Abstract
Background:
Stent grafts (SG) have gained the highest level of evidence for superior management over angioplasty of arteriovenous graft (AVG) venous outflow stenosis, which increases their use in hemodialysis patients. Migration to the heart and lungs is the most feared complication of SG of the venous system.
Method:
We present a previously unreported case of asymptomatic migration of a 10-cm-long Viabahn lodged in a U-shaped configuration in the main left pulmonary artery after AVG intervention in a 50 year old male hemodialysis patient.
Results:
Through a single femoral access percutaneous retrieval of migrated SG was accomplished. Technique of coaxial buddy snaring technique is described.
Conclusion:
Our technique may expand the endovascular likelihood of successful stent removal.
Keywords
Introduction
The current practice of endovascular therapy makes indications for stenting more common. Stents play a growing role in the endovascular percutaneous and hybrid management of vascular access malfunction in patients on dialysis. Stenosis of the central or peripheral venous outflow of native and prosthetic fistulae constitutes a routine stenting. After angioplasty, results, such as dissection, recalcitrant stenosis, elastic recoil, or vein rupture, may require the deployment of a stent. Stent grafts (SG) represent a significant improvement in this setting in that the biocompatible fabric supported by a metal scaffold can seal vessel rupture and impede trans mesh myointimal hyperplasia, delaying restenosis. Migration of foreign materials (vena cava filter, pacemaker lead, catheter venous access port, coils, etc.) into pulmonary arteries is classically reported in the literature. 1 Immediate or delayed post procedure stent migration in the venous system is of utmost concern because it is potentially fatal. Migrated stents may end up in the right cardiac chamber, causing dysrhythmia, cardiac failure, tricuspid insufficiency, hemopericardium, and death.2,3 More rarely, stents can pass the heart and dislodge in pulmonary vessels, even asymptomatically, leading to the dilemma of choosing long-life anticoagulant therapy or performing interventional retrieval or open surgical removal. 4 Herein, we present the case of a 50-year-old patient who underwent endovascular salvage of a thrombosed radio-basilic prosthetic loop using thrombolysis and stent grafting. Following the fourth occlusion in 2 years, a bent and thrombosed 10-cm-long stent graft Viabahn was discovered in the left main pulmonary artery. No comparable length, location, and appearance were previously reported among lung-migrated SGs published in the literature, and techniques applied in the past for the retrieval of bare metal stents from pulmonary vessels were not applicable. Adjunctive refinement to the single-access technique proposed by Zvavanjanja permitted percutaneous removal. 5 Details regarding the causes of migration and considerations of the retrieval technique are also discussed.
Case presentation
The patient was a 50-year-old male with ESRD secondary to a neurogenic bladder and myelomeningocele. At 33 years of age, the patient underwent a cystectomy with cutaneous ureteroileostomy (Bricker intervention). He started weekly hemodialysis through a left proximal radiocephalic fistula at the age of 45 years. Three years later, a radio-basilic prosthetic bridge was anastomosed to the hypertrophied left proximal radial artery. Subsequently, he had several thrombotic occlusions of the graft; the first, at 40 days after creation was a technical surgical failure, due to the short time elapsed from AVG creation. Thrombosis was treated with low-dose rTPA fibrinolysis and thrombectomy overnight. A 7 mm × 100 mm Viabahn was deployed on the underlying critical venous stenosis at the elbow, and the patient received dual antiplatelet therapy, which had to be discontinued following anemization and gastric bleeding within the next 2 months despite adequate therapy with proton-pump inhibitors (PPI). A second arteriovenous graft (AVG) thrombosis occurred 361 days later. Thrombolysis and thrombectomy were then performed. Edge stenosis on the first Viabahn was found to be recalcitrant to scoring and high-pressure angioplasty. A new 7 mm × 50 mm Viabahn was considered necessary to establish a brisk flow. A new circuit thrombosis occurred 498 days after the creation of AVG. Critical edge stenosis in the last Viabahn was appreciable following thrombolysis. The diameter of the prior stent graft was not checked; due to unsuccessful PTA result, a 6 mm × 50 mm Viabahn (the third) was overlapped distally for <1 cm with a 6 mm × 100 mm Viabahn (the fourth) to allow the coverage of further stenosis of the basilic vein (Figure 1(a)). Nephrologist was noticed that surgical revision was necessary before a new thrombosis could occur. After further occlusion 703 days after AVG creation, routine chest radiography on admission revealed a 100 mm long Viabahn on the left side of the heart. (Figure 1(b)) A spot radiogram of the left arm confirmed the disappearance and migration of the distal SG (Figure 1(c)). Prompt contrast Multi Slice Computed Tomography (MSCT)-localized stent dislodgement in the main left pulmonary artery without signs of pulmonary infarction (Figure 1(d)). The SG appeared occluded and transversally bent in a U-configuration inside the trunk of the left pulmonary artery. A left 14.5 Fr tunneled transjugular catheter was inserted for dialysis. The patient denied any past thoracic discomfort and remained eupneic and asymptomatic. After a multidisciplinary reunion with nephrologists, anesthetists, cardiac surgeons, and interventional radiologists, open surgery and conservative approaches were excluded. Percutaneous removal was then performed. Informed consent was obtained and invasive hemodynamic and electrocardiographic monitoring was performed. The patient was then placed under general anesthesia. The right groin was then prepped. After ultrasonography-guided femoral venipuncture, a 16 Fr 30 cm long sheath (Cook, Bloomington, IN, USA) was inserted with its distal tip in the inferior vena cava. A preliminary left pulmonary angiogram was obtained through a 5 Fr pigtail. (Figure 1(e)) Throughout the pigtail, the hydrophilic guidewire was exchanged with a 260 cm long 0.035″ stiff Amplatz (Boston Sc, Ma, USA) guidewire, which advanced in the left pulmonary artery. A 65 cm long 11 Fr armed sheath (Arrow, USA) was then coaxially placed inside the first 16F sheath and advanced upside the pulmonary valve into the left main left pulmonary artery. Leaving the Amplatz guidewire as a safety wire, a 20 mm Amplatz gooseneck was advanced alongside the safety wire inside the Arrow sheath, while carefully retrieving the Arrow tip nearest, but always beyond, the pulmonary valve. Multiple attempts to capture one end of the SG with the gooseneck failed. At this point, the gooseneck was pulled out and charged over a 5 Fr Glidecath Terumo precurved in the Judkins-Bentson configuration. This improved steering capability allowed the goose neck to easily capture the loose SG at one edge. Once the safety wire was removed, the firmly crimped and collapsed edge of the SG was gently oversheated under continuous fluoroscopy, advancing the sheath while withdrawing in counteraction with the SG (Figure 2(a)–(e)). After the completion of the SG ensnaring upside of the valvular plane, the armed sheath was withdrawn as a unit with the SG from the patient. The retrieved SG was intact with a thorough inspection (Figure 2(f)). Femoral hemostasis was achieved by manual compression after removal of the 16 Fr outer sheath. The patient remained hemodynamically stable during the procedure. No arrhythmias were observed. The findings of transthoracic echocardiography were unremarkable. The patient was discharged 2 days later after one dialysis session through the Central Venous Catheter.
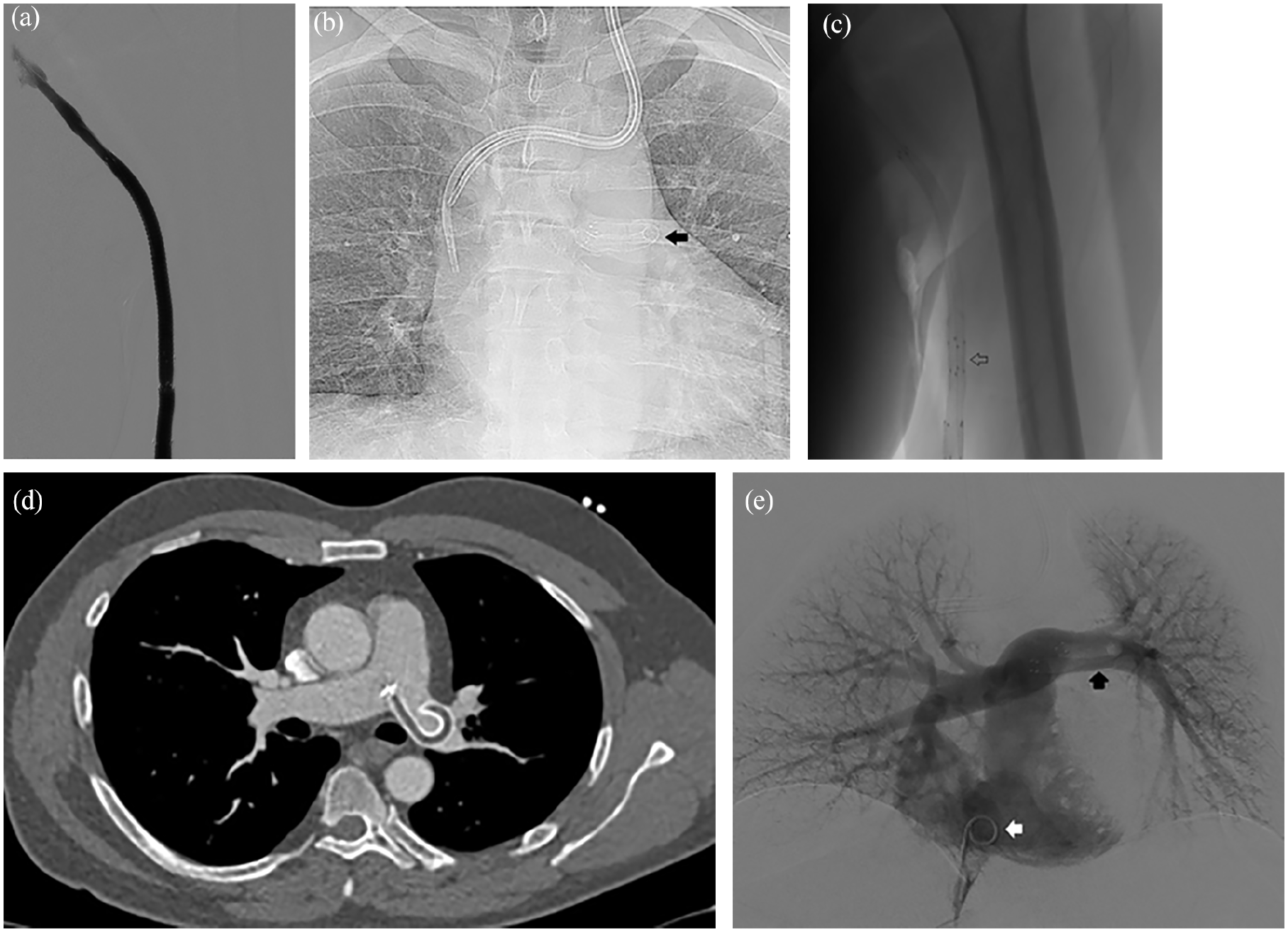
(a) Digital venography: regular flow after last Viabahn has been deployed in basilic vein, (b) chest X-rays: Bent SG on the left side (arrow), (c) fluoroscopy: short overlapping of distal markers (Arrow), (d) contrast Enhanced axial CT scan showing the thrombosed hypodense SG in the main left pulmonary artery, and (e) pulmonary digital angiography showing regular perfusion. (White arrow): pigtail catheter in right atrium. Black arrow. Thrombosed Viabahn.
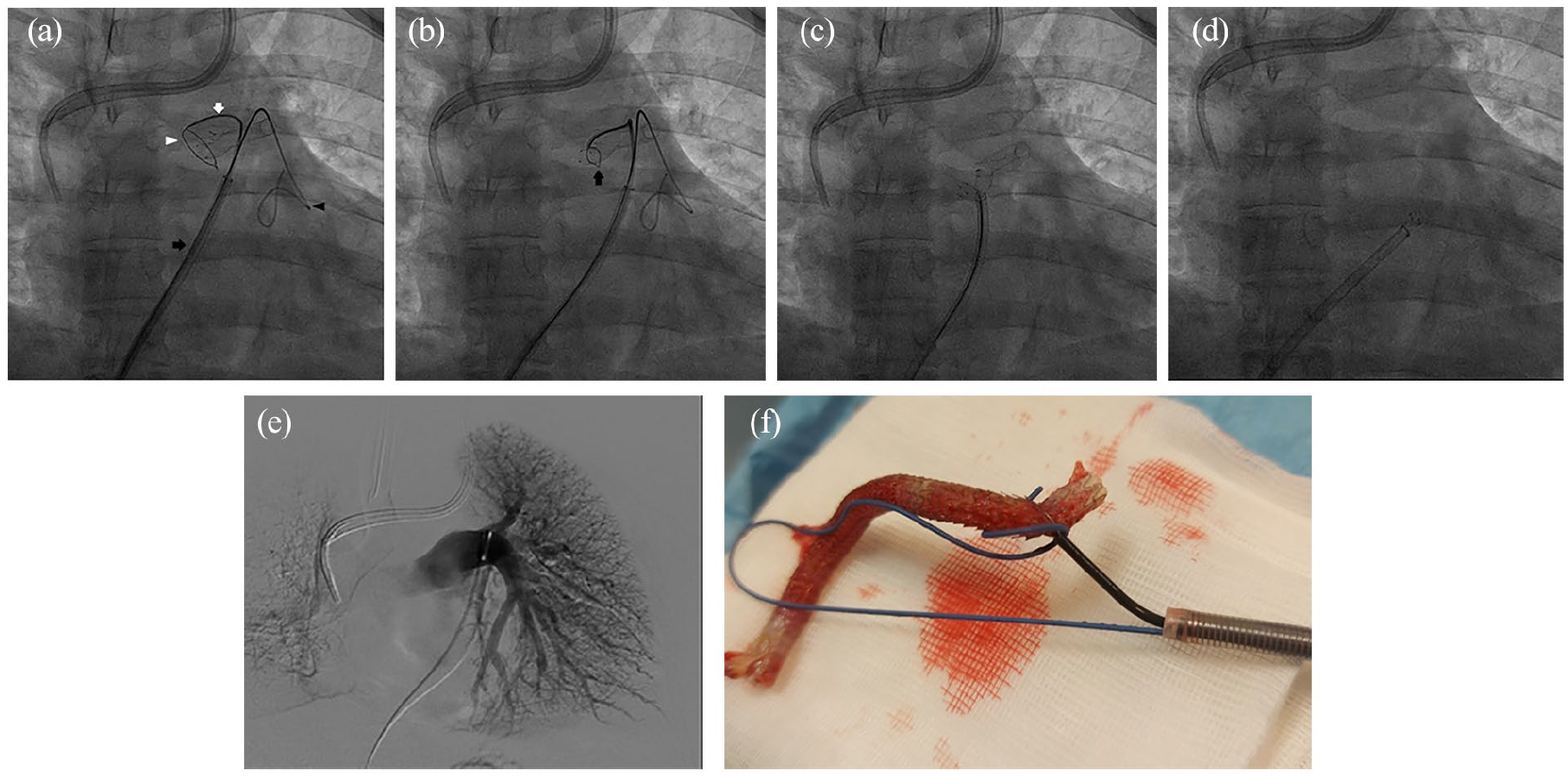
Procedural SG endovascular retrieval. (a) 11Fr armed sheath (Black arrow) upside the valvular plane alongside the safety wire (black arrow head). Curved catheter (White arrow). Open snare near one edge of SG (White arrow head). (b) SG inside the closed snare (black arrow). (c) After have tighten the snare, the retrieval of the bukled and fold on itself SG is accomplished. The safety wire has already removed. (d) SG inside the armed sheath. (e) Selective left pulmonary digital angiography after SG removal. (f) Ensnared Viabahn on the bench.
Discussion
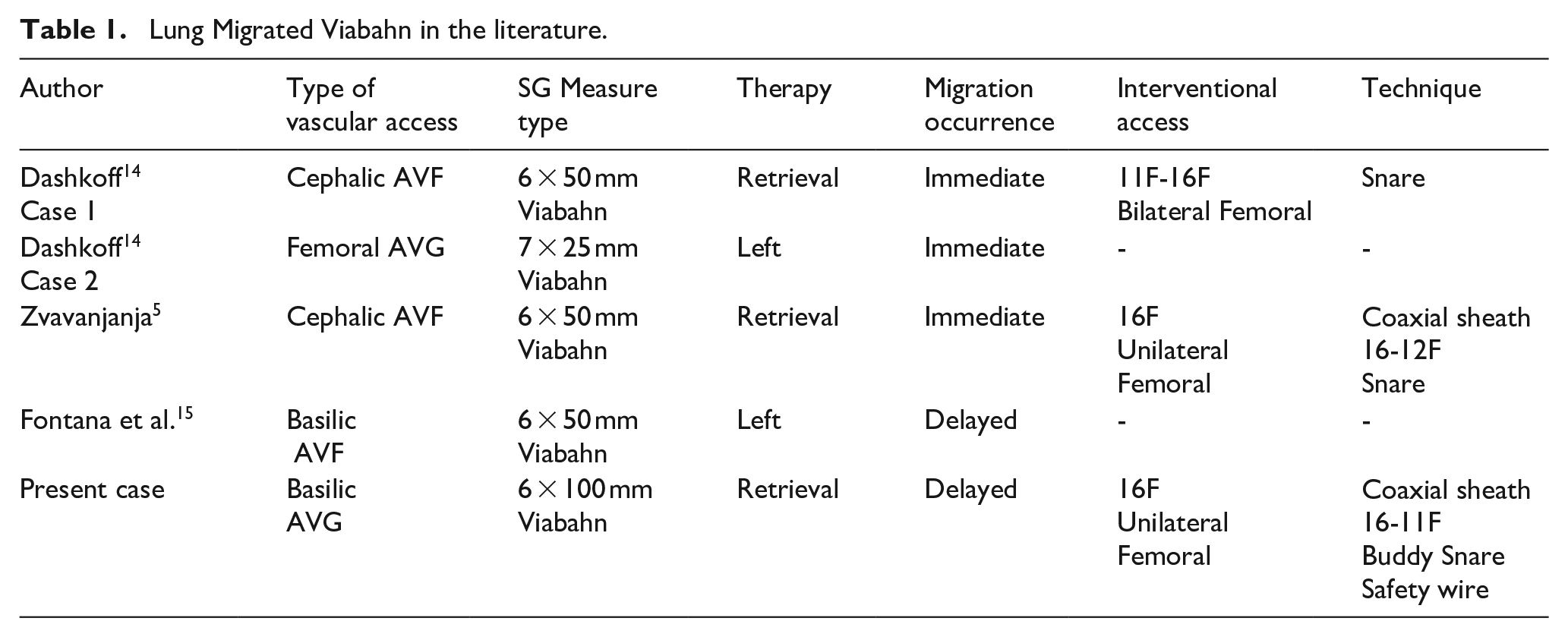
SGs have an established primary role in treatment of venous AVG stenosis. Less clear is the treatment of in-stentgraft restenosis. Leave nothing behind with DCB to prolong freedom from reinterventions is an accepted strategy. Successful predilation is a prerequisite for DCB application. Elastic recoil in fibrous in-stent restenosis after POBA/scoring balloons and delay in planning surgical revisions may lead to deploy several stentgrafts in the dialitic circuit. Stent migration can occur at the time of placement and subsequently. Treatment of cardiac and pulmonary stent migration may vary from anticoagulation therapy to surgical removal.6–8 Weighting factors, such as symptoms, risk of clots, age, comorbidities, and patient-expressed acceptance, are essential before clinical decision-making. Bare metal stents are the most commonly reported devices that migrate to the heart or pulmonary arteries.7,9–12 Due to increasing indications for the deployment of SGs in dialysis access, migration of covered stents has been reported in the last decade. Emergent pericardiocentesis followed by a thoracotomy was required for SG extraction after SG migration. 8 A piece of the fractured SG was left inside the heart, requiring anticoagulation therapy in another patient. 13 In addition to our case, there are four Viabahn (Table 1) SGs that were reported to migrate to the pulmonary arteries.5,14,15 Two were retrieved percutaneously, while the others were left in situ. 6 All migrated Viabahn SGs were small (⩽7 mm) and short (⩽50 mm) and lodged directly in the distal pulmonary artery without intraluminal thrombosis. In the present case, the thromboembolic risk associated with SG lodging in the proximal pulmonary artery and its flow-limiting transverse U-configuration was considered to offset the risk of an endovascular attempt. The caliber of our stent was also 6 mm, underscoring the role of downsizing as the most alleged factor favoring migration in venous stenting. Stent downsizing has been suggested to reduce vein wall shear stress and edge hyperplasia with potential benefits for minor stenosis recurrence.6,16 In contrast, calibrated SG oversizing of 10%–15% has been advocated as the first measure to avoid migration and summarized in the rule “measure twice and stent once” as SGs are known to provide minor vessel embedding. 17 In addition to downsizing, the short distal stent overlapping (<1 cm) and placement of a smaller SG within the preexisting SG, as already outlined by Ho et al., could have played a role in migration. 18 The coupling of SG downsizing and inadequate stent overlap may have synergistically caused the present complication. Clear reasons to explain the unusual stent length migration are questionable. In fact as suggested by the CIRSE quality assurance guidelines for SVC stenting longer stents are considered much less likely to migrate than short stents. We can speculate that the intrinsic flexibility of Viabahn and the large smooth curve at the basilic vein-axillary transition of vascular access could have favored the long stent migration.2,19 Regarding how we performed the Viabahn retrieval, we were obliged to add some changes to the single percutaneous access technique first described by Zvavanjanja (Figure 3(a)). 5 The ends of the SG were located near the pulmonary valve and floated inside the pulmonary artery. Moreover, as observed in MSCT, the thrombosed lumen of SG made other techniques not viable, since neither the guide wire-assisted snaring of the stent nor the angioplasty balloon-assisted snaring as outlined by Taylor et al. 2 was applicable (Figure 3(b)). We relied on the flexibility and collapsibility of the Viabahn. Due to the PTFE fabric properties, the frying of the SG ends was unlikely after the snaring and unraveling of SG. The modifications we added were a smaller 11 Fr sheath, stiff safety GW, and buddy gooseneck insertion through a pre-curved catheter left free inside the sheath. Considering the Viabahn features, we put inside the outer 16F Cook sheath a smaller but heavily braided 11F Arrow sheath over a long Amplatz GW. The stiff GW wire was left as a safety wire, allowing operators with continuous fluoroscopy and ECG monitoring to keep the sheath tip beyond the valvular plane. In case of under pulmonary valve dislodgment of the sheath tip, the pulmonary artery lumen could easily have been gained by inserting the vessel sheath over the GW. The gooseneck was advanced with its right-angled design in parallel with the Amplatz safety wire (not over) through the 11Fr sheath, a further technical variation (Figure 3(c)). The buddy snare configuration permitted the easy substitution of the original straight snare guiding catheter in favor of a pre-curved Glidecath Terumo, thus gaining the maximum steerability and torque control of the gooseneck nitinol cable to catch one Viabahn edge. This was necessary after recognizing that the original gooseneck was too distally located in an unfavorable position to catch the SG. Once captured, forced to pull outside the Amplatz guide wire due to the lack of room in the sheath, the Viabahn was completely ensnared inside the Arrow sheath and retired as a unit within the sheath without entanglement through the cardiac chambers (Video).
Lung Migrated Viabahn in the literature.
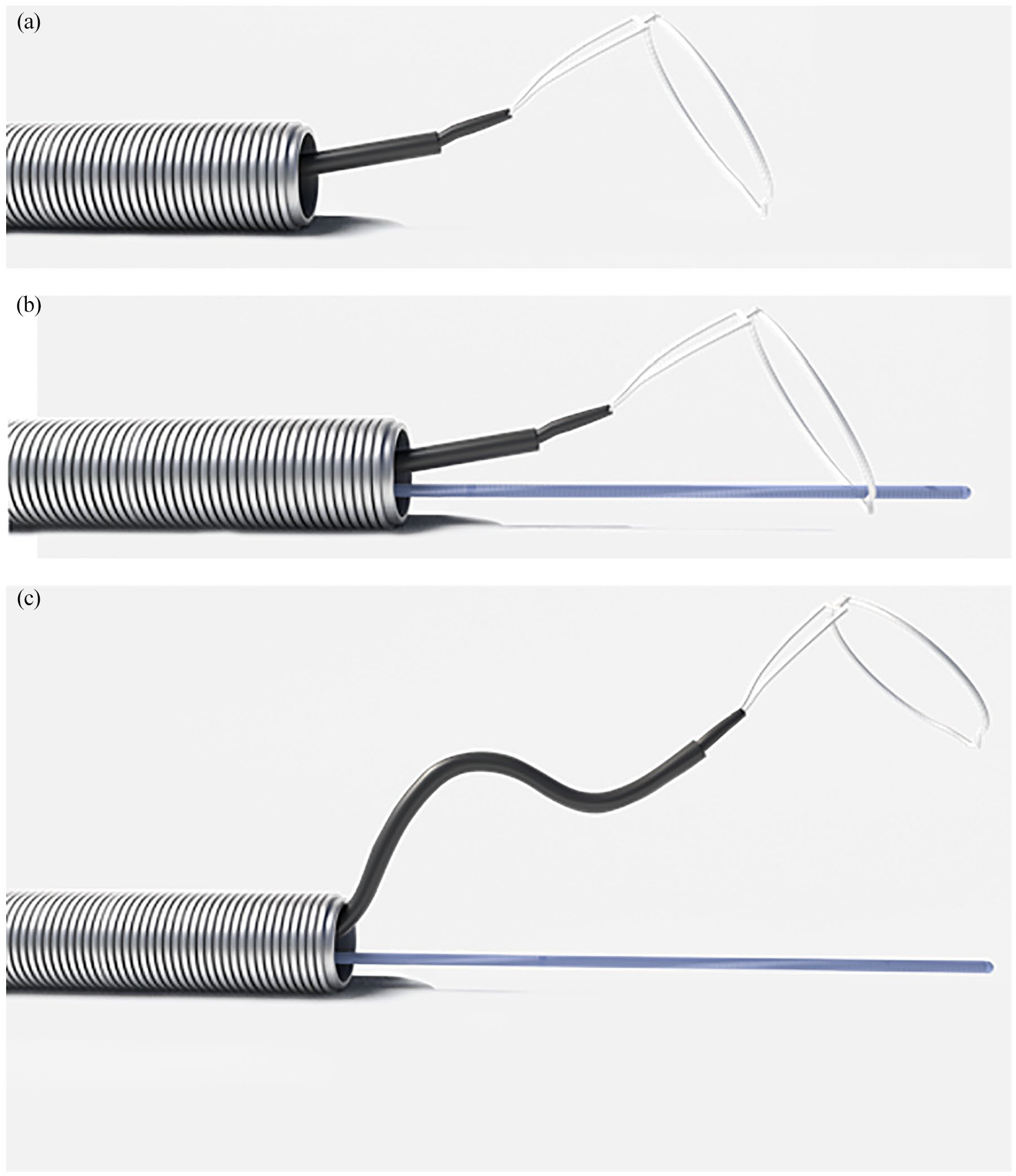
Scheme of buddy snaring. (a) Simple snaring outside the armed sheath. (b) Coaxial snaring. The snare is advanced over the guide wire. The free steering of snare all around the sheath is hampered. (c) Buddy snaring for improving snare steering. The snare is charged over a curved catheter alongside the safety wire. The snare steering is maximized.
Conclusion
This case report underlined the risk of SG downsizing in the venous system and showed that a long-migrated thrombosed Viabahn can be retrieved from the main pulmonary artery through a single percutaneous femoral access, avoiding damage to the cardiac valves and femoral vein.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298231158545 – Supplemental material for U-bent Viabahn stent graft in the main left pulmonary artery in a hemodialysis patient after asymptomatic migration: Percutaneous removal and technical considerations
Supplemental material, sj-pdf-1-jva-10.1177_11297298231158545 for U-bent Viabahn stent graft in the main left pulmonary artery in a hemodialysis patient after asymptomatic migration: Percutaneous removal and technical considerations by Pietro Quaretti, Riccardo Corti, Antonio Mauro D’Agostino and Nicola Cionfoli in The Journal of Vascular Access
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
