Abstract
Backgrounds:
Clinical monitoring is the recommended standard for identifying dialysis access dysfunction; however, clinical monitoring requires skill and training, which is challenging for understaffed clinics and overburdened healthcare personnel. A vascular access risk stratification score was recently proposed to assist in detecting dialysis access dysfunction.
Purpose:
Our objective was to evaluate the utility of using vascular access risk scores to assess venous stenosis in hemodialysis vascular accesses.
Methods:
We prospectively enrolled adult patients who were receiving hemodialysis through an arteriovenous access and who had a risk score ⩽3 (low-risk) or ⩾8 (high-risk). We compared the occurrence of access stenosis (>50% on ultrasonography or angiography) between low-risk and high-risk groups and assessed clinical monitoring results for each group.
Results:
Of the 38 patients analyzed (18 low-risk; 20 high-risk), 16 (42%) had significant stenosis. Clinical monitoring results were positive in 39% of the low-risk and 60% of the high-risk group (p = 0.19). The high-risk group had significantly higher occurrence of stenosis than the low-risk group (65% vs 17%; p = 0.003). Sensitivity and specificity of a high score for identifying stenosis were 81% and 68%, respectively. The positive predictive value of a high-risk score was 65%, and the negative predictive value was 80%. Only 11 (58%) of 19 subjects with positive clinical monitoring had significant stenosis. In a multivariable model, the high-risk group had seven-fold higher odds of stenosis than the low-risk group (aOR = 7.38; 95% CI, 1.44–37.82; p = 0.02). Positive clinical monitoring results and previous stenotic history were not associated with stenosis. Every unit increase in the score was associated with 34% higher odds of stenosis (aOR = 1.34; 95% CI, 1.05–1.70; p = 0.02).
Conclusions:
A calculated risk score may help predict the development of hemodialysis vascular access stenosis and may provide a simple and reliable objective measure for risk stratification.
Introduction
Long-term vascular accesses used for hemodialysis develop frequent complications, such as loss of access patency or the ability to deliver adequate flow for dialysis. The rate of using arteriovenous fistula (AVF) and graft (AVG) in prevalent hemodialysis patients has plateaued. Improving the survival of arteriovenous dialysis accesses has remained a long-standing problem. The development of progressive vascular access stenosis with subsequent access failure contributes to significant morbidity of patients on dialysis as well as economic burden. Conversion from a permanent vascular access to a catheter increases mortality by 80%, 1 and the incidence of primary AVF failures increases with each successive fistula placement. 2 Therefore, early identification of progressive stenosis and prompt intervention to preclude access failure are essential for improving patient outcomes in a value-based reimbursement environment.
Hemodialysis vascular access complications are monitored by either direct clinical examination or a device-associated surveillance methodology. The 2019 update to the National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI) Clinical Practice Guidelines for Vascular Access 3 recommends clinical monitoring as the standard to detect and correct stenosis and, in turn, to minimize or avoid dialysis interruption and to reduce the overall rate of thrombosis. This approach uses periodically observed clinical findings noted before initiating a dialysis treatment but fails to recognize that clinical monitoring is not standardized across different personnel (e.g. nurses and technicians), dialysis centers, and nations. Also, interpersonal and intrapersonal variability have not been well investigated. Any delay in identifying dysfunctional accesses can lead to complications like aneurysmal degeneration, thrombosis, and even catastrophic consequences like spontaneous rupture and death.4–7
Vasc-Alert is an FDA-approved device used for dialysis access dysfunction surveillance. It utilizes readily available intradialytic pressure and blood flow readings standardized to hematocrit and blood pressure and provides a derived static venous pressure in the form of a ratio to mean arterial pressure. 8 The vascular access (VA) risk score developed by Astor et al. 9 is a novel risk stratification method that uses an algorithm to analyze various Vasc-Alert measurements, including the rate of change in derived venous pressure, the number of recently derived venous pressure readings above threshold (alerts), inability to reach prescribed blood flow, and arterial pressure alerts. The VA score ranges from 1 to 10, and there is a monotonical increase in incidence of intervention with higher risk scores. Importantly, Astor et al. observed that the odds of intervention increased by two-fold between lowest and highest scores. While the study used intervention as an outcome measure, it lacked data on the presence or absence of venous stenosis as assessed by angiography. The extent to which the VA score can predict the occurrence of stenosis in dialysis accesses has not yet been prospectively evaluated. While the algorithm provides a score for each dialysis treatment, studies of other scoring systems suggest that a range of values is more valuable than an individual score in predicting an outcome measure. 10 Additionally, the agreement between VA score and clinical monitoring is undetermined.
Therefore, we conducted a prospective study to evaluate the utility of using risk scores for identifying dialysis access stenosis. We hypothesized that higher VA scores would be associated with the identification of significant stenosis in hemodialysis accesses. Our primary aim was to analyze the association between VA scores and stenosis detection in dialysis accesses, while our secondary aim was to determine the alignment between VA scores and clinical monitoring findings.
Subjects and methods
Study design
This study was a single-center prospective observational proof-of-concept validation trial to assess the association between VA score and detection of stenosis in dialysis access sites. This study was approved by the institution’s Institutional Review Board (IRB #14929).
Patients receiving hemodialysis through arteriovenous access (fistula or graft) at a single dialysis unit were prospectively enrolled and monitored from the beginning of August 2021 through October 2021. Inclusion criteria included any adult over the age of 18 receiving hemodialysis with arteriovenous access (either AVF or AVG) for more than 3 months. Exclusion criteria included any adult hemodialysis patients with use of a catheter as dialysis access or baseline risk score between 4 and 7. We did not include patients with VA scores 4–7 because of unclear clinical relevance and to minimize the occurrence of false positives. Patients were identified based on baseline VA score. The VA scores for patients at the study dialysis center are reported on a weekly basis from 2020. The scores are generated for each dialysis session but reported weekly based on the three-dialysis session. Identified patients were enrolled over a 3-month period from August 2021 to November 2021. Patients were categorized into two groups based on VA scores and assigned a probability risk for stenosis detection as follows: score 1–3, low-risk group; and score 8–10, high-risk group. The electronic medical records of the dialysis center contained vascular access data. All patients underwent an Informed Consent Process per the approved study protocol. The principal investigator and a research associate from the Clinical Trials section of the Division of Nephrology approached subjects for enrollment. All enrolled patients signed and consented to participate in the study.
All enrolled patients received a physical examination and point-of-care ultrasonography (POCUS) performed by an interventional nephrologist prior to the dialysis session. The patient’s primary nephrologist or designee received relevant information only if an impending risk for thrombosis was identified during the study examination. Other findings were not disclosed to the care team to minimize bias in referrals. Subsequently, clinical care was given per dialysis unit policy and by the respective clinical team. Subjects were referred to receive digital subtraction angiography of dialysis access by the primary care team as per clinical necessity only. Clinical monitoring included clinical symptoms and signs indicative of clinically significant lesions based on KDOQI 2019 guidelines as listed in Figure 1. The clinical monitoring was considered positive for stenosis if at least one of the signs or symptoms suggestive of stenosis was detected. Electronic health record review provided information on demographics, dialysis, VA creation, procedures, and interventional data. Significant stenosis was defined as a luminal narrowing of more than 50% found by ultrasonography or angiography. 11
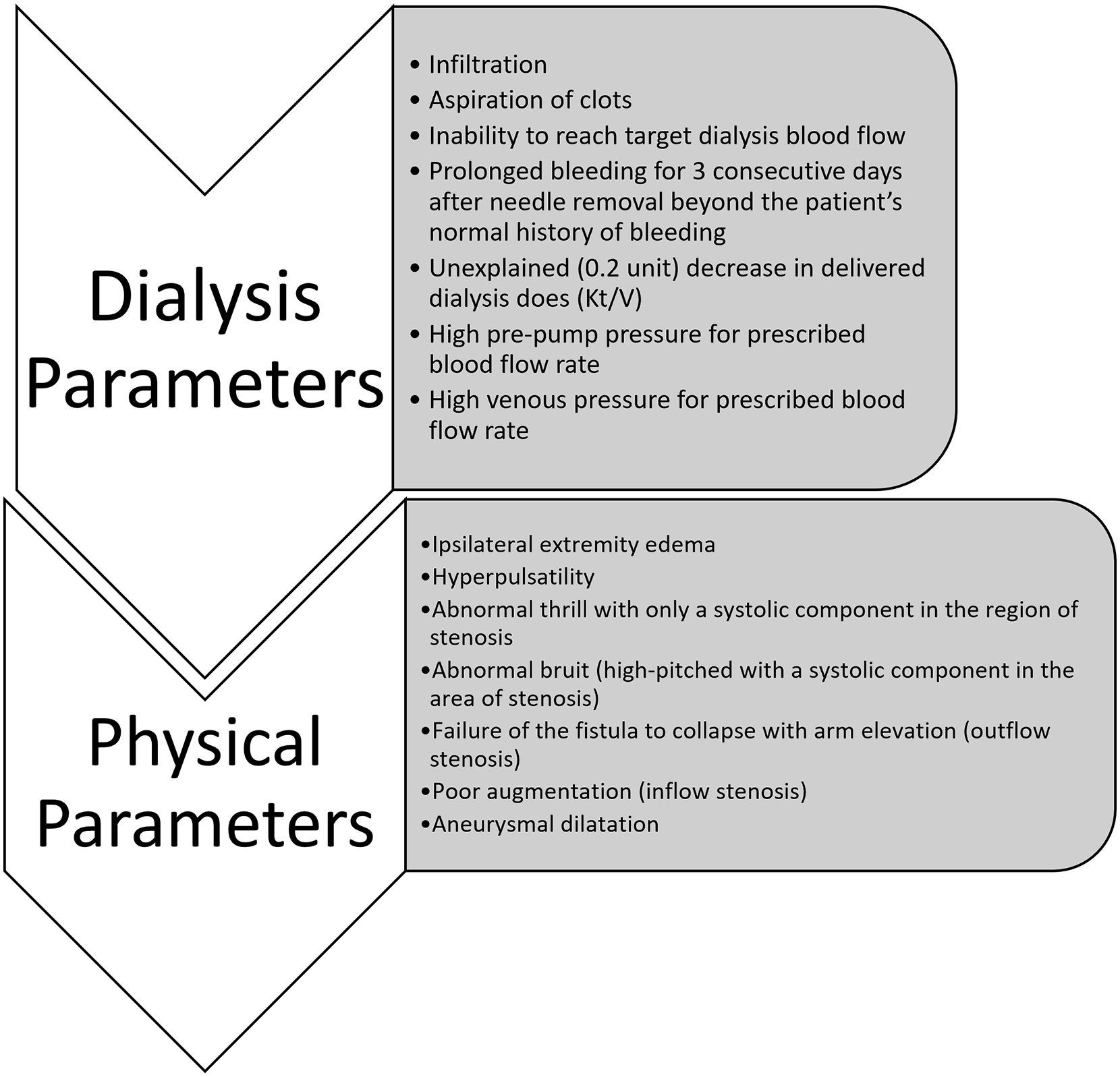
Clinical monitoring parameters used for assessing dialysis access stenosis.
Statistical analyses
The study tested whether high-risk VA scores were associated with a greater occurrence of clinically significant stenosis than low-risk VA scores. The primary analysis evaluated the proportion of patients who had significant stenosis within each of the two score groups. As appropriate, patient characteristics were compared across groups with chi-squared tests and t-tests. These analyses were repeated after stratification for access type and subsequently for the presence of clinical signs and symptoms. Similar analyses were performed to evaluate associations between clinical signs/symptoms and the presence of stenosis. Logistic regression models were developed to assess independent associations with stenosis. Statistical significance was set at p < 0.05. All analyses were performed using Stata Statistical Software Release 13.
Results
A total of 40 patients (20 high-risk VA score and 20 low-risk VA score) were enrolled, one was excluded due to a re-enrollment error, and another one was excluded for access thrombosis before the physical examination component. The final analysis included 38 subjects (20 female and 18 male) with 20 in the high-risk group and 18 in the low-risk group. There were 24 AVFs in the cohort: 13 (72%) in the low-risk group and 11 (52%) in the high-risk group (p = 0.27). Among the 14 AVG, nine were in high-risk group and five in low-risk group. The mean (SD) age of the patients was 66 (16.3) years for the low-risk group and 60.1 (13.1) years for the high-risk group. No significant differences in age, sex, diabetes, or hypertension between the groups were observed. Patients in the low-risk group had a higher prevalence of coronary artery disease than the high-risk group (56% vs 25%; p = 0.05) (Table 1).
Patient characteristics.
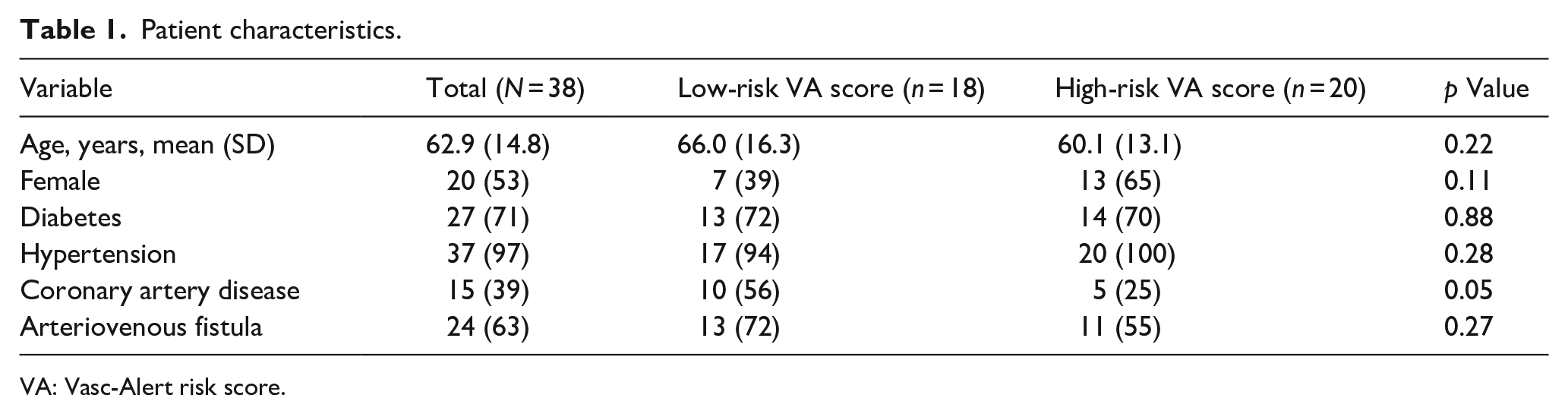
VA: Vasc-Alert risk score.
Categorical variables are presented as number (percentage) and continuous variables as mean (standard deviation).Clinical monitoring was positive in 37% of the low-risk group and 63% of the high-risk group (p = 0.19). Of all 38 subjects, 16 (42%) patients had significant stenosis. The high-risk group had more significant stenosis than the low-risk group (81% vs 19%; p = 0.003) (Table 2). History of stenosis was present in 20 (53%) patients: 8 (44%) in the low-risk group and 12 (60%) in the high-risk group (p = 0.15). The sensitivity and specificity of identifying stenosis in the high-risk group were 81% and 68%, respectively. The positive predictive value in the high-risk group was 65%, and the negative predictive value was 80%.
Association of surveillance score and clinical monitoring with significant stenosis.
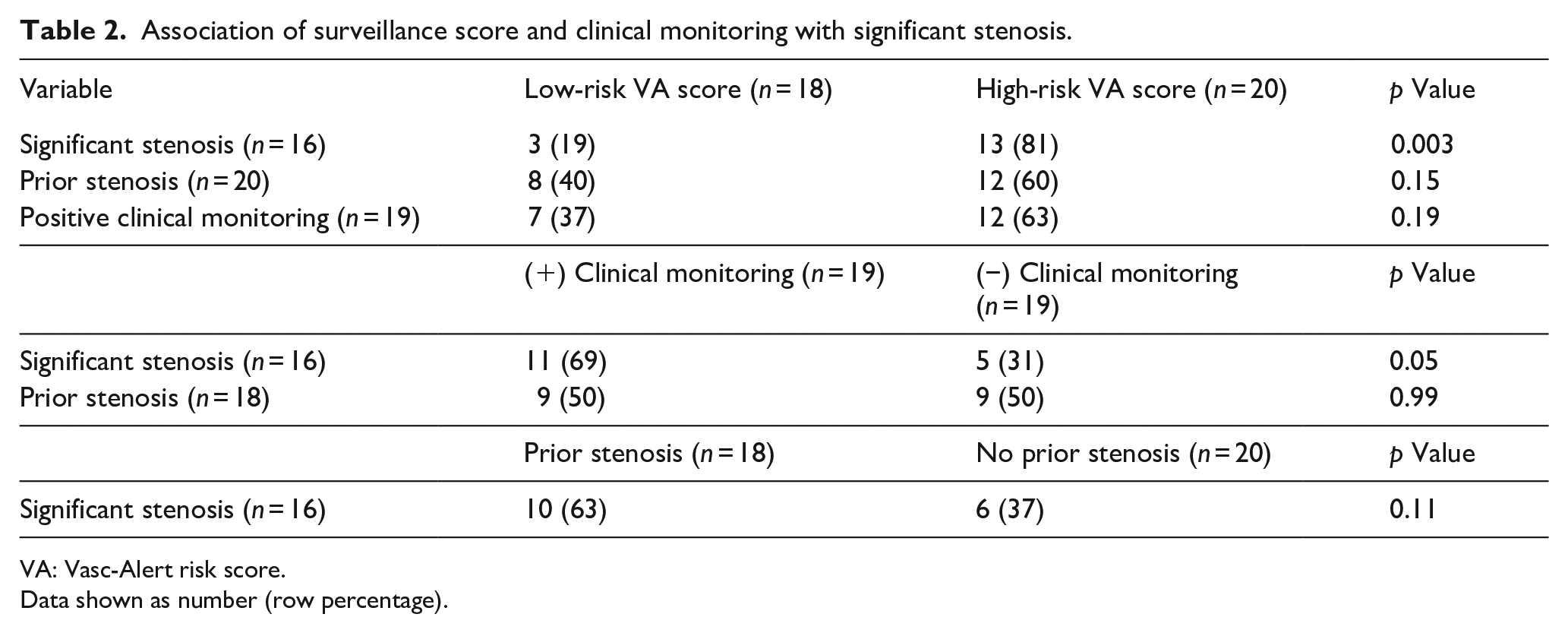
VA: Vasc-Alert risk score.
Data shown as number (row percentage).
A total of 19 patients had positive clinical monitoring results, and only 11 of the 19 patients (58%) with positive clinical monitoring had significant stenosis: 2 (11%) in the low-risk group and 9 (47%) in the high-risk group (p = 0.05). Among 19 patients with no positive clinical monitoring findings, 5 (26%) patients had significant stenosis: 1 (5%) in the low-risk group and 4 (21%) in the high-risk group (p = 0.05). Of the 24 patients who had an AVF dialysis access, 8 (33%) had significant stenosis: 2 (8%) in the low-risk and 6 (25%) in the high-risk group (p = 0.04). Of the 14 patients who had arteriovenous graft dialysis access, 8 (57%) had significant stenosis: 1 (7%) in the low-risk and 7 (50%) in the high-risk group (p = 0.04) (Table 3). Adjusted for risk score, a positive clinical monitoring finding and prior stenotic history were not significantly associated with significant stenosis. While the positive predictive value was 65% for VA high-risk score, clinical monitoring was 58%. In addition, the negative predictive value was 83% for VA high-risk score and while it was 74% for clinical monitoring. Notably, a high VA risk score identified four of the five accesses that were false negatives by clinical monitoring.
Association of surveillance score with significant stenosis, by clinical monitoring status and access type.

AVF: arteriovenous fistula; AVG: arteriovenous graft; VA: Vasc-Alert risk score.
Data shown as number (row percentage).Every unit increase in the score was associated with 34% higher odds of stenosis (adjusted odds ratio (aOR) = 1.34; 95% confidence interval (95% CI): 1.05–1.70; p = 0.02). Having a high-risk score was associated with seven-fold higher odds of stenosis than having a low-risk score after adjusting for other variables included in the model: high surveillance score, positive clinical monitoring, and prior stenosis (aOR = 7.38; 95% CI: 1.44–37.82; p = 0.02) (Table 4). The key study findings are graphically represented in Figure 2.
Logistic regression analysis of the association of surveillance score with stenosis.
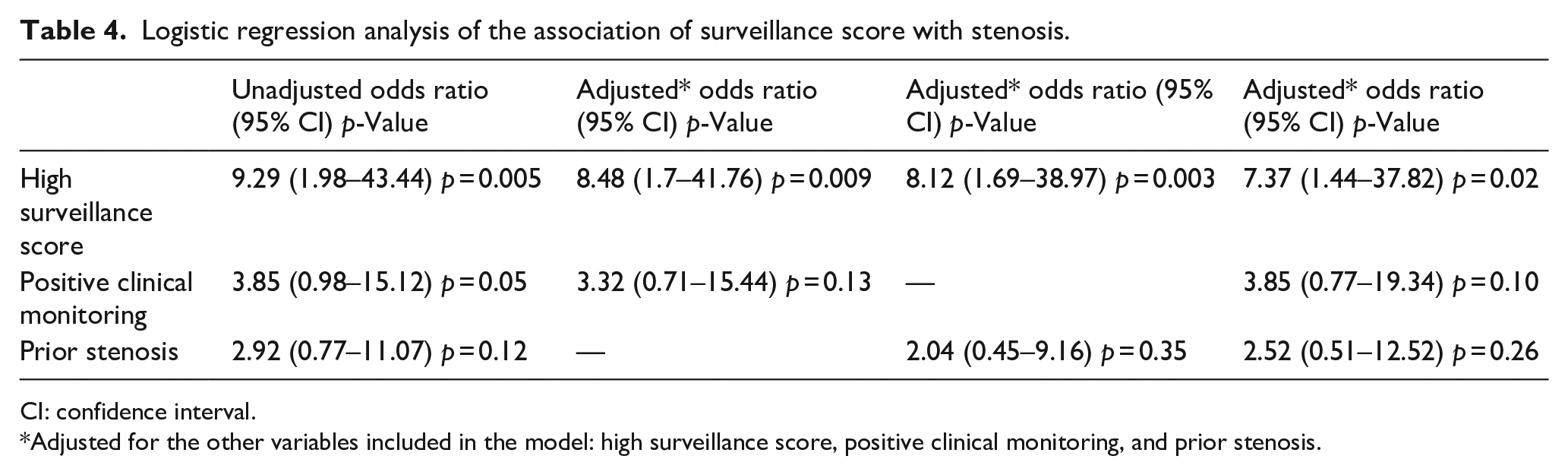
CI: confidence interval.
Adjusted for the other variables included in the model: high surveillance score, positive clinical monitoring, and prior stenosis.
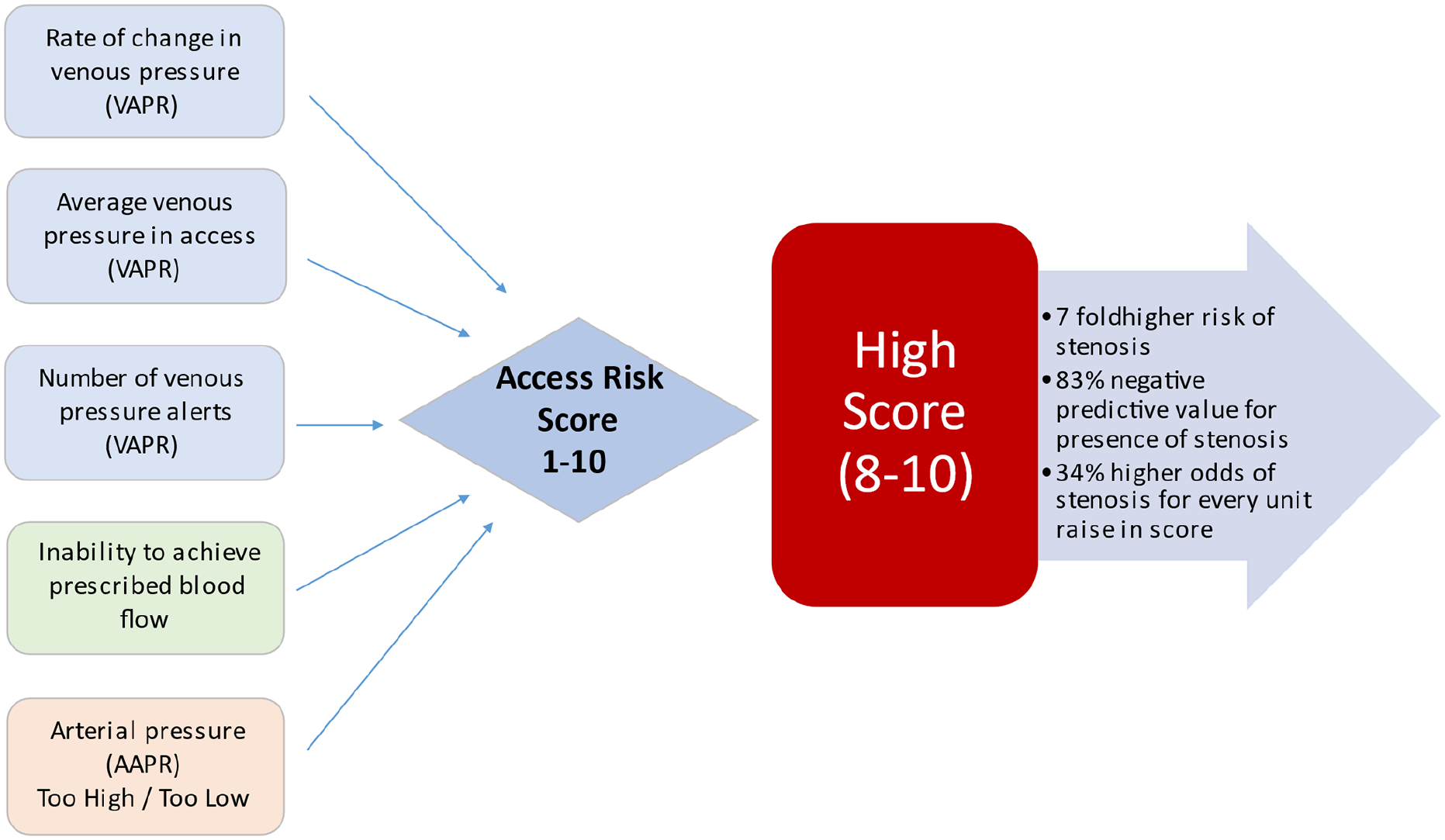
Graphical representation of key study findings.
Discussion
In this study, we observed that patients receiving dialysis through arteriovenous access with high-risk VA score were more likely to have significant stenosis than patients with a low-risk VA score. These findings suggest that a higher VA score may be useful for predicting which patients may have clinically significant stenosis as seen in imaging studies. We also observed that positive clinical monitoring results alone were insufficient for identifying stenosis. Thus, we recommend that an automated data-driven risk stratifying scoring system may have clinical utility for preventing and/or expediently treating access stenosis in patients who receive dialysis.
A brief discussion on clinical monitoring strategy with its inherent limitations is warranted. A clinical monitoring strategy includes physical examination supplemented with routine laboratory studies, dialysis adequacy, miscannulation, hemostasis post-needle withdrawal, and other clinical signs suggestive of dysfunctional dialysis access. Published studies that examined how these clinical indicators correlate with stenosis identified by angiographic imaging.12–14 The KDOQI 2019 guideline 15.1 recommends referral for confirmatory evaluation including imaging studies based on select clinical indicators, while attempting to provide a new roadmap for access surveillance. 3 The new “Access flow dysfunction” terminology distinguishes between stenosis-mediated access flow dysfunction and other causes, such as aneurysms. Guideline 13 recommends regular clinical monitoring by a “knowledgeable and experienced health practitioner” to detect clinical indicators of flow dysfunction of the AVF. However, the guidelines fail to define “knowledgeable and experienced health practitioners” and how to standardize clinical monitoring across people of various skill sets. Regardless, a robust clinical monitoring strategy needs a dedicated trained team available throughout the week for assessing patients’ dialysis accesses, but implementation of such a strategy is a logistical challenge. Additionally, the challenge of performing regular, thorough monitoring of patients’ dialysis accesses has exponentially worsened during the ongoing COVID-19 pandemic, with a high workforce attrition rate and difficulty with training new staff.
The evidence supporting clinical monitoring as a sole tool for identifying dysfunctional dialysis accesses has not been critically reviewed. Observational studies form the backbone of evidence for a clinical monitoring strategy. 15 Six studies published on this topic included physicians specializing in nephrology as the main providers who performed the physical examination of dialysis accesses, while we note that a single group published two of those studies.13,14,16–18 Also, studies included subjects who were referred for an angiography due to a clinical problem, which may have introduced some selection bias. Realistically, the current workflow in dialysis units involves non-physician dialysis staff performing access evaluation. The diagnostic value of clinical monitoring performed by non-physician personnel in identifying accesses with stenosis in a mixed cohort of functional and dysfunctional accesses has not been validated.
Our results highlight the limitations of using a clinical monitoring only strategy. In our study cohort, 50% of patients had positive clinical monitoring parameters, of which approximately two-thirds were in the high-risk score group. However, only a little over half of the positive clinical monitoring results were associated with a significant stenosis. Having a high-risk score alone identified stenosis in 21% of patients who did not have positive clinical monitoring findings. And adjusted risk scores indicated that a positive clinical monitoring finding and prior stenotic history were not significantly associated with significant stenosis. These findings caution against overreliance on clinical monitoring alone, as it may lead to missed dysfunctional, stenotic accesses. The poor performance of clinical monitoring in our study agrees with findings from a study by Maldonado-Carceles et al. 18 In that study, of 99 patients with dysfunctional AVF who were sent to a dedicated vascular radiology unit, the physical examination alone identified stenosis in only 62%. Further pragmatic studies on the utility of a clinical monitoring-only strategy to assess access outcomes in community-based dialysis populations are sorely warranted.
Vascular access surveillance is different from clinical monitoring. Vascular access surveillance methodologies requiring specialized equipment and operator skills are meant to detect stenosis before clinical indicators develop. 8 Surveillance methodologies are broadly divided into blood flow or pressure-based approaches, and ultrasound-based surveillance predates blood flow and pressure-based methods. 19 The reliability and reproducibility of surveillance methodologies, however, are frequently doubted, and each surveillance methodology has specific limitations beyond the scope of this discussion. A general principle of repeat measurements and trending surveillance results is vital to confirm any abnormal results.
The Vasc-Alert scoring system is a novel risk stratification algorithm based on Vasc-Alert vascular access surveillance technology. Our study provides preliminary clinicopathological evidence for practice-based risk classification using a scoring system. A high-risk score was not only significantly associated with stenosis, but it also identified stenosis in 21% of patients who did not have any positive clinical monitoring findings, suggesting that using a scoring system may be superior to clinical monitoring alone. The high-risk VA score range as a diagnostic tool appeared to be sensitive and relatively specific, with an excellent negative predictive value (sensitivity: 81%, specificity 68%, positive predictive value 65%, and a negative predictive value of 80%), which is very similar to physical examination alone as described by Maldonado-Carceles et al. 18 Coupled with increased odds of identifying a stenosis in a high score group provides a reliable stratification tool for vascular access coordinators (Figure 2).
The risk stratification strategy explored in the current study is apt in pragmatic dialysis access care. Dialysis access care is labor-intensive. The current recommendations of clinical monitoring alone fail to recognize the impact of the need for additional labor and the incurred administrative burden. Also, the mundane, repetitive clinical monitoring strategy may incur a high risk of staff fatigue and patient safety lapses. Many clinical monitoring features develop and evolve slowly, which further dampens staff perceptions and may decrease efficiency in identifying at-risk accesses. The ongoing COVID-19 pandemic adds to preexisting stressors in dialysis-related healthcare personnel. 20 A data-driven, practical surveillance program would aim to decrease staffing dependence while being able to identify at-risk accesses. We believe the time is right for an alternative, automated, data-driven risk stratifying strategy to improve vascular access care and help the overburdened dialysis frontline personnel. A tool to identify at-risk accesses would aid in efficacious management by dialysis staff by prioritizing resources for evaluation and referral for intervention.
Our study included POCUS as part of the evaluation. The KDOQI guidelines identify POCUS as an area for further research, and POCUS is proposed as a tool to aid in dialysis access care. POCUS can identify stenosis in the distal part of accesses with relative ease; however, its ability to identify proximal and central venous stenosis is limited, especially when the patient is in a sitting position, which is the case for most dialysis patients. Incorporating POCUS would best be prioritized for at-risk patients. Cost efficiencies of POCUS devices with advanced features for velocity and flow measurements and the need for skilled personnel for performing such studies mitigate many of the perceived benefits. A dedicated examination room to perform a POCUS study before cannulation of access raises questions about its feasibility in implementation. While theoretically achievable, POCUS as a standard approach would be limited to a minority of the dialysis population and have unknown benefits.
Our study has strengths and limitations. The study provides a single comprehensive VA evaluation strategy incorporating clinical monitoring, POCUS, and surveillance. A single experienced operator performed both clinical monitoring and POCUS, which is both a limitation and an advantage, and this may be more realistic in terms of clinical practice and may have minimized error from interpersonal variability. Our study employed a dedicated space in the dialysis unit for the study examination, like an access station. This novel concept of “access stations” (i.e. a dedicated access evaluation space in dialysis units) and its impact on access outcomes are worth exploring further. The small cohort size and short follow-up period were limitations, as many of the access outcomes could evolve. We believe that the temporal trend of a change in score over a longer time period could improve prediction scores which will be analyzed in future studies.
Conclusions
The vascular access risk stratification score may be an excellent, reliable, and efficient tool for identifying dialysis accesses that have significant stenosis. Our findings suggest that it may be comparable to or even better than clinical monitoring alone. The VA risk score provides an automated, simple-to-use, repeated, and reliable objective measure for risk stratification, even for patients without positive clinical monitoring findings. Patients identified by the risk score could be prioritized for review by access specialists, thus reducing the workload of COVID-19 burdened dialysis staff. Further studies to evaluate the impact of “smart surveillance” approaches on long-term dialysis access outcomes are logical next steps.
Footnotes
Acknowledgements
Authors offer special thanks to the late Dr. Jerry Yee for his leadership, vision, support, and contributions to this study. Authors also thank Karla Passalacqua PhD at Henry Ford Hospital for editorial and technical assistance.
Authorship declaration
All authors listed meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors, and all authors agree with the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L Kumbar and B Astor were members of medical advisory board of Vasc-Alert™, R Provenzano is a member of Board of Directors, A Besarab and J Yee are patent holders of Vasc-Alert™.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by a research grant of $13,950 from Vasc-Alert™ to cover the cost of conducting this study. The amount was paid to the hospital.
