Abstract
Background:
Guidewire-facilitated access to peripheral vessels is commonplace in vascular access, but guidewire insertion into small vessels, such as the radial and distal radial arteries, can still be challenging. Failure to gain access on the first attempt may contribute to increased risks of procedural complications, such as vessel dissection, spasm, and occlusion. This research assessed the safety and efficacy of radial and distal radial artery access using a novel, FDA-cleared, small-core-diameter guidewire with an articulating tip, under ultrasound guidance.
Methods:
This was a prospective, single-arm, single-center trial. Patients in need of vascular access were screened for participation and enrolled in the study. Guidewire insertion was attempted by four physicians (three interventional radiologists and an interventional nephrologist) at 162 arterial sites—65 radial and 97 distal radial, having a mean diameter of 2.0 mm.
Results:
First-attempt successful placement of the guidewire in the artery occurred at 87.6% of access sites (142/162) and differences in the success rate between the radial and distal radial arteries or between vessels with diameter smaller or larger than 2 mm were not observed (62/68 and 67/77, respectively; p = 0.6). Four of the five reported adverse events were unrelated to the study device or procedure. Two of the three distal radial artery spasms occurred before the guidewire was used. The other two events were a radial artery spasm, and a distal radial artery site hematoma. All adverse events resolved spontaneously.
Conclusions:
First-attempt placement of a novel articulating tip guidewire in the radial and distal radial arteries occurred at a high rate in our study and was not associated with safety concerns.
Introduction
Compared to the original femoral, the now-veteran radial 1 and the recently-pioneered distal radial 2 access have proven to be safer and more cost-effective access sites for transluminal diagnostic and interventional procedures.3–7 However, devotees of radial and distal radial access, experienced and inexperienced alike, 8 face more challenges, due to the smaller size of the vessels, compared with the femoral artery. One of the most common complications of radial artery access is radial artery occlusion (RAO).4,9,10
Distal radial artery (DRA) access is safer than radial artery access, is associated with lower time to hemostasis, and reduces incidence of RAO, but i also considered more challenging technically and shows a tendency for a greater number of access attempts, lower cannulation success rate, and a greater chance for crossover to an alternate route.11–17
Ultrasound guidance may improve the overall success rate of radial cannulation and reduce the odds of failure to gain access on the first-attempt,8,18 the latter being a globally recognized risk factor for vessel spasm.19–21 However, even with ultrasound, the first-attempt success rate may still be as low as 52%−65%.8,18
Vessel tortuosity and angulation of radial and distal radial arteries are known to negatively affect the access success rate22–24 and may contribute to occurrence of vessel perforation, hematoma, and compartment syndrome.25–27
Access guidewire is one of the determinants of success in both gaining access to a vessel and the frequency of preventable and ameliorable adverse events. Forcefully advancing the guidewire may result in spasm 28 or subintimal dissections of the accessed vessel, especially if the tip is too stiff or the vessel is tortuous or atherosclerotic. Broken tip of a guidewire can embolize downstream. 29
This study aimed to explore, prospectively, the safety and effectiveness of vascular access to radial and distal radial arteries utilizing a novel, small-core-diameter guidewire (TalWire; Embrace Medical Ltd., Tel Aviv, Israel) featuring an articulating tip envisioned to be deflected upon contact with the vessel wall or intraluminal obstacles. The tip articulation is facilitated by a structural element located 0.7 mm from the distal end of the guidewire, allowing this part of the guidewire tip to flex, upon contact with obstacles, in any direction and up to 180° relatively to the guidewire shaft axis. This functional attribute may be valuable, especially when securing vascular access in small arteries, by increasing the probability of guidewire placement and reducing the risk of vessel trauma. Figure 1 shows the TalWire schematics and showcases its functionality.
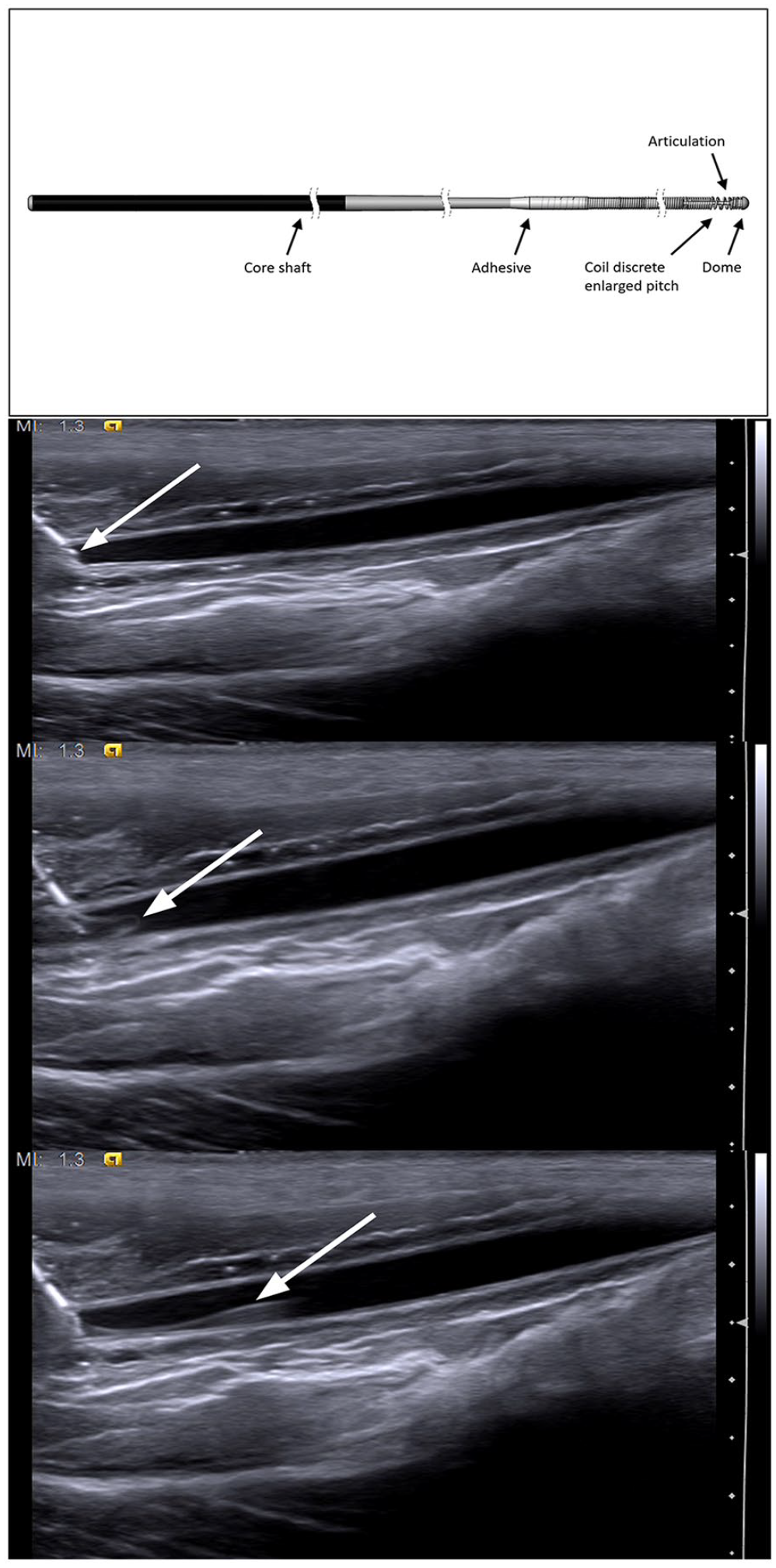
Top to bottom: TalWire structure; needle in the vessel (white arrow—needle tip); guidewire entering the vessel (white arrow—guidewire tip articulation); guidewire in the vessel (white arrow—guidewire).
Methods
This was a prospective, single-arm, interventional study conducted in line with the principles of the Declaration of Helsinki in a single center in the Dominican Republic. Ethics committee approved the research protocol. Informed consent was obtained from all participants.
Subjects
Enrollment was open to patients requiring vascular access. We included men and women, 18 years of age or older, referred to and scheduled for a procedure which required vascular access. We excluded pregnant or lactating women and patients with bleeding disorders.
Investigational device
TalWire is a novel, FDA-cleared, mandrel guidewire, 40–80 cm in length and 0.018 or 0.021 inch in diameter, with a proprietary articulating distal tip. The guidewire solid core shaft is made of Nitinol, with a ground tapered section at the distal end of the guidewire. A stainless-steel or tungsten micro-coil is wound with a lumen that is then placed over the tapered distal section.
Investigational procedure
Placement of the investigational device was attempted through a 21- or 22-G standard metallic entry needle (Cook Medical LLC, Bloomington, IN, USA and Terumo Medical Corporation, Shibuya City, Tokyo, Japan) under real-time B-mode ultrasound guidance (Q9 or ECO 3 EXPERT (CHISON Medical Technologies, Jiangsu, China) with Sonostar L6C linear wireless ultrasound transducer (Sonostar Technologies Co., Ltd. Guangzhou, China)). No intra-arterial spasmolytics or anticoagulants were used. Intravascular position of the guidewire was confirmed with ultrasound. Participation in the study ended after the guidewire was inserted and removed uneventfully or when its insertion was deemed unsuccessful by the investigator.
Outcomes
The performance outcome reported in this publication is the first-attempt guidewire insertion success rate.
Safety outcomes included the incidence of the device- and procedure-related adverse events (AEs), in general, and of vessel spasms, in particular, and incidence of device deficiencies.
Safety analysis
All AEs, whether considered related or unrelated to the study device and/or procedure, were reported and followed until resolution or until deemed resolving by the investigator. AEs were coded to the Medical Dictionary for Regulatory Activities (MedDRA) version 24.1, after consulting the International Medical Device Regulators Forum (IMDRF) IMDRF terminologies for categorized Adverse Event Reporting (AER): terms, terminology structure and codes Annex E (Health Effects – Clinical Signs, Symptoms and Conditions Terms and Codes). AEs were mapped to (where possible) and graded in accordance with the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Version 5.0.
Statistical analysis
Safety analysis was conducted on the intention-to-treat (ITT) dataset comprising all consented subjects. Efficacy analyses were conducted on the modified ITT (mITT) dataset comprising all access attempts during which the guidewire insertion was initiated, in all consented subjects. Continuous variables were summarized by a mean, standard deviation, minimum, and maximum, and categorical variables by a count and percentage. Fisher’s exact test calculating two-sided p value was used for analysis of potential risk factors for failure. Statistical analysis was performed with IBM SPSS Statistics version 29.0.0 (IBM, Armonk, NY, USA).
Results
Following the institutional ethics committee approval, 65 (23 female and 42 male) consecutive patients requiring vascular access were enrolled between November 2021 and March 2022. The mean age was 57 years (SD, 12.2; range, 29–83). Demographic data and medical history of the participants are shown in Table 1. Procedures were performed by four operators – three interventional radiologists, one of which had 15 and the other two – 20 years of experience, and one interventional nephrologist, with 8 years of experience. Only one of these (the interventional radiologist with 15 years of experience) had performed radial and distal radial access before this study.
Summary demographics and medical history.
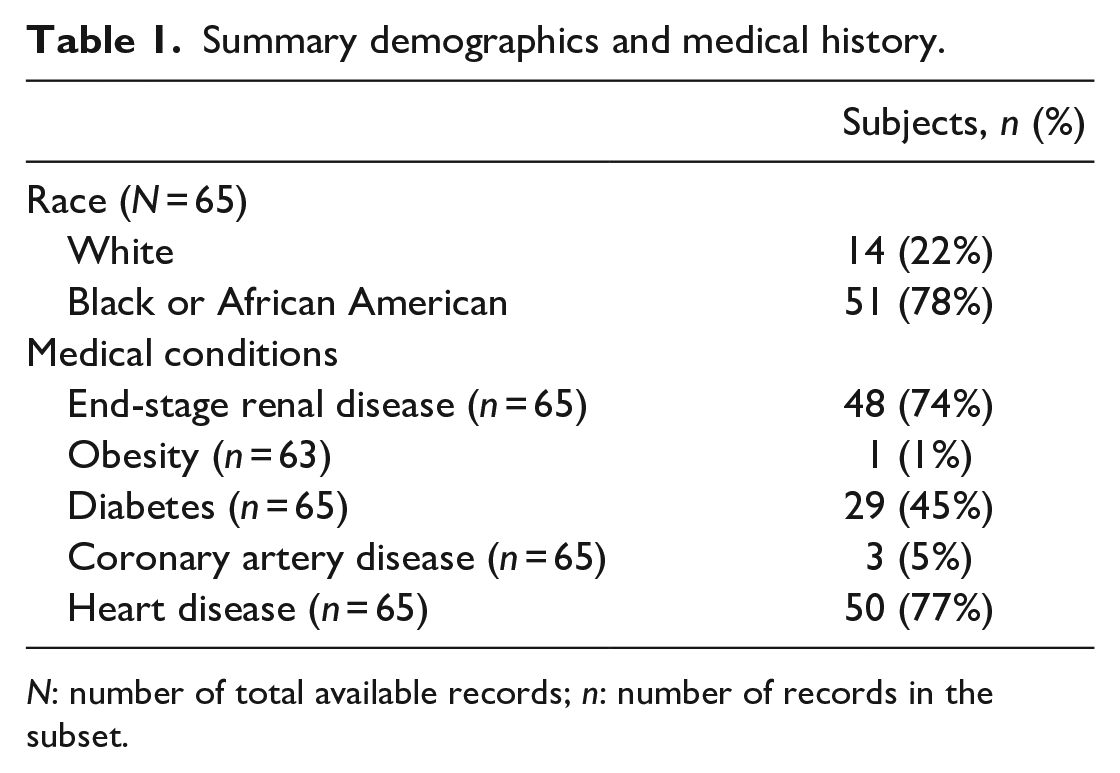
N: number of total available records; n: number of records in the subset.
Guidewire insertion was initiated in all participants, in at least one arterial site, during the study. Overall, we attempted to place the investigational device in 162 arterial sites, 107 of these in male and 55 in female participants. Of the 162 sites, 65 were radial (28 left and 37 right) and 97 distal radial (48 left and 49 right).
Efficacy
Guidewire placement on first attempt was achieved at 142 of the 162 (87.6%) of the radial and distal radial artery sites with mean diameter of 2.0 mm, where measurement was available (SD = 0.37; n = 145). We did not observe any discernible difference in the first attempt success rate between male and female participants (Table 2). There were also no statistically significant differences between the nominal values of this outcome for different target vessel types (radial vs distal radial), size (<2 vs ⩾2 mm), or laterality of access. Similarly, no differences were found in the assessment of significance of access laterality and participant’s gender when radial and distal radial sites were addressed separately (Table 3).
Overall percentage and rate of first attempt success.
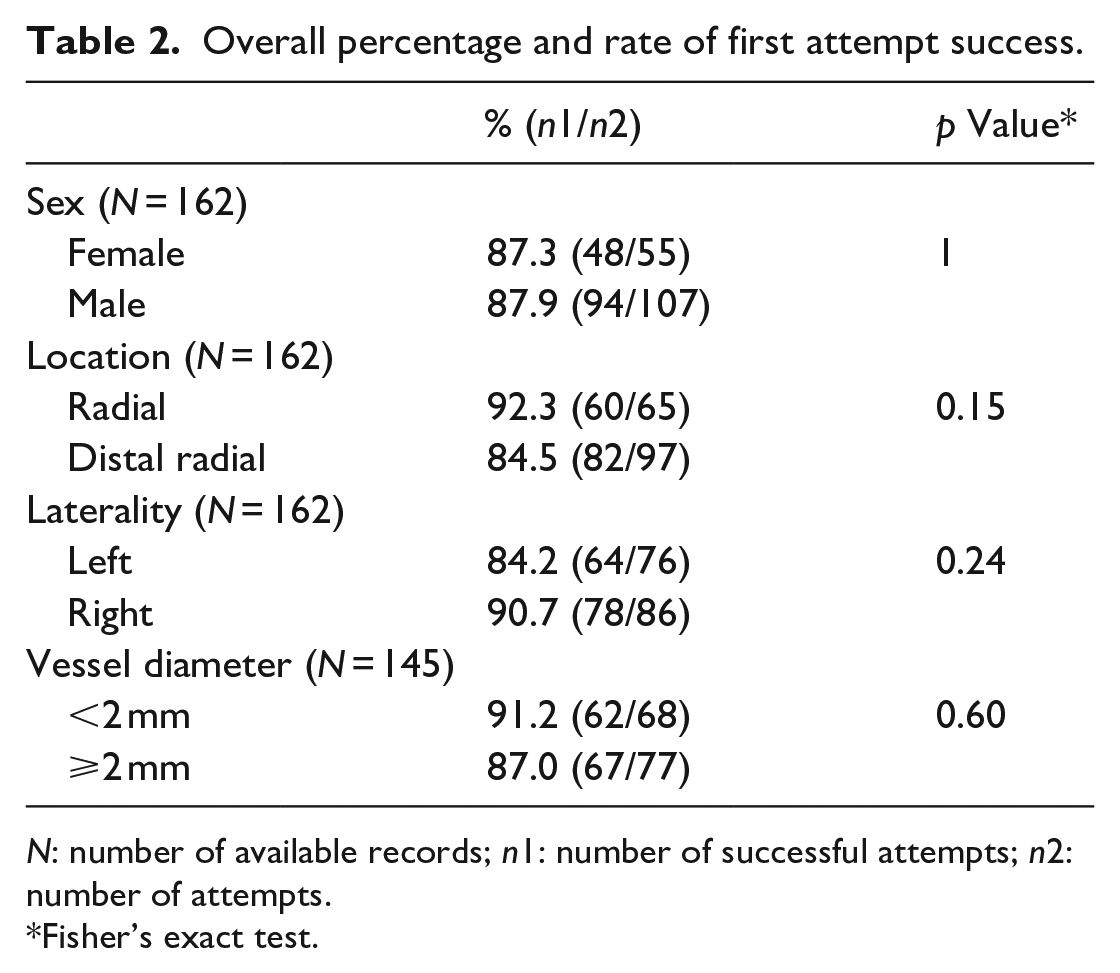
N: number of available records; n1: number of successful attempts; n2: number of attempts.
Fisher’s exact test.
Stratified percentage and rate of first attempt success per access site.

n1: number of successful attempts; n2: number of attempts.
Fisher’s exact test.
Safety
Serious adverse events and device deficiencies did not occur. Five NCI CTCAE Grade-1 (mild) adverse events were reported in 4 (3 male and 1 female) participants. These included three events of spasm of distal radial artery that occurred due to multiple attempts to access the vessel with the needle. All three events occurred during needle punctures and use of the investigational device was prevented in two of the three cases. Guidewire insertion was still attempted after the onset of the third event but was unsuccessful. All three events resolved spontaneously, and no intervention was required. These events were deemed not related to the study device by both the investigator and the sponsor. Hematoma was reported to coincide with one of the events and resolved without intervention or any additional procedure. In addition, a single event of spasm of the radial artery occurred in the study. This event also resolved spontaneously, and no intervention or additional procedure was required. The event was possibly related to the study device and the procedure in the opinions of the investigator and the sponsor.
Discussion
Experienced and inexperienced operators participating in this study were equally successful in first-attempt placement of the articulating-tip guidewire in the radial and distal radial access sites.
Comparison of the results of this study with reports in the state-of-art literature on vascular access should be interpreted with caution, as such comparison can be obscured by several factors, for example, heterogeneity of participants between the studies and differences in the procedural settings. Although systematic analysis of efficacy is not in the scope of this report, general observations, based on assessment of results from other trials, may still provide valuable insights.
Data abstracted in the recently published in the Cochrane Database of Systematic Reviews work of Flumignan et al. 8 on the radial access help put the radial artery portion of efficacy results in the current work in perspective. The 92.3% first-attempt success rate for radial artery achieved in our study is nominally superior to the results reported for radial artery access with ultrasound guidance in all studies appraised by Flumignan et al. 8 with the exception of two small-sized studies with 30 or less data points in each study arm. While the DRA access first-attempt success rate was numerically somewhat lower than its radial counterpart in our study, we did not find a reliable reference point for this outcome in the literature, since first-attempt success rate is missing from most reports of prospective studies on DRA access. Lower expected rates may be inferred from the lower DRA access overall success rates. Some studies show that the DRA diameter is smaller and the vessel tortuosity and angulation are greater compared to radial artery, an observation which may explain the greater incidence of failure.13,30,31
Safety-wise, insertion attempts did not result in adverse events occurring at an unexpected rate in our study, as evident from a nominal comparison of its results of with the published assessment of safety data from the state of the art. 8 The majority of spasms in our study could not be attributed to the investigational device as they occurred prior to its use. A single mild occurrence of hematoma in this study also compares favorably to the published evidence, especially considering the fact that only major hematoma events have been abstracted in the systematic review of Flumignan et al. 8
Summarizing the results, use of the articulating-tip guidewire was feasible in this study, the device was highly efficient in facilitating first-attempt access to the radial artery and DRA, and the nature of adverse events associated with use of the device was as expected from the state-of-the-art evidence. The articulating-tip guidewire was nominally equally efficient when used in both radial and distal radial arteries of men and women, regardless of the laterality of access or size of the target vessel. Nominal comparison of the success in securing first-attempt access observed in this study with the results reported in peer-reviewed publications may imply that use of the articulating-tip guidewire allows for performance superior to that possible with the state-of-the-art access guidewires.
Limitations of this study include its single-arm design and its conduct at a single site, features that may bias the results.32–36 However, consecutive enrollment of participants is likely to reduce the bias and contribute to patient sample being a representative one. 37
Vascular access is routinely performed for diagnosis and treatment of critically ill patients and the rate of occurrence of substantial harm associated with the procedure grows with the increase in the number of attempts required to establish access. Consequently, changes in the vascular access preferred anatomical locations require review of the physician’s toolkit, to which guidewires tailored to access sites may be a valuable addition. Specifically, the articulating-tip guidewire assessed in the present study may be such an addition for the radial and distal radial access. Further studies, including comparative trials would be required to validate this assumption.
Footnotes
Acknowledgements
We thank Dr. Igor Ruvinsky for his help in drafting the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael G Tal and Ron Livne have equity in Embrace Medical Ltd., which manufactures and markets TalWire. Other authors have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by Embrace Medical Ltd.
