Abstract
Background:
Intravenous (IV) therapy using short peripheral IV catheters (PIVC) is commonplace with neonatal patients. However, this therapy is associated with high complication rates including the leakage of infused fluids from the vasculature into the surrounding tissues; a condition referred to as, peripheral IV infiltration/extravasation (PIVIE).
Objective:
The quality improvement project aimed to identify the prevalence of known risk factors for PIVIE in the neonatal intensive care unit (NICU) and explore the feasibility of using novel optical sensor technology to aid in earlier detection of PIVIE events.
Methods:
The plan, do, study, act (PDSA) model of quality improvement (QI) was used to provide a systematic framework to identify PIVIE risks and evaluate the potential utility of continuous PIVC monitoring using the ivWatch model 400® system. The site was provided with eight monitoring systems and consumables. Hospital staff were supported with theoretical education and bedside training about the system operations and best use practices.
Results:
In total 113 PIVIE’s (graded II–IV) were recorded from 3476 PIVCs, representing an incidence of 3.25%. Lower birth weight and gestational age were statistically significant factors for increased risk of PIVIE (p = 0.004); all other known risk factors did not reach statistical significance. Piloting the ivWatch with 21 PIVCs using high-risk vesicant solutions over a total of 523.9 h (21.83 days) detected 11 PIVIEs (graded I–II). System sensitivity reached 100%; 11 out of 11 PIVIEs were detected by the ivWatch before clinician confirmation.
Conclusions:
Prevailing risk factors for PIVIE in the unit were comparable to those published. Continuous infusion site monitoring using the ivWatch suggests this technology offers the potential to detect PIVIE events earlier than relying on intermittent observation alone (i.e. the current standard of care). However, large-scale study with neonatal populations is required to ensure the technology is optimally configured to meet their needs.
Keywords
Introduction
The management of neonatal conditions relies heavily upon establishing and maintaining stable peripheral vascular access for administrating fluids, medications, nutrition, physiologic monitoring and diagnostic procedures.1,2 Neonates, especially those born preterm are at high-risk for intravenous (IV) therapy-related complications.1,3–5 This is in part, due to their immature immune system, underdeveloped skin barrier, smaller fragile blood vessels, and their exposure to numerous invasive procedures. 6
Despite innovations in IV therapy practice, the design and construction of peripheral vascular access catheters (PIVC) vascular access continues to be a concerning source of iatrogenic harm in neonatal units.3,7–10 Complications are common and include infection, thrombosis, occlusion, and peripheral IV infiltration/extravasation (PIVIE).1,4–6,8–11 The severity of leakage from the vasculature of non-vesicant (infiltration) and vesicant (extravasation) solutions into the surrounding tissue can be delineated clinically.12–15 For clarity in this article, we use the term PIVIE for both situations.
Recent literature1,3–6 supports the view that the incidence of peripheral IV complications has remained relatively constant over recent times. This situation remains in our neonatal ward despite quality improvement initiatives such as, establishing a team-based approach to IV therapy in 2008 and implementing several iterations of best evidence-based care bundles. Speculatively, it might reflect changes in our patient population’s inherent risk, changes in known, or the emergence of new risk factors. We might need to focus on other aspects of care such as accepting the prevalence of PIVIE and unmodifiable risk factors and adapting our IV therapy practices to reduce the severity of this harm. Consequently, it was agreed by our quality improvement committee that the next cycle of quality improvement in our unit would examine the current prevalence of known PIVIE risk factors and secondly, pilot a recent technology that has become available to aid earlier detection of PIVIE.
Methods
Design and setting
The plan, do, study, act (PDSA) quality improvement (QI) model is a widely adopted and adapted model of cyclical QI initiatives in healthcare situations.16,17 It is the one used in this study site. The intent of this QI cycle was twofold. Firstly, to gain data about the risk factors for PIVIE in our neonatal population and secondly, to evaluate the potential for using a novel sensor technology (ivWatch Model 400®, ivWatch LLC, VA, USA) to detect and provide earlier notification of PIVIE events than the current standard of care in our neonatal patient population.
Data was collected at a 24-bed tertiary-level NICU located in the Wilhelmina Children’s Hospital (WKZ), part of the University Medical Centre of Utrecht (the Netherlands) by members of the IV team. This unit averages 600 admissions annually, with approximately 220 PIVCs placed per month.
Ethical considerations
Ethical review determined that there were no risks related to participation, and no burden. Any benefit for participants was indirect as the study provided information to improve the quality of care and patient outcomes. Consequently, formal ethical approval was not required. However, this QI project was conducted according to prevailing governance structures for clinical audit and quality improvement. This included adherence to the Central Committee on Research Involving Human Subjects (CCMO) regulations. For privacy and anonymity of all participants, the Dutch General Data Protection Regulation was followed (GDPR) as per hospital regulation.
Patient and public involvement statement
Neither study participants nor their parents were directly involved in the design, conduct, or reporting of this study. However, the topic of focus (PIVIE) was chosen by staff and reflects parental concerns highlighted in 1:1 encounters with healthcare staff and our unit parent support group.
Participants and sample size
The study population comprised preterm and term infants admitted to the NICU requiring IV therapy provided by PIVCs. Infants were excluded if they had a major congenital malformation involving the veins or if they required total body cooling for the management of hypoxic-ischemic encephalopathy. Data related to the first objective was collected in the NICU between February 1st, 2018, and May 1st, 2019. For the second part, relating to the pilot use of the sensor technology data collection took place between 25th and 31st of October 2022; this delay was due to the impacts of the COVID pandemic.
Procedure
Peripheral intravenous cannulation was performed according to hospital policy based on international guidelines. 18 In the study setting, peripheral intravenous cannulation is routinely performed by a selected team within the NICU, a so-called IV-team. The IV-team is a group of nurses and clinicians who have received additional training to insert PIVCs, oversee the use, manage complications, and monitor IV therapy-related data collection. At the time of data collection vein selection for PIVC insertion was conducted by the IV-team. Vein selection considered the placement of other medical devices (e.g. preductal monitoring of oxygen saturation), intended infusate and flow rate, and pre insertion vein visualization. Vein visualization was generally by direct viewing though optical vein visualization technologies were also available for use.
Internationally, the current standard of care for PIVC sites for the emergence of PIVIE involves intermittent practitioner visual checks of the site using PIVIE assessment tools and treatment algorithms13–15,19 combined with techniques such as “touch, look, compare” to confirm patency.15,18 Despite its benefits this approach clearly has limits and a technological response that provides continuous monitoring and earlier notification of potential PIVIE is desirable.
The company ivWatch LLC located in the USA was the first to bring to commercial realization an approved medical system for everyday and continuous monitoring of IV sites for PIVIE. 20 The system sensor is attached adjacent to the cannula insertion site (Figure 1 shows a typical PIVC set up with the system sensor in place) and works by detecting changes in the absorption and scattering of different wavelengths of light between healthy and infiltrated tissue and analyzes these to determine if a PIVIE event has occurred and then alerts the clinician with a yellow (possible PIVIE) or red (probable PIVIE) check site notification depending on the urgency. 20 The system has been clinically tested in laboratory and real-world clinical use for sensitivity and specificity amongst adult, pediatric, and neonatal populations with reassuring findings.20–23 Eight ivWatch systems and related consumables (sensors) of the most recent iteration of the product known as the ivWatch Model 400® were loaned for this evaluation. This system uses a near-infrared and visible light LED sensor combined with an interpretive algorithm to notify clinicians to check the site for possible (yellow alerts) and probable (red alerts) instances of infiltration around the infusion site. 21
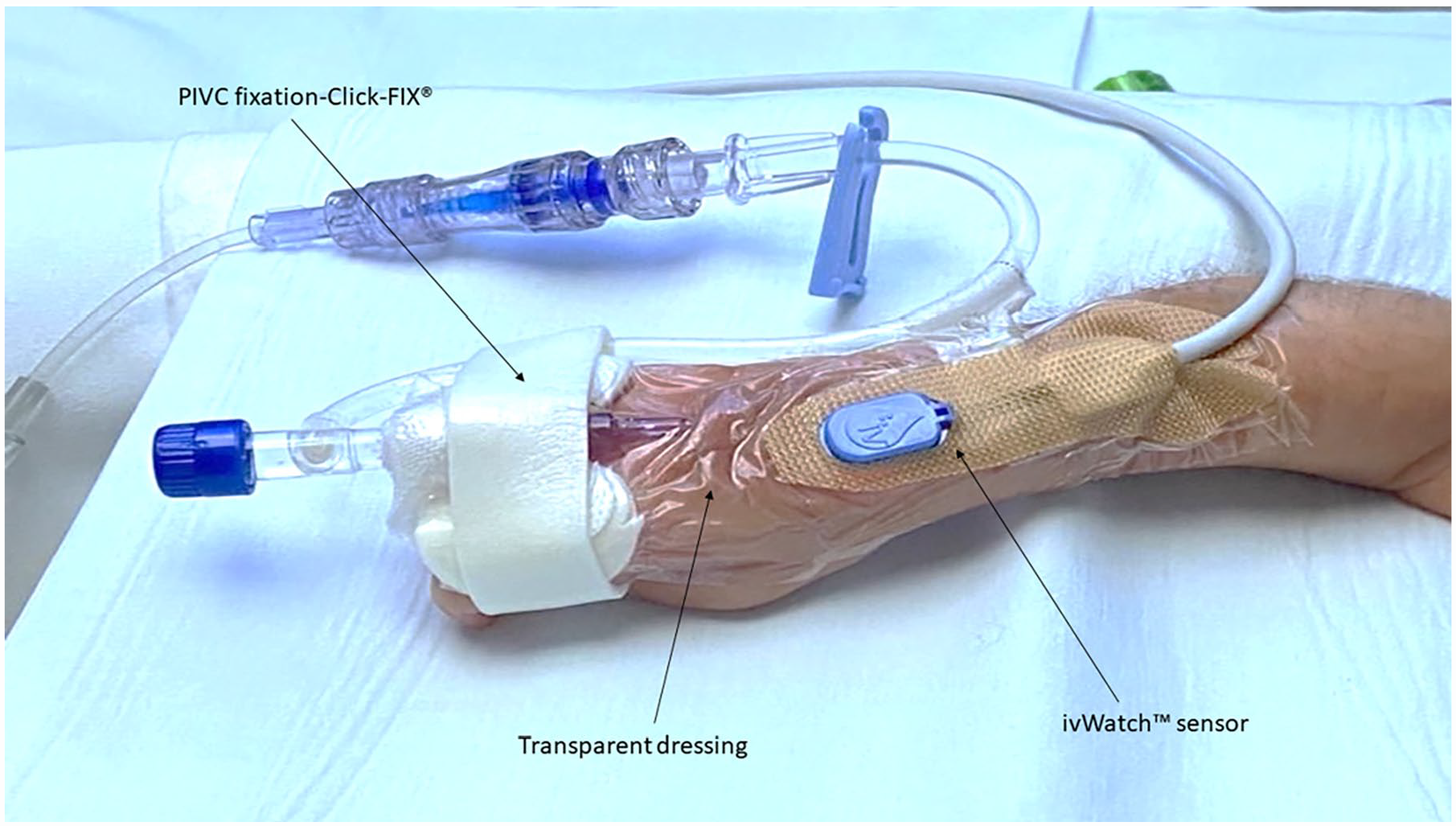
Typical IV infusion set up with ivWatch sensor in place.
The IV-Team routinely collects IV therapy-related data. Data included anonymized patient demographics (including sex, gestational age at birth in weeks and days, age in days, birth weight, and current body weight in grams). Data relating to the procedure of intravenous cannulation included: date and time of cannulation, cannulation side (left or right), extremity of cannulation, and the site on the extremity (dorsum of the hand, wrist and lower arm, elbow crease and upper arm, foot, ankle and lower leg, or knee and upper leg), the type intravenous fluids (clear or non-clear fluids), the date and time of removal of the PIVC, total dwell time of the PIVC in hours (calculated as the removal date and time minus the insertion date and time).
In the event of a clinician confirmed PIVIE medical imagery was taken by the IV-team and the site was classified using an adapted scale after Millam 12 and others13,14 and graded using the Cincinnati Children’s hospital tool 15 for PIVIE assessment to determine the optimal treatment. Treatment of confirmed PIVIE followed local evidence-based protocols developed from international consensus and case reports.14,18,19,24,25 As per unit documentation protocol, a form developed after Mason-Wyckoff and Sharpe 26 was used to record incidents, assessments, actions taken and their evaluation in individual patient electronic health records (HIX, Chipsoft).
Statistical analyses
Descriptive statistics (mean, standard deviation) were used for continuous variables and numbers and percentages for discrete variables to summarize the data. Assumptions about normal distribution were determined by Kolmogorov-Smirnov testing. Any relationship between variables and the outcome of interest (PIVIE occurrence) was identified using univariate logistic regression analysis with p < 0.05 determined statistically significant. SPSS Statistics (version 26.0, IBM Armonk, NY, USA) was used.
Coded data collected during the ivWatch pilot was analyzed using a logistic regression script in the MATLAB software (The MathWorks Inc. Natick, MA, USA) to model patient variables with the occurrence of PIVIE and demonstrate performance sensitivity for detecting early-stage infiltrations.
Results
Risk factors for PIVIE
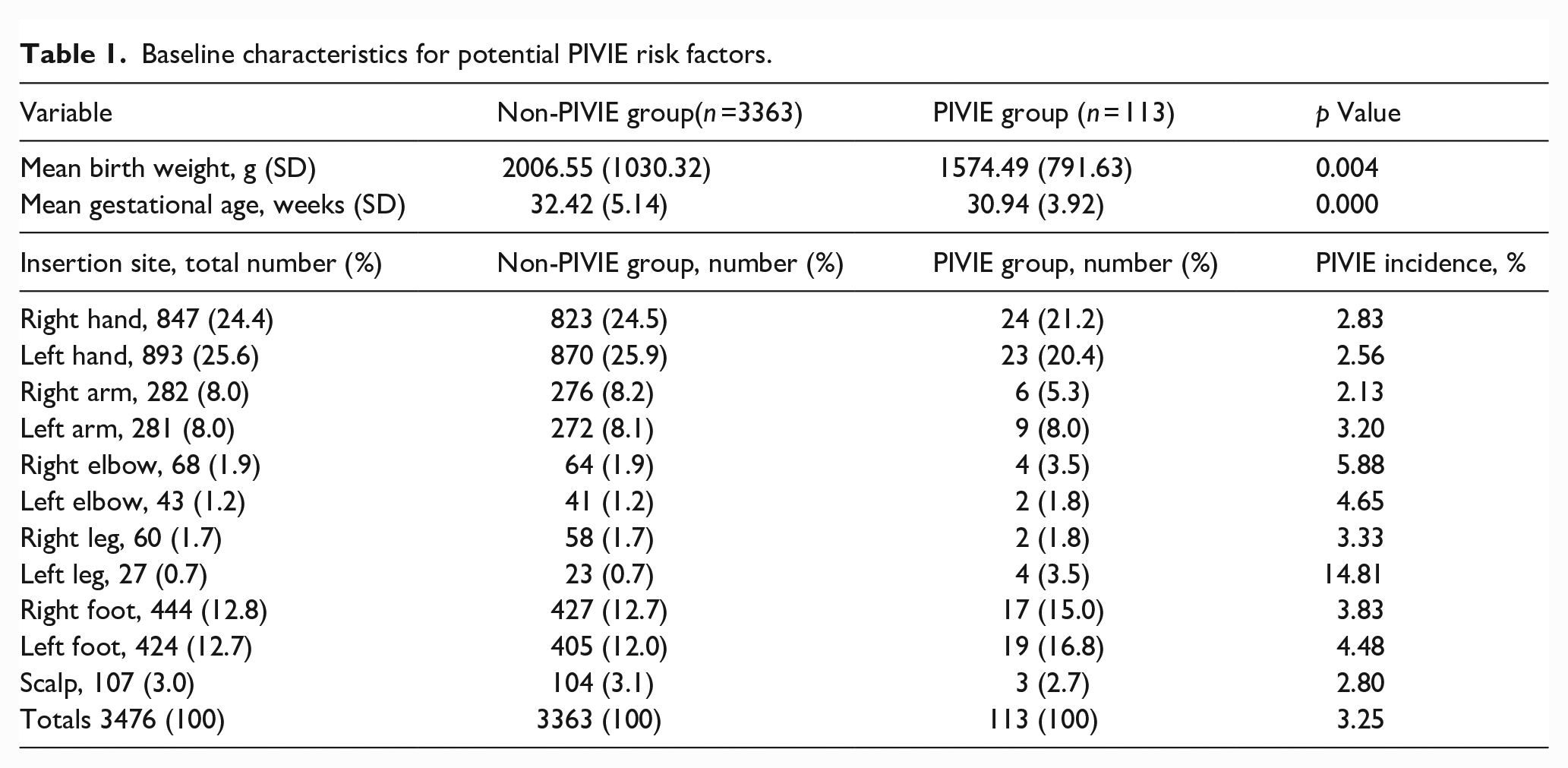
During the first data collection period 113 clinically confirmed PIVIE’s (severity graded between II and IV) were recorded from 3476 PIVCs. This represents an PIVIE incidence of 3.25%. Demographic and IV therapy-related characteristics for this study population are shown in Table 1. The right or left hand was the preferred insertion site for PIVC insertion in half of cases (24.4% and 25.6% respectively) and these sites accounted for 41.6% of all PIVIEs reported, with a combined site incidence of 2.7%. The second most popular locations were the right or left foot representing 16% of insertions and a combined PIVIE incidence of 4.1%. Other less popular insertion sites had greater variability in both the number of times they were accessed and the instances of PIVIE; the left leg insertion site had the highest incidence of PIVIE cases (14.81%). Insyte (24G, BD), Neoflon (26G, BD), and Safety (24G, Introcan B Braun) were the most used catheters in the PIVIE group representing 41, 25, and 30 PIVIEs, respectively. Glucose 10%, Glucose 15%, Total Parenteral Nutrition (TPN) (with intralipid), and erythrocytes were the most common IV fluids used in the group experiencing a PIVIE event, a situation reflecting the whole unit use of these different PIVC devices and type of prescribed infusate. Lower mean birth weight and gestational age were statistically significant factors for increased risk of PIVIE (p = 0.004).
Baseline characteristics for potential PIVIE risk factors.
The 113 Infants who experienced a PIVIE were treated according to local policy, based on evidence-based literature and consensus guidelines.14,18,19,24–26 However, outcome data for some of these infants was incomplete in the electronic health record (HIX, Chipsoft); improvements in the completeness of records are the topic of a separate QI project.
ivWatch
The ivWatch evaluation was conducted from 25th to the 31st of October 2021. The total monitoring time was 523.9 h (equivalent to 21.83 days), with an average monitoring time of 24.94 h per PIVC. During this period, a total of 13 preterm infants (nine male and four female) having a total of 21 PIVCs (i.e. some infants had more than one PIVC inserted during the study period) were continuously monitored by the loaned ivWatch Model 400® systems. The gestational age at birth ranged from 27 to 41 weeks plus 1 day. Infants’ ages at the time of PIVC insertion ranged from 1 to 44 days, and their current weight at the time of PIVC insertion was recorded as between 1044 and 5020 g. For consistency all the 21 monitored PIVCs used a 26-gauge BD Neoflon™ [Becton, Dickinson and Company] cannula. The decision to use the smallest size device was in accord with international guidelines. 18 In total nine catheters were inserted in the hand, six in the foot, three in an ankle and two in the lower arm and one in the wrist.
In statistical modeling, none of the known PIVIE predictor variables had a significant (p < 0.05) effect on the occurrence of PIVIE in this study population. The variable closest to statistical significance (p = 0.07) was dwell time. During the monitoring timeframe, 11 infiltrations were detected among the PIVs monitored, suggesting an infiltration rate of 52.4%. However, despite all the infusates being vesicant, all PIVIEs were graded as minor. Notifications were issued for 11 of the 11 PIVIEs, corresponding to 100% sensitivity and, importantly, all before the clinician identified the event.
Yellow check IV notifications were issued at a rate of 0.056 notifications per hour, and Red check IV notifications were issued at a rate of 0.082 notifications per hour. The notification rate for non-PIVIE cases was less than one per day for both yellow and red notifications (0.9 and 0.7 respectively); figures similar to that reported in the literature.20–23
Discussion
Newborn infants are inherently vulnerable to IV therapy-related complications, including PIVIE which can lead to serious harm.3–6 Therefore, recognition of known intrinsic and extrinsic risk factors together with preventative and remedial strategist and treatments are essential components of quality care. PIVIE is one of the most common causes of IV therapy failure resulting in unplanned removal of the PIVC.1,4–6 The incidence of PIVIE in the whole unit population data was 3.25%. This figure is low compared to some studies 6 but is comparable with the incidence of 3.8% reported by Atay et al. 5 It might be that low rates of reported PIVIE reflect underreporting of minor classifications of PIVIE either due to non-recognition, or the presence of confounding factors, or that documentation of the reason for PIVC removal fails to capture this explanation; situations reported elsewhere. 23 A focus on providing additional education on recognition of early stage PIVIE, PIVIE classification and documentation might affect reported incidences regardless of the introduction of technology-based monitoring of PIVC sites.
In contrast the PIVIE incidence reported during the ivWatch pilot was 52.4% which is higher than that seen in the whole unit sample but similar to rates reported in other studies with or without the ivWatch.6,23 This situation might represent a statistical anomaly due to the small sample size, or advances in the sensitivity of the optical sensor and interpretive algorithm technology. It might also reflect differences in the PIVIE risk in this study group or underreporting of PIVIE as the reason for PIVC removal in the whole unit sample. Naramore et al., 27 report increased sensitivity of the ivWatch model 400® system to detect infiltration events when the PIVC tip is nearer the skin surface and thus closer to the sensor. It is feasible that because of neonatal vascular anatomy PIVC tips are inherently physically closer to the sensor and this might account for the increased sensitivity to detect PIVIE reported here and elsewhere in neonatal and pediatric populations.22,23 However, further detailed study is required to explore this relationship.
The ivWatch system detected 11 of the 11 clinically confirmed PIVIE events, suggesting a 100% sensitivity. All PIVIEs were graded minor despite the presence of vesicant fluids, suggesting early detection of infused fluid leakage into the surrounding tissue. This figure is higher than previously reported in neonatal and pediatric study using a previous version of the sensor22,23 but is comparable with reports from adult studies.20,21 This might reflect improvements in sensor technology and PIVIE detection algorithms. False system check site notifications (red, probable, and yellow possible PIVIE) about the presence of PIVIE occurred on small number of occasions (n = 12). These occasions included incorrect placement of the sensor (n = 2), inadvertent removal of the sensor whilst it was still monitoring (n = 1), and pressure applied to the sensor site during kangaroo care or infant position (n = 5). It is also possible that the system identified infiltration undetectable by inspection (n = 4) which was documented under a different reason for PIVC removal. Anecdotal reports from staff suggest that these “false alarms” did not overly contribute to alarm fatigue. The lack of statistical significance in any of the patient and IV therapy-related variables considered and PIVIE, apart from birth weight and gestation is at odds with analyses published elsewhere.1,5,6,9,10 It might be that this absence of relationship is real or a larger more statistically powered data set is required to determine these relationship effects. Further study to clarify this is required.
Strength and limitations
All eligible infants meeting the inclusion criteria were included in the data set, a sample that was representative of the NICU population requiring a PIVC. However, this was a single unit study carried out over a comparatively short data collection timeframe.
A strength of the chosen PDSA methodology is based on its simplicity, formalized approach, and proven effectiveness in bringing about improvements in care in real-world clinical settings. However, evaluations of the method’s effectiveness in providing long-lasting QI is mixed. 28 This situation is, in part due to the small scale focus of changes, poor adherence to the four key steps in subsequent cycles, objective drift, and over simplification of the processes involved in QI. 28 Nevertheless, our findings and experiences provide a basis for comparison with other neonatal and other patient care settings where IV therapy is conducted and where staff are keen to explore the potential to address modifiable risk factors for PIVIE and its earlier detection.
Conclusion
The incidence of PIVIE in our unit was comparable with low figures reported elsewhere, however this might reflect underreporting of the condition. Prevailing risk factors for PIVIE, low birth weight and lower gestational age confirm findings from other studies, but other known risk factors were not statistically significant in this study sample.
Continuous infusion site monitoring using the ivWatch system as an add on suggests that this technology offers the potential to detect PIVIE events earlier than relying on intermittent observation alone (i.e. the current standard of care), this might help to explain the higher incidences of PIVIE reported by us and other users during use of this system. Lack of statistical significance of patient and IV site variables versus PIVIE incidence during the ivWatch evaluation period is unsurprising given the sample size during this period (n = 21) versus the sample size during the study of the general NICU population (n = 3476). However, the reported 100% sensitivity is reassuring. Further large-scale studies with adequate statistical power are required to determine relationships between variables and ensure that the technology is optimally configured for this population.
Footnotes
Acknowledgements
The authors would like to acknowledge Jozua A. van Duuren and the neonatal vascular access team and all our colleagues in the Department of Neonatology of the Wilhelmina Children’s Hospital and ivWatch LLC who have supported this quality improvement project.
Author contributions
MvR, DV, and AvdH conceptualized and designed the study. SB supervised data collection and with AvdH, and FHJvL oversaw data analyses. MvR drafted the initial manuscript, and KH reviewed and revised the manuscript. All authors critically reviewed and revised the manuscript for important intellectual content. All authors approved the final manuscript as submitted.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
