Abstract
Introduction:
We have previously evaluated the usefulness of venocutaneous fistula (VCF), also called “dermatorrhea.” VCF is a technique of blood removal/return by advancing a dialysis cannula to the femoral vein for each hemodialysis session using a fistula created between the great saphenous vein and skin.
Methods:
In this study, we evaluated 46 limbs of 40 patients for whom VCF was created at our hospital between May 2017 and April 2022. In all the patients, it was difficult to construct an arteriovenous fistula or arteriovenous graft because of the general/vascular conditions. The usefulness of this method was evaluated based on the results of dialysis treatment after fistula creation and the use of fistula.
Results:
Fundamental evaluation confirmed the progression of vascular wall thickening over time. During clinical review, no serious complications were found in any patient during or after fistula creation surgery. The infection rate was 0.30/1000 days of fistula maintenance. Secondary patency rates by the Kaplan–Meier method were 87.0% at 1 year and 42.6% at 3 years.
Conclusion:
This method has demonstrated a good patency rate, low infection rate, and seems to be a potentially useful alternative in patients in whom it is difficult to establish vascular access.
Introduction
Tunneled hemodialysis catheters are used for dialysis access in cases where arteriovenous fistula (AVF) or arteriovenous graft (AVG) placement is not feasible.1,2 Problems with infection and fibrin sheath formation are often encountered with catheters. 2 In this study, we devised a method of creating a fistula between the great saphenous vein and skin; a dialysis catheter can be advanced into the femoral vein using a guide wire for each dialysis session for blood removal/return. We call this access a venocutaneous fistula. This method provides a new access for hemodialysis that uses the backflow prevention mechanism of venous valves for hemostasis. We performed the first ever surgery using this method in May 2017. In 2019, we reported 10 patients who were subsequently treated with this method. 3 Here, we report the results of fistula creation and routine dialysis management after refining the method and the device and after obtaining mid- and long-term results. The previous report was characterized by a short time to hemostasis and low incidence of infections. We have now added data supporting shorter time to hemostasis and lower infection rates, focusing on the progression of vascular wall thickening over time after surgery.
Methods
Subjects
Fundamental evaluation
Three consecutive patients who underwent VCF creation between May 2021 and April 2022 underwent an evaluation of the progression of vascular wall thickening.
Clinical analysis
We investigated 40 patients on maintenance hemodialysis who underwent VCF creation at our hospital between May 2017 and April 2022. Inadequate arteries were noted in all patients, and it was difficult to create an AVF or AVG in these patients.
In all, 16 men and 24 women aged 77.5 ± 11.2 years were included. The patients had a history of dialysis for 7.3 ± 8.0 years. The primary disease was diabetes mellitus in 11 patients, chronic nephritis in eight patients, nephrosclerosis in seven patients, collagen disease in two patients, other etiologies in three patients, and unknown etiologies in nine patients. The VCF site was the right lower limb in 15 patients, left lower limb in 20 patients, both lower limbs in four patients. Among these patients, blood removal from the VCF created on one side was insufficient in one patient, and blood removal was switched to both sides after creating another VCF on the other side. In the remaining three patients, blood removal from one side became difficult, and blood removal was switched to the other side. The left upper limb basilic vein was used in addition to both lower limbs in one patient.
Fundamental evaluation
The cross-sectional area of the VCF was measured at regular intervals in three consecutive patients, starting immediately after creation of the access. The measurement was performed under the same conditions. The cross-sectional area of the great saphenous vein was measured by ultrasonography at the intermediate area between the fistula site and the femoral vein junction [LOGIQ S8 system, GE Healthcare, Chalfont St Giles, UK was used for the measurements.] An 11 MHz linear probe was also used.
Clinical analysis
(1) For fistula creation, the initial success rate and the incidence of complications were determined.
(2) For dialysis management using a fistula, the incidence of infection and patency rate were determined.
The endpoint of primary patency was defined as intervention of the initial endovascular treatment (except for dilation with a dilator.) The endpoint of secondary patency was defined as the discontinuation of the use of fistula. All endpoints were calculated using the Kaplan–Meier method. In patients receiving multiple treatments, the details of the initial treatment were analyzed. The statistical software “EZR” (Easy R) was used for statistical analysis.
Definition of fistula creation, usage, and terms
Fistula creation
Figure 1 shows how a great saphenous venocutaneous fistula (Figure 1(a)) and basilic venocutaneous fistula (Figure 1(b)) were created. The surgery was performed by three physicians with over 10 years of experience.
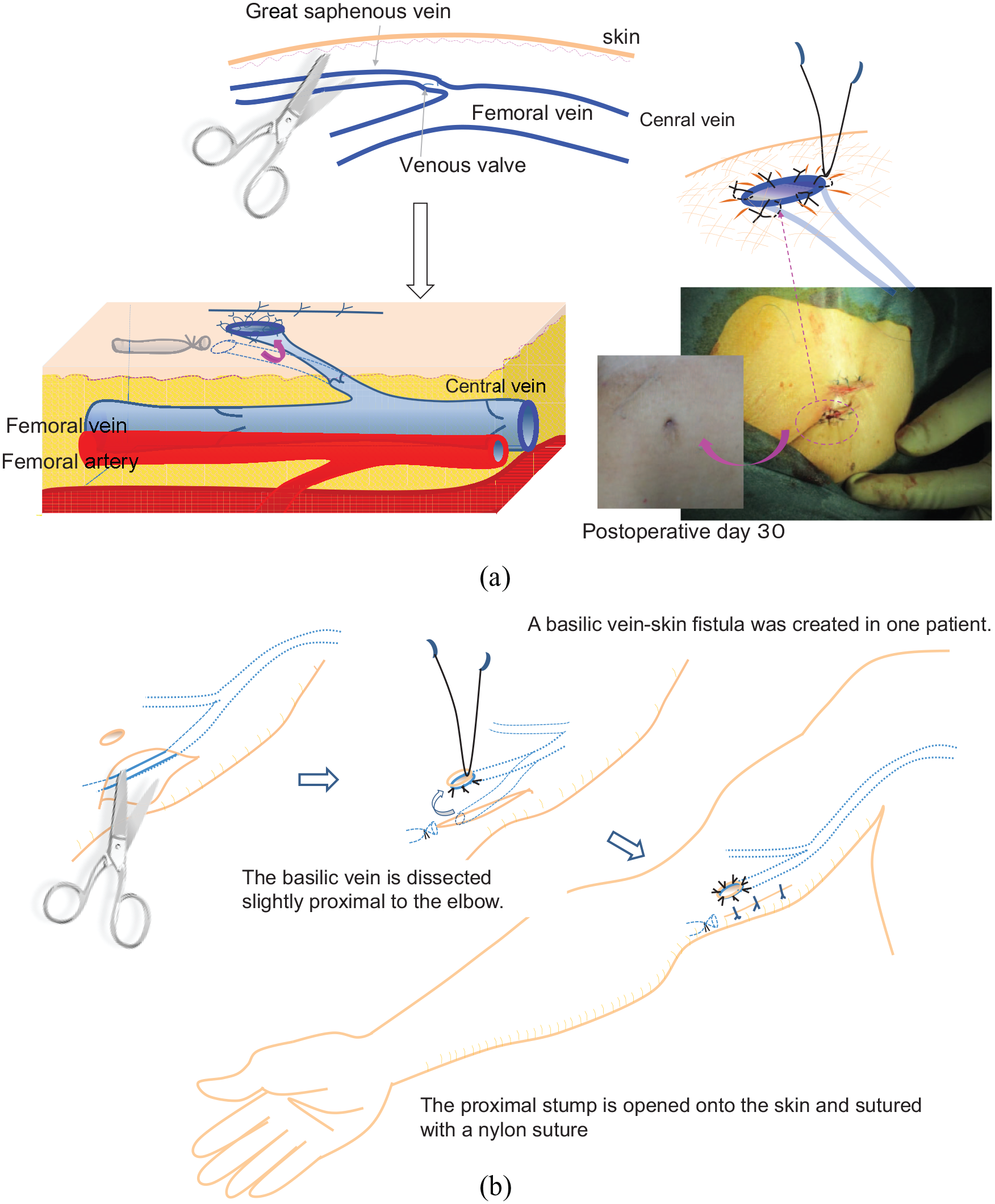
(a) Creation of a great saphenous venocutaneous fistula and (b) creation of a basilic venocutaneous fistula.
Daily management
(1) Early postoperative period
In principle, patients had to be hospitalized for 7 days after fistula creation. Bathing was not allowed during this period. The patients were asked to rest in supine position for 3 h after dialysis, after which there were no restrictions on body posture and duration of rest.
(2) Stable period
From 8 days after the fistula creation, there were no restrictions on posture if hemostasis was confirmed in the standing or sitting position after completing dialysis. On non-dialysis days, there were no restrictions on exercise or body posture, and bathing and shower were allowed as long as the fistula was covered with a waterproof tape.
Dialysis cannulation
The cannulation method for dialysis is shown in Figure 2. The cannulation procedure was performed by a physician or nurse who was professionally trained for the procedure. The VCF was used from the next day after its creation. It was used for blood removal and return during maintenance dialysis performed 1–3 times a week.
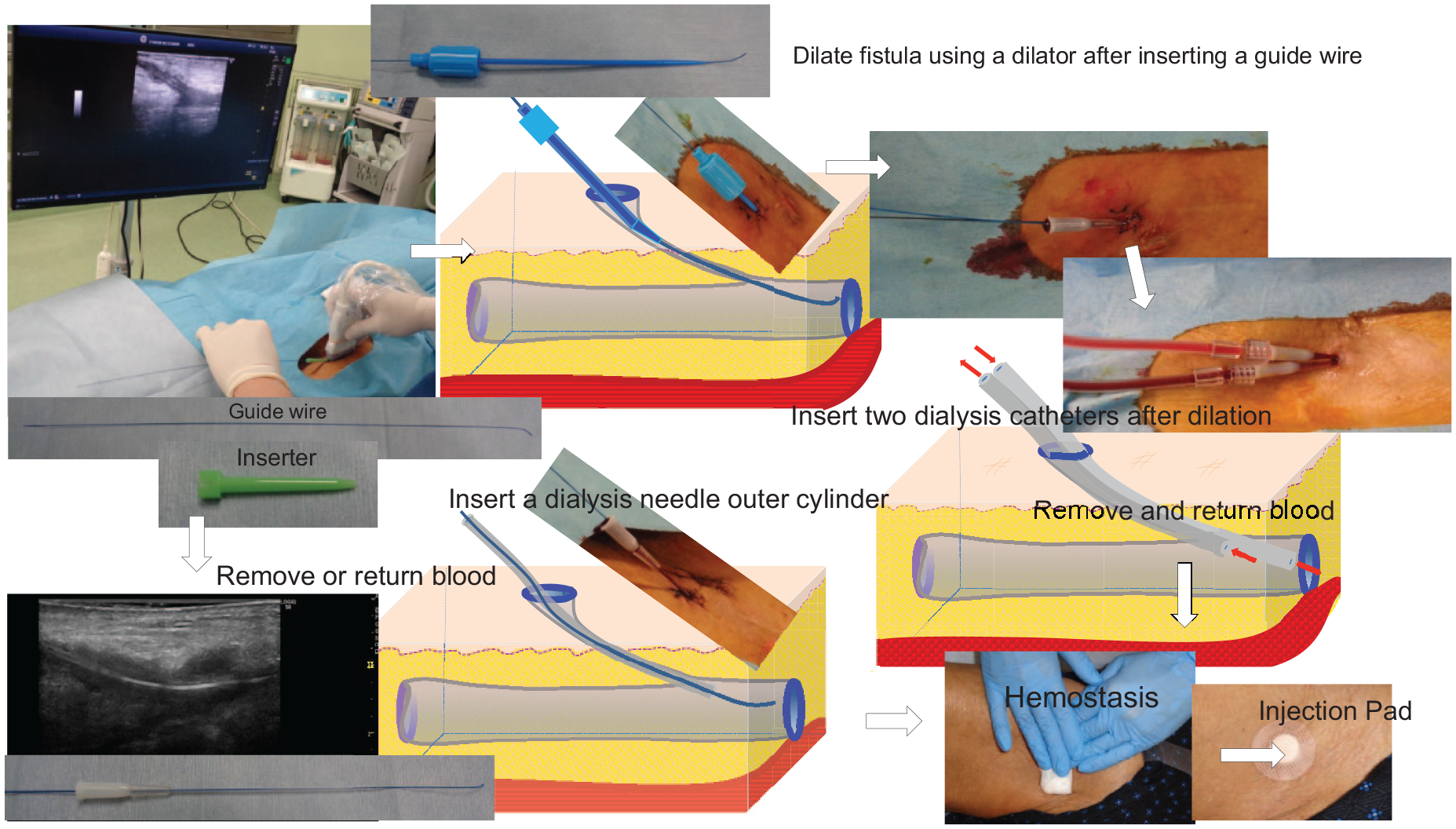
Cannulation to VCF.
Hemostasis at the end of a dialysis session is performed in the same manner as that after removing needles from the AVF, where manual compression is applied for approximately 5–15 min, the absence of bleeding is checked during postural changes and leg bending, and injection pads are applied and fixed onto the fistula until the next morning.
As shown in the Figure 3, cannulation to VCF was used in combination with other points of access depending on the situation (Figure 3).
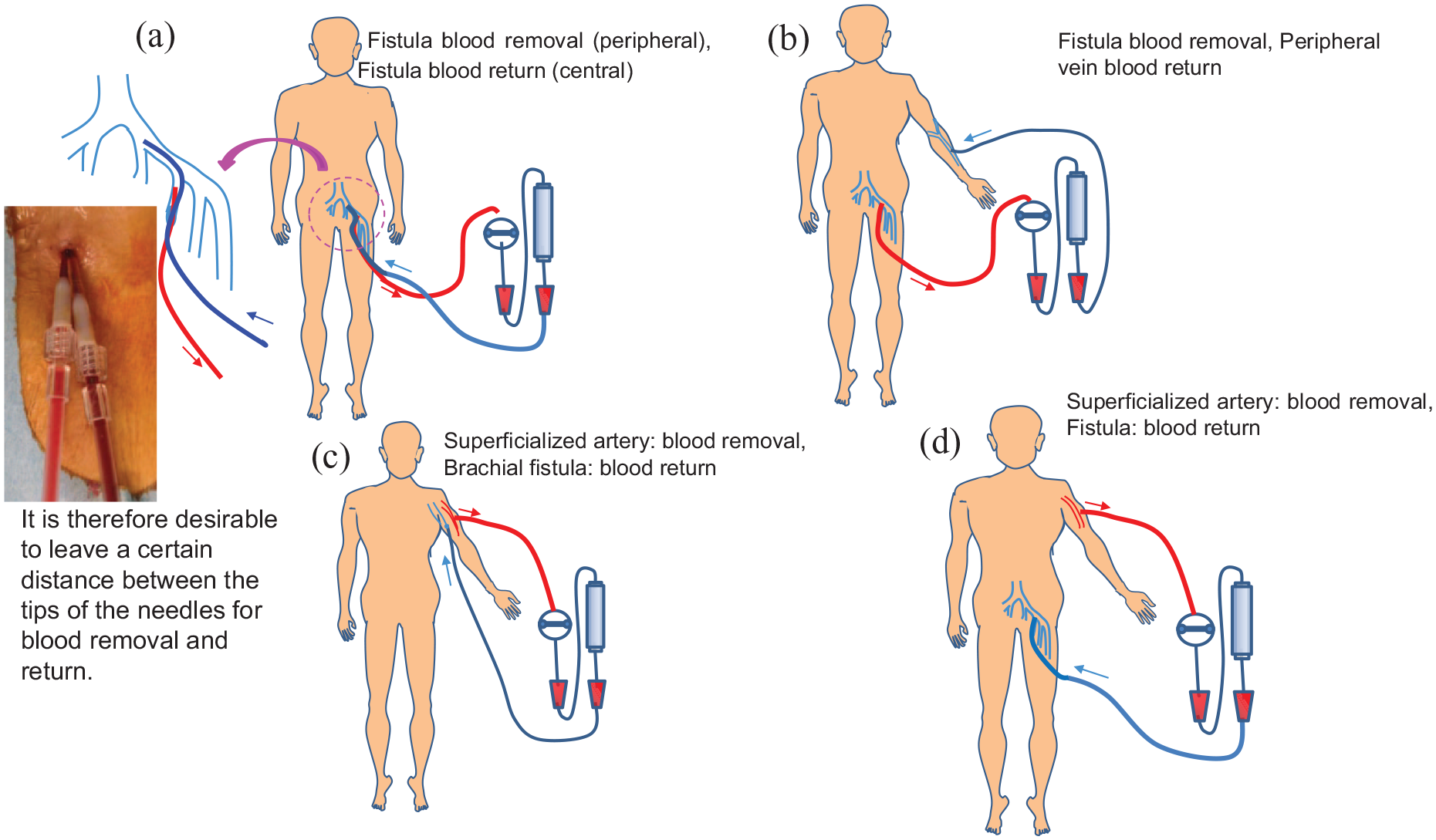
Actual use of VCF.
In six patients who developed thrombosis of the femoral vein during the study period, warfarin was administered aiming at PT-INR of 1.5–2.5. After dialysis, 60,000–120,000 units of urokinase was intravenously administered as needed.
Thrombosis refers to a complete thrombosis in the femoral vein, rendering it unusable unless it is invasively salvaged. The cause of the complete thrombosis is deeply related to intimal injury from stimuli with cannulation. It was expected to be difficult to achieve surgical or endovascular recanalization. All patients with thrombosis had swelling of the legs and poor performance status, and were at a higher risk of complications from invasive treatments. Warfarin and urokinase were used in expectation of symptom relief and recanalization from thrombolysis.
Infection
(1) Definition of infection
The mode of VCF infection closely resembles that of a cuffed catheter infection. It was therefore defined, based on the guidelines stated in KDOQI 2019, 1 CDC for cuffed catheter infection. 3
(2) Infection rate
The infection rate was calculated as the number of events/total number of days of fistula maintenance × 1000. The observation date was set as April 30, 2022, and the number of infection events and the total number of days of fistula maintenance were calculated accordingly. The number of days of fistula maintenance was defined as the period during which blood removal and/or return is performed at least once a week.
Results
Fundamental evaluation
In all patients, the vascular wall thickness progressed over time after the creation of VCF (Figure 4).
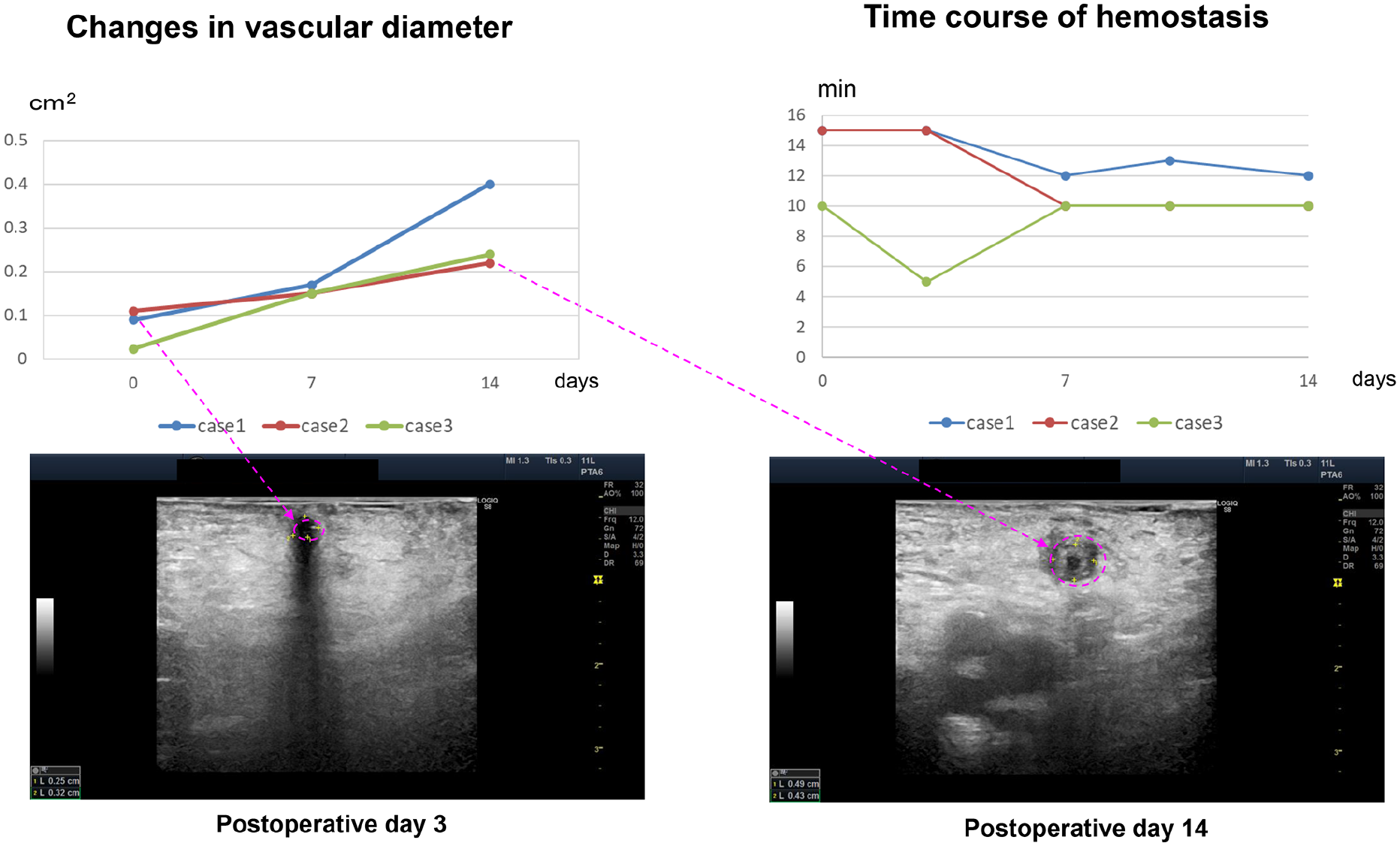
Changes in the vascular wall of VCF over time.
Clinical analysis
(i) Fistula creation
Early success (enabling dialysis) was achieved in all patients who underwent fistula creation. The amount of intraoperative blood loss was small in all patients, and the operation duration was 50.6 ± 21.6 min. No serious postoperative complications such as bleeding or infection associated with the procedure were observed.
(ii) Dialysis management using a fistula
(1) Infection rate
A total of four infections were reported in 40 patients. The infection rate was 4/13,420 total fistula maintenance days × 1000 = 0.30/1000 days of fistula maintenance. The shortest time from fistula creation to the onset of infection was 118 days and the mean time was 142.5 days.
In all cases, the use of a fistula for dialysis was discontinued for 7 days after the onset.
(i) Fistula exit-site infection
Exit-site infections were observed in two patients. In one patient, the responsible bacteria was identified as Staphylococcus aureus, and in the other, the bacteria could not be identified. In both cases, an intravenous drip with antibiotics resulted in prompt recovery.
(ii) Tunnel infection
Tunnel infection was reported in one patient. It was attributable to methicillin-resistant Staphylococcus aureus (MRSA). The event was resolved with local irrigation and intravenous drip with antibiotics. It seemed to have spread from the fistula exit-site.
(iii) Bloodstream infection
One event of bloodstream infection was observed in the study. It was attributable to MRSA. The event was resolved with an intravenous drip with antibiotics. The bloodstream infection rate was 0.075/1000 days of fistula maintenance.
(2) Patency rate
The primary patency rates were 78.9%, 61.5%, and 41.0% at 1, 2, and 3 years, respectively. The median patency duration was 25 months. The secondary patency rates were 87.0%, 63.8%, and 42.6% at 1, 2, and 3 years, respectively (Figure 5).
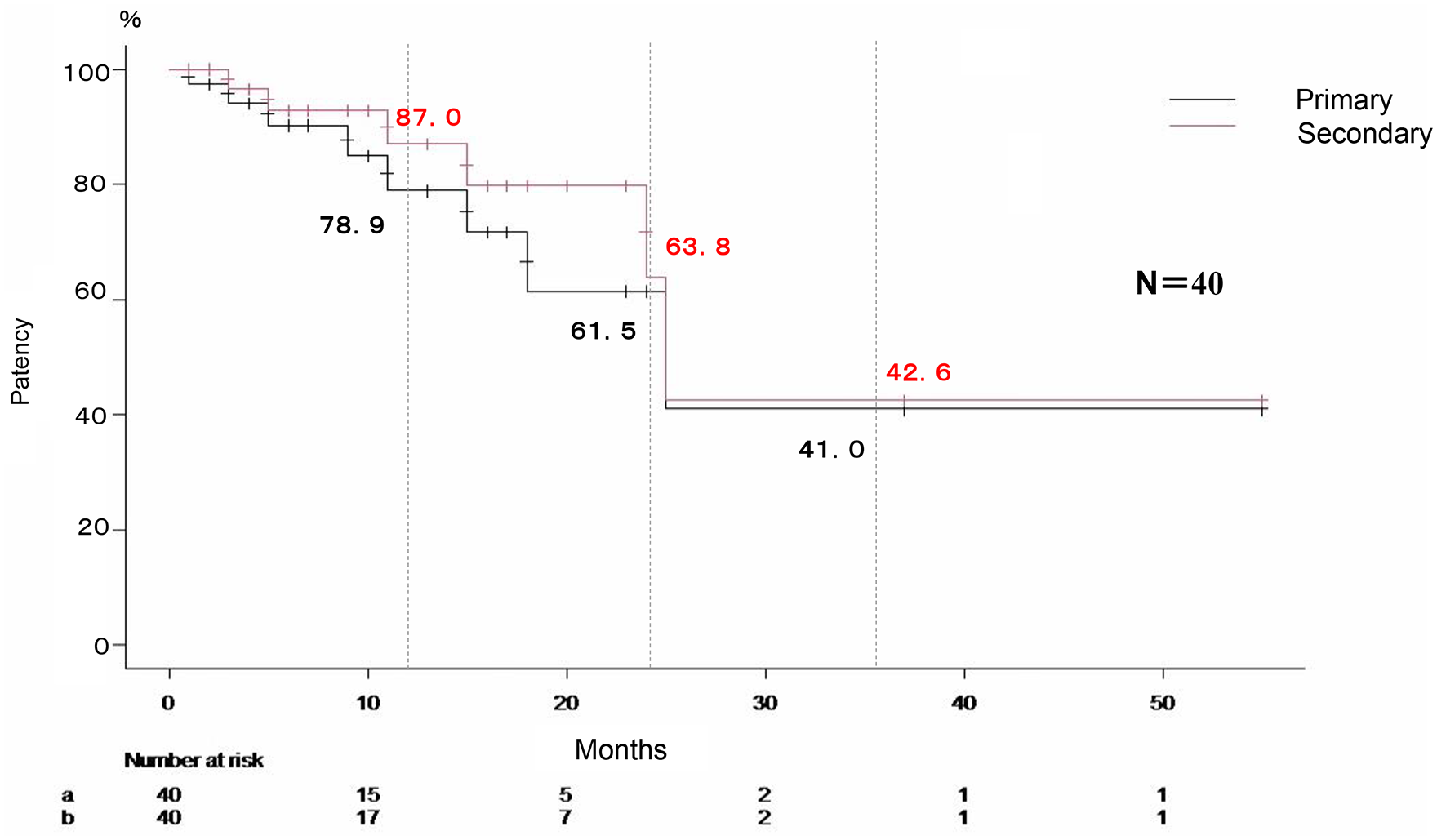
Primary and secondary patency rates of VCF.
The use of fistulas had to be terminated in four patients (nine limbs, including upper limbs) due to femoral venous thrombus occlusion associated with thickening and irregularity of the deep vein intimal membrane.
Discussion
The short- to long-term results of the venocutaneous fistula, a newly devised technique for patients who have difficulty creating or maintaining vascular access, are reported in this study. Long-term evaluations were performed to determine patency and infection rates. The results of this study were then compared to the results of previous studies in the literature.
The tunneled great saphenous vein had progressive intimal thickening in our study.
Fistulas created between the great saphenous vein and the skin rapidly develop a type of seal, and appear like a buttonhole4,5 at the puncture site. There are some reports in the literature on shear stress caused by great saphenous vein grafting after bypass surgery, venous intimal hyperplasia caused by internal pressure load, and intimal thickening.6,7 With VCF, reflux prevention by the venous valves and the thickening of the vessel walls create a type of seal between the vein and the skin surface. Vascular catheters penetrate this seal during cannulation, and re-sealing occurs when the catheters are removed.
Fistula creation was completed in a short period of time without complications in all cases; thus, we consider that the surgery itself has few problems if a skilled surgeon performs it.
Infection was not observed in the perioperative period, and all events occurred during the stable period once adequate wound healing and adhesion were achieved. The infections at the exit-sites observed in two patients presumably were instigated at the skin surface. Tunnel infection and bloodstream infection were observed in one patient each, and the cannulation procedure may be involved in the onset of these events; With regard to the infection rate in tunneled cuffed catheters, Uchino et al. 8 reported exit-site tunnel infection rate of 0.27/1000 catheter days, and bloodstream infection rate of 0.18/1000 catheter days. Murea et al. 9 have reported that the incidence of bloodstream infections varies by age within a range of 0.55–1.97/1000 catheter days. Righetti et al. 10 have reported that the incidence of catheter-related infection rates varies by the type of wound dressing within a range of 0.28–1.21/1000 catheter days, whereas that of bloodstream infection rate varies between 0.09 and 0.65/1000 catheter days. The KDOQI 2019 guidelines 1 set the control target for bloodstream infection caused by cuffed catheters as 1.5/1000 catheter days or less.
With this method, the overall incidence of infection was 0.30/1000 days of fistula maintenance, and that of bloodstream infection was 0.075/1000 days of fistula maintenance, which were both much lower than the reported incidence rates of infection with cuffed catheters or the control target.1,8–10 It is said that thrombi in central venous catheters are closely associated with infections. 3 With the VCF method, a closed venous lumen was observed because of the progression of vascular wall thickening associated with venous valve function and mechanical stimulation. Therefore, there was no involvement of foreign matter such as dead spaces or thrombi in the occurrence and spread of infection, and this resulted in the low incidences of tunnel infection and bloodstream infection.
With regard to the patency rate, Obialo et al. 11 have reported a patency rate of cuffed catheters as 42%–67% at 4 months. Further, Kakkos et al. 12 have reported a 6-month patency rate of 61%–94%, depending on the type of catheter used. Our method achieved significantly better patency rate (the secondary patency rates were 87.0%, 63.8%, and 42.6% at 1, 2, and 3 years, respectively.).
According to the “Japanese Society for Dialysis Therapy Guidelines for Vascular Access Construction and Repair for Chronic Hemodialysis,” 13 the secondary patency rates for AVG are set at 80% at 1 year, and 60% at 3 years after surgery. The patency rate achieved with VCF is thus comparable to these.
From the patient perspective, a VCF offers a lower risk of infection or thrombosis in comparison to a dialysis catheter. Additionally, VCFs offer a better cosmetic appearance and allow for showering and a greater degree of freedom in daily activities.
Study limitations
The possibility that the prognosis may have been influenced by the fact that the target patients were limited to elderly patients and long-term dialysis patients who had difficulty creating and maintaining AVFs and AVGs cannot be ruled out. The present data was a single-center, retrospective study. Proof of the usefulness of this method will require prospective comparative data with other methods for patients who have difficulty creating or maintaining AVF or AVG.
Conclusion
Since VCF has a favorable patency rate and a low infection rate, we consider that this is a promising alternative method for patients who have difficulty in vascular access creation via AVF and AVG.
Footnotes
Acknowledgements
We thank all the investigators and staffs participated in this study.
Author contributions
Study conception: MW; Writing: MW; Critical review and revision: YW; Final approval of article: MW, KI, KW, YW; Accountability for all aspects of the work: MW, KI, KW, YW.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by Bousei Daiichi Clinic Ethics Committee (Approval No. 2017-01).
Informed consent
Written informed consent was obtained from all individual participants included in the study.
