. Alongside the in-house SPEAR sign-off procedures, this portfolio is also used to fulfil the requirements of the Royal College of Emergency Medicine Level 1 accreditation for ultrasound-guided vascular access. This allows EAAA clinicians, if they have not already, to achieve a nationally recognised qualification that permits them to independently perform ultrasound-guided vascular access in emergency settings.
Governance
All SPEAR insertions are subject to a rigorous clinical governance process. Cases are automatically flagged for review in a ‘death and disability’ meeting as part of routine EAAA clinical governance. Identified learning points are highlighted for presentation and discussion at clinical governance days, which are open to the wider prehospital community. Furthermore, physiological data from SPEAR cases are recorded in real-time using time-calibrated monitors (X Series®, ZOLL Medical Corporation, Runcorn, UK) and uploaded at 30-s intervals to the service’s electronic patient record (HEMSbase, MedicOne Systems Ltd, UK). These data are interrogated retrospectively for each SPEAR case by the EAAA Research, Audit, Innovation and Development (RAID) team. Lastly, ultrasound images acquired during SPEAR insertion are reviewed within 24 h by an on-call prehospital consultant with a specialist interest in POCUS, and feedback provided on ultrasound/vascular access technique if required.
Future directions
Research is planned to analyse the success of SPEAR insertion, and its effect on clinical care, scene times and patient outcomes. Furthermore, continuous IBP measurements from SPEAR cases will be interrogated to develop an improved understanding of the haemodynamic effects of goal-directed CPR and titrated vasopressor therapy in low-flow states. This may help develop nuanced and haemodynamically-targeted OHCA algorithms, which move beyond protocolised regimens with a uniform position and depth of chest compressions and fixed interval bolus dosing of vasopressors to one which uses real-time feedback to enhance cerebral and coronary perfusion.
Conversely, a lack of effectiveness of these treatments may demand a change to resuscitation efforts in refractory OHCA. These include advanced endovascular techniques such as the mechanical augmentation of AoDBP using prehospital resuscitative endovascular balloon occlusion of the aorta (REBOA), or prehospital eCPR. Rapidly, safely and reliably securing femoral arterial access is a primary and essential step towards delivering these advanced endovascular techniques. Using our experience with SPEAR, our next step is to deliver the first pilot study of prehospital REBOA for patients with refractory, non-traumatic OHCA in the UK – the ERICA-ARREST trial (NCT06071910).
Footnotes
Acknowledgements
The authors would like to thank all individuals involved in the development of SPEAR, and to the HEMS teams providing care to patients at EAAA. The authors would also like to extend thanks to the EAAA Clinical Operations team and the Research, Audit, Innovation and Development team for their support with this project.
Abbreviations
AoDBP Aortic diastolic blood pressure
DBP Diastolic blood pressure
EAAA East Anglian Air Ambulance
eCPR Extracorporeal cardiopulmonary resuscitation
EVTM Endovascular Resuscitation and Trauma Management
HEMS Helicopter Emergency Medical Service
IBP Invasive blood pressure
NHS National Health Service
OHCA Out-of-hospital cardiac arrest
RAID Research, Audit, Innovation and Development
REBOA Resuscitative Endovascular Balloon Occlusion of the Aorta
SBP Systolic blood pressure
SPEAR Specialist Percutaneous Emergency Aortic Resuscitation
Author contributions
The first and subsequent drafts were written by Dr Shadman Aziz. Dr Jon Barratt and Dr Paul Rees led the development and implementation of the SPEAR project. The subsequent drafts were critically revised by Dr Kate Lachowycz, Dr Rob Major, Dr Jon Barratt and Dr Paul Rees. All authors approved the final draft. Dr Shadman Aziz is the guarantor of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
