Abstract
Rationale and objective:
This study aimed to develop a cosmesis scale to evaluate the cosmetic appearance of hemodialysis (HD) arteriovenous (AV) accesses from the perspective of the patient and clinician, which could be incorporated into clinical trials.
Study design:
Using a modified Delphi process, two AV access cosmesis scale (AVACS) components were developed in a four-round Delphi panel consisting of two surveys and two consensus meetings with two rounds of patient consultation.
Setting and participants:
The Delphi panel consisted of 15 voting members including five interventional or general nephrologists, five vascular surgeons, three interventional radiologists, and two vascular access nurse coordinators. Four patients experienced with vascular access were involved in patient question development.
Analytical approach:
For a component to be included in the AVACS, it had to meet the prespecified panel consensus agreement of ⩾70%.
Results:
The clinician component of the AVACS includes nine questions on the following AV access features: scarring, skin discoloration, aneurysm/pseudoaneurysms and megafistula appearance. The patient component includes six questions about future vascular access decisions, interference with work or leisure activities, clothing choices, self-consciousness or attractiveness, emotional impact, and overall appearance.
Limitations:
Delphi panel methods are subjective by design, but with expert clinical opinion are used to develop classification systems and outcome measures. The developed scale requires further validation testing but is available for clinical trial use.
Conclusions:
While safety and efficacy are the primary concerns when evaluating AV access for HD, cosmesis is an important component of the ESKD patient experience. The AVACS has been designed to assess this important domain; it can be used to facilitate patient care and education about vascular access choice and maintenance. AVACS can also be used to inform future research on developing new techniques for AV access creation and maintenance, particularly as relates to AV access cosmesis.
Keywords
Introduction
In the United States, an estimated 500,000 patients were undergoing hemodialysis treatment by the end of 2019, with an estimated 113,000–115,000 incident end-stage kidney disease (ESKD) patients initiating hemodialysis. 1 To provide access to hemodialysis, the Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines recommend arteriovenous (AV) access (either AVF or AVG) over a CVC due to the lower rate of infections associated with AV access use. 2 However, up to 30% of patients are estimated to refuse the creation of an AVF for a variety of reasons including poor previous personal or vicarious experiences with AVF and personal or perceived lack of complications from other vascular accesses. 3
Cosmesis in the context of hemodialysis AV access is defined as the preservation of, or change in, limb physical appearance after AV access creation. It has been reported that cosmesis has an impact on vascular access decision making and the cosmetic appearance of the vascular access is a key concern for patients.3–6 A systematic review of 46 qualitative studies that explored expectations among patients with any-stage chronic kidney disease identified disfigurement from having a vascular access as one of the key themes among the types of patient concerns identified. 4 Patients reported strong preferences to preserve normal body appearance, to avoid stigma from having a visible vascular access, and to avoid an obvious reminder of their health condition from having a vascular access. 4
There is a paucity of reports about the effect of AV access cosmesis on patients’ quality of life, dialysis experience, and decision making about choice of vascular access type and creation; furthermore, the importance of developing patient reported outcome measures (PROMs) to assess this has been raised. 6 By understanding which factors are associated with better or worse cosmesis for various AV access types, both patients and vascular access care teams can make more fully informed decisions around access creation and utilization, and with more accurate expectations. However, an important limitation for research in this area is the lack of a standardized instrument that specifically assesses cosmesis of vascular accesses. Thus, the objective of this study was to develop a cosmesis scale for evaluation of hemodialysis arteriovenous accesses from the perspective of the patient and clinician, which could be incorporated into clinical trials.
Methods
A Delphi process is a structured communication technique to address complex issues where gaps in knowledge or problems with uncertain solutions are evaluated by experts in an iterative manner; key components include anonymity, multiple rounds of consensus building, and coordinated structured processes for information gathering and feedback. 7 This study used a modified Delphi approach where a literature review and information gathering surveys were used to inform topics for consensus building through multiple rounds, which included online and virtual face-to-face rounds. Questions to include in the scale were determined by consensus voting at a Delphi panel meeting. Surveys were repeated to refine the cosmesis scale with a second Delphi panel meeting convened to finalize the scale.
The goal was to develop a scale that can be completed in a reasonable timeframe, can be easily integrated into clinical trials, and is reproducible with a low inter-rater variability. For a comprehensive assessment of the cosmetic appearance of AV access, a scale with two components was designed with a clinician component and patient component. The clinician component was developed specifically for clinicians to assess objective aspects of cosmetic appearance. The patient component was developed specifically for patients to assess more subjective aspects related to appearance and perception of the AV access. Figure 1 shows an overview of the Delphi process for cosmesis scale development.
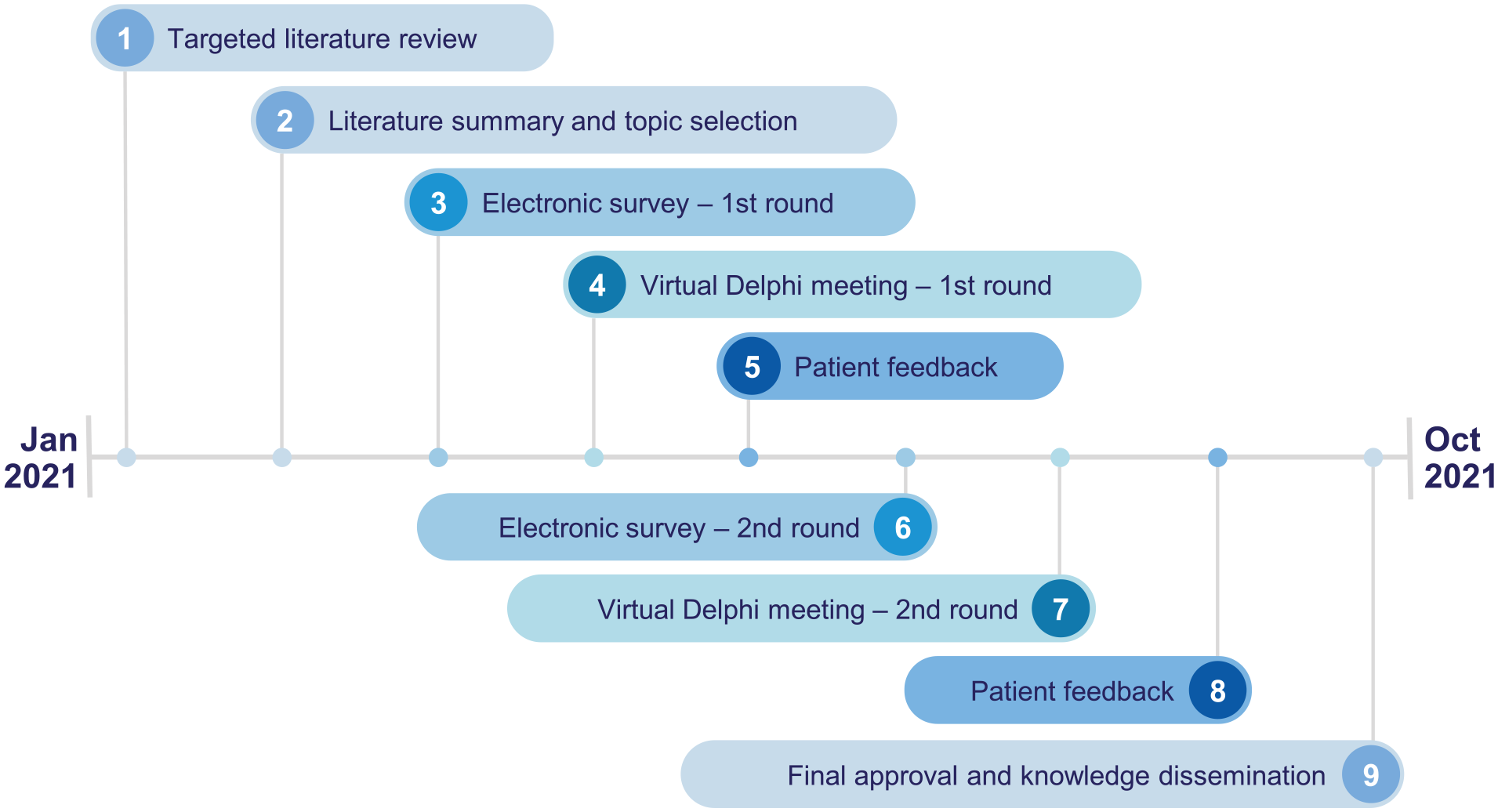
Overview of the Delphi panel process for arteriovenous access cosmesis scale (AVACS) development.
Participant selection and recruitment
A multidisciplinary steering committee was established and consisted of a vascular surgeon, interventional radiologist, and nephrologist with expertise in the management of hemodialysis vascular access and hemodialysis patients. Expert panelist selection was based on the goal of having a multidisciplinary team with members from various geographic regions, a mix of academic and community practices, and different clinical specialties represented. Twelve panel members were invited (four vascular surgeons, four interventional/general nephrologists, two interventional radiologists, and two vascular access nurse coordinators), for a total of 15 voting members in the Delphi panel. An additional four American Association of Kidney Patients (AAKP) Patient Ambassadors participated on topics important to patients and provided input to ensure patient-friendly wording of scale questions and answers for the patient scale. The AAKP Patient Ambassadors also reviewed and approved the final patient scale questions after their feedback was implemented and confirmed by the Delphi panel.
Data collection
Literature review
A targeted literature search was performed in PubMed to identify cosmesis scales and assessment instruments from a wide variety of areas including plastic surgery, hepatopancreatobiliary surgery, and vascular diseases (See Supplement 1 for search terms used). Key cosmesis categories of interest were identified by the steering committee upon which scale questions and potential responses were developed.
Survey 1
Panelists were sent an online survey with a list of potential cosmesis scale topics drafted by the steering committee and asked to rank the importance of each topic on a scale of 1–5: 1 = very important, 2 = important, 3 = neutral, 4 = low importance, 5 = not important at all. The online survey also included proposed scale questions and answers derived from the literature review and steering committee, as above. Panelists were asked to provide their level of agreement with inclusion of the proposed questions in a scale for assessing AV cosmesis. Responses were provided on a Likert scale of 1–5 where 1 = strongly agree, 2 = agree, 3 = neither agree nor disagree, 4 = disagree, and 5 = strongly disagree. Panelists with a response of “neutral” to “strongly disagree” were asked to suggest how the question could be revised/reworded for improvement. Panelists with a response of agree or strongly agree were given an opportunity to refine the response categories of the scale questions. Panelists were also able to suggest any additional topics not originally included in the scale that could be important to assess AV cosmesis. Consensus was defined a priori as greater than 70% of the panelist responses being either “agree” or “strongly agree” on a 5-point Likert scale.
Virtual meeting 1
The steering committee shared the initial results from the first survey during virtual meeting 1. This was followed by a robust discussion on the results and suggested revisions to the scale questions were made. Minor changes to the questions were made only after agreement was obtained by virtual consensus polling where the panelists provided anonymous responses. Consensus was defined a priori as greater than 70% of the panelist responses being either “agree” or “strongly agree” on a 5-point Likert scale.
Patient feedback 1
AAKP Patient Ambassadors provided feedback via individual interviews on the patient scale. Interviews were conducted virtually via Microsoft Teams by NF and GW using a standardized slide deck with draft versions of the patient component questions for discussion. Patient Ambassador feedback was recorded via Teams for subsequent review and incorporation into the patient component of the scale. The Patient Ambassadors provided input on the wording of the scale questions and response categories and suggested additional AV access cosmesis topics from the patients’ perspective. They were asked for their input on whether specific cosmetic features, such as bruising, should be included in the scale questions.
Survey 2
Panelists were sent a second online survey in which they were asked whether each question should be included in the scale with the following three response categories: agree, agree with revisions, or disagree. Similar to the first survey, panelists could also suggest any additional topics important to assess AV cosmesis that were not included in the scale if they responded, “agree with revisions.” Consensus was defined a priori as greater than 70% of the panelist responses being “agree” for inclusion of the question.
Virtual meeting 2
Led by the steering committee, results from the second survey were shared with the panelists in the second virtual meeting. As in the first virtual meeting, this was followed by a discussion on the results and suggested revisions to the scale questions. Anonymous virtual consensus polling was conducted to reach agreement on the revised scale. Consensus was defined a priori as greater than 70% of panelist responses being either “agree” or “strongly agree” on a 5-point Likert scale.
Patient feedback 2
The revised patient scale was then shared with the AAKP Patient Ambassadors for their final feedback and approval. In the final meeting with the Patient Ambassadors via teleconference, they were asked whether the revisions captured their suggestions, and whether they were satisfied with the overall patient scale. Responses were captured via anonymous electronic voting with response categories of “yes” or “no.”
Final approval
All members of the Delphi panel confirmed final approval on both scales.
Results
Several key cosmesis topics were consistently identified from the targeted literature review, steering committee discussions, and clinical discussions during the Delphi process. The literature review also identified several scales on the cosmetic appearance of scars broadly applicable to multiple specialties.8–10 The identified key topics were ranked in the following order of importance based on the average score from the first panelists survey: lumps/bulging and/or related tortuosity (1.13), oversized vascular access (e.g. “mega access”) (1.20), combined score from all questions (1.47), AV access-related scarring (1.60), noticeability of the vascular access (1.60), overall satisfaction with appearance (1.60), number of scars (1.93), localized swelling (2.00), pigmentation and discoloration (2.07), scarring length (2.13), scarring height (2.13), scarring width (2.20), and bruising (2.47). Bruising, due to its temporary nature, was rated lowest in importance by the panelists whereas lumps/bulging and/or related tortuosity were rated highest. Additional topics suggested by the panelists that were discussed and evaluated throughout the Delphi process included location of AV access, cannulation patterns, dialysis needle scabs, keloid formation, skin surface area of the disfiguration, permanence of the disfiguration, ability to hide disfiguration, significant whole-arm swelling, and comments or feedback from other people about the AV access appearance.
Through the Delphi process, panelists discussed and reached agreement that the scale should focus on the current AV access. Scarring was confirmed to be one of the most critical topics in both patient and clinician components of the scale, with the patient component assessing the perceived impact of the scars, and clinician component assessing objective measures such as the number and size of the scars. The panel agreed that the clinician component should also include the assessment of visible skin discoloration related to the current AV access. Through lengthy discussions across the surveys and meetings, it was determined bruising was not appropriate for inclusion in the clinician or patient components given its transient nature and occurrence dependent on cannulation skill, concurrent anticoagulant medications, and other patient-specific factors.
Patient input from AAKP Patient Ambassadors agreed that the proposed scale questions were relevant. They also provided suggestions on the wording of several scale questions to improve comprehension. These suggestions included adding directionality to the question on decision regarding future vascular accesses, adding the topics of attractiveness and emotional impact to the questions, and adding the clause about “disguising the AV access” to the question on avoiding certain types of clothing. All suggestions were integrated into the scale.
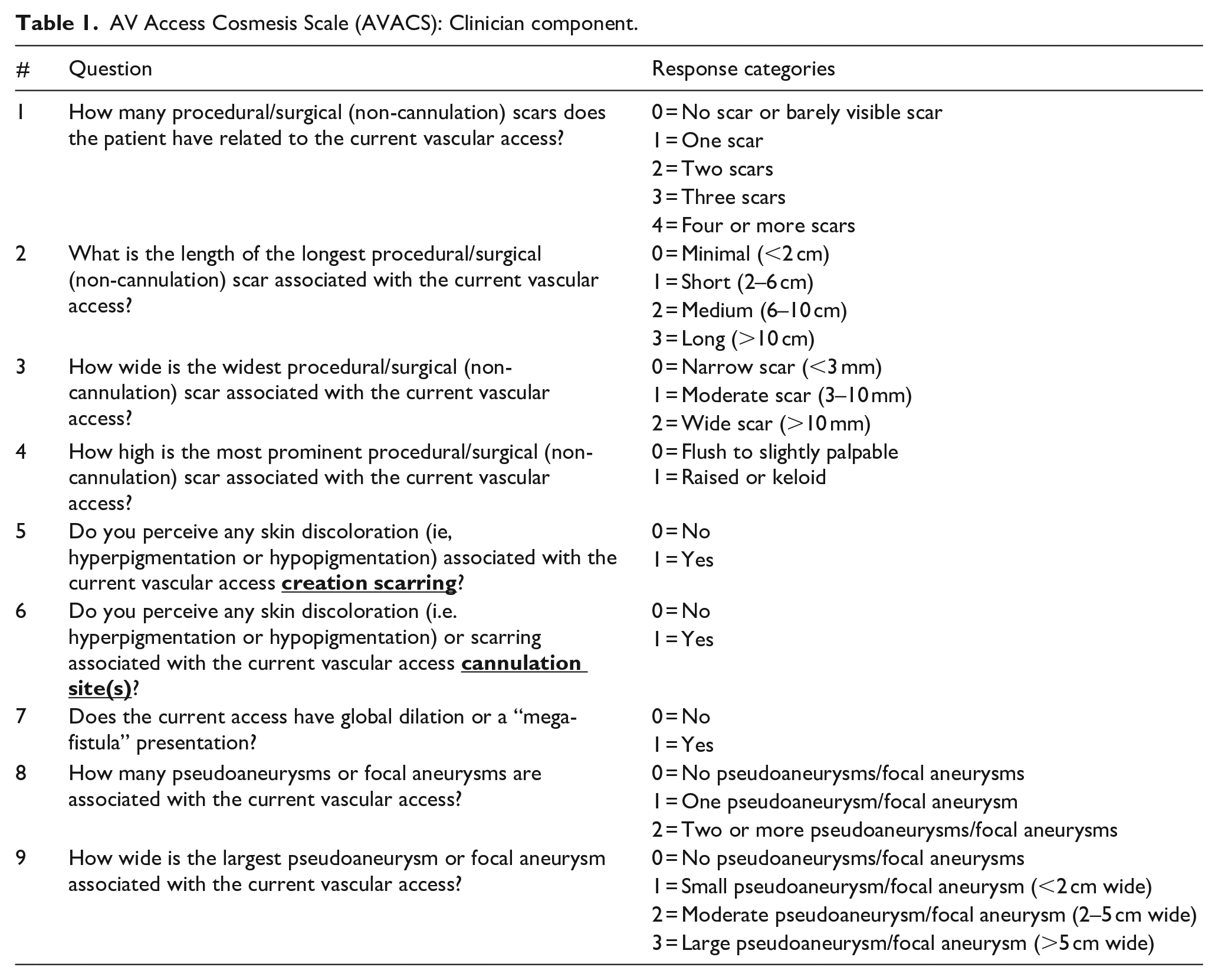
Over the course of scale development involving the two surveys and two Delphi panel meetings, 56 versions of questions for the patient and clinician components were polled for inclusion. The surveys, Delphi panel meetings, and two rounds of consultation with the Patient Ambassadors led to the development of clinician and patient components of the cosmesis scales called the AV Access Cosmesis Scale (AVACS), presented in Tables 1 and 2, respectively.
AV Access Cosmesis Scale (AVACS): Clinician component.
AV Access Cosmesis Scale (AVACS): Patient component.

In order to illustrate use of the AVACS Clinician Component, multiple example images are given and scored in Figure 2. Different methods for AV access creation result in permanent scars of various sizes and locations on a patient’s upper extremity. Single-catheter percutaneous or radiocephalic AVF that do not require superficialization procedures may result in relatively small surgical scars (Figure 2(a) and (b)). In contrast, a patient requiring secondary surgical procedures typically generates additional scarring; as an example in Figure 2(c) is a transposed brachiobasilic AVF in which the vein harvest incision wound broke down and created a broad cicatrix. Similarly, multiple incisions required for ligation of competing outflow branches will generate additional scars [not shown]. Figure 2(d) depicts an example of a brachiocephalic AVF that has degenerated into a mega-fistula.
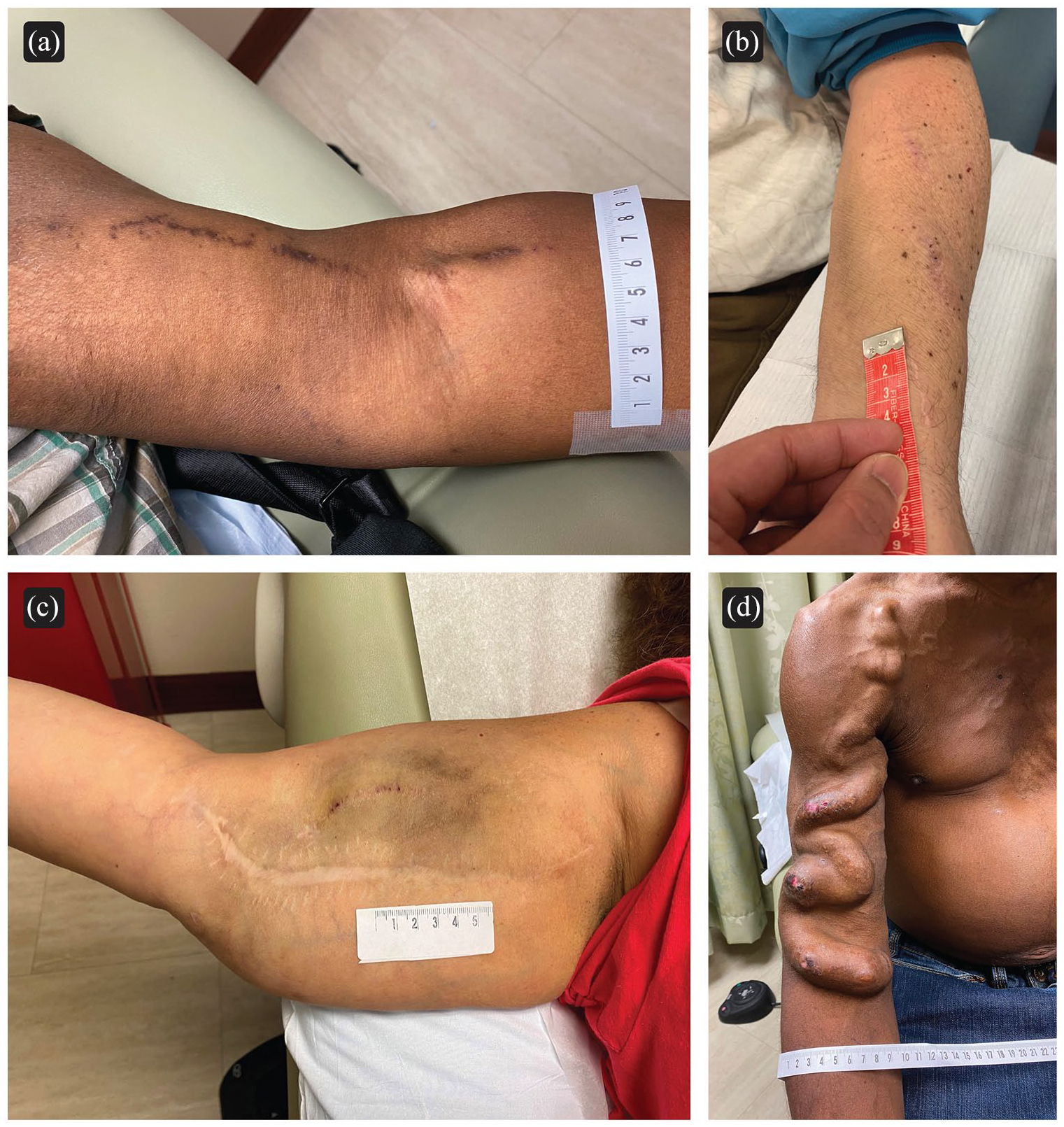
Four examples of AV accesses with varying levels of cosmesis: (a) AVF created with single catheter percutaneous technique. This was scored 0,0,0,0,0,1,0,0,0 = 1/18, (b) Radiocephalic surgical arteriovenous fistula. This was scored a 1,1,0,0,0,0,0,0,0 = 2/18, (c) Brachiobasilic AVF shown after surgical transposition. This was scored 1,3,2,0,1,1,0,0,0 = 8/18, and (d) Brachiocephalic arteriovenous megafistula. This was scored 1,1,0,0,1,1,1,2,3 = 10/18.
A more detailed explanation of the AVACS clinician component scoring is provided by Figure 3 which shows an endovascular AVF (endoAVF) created with a two-catheter percutaneous technique accessed at the wrist. While the endoAVF is cannulated in the basilic vein, there are no visible scars, and therefore no points are given for the first five questions. A point is given for question 6 due to the presence of skin discoloration at the cannulation sites. Otherwise, no points are given for the remaining questions (7–9), since no aneurysms are present. A similar analysis applies for the percutaneous AVF created with the single catheter technique (Figure 2(a)), and so it is also graded 0,0,0,0,0,1,0,0,0 = 1/18. Descriptions of Figure 4, and Supplemental Figures 1 and 2 and their respective scoring rationales are in Supplement 2.
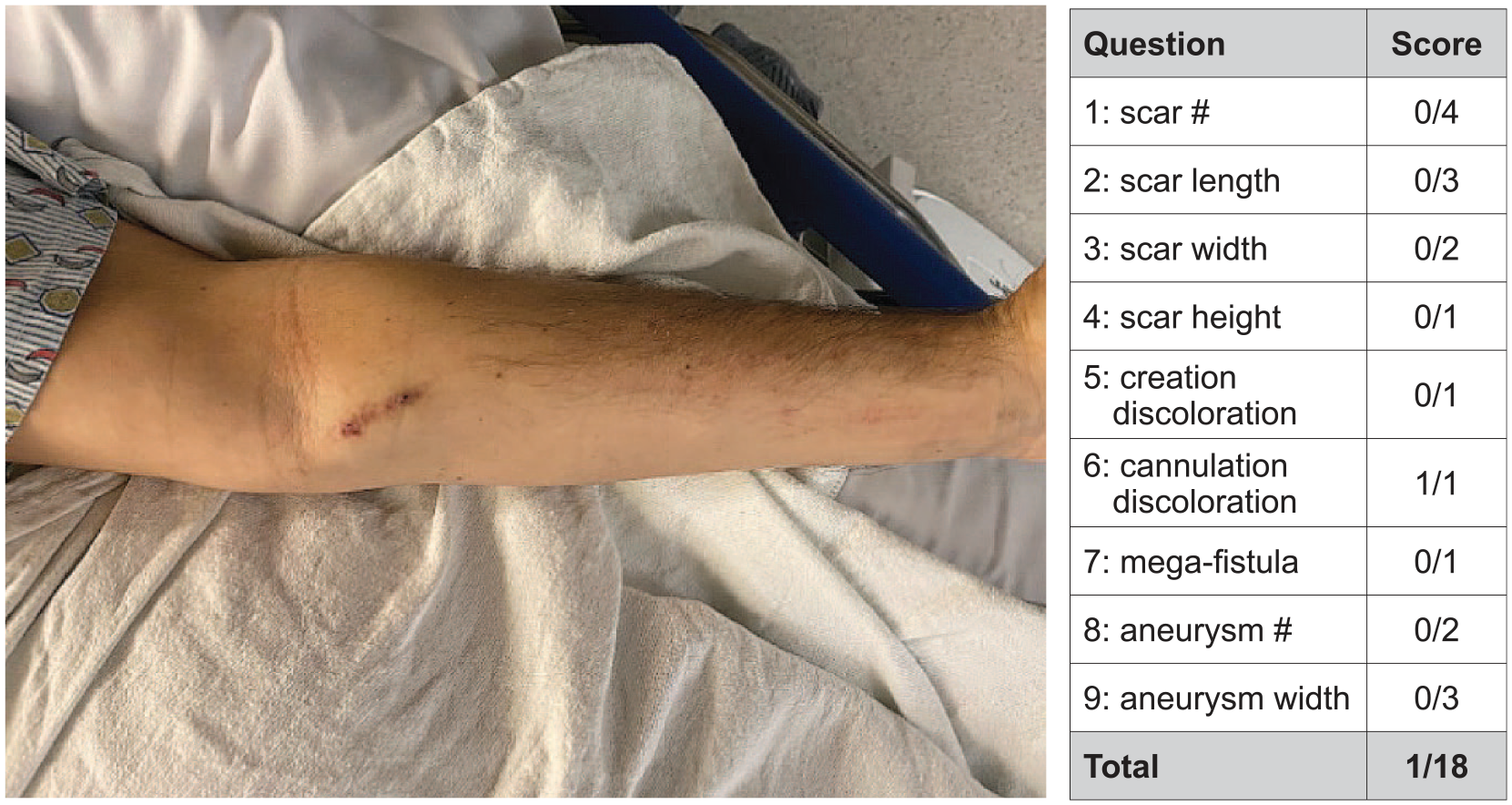
AVF created with two catheter percutaneous technique.
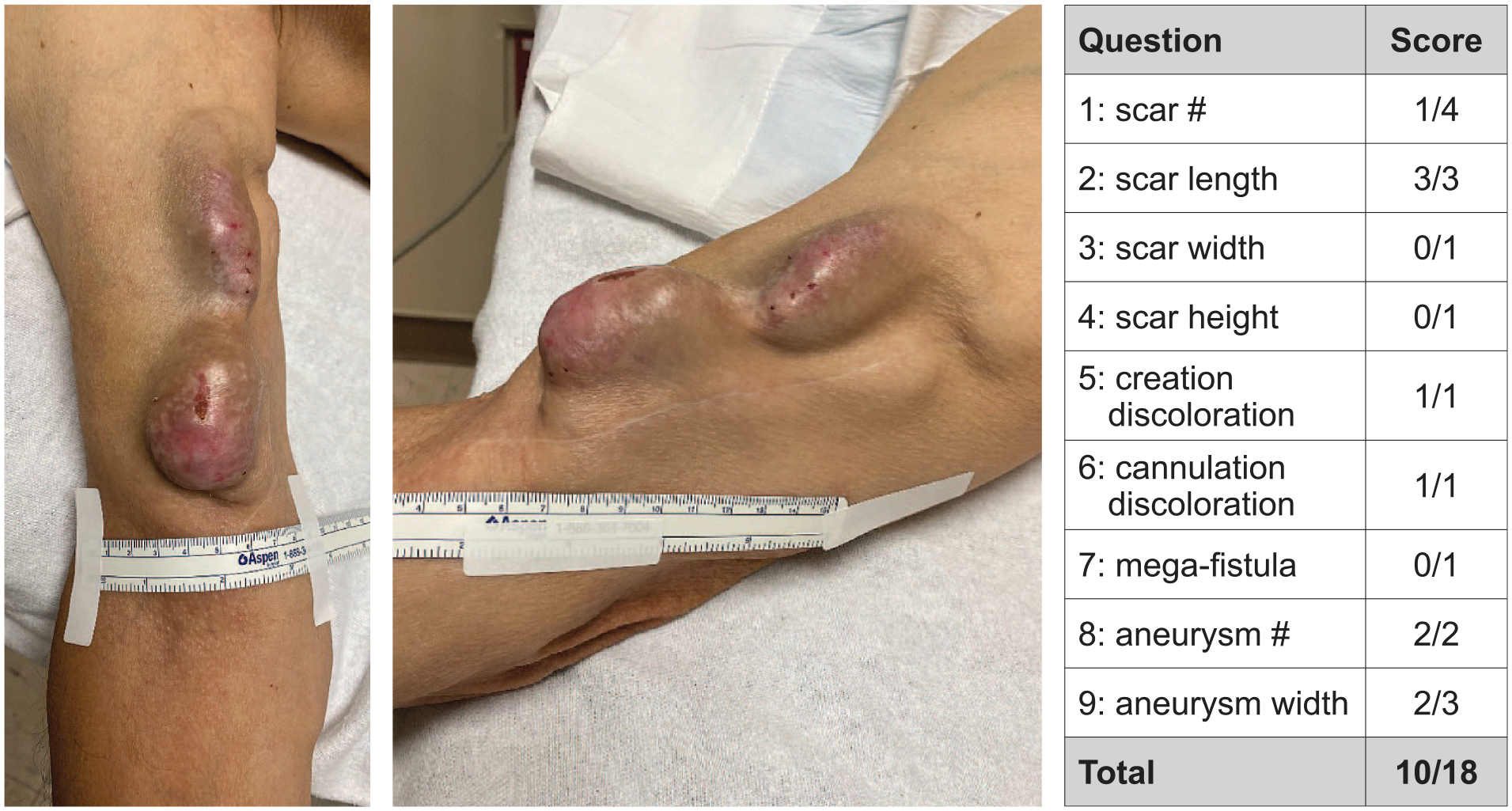
Basilic vein transposition AVF.
Discussion
The main outcome of our multidisciplinary efforts was the design and creation of a cosmesis scale that could be used to evaluate various forms of arteriovenous access for hemodialysis. The final AVACS consists of a clinician component, which is designed to be objective and quantitative, and a patient component, which explicitly incorporates the patient’s subjective component of the cosmesis experience. The cosmesis experience of an ESKD patient receiving an AV access includes the various effects that the appearance of a vascular access has on the patient and the way patients perceive and respond to changes in their appearance after an AV access is created. The clinician component includes nine questions that cover aspects of scarring, discoloration, cannulation scarring, pseudoaneurysms, focal aneurysms and mega fistula presentation. The patient component includes six questions that cover aspects of decision making regarding future vascular access, work and leisure activities, self-consciousness about the AV access, clothing and disguising of the AV access, emotional impact of the AV access, and an overall rating of the AV access appearance. The clinician component should be completed by relevant physicians, nurses, or vascular access coordinators and the patient component should be completed by patients themselves. Both components can be completed at any point in the AV access lifecycle.
In developing AVACS, we used a modified Delphi approach, which has been utilized frequently for the development of PROMs.11–13 We found that key issues became important depending on the timeline of the AV access lifetime. For example, with AV access creation, one key issue was the cosmesis effect of scarring, which was deemed important by both clinicians and patients. Scarring associated with cannulation and aneurysm development is also captured in the scale and will likely be present regardless of the technique used to create the AV access. Over the lifetime of an AV access, the presence of lumps, bulging and related tortuosity associated with the body of the fistula AV access were identified as key cosmetic features of AV access that should be captured. In addition to the timeline of the AV access, the permanence of the cosmetic features was an important topic; panelists agreed that bruising, while cosmetically displeasing and often associated with failed cannulation, is typically transient and should not be included in the scale. Similarly, the panelists felt that whole arm swelling, most often related to central venous stenosis and can often be treated, should also not be included.
While several quality-of-life instruments and vascular access focused questionnaires have been previously published, the negative cosmetic impact of AV access is not well understood. The VASQoL defined by Richarz et al. 14 recognizes the impact of cosmesis, but only investigated this at a high level addressed by a single question. The vascular access questionnaire (VAQ), 5 a measure of patient satisfaction with their vascular access, covers a variety of topics related to problems or complications with the access as well as negative effects of appearance or function of the access on the patient’s activities and hemodialysis routines. However, like the VASQoL scale, only one cosmesis-related question was asked (Supplemental Table 1). The Hemodialysis access-related quality-of-life (HARQ) instrument 15 included some similar topics to those incorporated in AVACS (e.g. patient/self-consciousness about vascular access appearance). However, AVACS more comprehensively included additional topics compared to HARQ such as future AV access decisions, disguising the AV access, negative emotional impact of AV access appearance, and overall appearance rating. The focus of the AVACS patient component on the patient’s own perceptions of their AV access rather than the responses of others make it well suited to assess patient reported AV access cosmesis. While these previous scales are consistent with AVACS highlighting that the cosmetic considerations of AV access creation is an important aspect of the patient experience, the AVACS focuses on and allows for a more granular evaluation of cosmesis than previous scales.
The resulting AVACS facilitates discussion around AV access care, including the choice of vascular access and cosmetic implications. Cosmesis is one of many important factors to be considered in choosing vascular access within the context of the patient’s ESKD Life-Plan. By creating a scoring instrument that allows stratification of cosmesis of AV accesses, factors that affect cosmesis can be assessed. In doing so, both clinicians and patients will be better informed to make decisions about AV access, based on a patient’s priorities. For example, different techniques used to create AV access, such as different endovascular and open surgical AVF and AVG creations may vary in terms of their cosmetic impact. Numerous other AV access factors, such as location of the AV access, surgical techniques utilized, patient factors such as ethnicity, and process factors such as wound management algorithms, cannulation strategies, and others, also impact on AV access cosmesis. By better understanding factors that correlate with cosmesis, strategies to support patient satisfaction with their AV access cosmesis can then be developed. In addition to informing decision making, that is, understanding the potential cosmetic effects associated with vascular access choices, along with other factors and how AV access cosmesis may change over time, patients can be provided with more accurate expectations. Furthermore, the scale is user friendly, designed to be allows it to be completed rapidly (less than 5 min), and is while providing a standardized patient outcome measure that can be used in practice and in clinical trials of vascular access that addresses patients concerns. Importantly, this cosmesis scale answers the call to action by patients and healthcare providers to develop new patient reported outcomes specific for HD vascular access. 6
Limitations
Cosmesis is inherently subjective, with different parameters or features being more or less important to a given individual, hence we tried to reflect this by including a variety of expert clinicians involved in AV access management, as well as patients.
Further, assessment parameters derived from studies using Delphi consensus methods are subjective by nature, given the lack of data for parameter generation. However, Delphi-obtained expert opinion is frequently used to define and develop classification systems and is more rigorous than those developed by a single author or group not using the Delphi method.
In the development of the cosmesis scale, we decided to focus on upper extremity AV accesses as they are most likely to be used as primary long term hemodialysis accesses. It may be possible to use the scale for lower extremity AV accesses, but we have not included examples here. Future validation studies should examine the applicability of the AVACS to these AV accesses and determine whether modifications are required to capture other relevant factors.
While panel members agreed that cosmesis associated with the cannulation segment and cannulation technique could negatively impact cosmesis, we also recognized that such details within a scale could be complex and problematic. Due to issues such as difficulty of clearly defining where the cannulation segment begins and ends for many patients, a simple dichotomous evaluation of the presence or absence of cannulation scarring was deemed sufficient.
The cosmesis scale does not include questions on transient bruising or bandages that may be present at certain points during the natural history of the AV access. This could be addressed by coaching patients completing the patient component to consider the impact of bandages and transient bruising when rating their overall current access appearance.
Lastly, the panelists and steering committee members were from North America (US and Canada), and the scale is in English. Future research should investigate the generalizability of the AVACS in other countries and languages. The clinician and patient scales outlined here are yet to be validated and the scale is available for use by other researchers. To address these limitations, there are current plans to validate AVACS with input from both clinicians and patients.
A key strength of our scale is that it incorporates two components that can work in concert to create a holistic view. However, while the clinician component was designed to be as objective as possible to reduce interobserver variability (e.g. obtaining scar length measurements), an element of interobserver variability is inevitable due to subjective evaluation of the absence or presence of some items, for example, discoloration. In contrast, the patient component is purposely subjective, eliciting opinions on multiple dimensions of the patient experience.
Conclusions
While safety and efficacy are clinician priorities for the evaluation of AV access for HD, cosmesis is an important component of the ESKD patient experience. The AVACS is a two-component scale that incorporates clinician and patient evaluation designed through a multidisciplinary Delphi process to assess this important domain. AVACS is tool that can be used to help inform patient decision making and future research on AV access creation and maintenance.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298221141499 – Supplemental material for Hemodialysis Arteriovenous Access Cosmesis Scale (AVACS): A new measure for vascular access
Supplemental material, sj-pdf-1-jva-10.1177_11297298221141499 for Hemodialysis Arteriovenous Access Cosmesis Scale (AVACS): A new measure for vascular access by Theodore H Yuo, Charles Y Kim, Dheeraj K Rajan, Vandana D Niyyar, Marianna Murea, Ellen D Dillavou, Peter R Bream Jr, Lesley C Dinwiddie, Stephen E Hohmann, Karen Woo, Tushar Vachharajani, Cynthia Roberts, Christie Gooden, George WJ Wright, Andrew J Hogan, Nicole C Ferko, Erin Kahle, Diana Clynes and Charmaine E Lok in The Journal of Vascular Access
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of Dr. Michael Allon and thank him for participating in the first and second clinician surveys for the Delphi panel process.
The authors would also like to acknowledge the contribution of the American Association of Kidney Patients (AAKP) Patient Ambassadors Cecilia Santana, David Rodriguez, Fiona McKinney, and Melissa Bensouda and thank them for their feedback and insights on the patient component of the AV cosmesis scale.
The authors would like to acknowledge the partnership with the AAKP and thank the AAKP Center for Patient Research and Education for their patient data collection. We appreciate their insights and collaboration on this project.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that they have no conflicts of interest regarding patents or royalties, stock/stock options, or first-degree relatives with any of these relationships related to this work. No reimbursement was received for the development of the manuscript.
DR, VN, MM, CR, ED, CG, TV, SH, PB, LD and patients CS, DDR, FM, and MB (see acknowledgements) received remuneration from Becton Dickinson (BD) for their participation in the Delphi process.
GW, AH, and NF are employees of CRG-EVERSANA Canada Inc., which was contracted by BD to facilitate the Delphi panel process and analyze the results.
EK and DC are employees of the American Association of Kidney Patients (AAKP) which was contracted by BD to advise on patient input and recruited the Patient Ambassadors.
DR is a consultant for BD and Gore Medical. VN is a consultant for the NACCME – Moderator for AV Access Webcast, supported by an educational grant from Medtronic. SH is a consultant/speaker for BD, Merit Medical, Medtronic, and Gore Medical. ED is a consultant for WL Gore, Angiodynamics, and 3M/KCI. ED is a scientific advisor for Boston Scientific. TY is a consultant for BD, WL Gore and Merit Medical and a scientific advisory board member of BD and Medtronic. CK is a consultant for BD and ACI/Humacyte and advisory board member for Boston Scientific. CL is a consultant for BD, Gore, and Medtronic.
ED, KW, and TY are authors and ED is an editor for UpToDate.
Funding
The author(s) disclosed the receipt of the following financial support for the research from Becton Dickinson.
Becton Dickinson provided support for publishing fees.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
