Abstract
Background:
Vascular stenosis commonly leads to dysfunction in hemodialysis vascular access. Although percutaneous transluminal angioplasty is an established treatment, stent utilization has increased in the last decade as an alternative solution to extend the access function. This study evaluated the safety and initial results of a new impermeable covered stent for treating vascular access outflow stenosis.
Methods:
Investigators retrospectively analyzed 114 hemodialysis patients treated with polytetrafluorethylene-covered stents from September 2018 to September 2022 across four centers. Lesions treated were de novo or restenotic and located in the venous graft anastomosis, outflow segment, cephalic arch, and basilic swing point. Patients were followed by in-person physical examination at 1, 3, and 6 months, and Duplex ultrasound was performed to evaluate the vascular access circuit and in-stent restenosis. The primary efficacy endpoint was target lesion primary patency at 1, 3, and 6 months. Secondary endpoints included access circuit primary patency and secondary patency at 1, 3, and 6 months. The primary safety endpoint was freedom from local or systemic serious adverse events through 30 days post-procedure.
Results:
Forty-four patients had thrombosed access at the initial presentation, and 41 patients presented with recurrent stenosis. The target lesion primary patency rates at 1, 3, and 6 months were 100%, 89.4%, and 74%, respectively. The access circuit primary patency rates were 100% at 1 month, 85% at 3 months, and 62.7% at 6 months. The secondary patency rates at 1, 3, and 6 months were 100%, 96.4%, and 94.6%, respectively. In the adjusted multivariate Cox regression analysis, only recurrent lesions and female gender were associated with reduced primary patency rates. No serious adverse event was observed through the first 30 days post-procedure.
Conclusion:
In this retrospective analysis, a new covered stent was shown to be safe and effective for treating peripheral outflow stenosis in vascular access.
Introduction
In the United States, nearly 500,000 patients with end-stage renal disease receive maintenance dialysis treatments. 1 Brazil, the largest Latin American country, ranks third in the list of nations with the largest number of dialysis patients. 2 About 148,000 patients were on dialysis in Brazil in 2021, with 94% of them undergoing hemodialysis as their renal replacement therapy. 3
Three types of hemodialysis vascular access are currently used: autologous arteriovenous fistulas (AVF), arteriovenous grafts (AVG), and central venous catheters. AVFs remain the first choice for chronic hemodialysis due to their longer patency, lower complication rates, and reduced overall cost compared to AVGs and hemodialysis catheters.4–7 Despite their favorable outcomes, between 23% and 50% of AVFs show maturation problems,8–10 resulting in difficulties in achieving the appropriate vein caliber and flow for hemodialysis. In addition, both patients with AVFs and AVGs can develop stenosis and thrombosis associated with neointimal hyperplasia. 11 Venous stenosis is the primary cause of vascular access failure, and it occurs in the peri-anastomotic region and outflow segment of AVFs or at the graft-vein anastomosis of polytetrafluorethylene (PTFE) grafts. 9
For years, first-line treatment of AVFs stenoses has been percutaneous transluminal angioplasty, generally with high-pressure or cutting balloons. However, restenosis and reintervention rates associated with this technique remain incredibly high; only 26%–53% of AVFs still function without subsequent interventions 12 months after percutaneous transluminal angioplasty.12,13
The use of stent grafts (i.e. covered stents or endoprostheses) has gained significant interest as a primary treatment for failing AVFs. Studies have shown that stent grafts achieve more favorable patency rates than other treatments.14,15 The application of a stent graft provides protection against vein rupture and creates a physical barrier that may prevent the development of neointimal hyperplasia.16,17 Furthermore, stent grafts with improved flexibility can better adapt to the challenging anatomy of segments like the cephalic arch, thereby reducing the risks of stent migration, stent collapse, and in-stent stenosis. 18
Solaris, a new covered stent with an impermeable electrospinning PTFE wall, has the proposal to reduce the neointimal response and restenosis rates. The performance of this stent and the vascular response to it were first characterized in a porcine restenosis model. The stent exhibited resistance to fracture as well as increased flexibility, navigability, and better conformability to artery curvature compared to a commercially available PTFE-covered stent. The release system enabled precise geographical delivery, and the device produced a lower neointimal response than the control group. 19 These distinctive features make it an attractive device for effectively treating neointimal hyperplastic stenoses.
In this retrospective study, we evaluated the safety and effectiveness of Solaris® endograft in patients who had undergone angioplasty of the autologous AVF and AVG for vascular access dysfunction.
Material and methods
Patient selection
We performed a retrospective analysis of patients treated for hemodialysis vascular access outflow stenosis using Solaris® endoprostheses between September 2018 and September 2022 at four centers: (1) Centro Clínico Livcare (Niterói, Rio de Janeiro, Brazil), (2) Instituto de Angiologia e Cirurgia Vascular (Rio de Janeiro, Rio de Janeiro, Brazil), (3) Clínica Inteligência Vascular Avançada (Salvador, Bahia, Brazil), and (4) Dermavasc (Brasília, Distrito Federal, Brazil).
Patients included in the study underwent angioplasty of dysfunctional AVF or AVG access, had significant angiographic stenosis (luminal narrowing ⩾50%), clinical dysfunction (including access thrombosis), a target lesion’s length less than 8 cm, and a vessel diameter falling within the range of 4.5–8.5 mm. Individuals with associated central venous stenosis and lesions crossing the elbow were excluded from the analysis. During the study period, we reviewed data from electronic medical records to analyze the profile of patients, causes of access failure, access patency, need for reintervention, proper access functioning, predictors of procedure success, complication rates, and mortality rates.
This study was approved by the research ethics committee at the Department of Medicine, Universidade Federal Fluminense, Niterói, Rio de Janeiro, Brazil (protocol number 63527022.0.0000.5243). No written informed consent was required because of the retrospective nature of the study.
Study device
The stent used in all procedures was Solaris® (Scitech Medical©; Goiania, Goiás, Brazil). Solaris® is a flexible, self-expanding nitinol stent comprised of a thin, durable electrospinning PTFE membrane (Figure 1(a)). Electro-spinning is a method used to create nanoscale fibers. This technique arranges the fibers in a multidimensional and multidirectional pattern, resulting in a PTFE membrane with micropores measuring 1–2 microns in size. These pores are smaller than those found in expanded PTFE membranes used in other devices (15–30 microns). The PTFE membrane aims to cover and seal off diseased tissue with a higher multidirectional resistance force (the water pressure resistance of Solaris® is 180 vs 2–40 mmHg from competitors that use expanded PTFE).
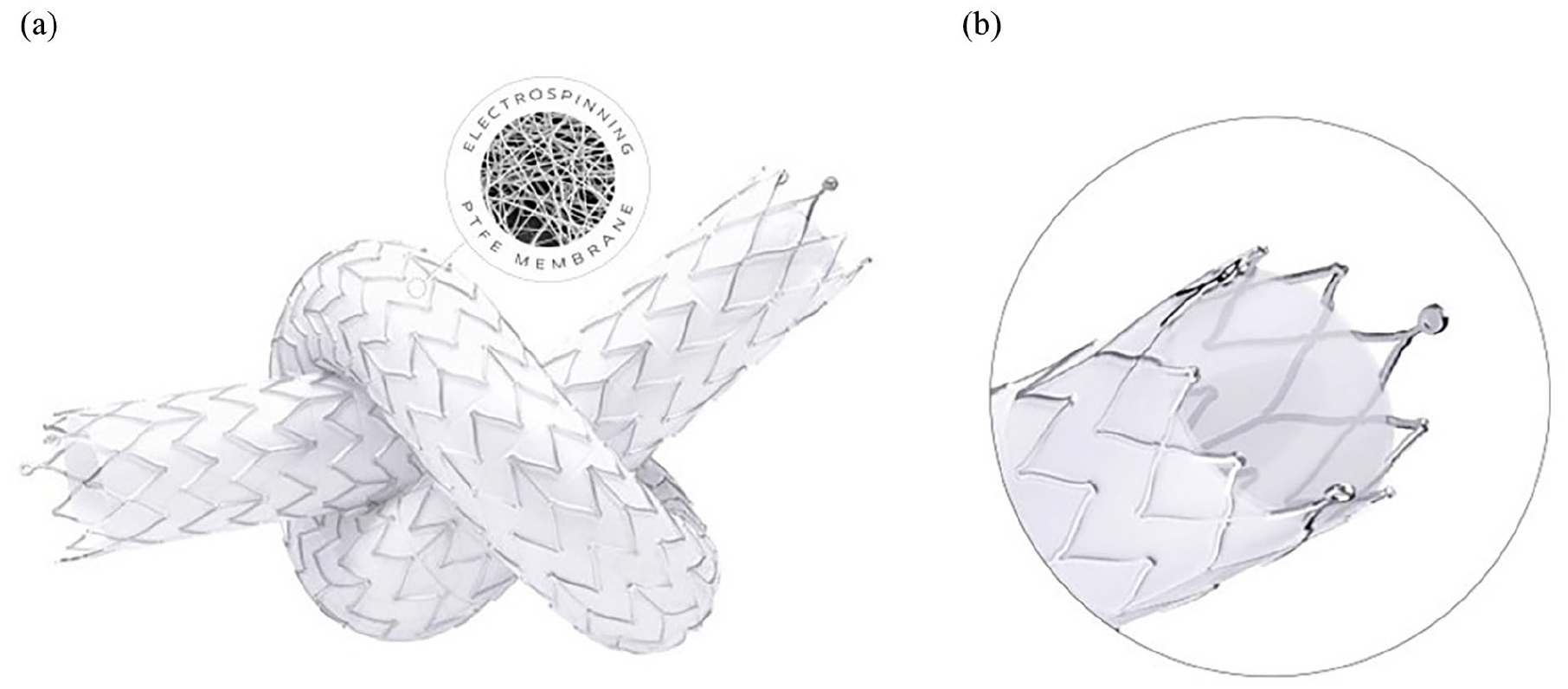
Study device: (a) SOLARIS multidirectional, impermeable electrospinning PTFE membrane and (b) three tantalum marker bands (distal/proximal).
The device has a pullback delivery system that is easy to operate. The operator must maintain the position of the stent graft by firmly holding the end of the delivery system and retract the sheath that encapsulates the crimped stent. Solaris® has been available in Brazil for the treatment of arterial peripheral disease, venous and vascular access stenosis since 2016.
The stent grafts come in diameters of 5–9 mm and lengths of 40, 60, 80, and 100 mm (limited to straight configuration), enabling the treatment of vessels ranging from 4.5 to 8.5 mm in diameter. The delivery catheter was designed with a 0.035-inch guide wire platform and can be introduced through either an 8F or 9F sheath, depending on device size. The device has three radiopaque tantalum markers positioned within the final 5 mm of the bare metal edges, providing a good radiopacity (Figure 1(b)).
Study treatment and follow-up
All procedures were conducted by four vascular surgeons under local anesthesia and sedation. Surgeons accessed the access circuit via the femoral vein in cases involving the treatment of graft-vein anastomosis without thrombosis and in situations where there was no vein segment with at least 9 mm in the AVF for access. This approach prevents stenosis in the site of the access after the sheath removal and close of the site puncture with a hemostatic stitch.
In cases with thrombosis at initial presentation, the thrombectomy with complete flow restoration was performed before any angioplasty procedure. If the thrombectomy was performed through open surgery, the access for performing the outflow stenosis angioplasty was done using the same vein segment used for the thrombectomy procedure. If an endovascular procedure was chosen for the thrombectomy, the access for the outflow stenosis angioplasty was performed in the same site used to perform the thrombectomy.
The stenosis was crossed using a 0.035 hydrophilic guidewire and pre-dilated with a high-pressure non-compliant balloon (type and size chosen by the operator). The balloon was inflated to a pressure sufficient for full expansion of the pre-dilation balloon and to efface the lesion. There was no specific recommendation regarding the balloon inflation time. After pre-dilation and angiography reassessment, patients underwent the implantation of the stent graft.
To ensure adequate fixation and vessel wall apposition, devices were deployed in a 10%–25% oversized configuration in the distal segment of the stenosis (vs adjacent healthy vessel) with at least 1 cm overlap with the healthy vessel or synthetic graft. Subsequently, the devices were post-dilated with a balloon that did not exceed the stent graft diameter. If multiple devices were deployed in an overlapped configuration, post-dilation of the first device did not occur before deploying the second and the difference between the overlapped stent graft diameters was 1 mm. Additionally, the smaller device was deployed first if different-sized devices were overlapped to accommodate vessel diameter changes. In the treatment of the venous graft anastomosis stenosis, the stent graft diameter was 1 or 2 mm larger than the diameter of the PTFE used in the AVG and its length should cover the entire stenosis. Moreover, surgeons ensured the stent graft was situated in a vein with a diameter greater than that of the device, thereby avoiding contact of the stent with the vein wall. Post-procedure, all patients were administered daily aspirin (100 mg) and clopidogrel (75 mg) for 1 month, followed by aspirin as a maintenance regimen thereafter.
Physical examinations and a Duplex ultrasound were scheduled at 1, 3, and 6 months after the procedure, according to the routine protocol of the centers, with extra visits as needed to address access circuit dysfunction. These follow-up times were selected to facilitate analysis over the critical period when current interventions fail. Physical examinations and dialysis parameters included, at minimum, an assessment of thrill, prolonged bleeding, high venous pressure, pulsatility, difficult needle cannulation, low Kt/V achievement, hand, arm, neck, and trunk edema, as well as evaluations for pain related to the dialysis circuit, respiratory and neurological symptoms, skin changes, and any adverse effects.
The objective of the Duplex ultrasound was to evaluate the volume flow in the brachial artery, the vascular access circuit, and in-stent restenosis. Evaluating the circuit with Duplex ultrasound enhances the reliability of patency rate analysis because sometimes there might be restenosis in the target lesion without noticeable symptoms, which would not be accounted for in the target lesion primary patency loss analysis. Restenosis on the Duplex ultrasound was determined by assessing either of the following criteria: when the diameter reduction calculated at the B Mode ultrasound among the narrowed segment and the adjacent normal vessel was greater than 50% or the Peak Systolic Velocity (PSV) ratio calculated by dividing the PSV at the stenosis by the PSV obtained 2 cm caudal to the narrowed segment was two or more.
Study endpoints
Clinical results were retrospectively assessed for 6 months following the initial stent placement. The primary study endpoint was target lesion primary patency at 1, 3, and 6 months and freedom from local or systemic serious adverse events through 30 days. The secondary study endpoints included access circuit primary patency and target lesion secondary patency at 1, 3, and 6 months.
Target lesion primary patency comprises the time between stent placement and any repeat intervention within the stent-implanted region (i.e. stent-implanted region ± 5 mm on either edge) or access circuit thrombosis, as previously described. 20 Access circuit primary patency was defined as the time between stent placement and the first intervention anywhere in the access circuit or access circuit thrombosis. 20 Target lesion secondary patency comprises the time from initial stent placement until the graft or AVF was abandoned. 20 For stenoses without thrombosis, anatomic success was defined as <30% residual diameter stenosis of the treated vascular segment. 21 For thrombosed accesses, anatomic success was defined as restoration of flow combined with a <30% residual diameter stenosis for any significant underlying stenosis. 21 Clinical success refers to the resumption of normal hemodialysis, and procedural success was defined as anatomic success plus at least one indicator of hemodynamic or clinical success. 21
Statistical analysis
Kolmogorov-Smirnov was used to test for the distribution of variables. Variables with normal distribution were expressed as mean ± standard deviation, otherwise as median and interquartile ranges. Survival analysis was performed for patency as a Kaplan–Meier survival curve and, when applicable, compared by using the log-rank test. The hazard ratios for patency loss were estimated by Cox proportional hazards regression. Initially, univariate analysis was performed for each demographic, clinical, and anatomical variables. Subsequently, only those variables that showed p-values <0.20 in the univariate assessment were included in the multivariate model. p-Values <0.05 were considered statistically significant. Data were analyzed using SPSS software version 18.0 for Windows (IBM, USA).
Results
One hundred fourteen patients fulfilled the inclusion criteria and underwent a retrospective review as part of the study.
Patient and lesion characteristics
Table 1 details patient demographics, concurrent medical conditions, and dialysis access circuits.
Baseline patient demographics and details of the dialysis access circuits (n = 114).
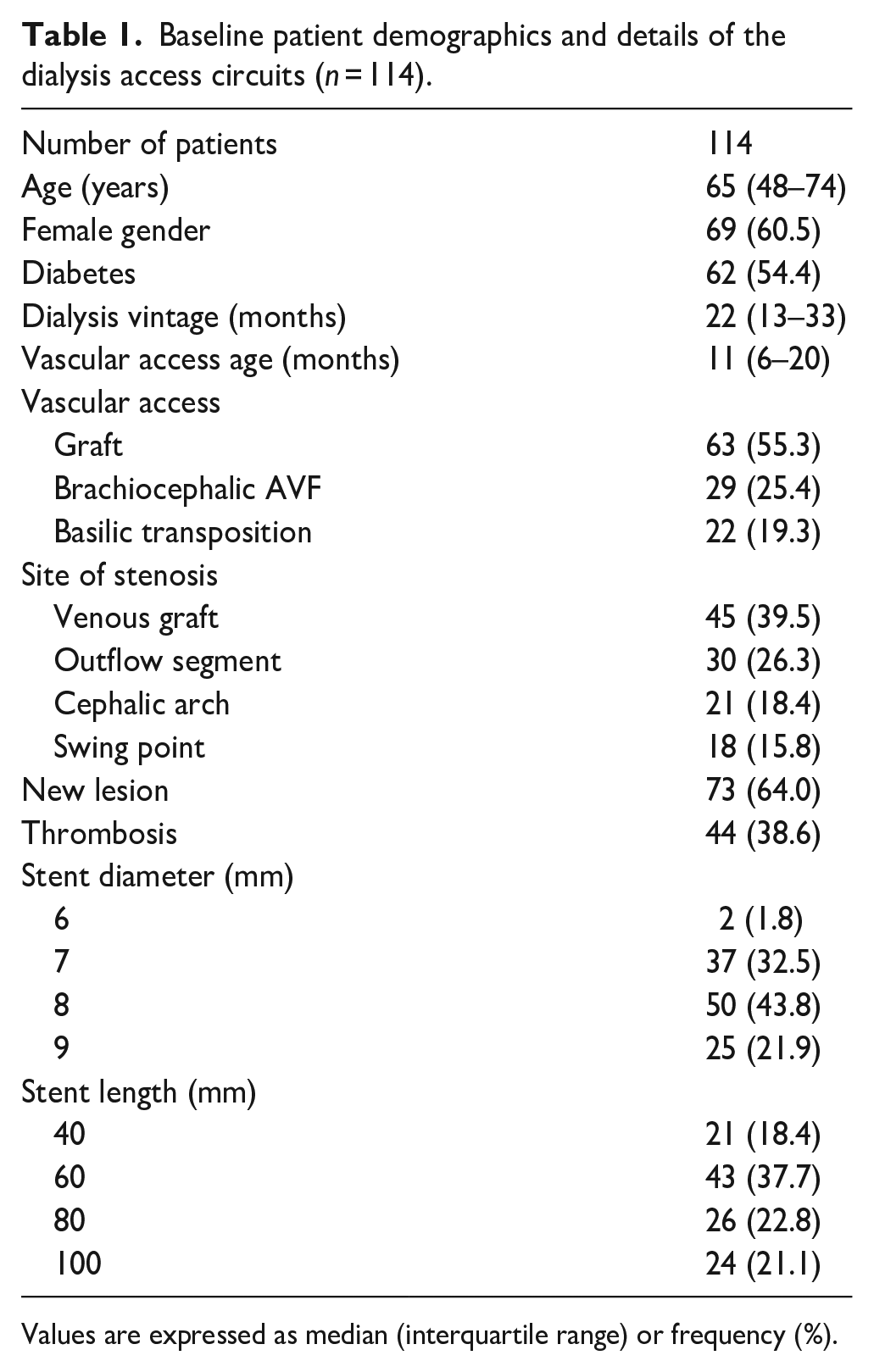
Values are expressed as median (interquartile range) or frequency (%).
The mean age of patients was 65 ± (48–74 years), and 61% were female. Significant comorbidities included hypertension (81%), diabetes (54%), and coronary heart disease (21%). Forty-four patients (39%) had thrombosed access at the initial presentation. Forty-one patients (36%) presented with recurrent stenosis (lesions that had undergone prior treatment), and 73 (64%) with de novo lesions. Stenotic lesions were located in the graft-vein anastomosis (39.5%), outflow segment (26.3%), cephalic arch (18.4%), and basilic swing point (15.8%). The types of access treated were AVG in 55.3% of patients, brachiocephalic fistulas in 25.4%, and brachiobasilic AVF in 19.3% of patients. The procedures were performed through the femoral vein in 58 patients (51%) and via anterograde access directly to AVF or AVG in 56 patients (49%).
Procedural and safety outcomes
Anatomical, clinical, and procedural success were achieved in all 114 patients. Figure 2 shows the procedural outcomes of an example case. No local or systemic serious adverse event was observed through the first 30 days post-procedure.
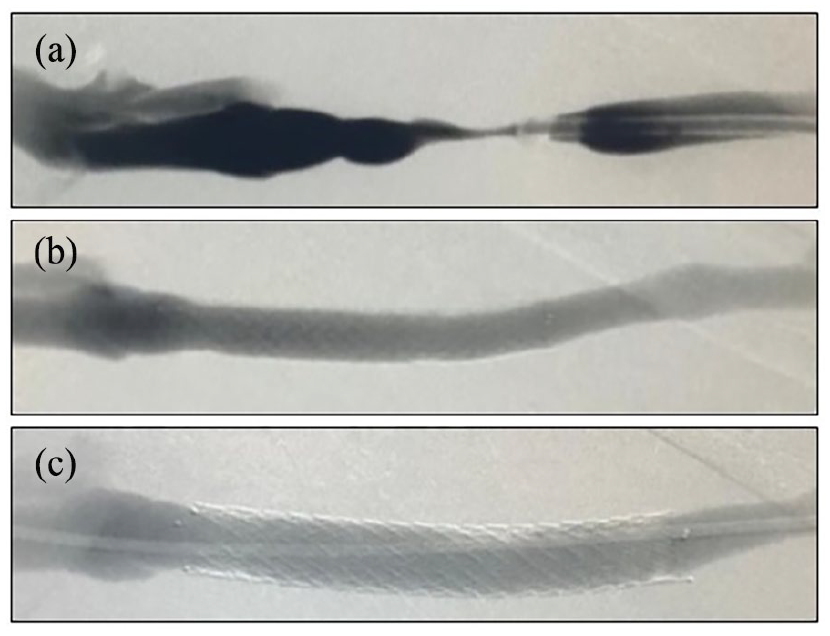
Example of procedural outcomes in a patient with brachiobasilic AVF with stenosis at the swing point: (a) angiography of a basilic vein transposition with severe stenosis at the swing point identified during a thrombectomy procedure, (b) final angiography after implantation of the Solaris 9 × 60 mm coated stent (stent graft), and (c) control angiography 7 months after the initial procedure without evidence of restenosis.
Performance outcomes
Figure 3 demonstrates patency for the study endpoints displayed as Kaplan–Meier survival curves over the 6-month follow-up period.
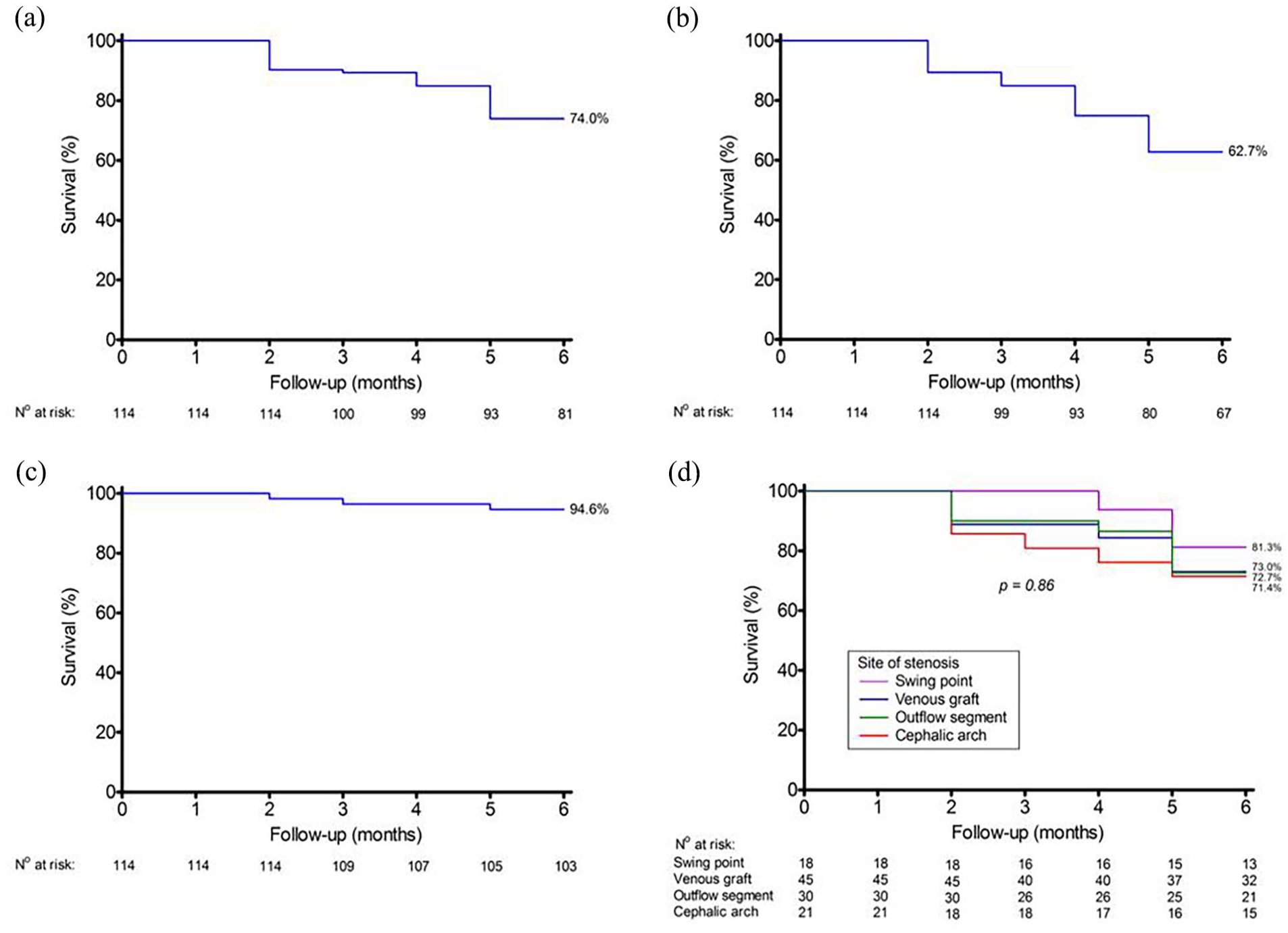
(a) The target lesion primary patency rates were 100% at 1 month, 89.4% at 3 months, and 74% at 6 months, (b) the access circuit primary patency rates were 100% at 1 month, 85% at 3 months, and 62.7% at 6 months, (c) the target lesion secondary patency rates at 1, 3, and 6 months were 100%, 96.4%, and 94.6%, respectively, and (d) target lesion patency by segments 6 months after stent implantation.
The target lesion primary patency rates (Figure 3(a)) at 1, 3, and 6 months were 100%, 89.4%, and 74%, respectively. The access circuit primary patency rates (Figure 3(b)) were 100% at 1 month, 85% at 3 months, and 62.7% at 6 months. The target lesion secondary patency rates (Figure 3(c)) at 1, 3, and 6 months were 100%, 96.4%, and 94.6%, respectively.
During the initial 3 months of follow-up, 17 patients required reintervention to maintain the access circuit patency (12 patients with restenosis at the target lesion and 5 with new lesions in the cannulation zone or inflow site). At 6 months, 41 patients underwent new procedures to maintain access functionality (28 patients with restenosis at the target lesion and 13 with a non-target lesion in the cannulation zone or inflow site). In all cases of target lesion restenosis, it occurred at the stent graft’s edge. Six patients had their access abandoned during the study period due to failure to recanalize the non-target lesion (n = 3), impossibility of performing thrombectomy (n = 2), and failure to recanalize the target lesion (n = 1).
As shown in Figure 3(d), we did not observe differences between segments in the target lesion patency rates 6 months after stent implantation (basilic swing point = 81.3%, venous graft anastomosis = 73%, outflow segments = 72.7%, and cephalic arch 71.4%, p = 0.86).
In multivariate Cox regression analysis with adjustment for gender, age, diabetes status, size and diameter of the stent, AVF site, thrombosis at initial presentation, recurrent lesions or stenosis sites (Table 2), only new lesions with a hazard ratio of 0.44 (95% CI, 0.21–0.93; p = 0.030) and female gender with a hazard ratio of 3.79 (95% CI, 1.31–10.93; p = 0.014) were associated with better and reduced primary patency rates, respectively.
Cox regression analysis for prediction of 6-month vascular access primary patency failure.
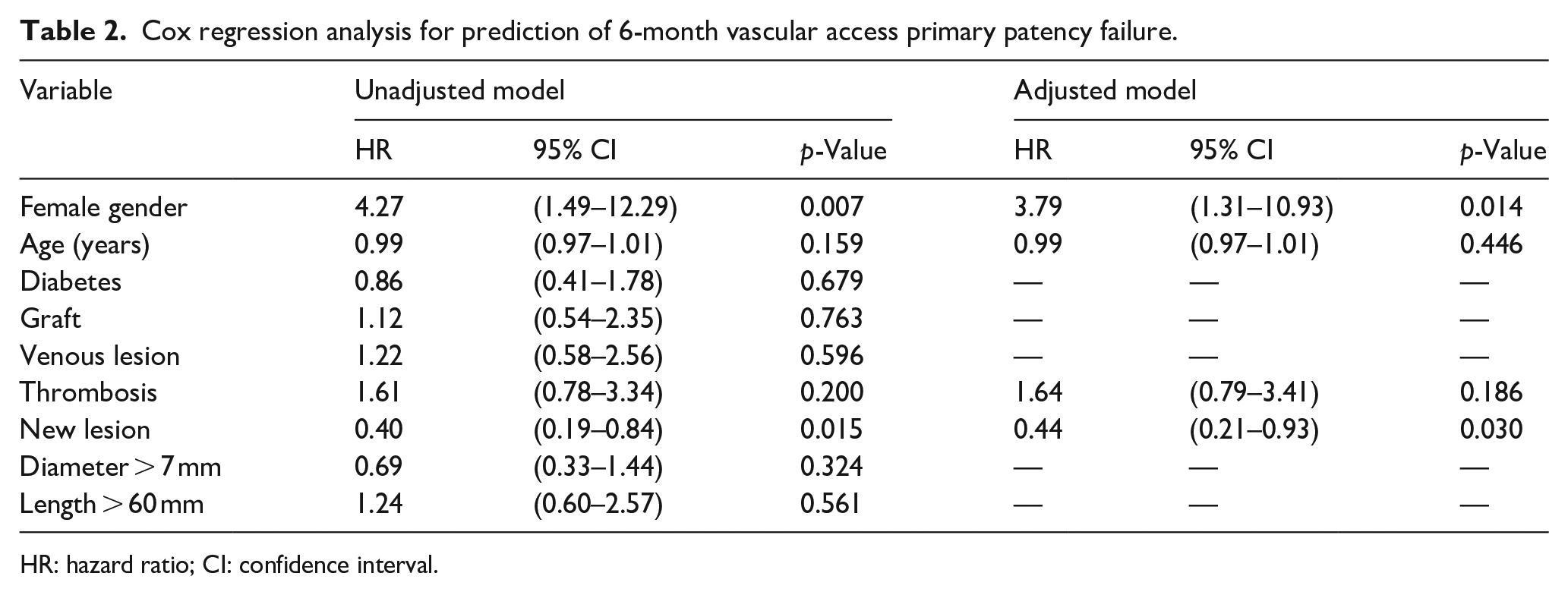
HR: hazard ratio; CI: confidence interval.
Discussion
The present study demonstrated favorable results with the use of a self-expanding covered stent to treat hemodialysis vascular access outflow stenosis. The procedural and safety outcomes were excellent, with 100% achievement of anatomical, clinical, and procedural success. No deaths related to the device or procedure and no serious adverse events, either local or systemic, were observed during the 6-month post-procedure, indicating the safety and feasibility of this approach. Five deaths occurred during the study period, all attributed to cardiovascular causes.
In our study, we analyzed 114 patients treated with the stent and observed a target lesion primary patency rate of 74% at 6 months. This rate not only surpasses the 50% goal set by the K/DOQI guidelines 22 but also outperforms the patency rates reported in studies using percutaneous transluminal angioplasty 20 or other types of stent grafts.20,23,24 Similarly, the access circuit primary patency rate was 62.7% at 6 months, indicating good long-term patency of the vascular access circuits. Among patients who lost target lesion patency, recurrences occurred at the edge of the stent graft. This suggests the effectiveness of the multidirectional electrospinning PTFE membrane in preventing trans-graft cellular migration and restenosis within the graft, as previously demonstrated by Tellez et al. 19
After 6 months of stent implantation, we observed no significant differences in the patency rates of target lesions across the segments evaluated (venous graft anastomosis, basilic swing point, cephalic arch, and outflow segments). This similarity in patency rates across segments demonstrates the consistent performance of the studied stent and its ability to maintain target lesion patency irrespective of the specific segment where it is deployed.
While a more in-depth analysis is necessary to achieve an accurate comparison, it is worth noting that the patency rate of Solaris® in the cephalic arch (71.4%) appears comparable to the results obtained by Jones et al. 25 and Miller et al., 17 where the Viabahn® stent graft achieved 67% and 74% 6-month target lesion primary patency in arteriovenous access. The 6-month target lesion primary patency rate of the stent in the outflow segment (72.7%) achieved in the present study is also comparable to results reported in previous studies evaluating covered stents.26,27
In the venous graft anastomosis segment, our study revealed a target lesion primary patency rate of 73% at 6 months. This rate is similar to the one reported by Dolmatch et al. 28 for the Covera® vascular covered stent and exceeds the patency rates reported for Viabahn®24 and Flair® stent grafts. 20 In the basilic swing point, our study achieved a target lesion primary patency rate of 81.3%, which compares favorably to the rate reported by Nassar et al. 23 In their study, stent grafts (Flair®, Fluency®, and Viabahn®) achieved a 57% 6-month target lesion primary patency. These findings suggest that Solaris® performs well compared to other devices in both the venous graft anastomosis and basilic swing point segments. Such positive results may benefit patients requiring vascular access interventions in these areas.
In our analysis, diabetes status, size and diameter of the stent, AVF site, and thrombosis at initial presentation did not negatively affect the 6-month patency of the stent graft. Thrombosis is usually associated with worse target lesion primary patency rates after angioplasty. 29 However, in our analysis, thrombosis at the initial presentation was not associated with a significantly higher risk of patency loss. Maybe the immediate thrombectomy protocol (before 24 h) used at the centers can avoid endothelial damage and justify these findings, or simply our sample size did not have statistical power to demonstrate it.
Conversely, having recurrent lesions and being female was associated with a higher risk of graft patency loss within 6 months. In the present study, 6-month primary patency rate in recurrent lesions was 60.3% compared with 81.7% in de novo stenosis (p = 0.013). Similarly, the 6-month primary patency rate in female patients was 64.4% versus 88.6% in male individuals. Patients with recurrent lesions often have preexisting vascular diseases, such as intimal hyperplasia, calcification, and increased vascular stiffness.30–32 These lesions are usually hyper-reactive and have worse patency rates than de novo lesions. 33 These conditions can subject the stent graft to increased stress and strain, potentially compromising its integrity over time and leading to decreased patency rates. Women are often smaller in stature than men and have veins with smaller diameters. Moreover, males and females have differences in vascular physiology and hormonal variations. 34 These anatomical and physiological differences might contribute to the decreased target lesion primary patency rate in women. However, further research is needed to fully understand sex differences in the performance of stent grafts.
Our study has limitations due to its retrospective design. One major limitation is the lack of a standardized protocol for various aspects of the intervention, including stenosis measurement and quantification of arteriovenous access dysfunction. Additionally, the decision to use stent grafts was at the interventionalist’s discretion, without predefined indications. The study design and lack of standardized criteria for intervention make it difficult to compare our results with other AVFs salvage methods. A randomized study would be necessary to determine patency differences between different salvage methods within the same demographic population. Furthermore, it would be valuable to conduct future studies comparing the effectiveness of stent grafts and drug-coated balloons delivering paclitaxel directly at the angioplasty site for treating dysfunctional AVFs. This is especially important in light of encouraging results demonstrating that drug-coated balloons have superior patency compared to standard percutaneous transluminal angioplasty, particularly in cases of stenosis in the AVF anastomosis and within the cephalic vein arch.35,36
Conclusion
We observed high technical and clinical success when using Solaris®, a new PTFE-covered stent, to treat hemodialysis vascular access outflow stenosis in 114 patients. The procedure was safe, and the patency outcomes were comparable to or even better than those reported for other covered stents. While more standardized and prospective research is required to draw definitive conclusions on the efficacy of this stent, our findings suggest that it holds promise as an option for treating peripheral outflow stenosis in vascular access.
Footnotes
Acknowledgements
The authors thank Morgana Moretti, PhD, for providing medical writing assistance during the elaboration of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.O.H. reports receiving research grants from Merit Medical Systems and Bard BD and consulting or lecture fees from Medtronic, Bard BD, Merit Medical Systems, Scitech Medical©, and Gore. T.A.B., M.G.F., and L.C.A. report receiving research grants from Merit Medical Systems, Scitech Medical©, and Bard BD, as well as consulting and lecture fees from Medtronic, Bard BD, Merit Medical Systems, Scitech Medical©, and Gore. J.P.S.M. received consultancy fees from Fresenius Medical Care Brazil.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the article publication was provided in part by the sponsor, Scitech Medical©.
