Abstract
Background:
In elderly and diabetic patients, arterial calcifications are prevalent and result in worse outcomes of arterio-venous fistulas (AVFs). Optimal ultrasound criteria for assessment of calcified arteries are unknown. We report our experience with ultrasound assessment of calcified arteries prior to placement of radio-cephalic (RC) AVFs.
Material and methods:
We included 85 patients in whom a RC-AVF placement was planned. Patients were divided according to the presence of radial artery calcifications into a calcified group (moderate/severe calcifications,
Results:
In the calcified group mean patients’ age was 72 ± 11 years, 72% were diabetics, mean resistance index (RI) during RH was 0.78 ± 0.09 (range 0.63–0.90). In 14 (78%) patients an AVF was successfully placed and in 12 it matured (67% maturation rate). A lower RI at rest was observed in the primary failure sub-group with high area under the ROC curve (0.89) and a cut-off value of 0.90 for RI. There was no difference in RI at RH between failed and matured AVFs. 1-year secondary patency was 66% and in eight patients with follow-up ultrasound available, blood flow was 350–1300 ml/min. For comparison, in the control group a radio-cephalic AVF was placed in all patients, primary failure rate was 6%, maturation rate 85% and 1-year secondary patency was 77%.
Conclusions:
Our very limited experience suggests that successful RC-AVF placement and maturation with good 1-year patency rates is possible in patients with moderately/severely calcified arteries and RI during RH of up to 0.90.
Introduction
In patients approaching end-stage kidney failure ultrasound/Doppler examination of vessels prior to an arterio-venous fistula (AVF) placement is recommended1,2 and plays an important role in planning of surgical procedure and improving AVF outcomes.3–5 With increasing age and prevalence of diabetes, increased incidence of arterial calcifications can be expected,6,7 which makes AVF placement technically more challenging or even impossible. In case of successful placement, AVF may not mature, due to the inability of the feeding artery to provide sufficient inflow. Arterial calcifications were shown to adversely affect AVF blood flow 8 and long-term patency. 9
While ultrasound examination of the veins mainly includes confirming their presence and measuring their diameter, examination of the arteries is more complex and often changes the surgical plan. 4 When arterial calcifications are present, there are no established ultrasound Doppler criteria for evaluating the suitability of the artery for successful AVF placement in addition to its diameter. As a functional test that mimics conditions after anastomosis formation, resistance index (RI) during reactive hyperemia was suggested as a good Doppler index for assessing suitability of the artery and RI during reactive hyperemia >0.7 was associated with a poor outcomes. 10
The aim of this observational study was to report our experience with Doppler ultrasound assessment of calcified radial arteries, related radio-cephalic AVF outcomes and comparison to noncalcified arteries.
Patients and methods
This was a retrospective observational case-control study focusing on patients with calcified radial arteries in whom placement of an AVF was attempted. The study was performed in accordance with the Declaration of Helsinki and approved by the National Medical Ethics Committee (No. 0120-51/2021/6), informed consent was waived due to retrospective study design.
We screened all patients referred for mapping between Dec 2017 and Oct 2020 in whom a plan for radio-cephalic AVF placement was made. We included in the calcified group 18 patients with planned radio-cephalic AVF in whom a suitable cephalic vein and radial artery (by diameter criteria) were found, but the artery had moderate/severe calcifications with at least partially homogeneous color Doppler signal (see below). In patients with very patchy/absent color Doppler signal, no attempt at AVF placement was made. The control group consisted of 67 patients in whom a radio-cephalic AVF was planned after mapping but who had no/mild radial artery calcifications (Figure 1).
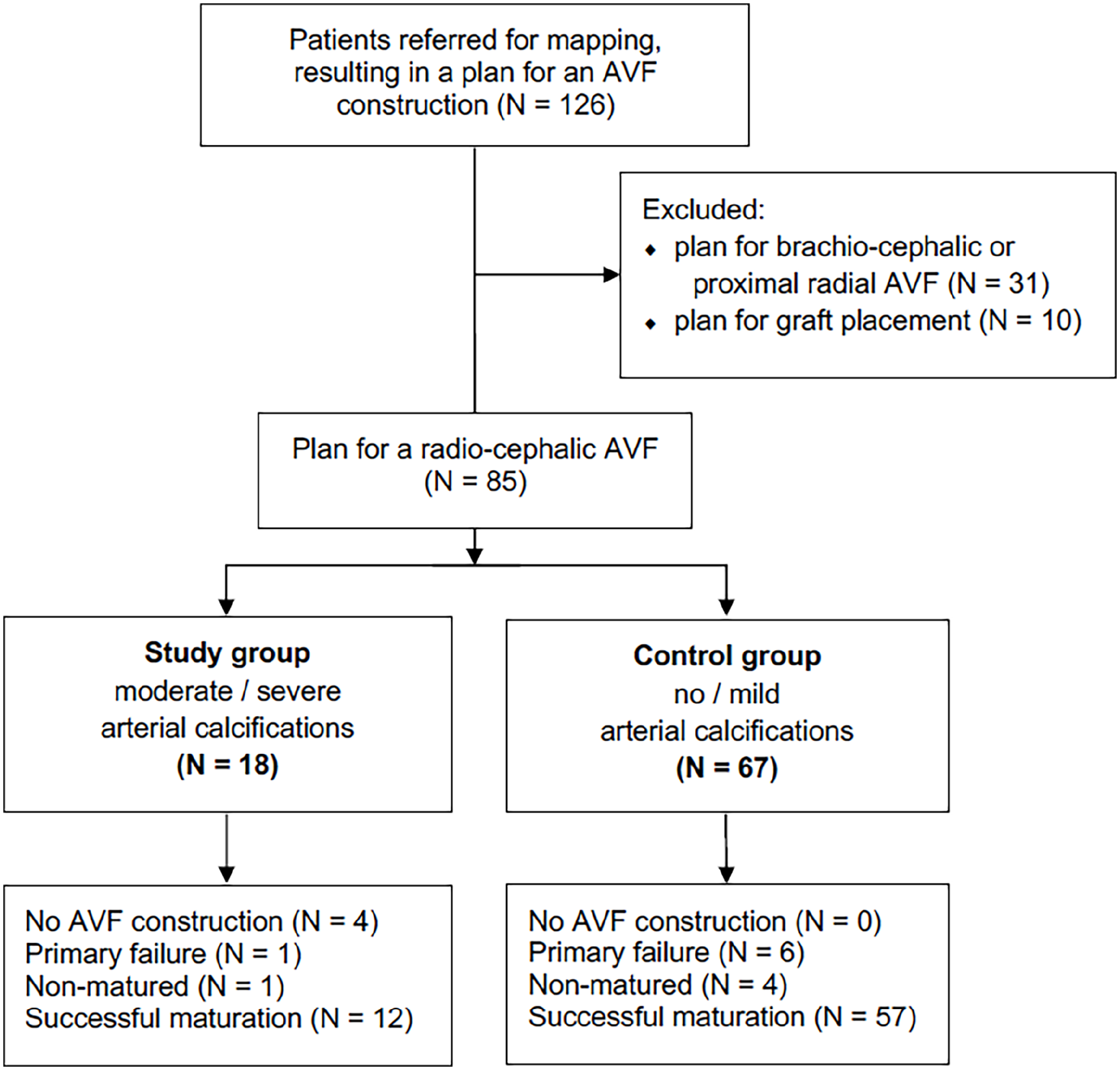
Patient selection flow chart and main outcomes.
All AVFs were placed under local anesthesia. In the calcified group, when the artery was exposed, a compressible part was found for clamps and anastomosis. If the arterial wall was completely noncompressible, no attempt was made to place an AVF. The size of the anastomosis was about 8–10 mm in both groups. Low-dose Aspirin was not discontinued before surgery.
The immediate outcome of AVF placement, clinical maturity of AVF, volume flow measurement (when available), and 1-year primary and secondary patency
11
were calculated using a Kaplan-Meier survival analysis. The analysis was by intention-to-treat principle; as the aim of the study was to identify ultrasound predictors of successful AVF placement, attempted surgeries, where no anastomosis was created because the artery was too calcified, were counted as primary AVF failures.
11
Descriptive statistics were used for other data, which are reported as mean ± standard deviation or frequency/percentage as appropriate. Calcified versus non-calcified groups and within calcified groups the matured AVFs versus primary failure (non-matured, thrombosed, and never created AVFs) sub-groups were compared with Student’s T test and
Ultrasound Doppler assessment
Diameter of cephalic vein (under tourniquet) and radial artery was measured with a linear probe (either 14 MHz probe, Acuson S2000, Siemens or 8 MHz probe, Vivid S6, General Electrics). Arterial wall was examined for presence of atherosclerotic changes. Based on clinical experience and other classifications in the literature,8,10,12,13 atherosclerotic changes were classified as “mild” if the arterial wall was hyperechogenic but without distal shadowing (mild atherosclerosis), “moderate” if the arterial wall was hyperechogenic and had more intense white spots with distal shadowing present (patchy calcifications), and “severe” if calcifications were continuous and had severe distal shadowing (linear calcifications). Lumen of the artery was examined with color Doppler13,14 and the signal was classified as “homogeneous” if the signal was present over the entire length of the artery, “partially homogeneous” if the signal was present along more than approximately two-thirds of the visible length of the artery, “very patchy” if less than two-thirds of the length of the artery had Doppler signal, or “absent” (Figure 2). In patients with calcifications, a pulsed wave Doppler signal was assessed (peak systolic velocity (PSV), end diastolic velocity (EDV) and resistance index (RI)) at rest and during reactive hyperemia (achieved by 30 s of fist clenching) (Figure 3). 10 All ultrasound examinations and surgeries were performed by the author.
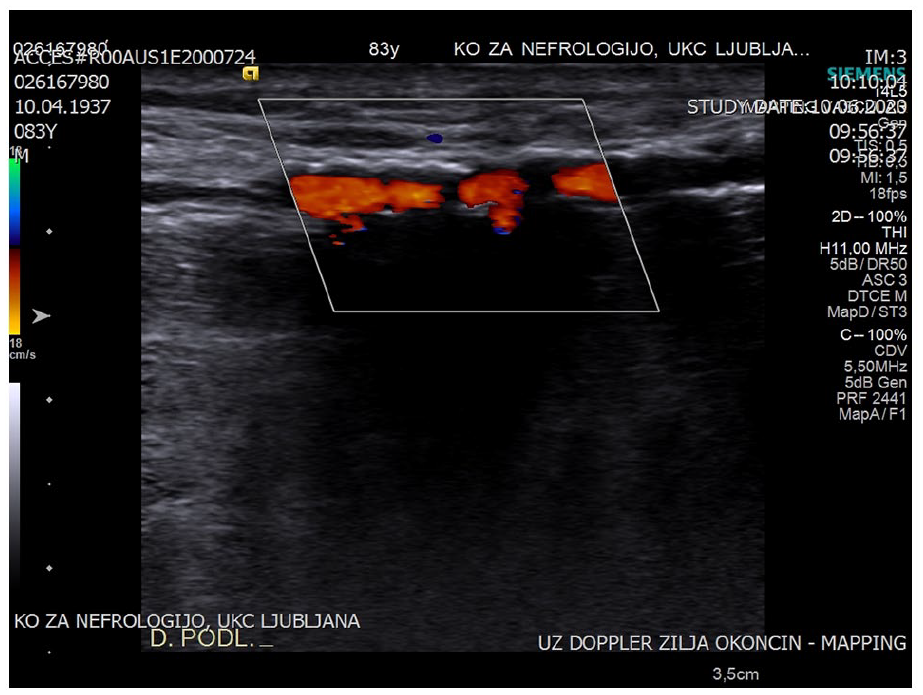
Ultrasound image of a severely calcified radial artery with linear calcifications (and distal shadow) and partially homogeneous color Doppler signal.
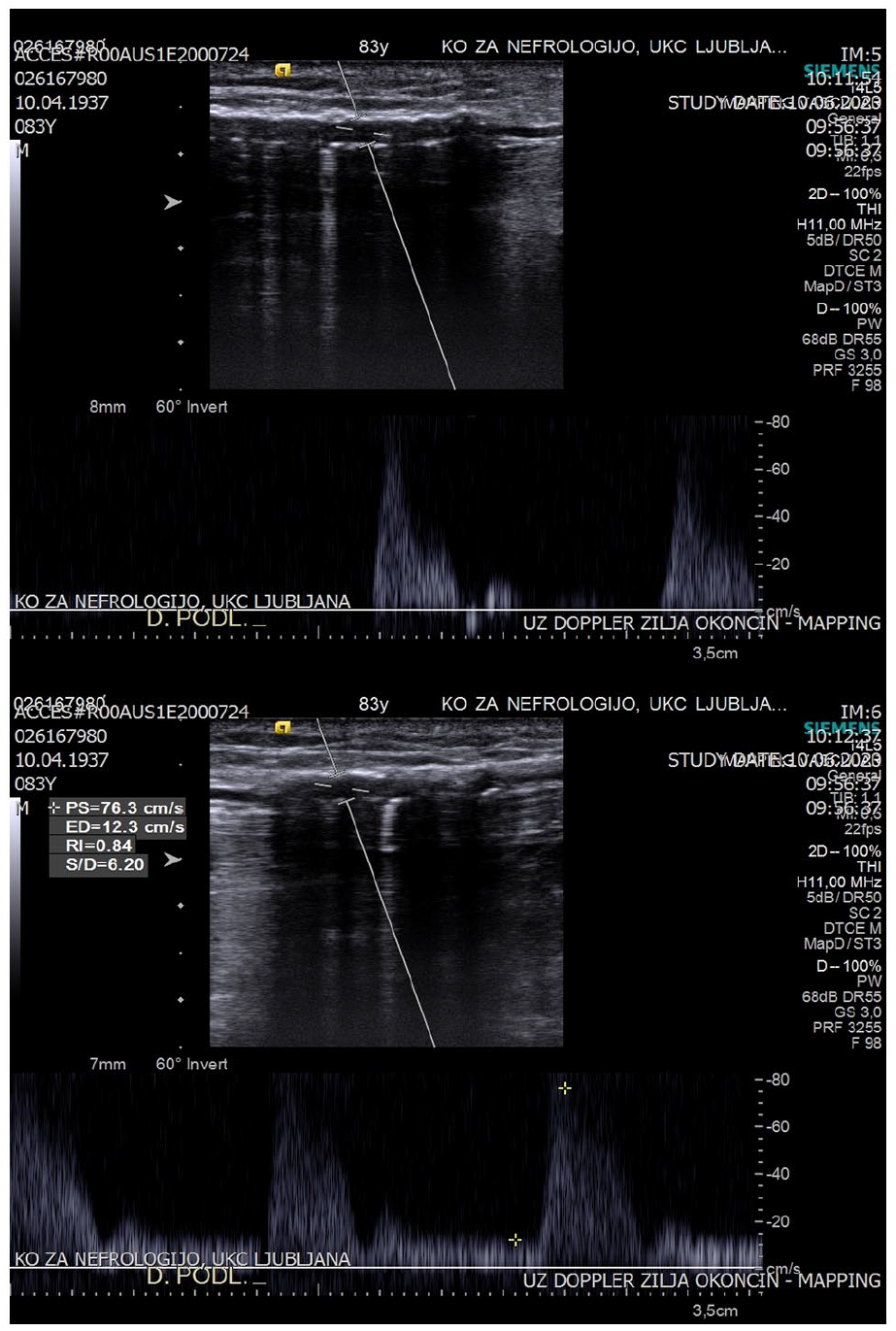
Pulsed wave Doppler exam of a radial artery showing triphasic, high-resistance curve with resistance index (RI) of 1.0 at rest and increased diastolic flow during reactive hyperemia (after 30 s of fist clenching) with a decrease in RI (RI 0.84).
Results
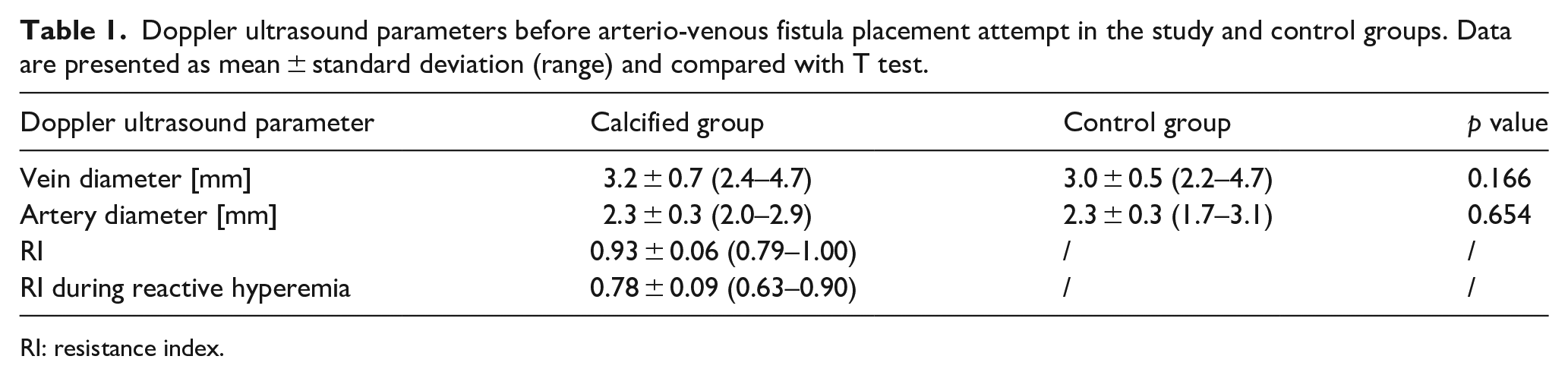
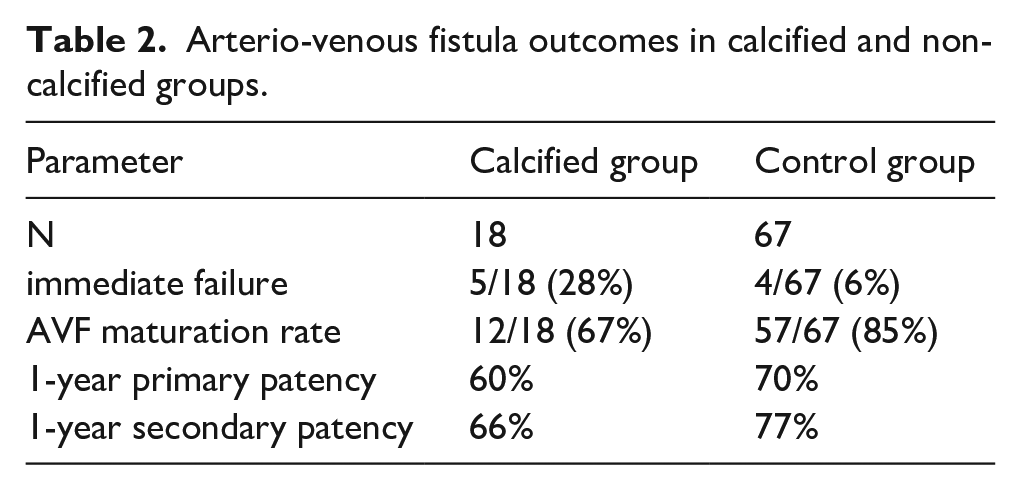
Altogether, 85 patients were included in the study, of them 18 patients in the calcified group (2 females, mean age 72 ± 11 years, 72% diabetics, 44% current/past smokers) and 67 in the control group (23 females, mean age 67 ± 14 years, 25% diabetics, 33% smokers). Doppler/ultrasound parameters of both groups are presented in Table 1. In 14/18 (78%) of patients in the calcified group, an AVF was successfully placed, while in the remaining four, the artery was too calcified to attempt making an anastomosis. Among 14 patients in whom an AVF was placed, there was one immediate AVF failure and another AVF failed to mature. The other 12/18 (67%) AVFs matured; 11 were successfully used for dialysis and one was clinically assessed as mature but was never used for dialysis as the patient had a preemptive kidney transplantation. Follow-up Doppler ultrasound was available in 8/18 (44%) patients, which was performed between 10 days and 6 months after AVF placement. Median measured flow volume through the feeding brachial artery was 600 (range 350–1300) ml/min. After 1 year, two patients died with working AVF, one needed an angioplasty, nine had a working AVF. The Kaplan-Meier estimated 1-year primary patency was 60% and secondary patency was 66% (Table 2, Figure 4). There were no cases of clinically significant steal syndrome. In the control group, an AVF was placed in all patients, primary failure rate was 6% and maturation rate was 85%. After 1 year, one patient died, of the other 56 functional AVFs, four were abandoned/thrombosed and three required an angioplasty, thrombectomy or re-anastomosis. Resulting Kaplan-Meier estimated 1-year primary patency was 70% and secondary patency was 77% (Table 2, Figure 4).
Doppler ultrasound parameters before arterio-venous fistula placement attempt in the study and control groups. Data are presented as mean ± standard deviation (range) and compared with T test.
RI: resistance index.
Arterio-venous fistula outcomes in calcified and non-calcified groups.
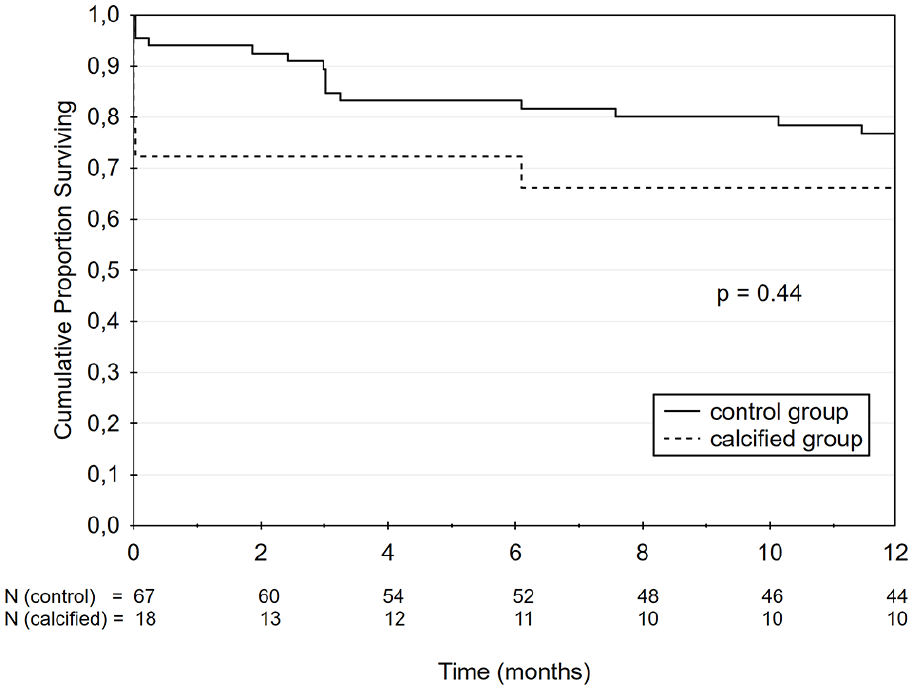
Kaplan-Meier analysis of secondary patency for calcified and non-calcified group.
In the calcified group, we further compared AVFs that matured successfully to those that had primary failure (were not placed, thrombosed or unmatured) (Table 3). There was no difference in vessel diameters or RI during reactive hyperemia. Rather, there was a significant difference in the initial RI and EDV at rest with lower RI and higher EDVs observed in the primary failure group. ROC curve analysis (Figure 5) showed that RI had higher area under the curve (AUC 0.89) than EDV (AUC 0.84). The optimal cut-off value for RI at rest predicting primary failure was 0.90, with sensitivity of 0.80, specificity of 0.83, positive predictive value of 0.67 and negative predictive value of 0.91.
Comparison of the Doppler/ultrasound parameters between the subgroup with matured arterio-venous fistula (AVF) and primary failure (not placed, thrombosed or non-matured) within the calcified group. Data are presented as mean ± standard deviation (range) and compared with Student’s T test.
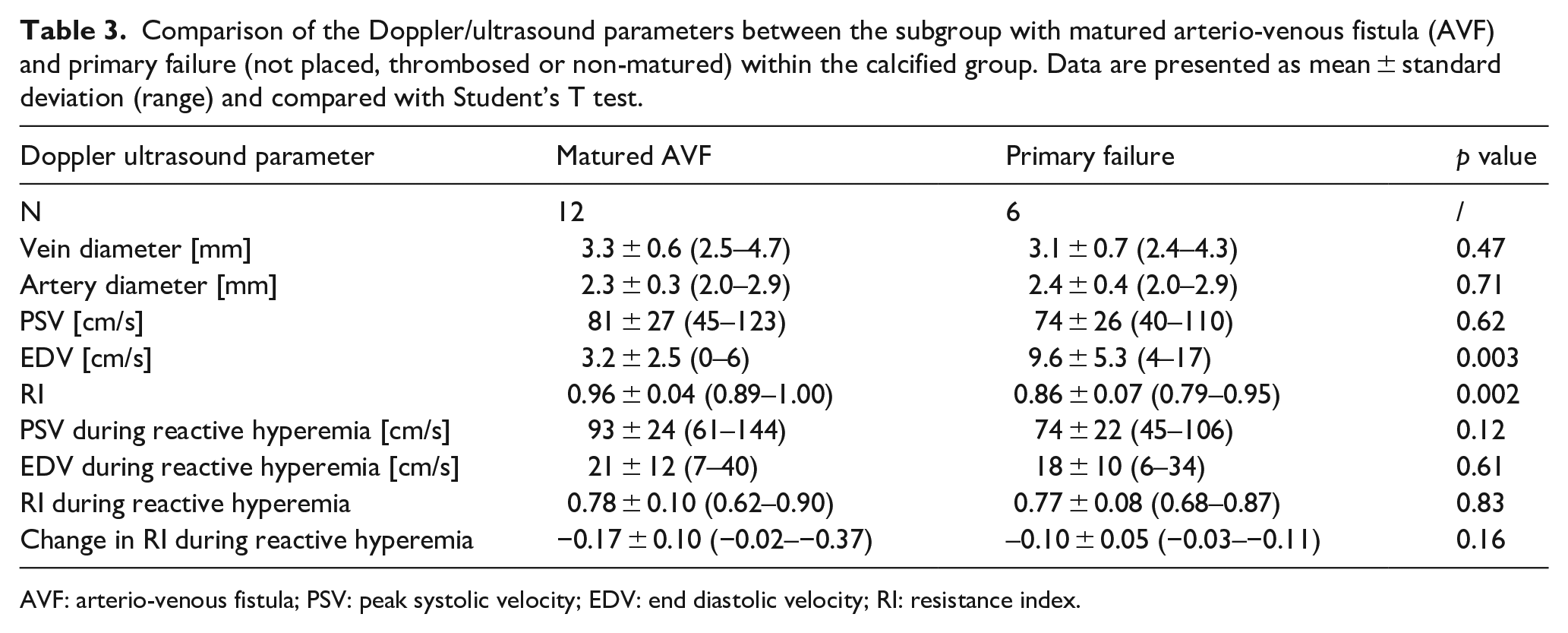
AVF: arterio-venous fistula; PSV: peak systolic velocity; EDV: end diastolic velocity; RI: resistance index.
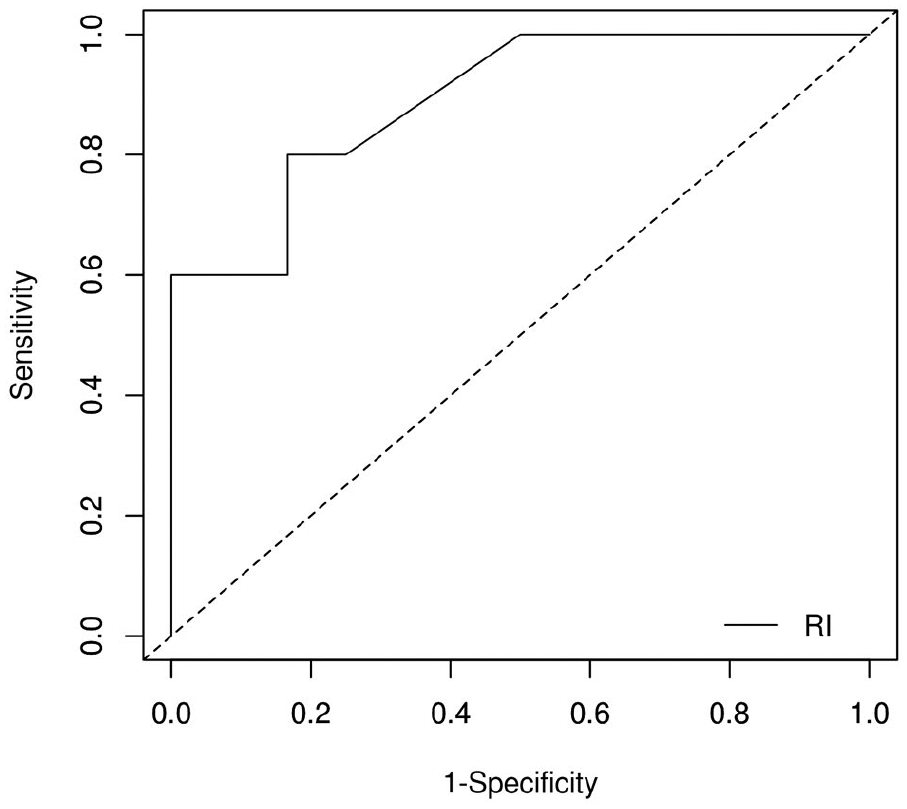
Receiver-operator characteristic (ROC) curve for initial radial artery resistance index (RI), predicting primary arterio-venous fistula failure in the calcified group.
Discussion
Our very limited experience suggests that a significantly higher RI during reactive hyperemia (up to 0.90) than has previously been suggested (>0.70) can confirm the ability of the artery to provide sufficient flow for AVF. RI at reactive hyperemia was not able to discriminate failed and successful AVFs, while a low initial (“at rest”) RI was associated with primary failure. Finally, visual assessment of the continuity of the color Doppler signal may also provide an assessment of the severity of calcifications.
Arterial calcifications are more common in patients with diabetes, advanced age and long-standing kidney disease, and adversely affect cardiovascular morbidity and mortality. 15 There are relatively few reports in the literature on AVF outcomes in patients with significant macroscopic arterial calcifications and even in larger studies, the number of patients with significant calcifications actually present is low. Moderate, spotty calcifications were shown to provide good clinical outcomes with no effect on clinical maturation and moderate reduction in blood flow. 8 Severe calcifications, on the other hand, are often considered at least a relative contraindication for a distal AVF.16,17 When AVF is placed under such conditions, a significantly worse primary patency rates have been reported as compared to noncalcified arteries, but still reaching 50% at 12 months, 9 which in our opinion makes it a worthy attempt. Furthermore, the secondary patency, which is even higher, is important for the patient, as some of the non-matured AVFs can be salvaged with secondary procedures. A brachio-cephalic AVF can result in distal ischemia even in the absence of high-flow AVF. 18 On the other hand, no distal ischemia was reported in distal AVFs despite presence of calcifications. 9 An anastomosis to the proximal radial or ulnar artery is also an excellent possibility in these cases.
Ultrasound/Doppler assessment of calcified arteries and related clinical decision making is often not elaborated on in the literature. When is an artery too calcified to provide sufficient inflow for an AVF? Vascular access guidelines do not give any recommendation1,2,19 and the new European guidelines do not even discuss pre-operative issues. 20 In addition to a suitable diameter, which is a precondition, a relatively homogeneous color Doppler signal might indicate that the arterial wall is still penetrable for ultrasound and therefore not completely calcified. Furthermore, the artery should be able to provide sufficient inflow. 21 To mimic conditions after creation of an anastomosis, the use of functional tests have been proposed, at least in borderline cases.10,22 RI is less prone to measurement errors as absolute velocities (PSV, EDV), 22 therefore RI during reactive hyperemia might provide the best functional assessment of the artery. Malovrh 10 showed that RI during reactive hyperemia ⩾0.70 was a predictor of poor outcome (40% vs 95% success rate). Our preliminary and limited results suggest that successful placement of AVF with outcomes that compare favorably with noncalcified arteries is possible even with a much higher RI during reactive hyperemia than 0.70 (up to 0.90).
Furthermore, a new potential prognostic Doppler parameter, namely the initial (“at rest”) RI of the radial artery, was identified for the calcified group with a cut-off value of <0.90. A lower PSV, higher EDV and resulting lower RI is the typical Doppler pattern occurring distal to hemodynamically significant arterial stenosis, 23 presumably as a result of distal vasodilation in the ischemic area. 23 However, RIs we observed (minimum value of 0.79) are higher than typical post-stenotic values 23 and the PSVs in our calcified group were comparable to those in the non-calcified group. Therefore, it is unlikely that our Doppler findings resemble hemodynamically significant proximal stenosis as a cause of AVF failure. Rather, we suggest that a long calcified segment of the artery produced a “cumulative stenosis” that was only relative (hemodynamically non-significant) and therefore produced a comparatively lower RI compared with calcified arteries with successful AVF maturation. Given very low number of patients, these observations should be considered hypothesis-generating and need to be confirmed in larger prospective studies.
Limitations of the study
Limitations of our study include its observational design and small number of patients; the results should therefore be considered preliminary. In depth Doppler assessment was only made in the presence of arterial calcifications. It should be noted that vessel diameters in our study were higher than required by the guidelines, although comparable in both groups, which might have contributed to the relatively good outcomes observed.
Conclusions
Successful AVF placement and maturation is possible with good 1-year patency rates in patients with moderately/severely calcified radial arteries of a suitable diameter, relatively homogeneous color Doppler signal and RI during reactive hyperemia of up to 0.90, much higher than the literature suggests. Although functional tests might seem better at discriminating poor AVF outcomes, a low initial RI of the radial artery was associated with primary AVF failure in this study. Further prospective studies are needed to confirm these findings in this challenging subgroup of patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author acknowledges the financial support from the Slovenian Research Agency (research core funding No. P3-0323).
Ethical approval
The study was approved by the National Medical Ethics Committee (No. 0120-51/2021/6), informed consent was waived due to retrospective study design.
