Abstract
Background:
Arteriovenous fistula (AVF) is the preferred angioaccess for haemodialysis but suffers from a high stenosis rate, juxta-anastomotic stenosis (JAS) being the most frequent. Percutaneous transluminal angioplasty (PTA) of JAS would have some advantage (such as mini-invasive and vein sparing treatment), but higher recurrence rate is observed as compared to surgery. We report results of juxta anastomotic stenosis PTA using the ‘double guide technique’ (DGT) as described by Turmel-Rodrigues, in a selected cohort from our Vascular Access Centre.
Patients and methods:
From January to June 2018, 25 consecutive patients were treated by DGT. By means of retrograde access through the outflow vein by a 6 F introducer, two guide wires were navigated: one into proximal radial artery (GW1), the other into distal artery (GW2). GW2 was used to dilate juxta-anastomotic vein and anastomotic area with 6 mm high-pressure balloon, while by GW1 juxta-anastomotic artery was dilated with 4 mm semi-compliant balloon. Mean diameter of balloons were 6.7 and 4.1 mm for venous and arterial tract dilatation. Follow up was carried out up to 12 months. Prospectively collected data were analysed retrospectively.
Results:
One-year primary and secondary patency was 52% and 95% respectively. Recurrence rate was 0.56 procedure/pt/year. Mean access blood flow at 12 months was 830 ml/min.
Conclusion:
Double Guidewire Technique is an effective and minimally invasive procedure. By avoiding under dilation of JAS the recurrence rate resulted quite satisfactorily in our population.
Keywords
Introduction
Arteriovenous fistulas (AVF) are the preferred vascular access for haemodialysis due to their better long-term patency along with lower rate of complications. 1 However, despite their advantages, AVFs are plagued by high stenosis rates. Therefore, a maintenance, by means of treatment of those stenosis, is needed to maintain functional patency. The most significant and common type of stenosis is juxta-anastomotic stenosis (JAS), which is found in 41%–64% of cases of stenosis complicating autogenous radial-cephalic fistulas (RCFs).2,3 One study from Grogan et al. 4 would infer, based from their results, that nearly 34% of AVFs will develop a hemodynamically significant juxta-anastomotic stenosis within 3 months from its creation. JAS is variably defined in the literature as a more than 50% reduction of the luminal diameter of the outflow vein within 2 cm5 or 5 cm6 from the arteriovenous anastomosis. The etiologic mechanism of JAS is unclear, multiple hypotheses exist as including the loss of the vasa vasorum during dissection for mobilisation of the vein, 7 kinking and increased turbulence at the anastomosis, torsional stress all along with low and fluctuating shear stress at this location. 8 A combination of these features on this segment of vein leads to intimal injury causing a cascade that result in neointimal hyperplasia and subsequent stenosis. 9 A debate still exists whether to treat all these lesions: the most recent DOQI guidelines 10 for example recommend to treat just stenoses greater than 50% only when there are concomitant clinical abnormalities such flow-rate reduction or dialytic circuit pressure changes. In general, the literature shows that treatment of hemodynamically significant stenosis would reduce the rate of thrombosis and graft loss and prolongs the average lifespan of the access. 11 Percutaneous transluminal angioplasty (PTA), compared to surgery, is by far the preferred treatment for vascular access stenotic complications. The main inconvenient of endovascular treatment is the high recurrence rate that can significantly reduce primary patency.
In the picture of JAS, the study from Tessitore et al. 12 has outlined how surgery showed a better primary patency compared to PTA. Secondary patency however did not differ between the two techniques, with a significant higher rate of re-intervention needed to maintain functional patency in those treated by PTA. Despite these results, endovascular approach may have some advantage for JAS resolution because PTA treatment is minimally invasive, spare AVF length suitable for venipuncture and is less prone to develop high fistula flow.
A special technique to treat JAS by PTA – named Double Guide Wire Technique – have been described by Turmel-Rodrigues et al.,13,14 aiming at the optimisation of early and long-term results. We report our single-centre experience on endovascular treatment of JASs by DGT in a selected cohort of patients.
Methods
We collected prospectively data of all consecutive patients treated for JAS by DGT, from January to June 2018; data were analysed retrospectively. All endovascular treatments (DGT) were performed by a single operator in a single centre. All patient were evaluated clinically and by colour-Doppler ultrasound (CDU) because of clinical abnormality arising from surveillance in dialysis. Inclusion criteria for endovascular treatment of JAS by DGT (vs surgical proximalization of the anastomosis), were: first occurrence of haemodynamically significant JAS in mature fistulas in patients with either (1) an anastomosis up to the mid third of the forearm or (2) risk of Haemodialysis Access Induced Distal Ischaemia (HAIDI) after excessive increase of blood flow as expected from surgical proximalisation. Haemodynamically significant JASs were defined as stenosis located at the anastomosis, or within the first 2 cm of the outflow vein, causing greater than 50% reduction in their diameter along with absolute low flow or flow reduction >30%. We judged also as haemodynamically significant stenosis with residual inner diameter <1.9 mm independently from flow data.
Risk of HAIDI syndrome was based on presence of low basal distal arterial perfusion (without HAIDI sign nor symptoms) which increased significantly after AVF compression. 15 Concomitant clinical abnormalities such inadequate flow (Qb below 600 ml/min) and consequent pressure change of the dialytic circuit were taken into account.
Among the participants at baseline, mean age was 71 years; 8 patients (32%) had diabetes and 21 (84%) had hypertension. Mean dialysis vintage (length of time on dialysis) was 31 months, with a range of 1–200 months, while the mean AVF vintage was 31 months, with a range of 2–204 months. Distribution of VA type was as follow: 8 patients (32%) had distal radio-cephalic fistulas, 13 patients (52%) had proximal radio-cephalic fistulas, 2 patients (8%) had distal ulno-basilic fistulas and 2 patients (8%) had brachial-basilic fistulas. The majority of patients (68%) have low flow (Qb < 600 ml/min) associated to JAS, while eight patients (32%) have a Qb > 600 ml/min but residual inner diameter <1.9 mm of stenosed tract. Mean flow rate of vascular access pre-PTA was 540 ml/min (Table 1).
Patient’s data.
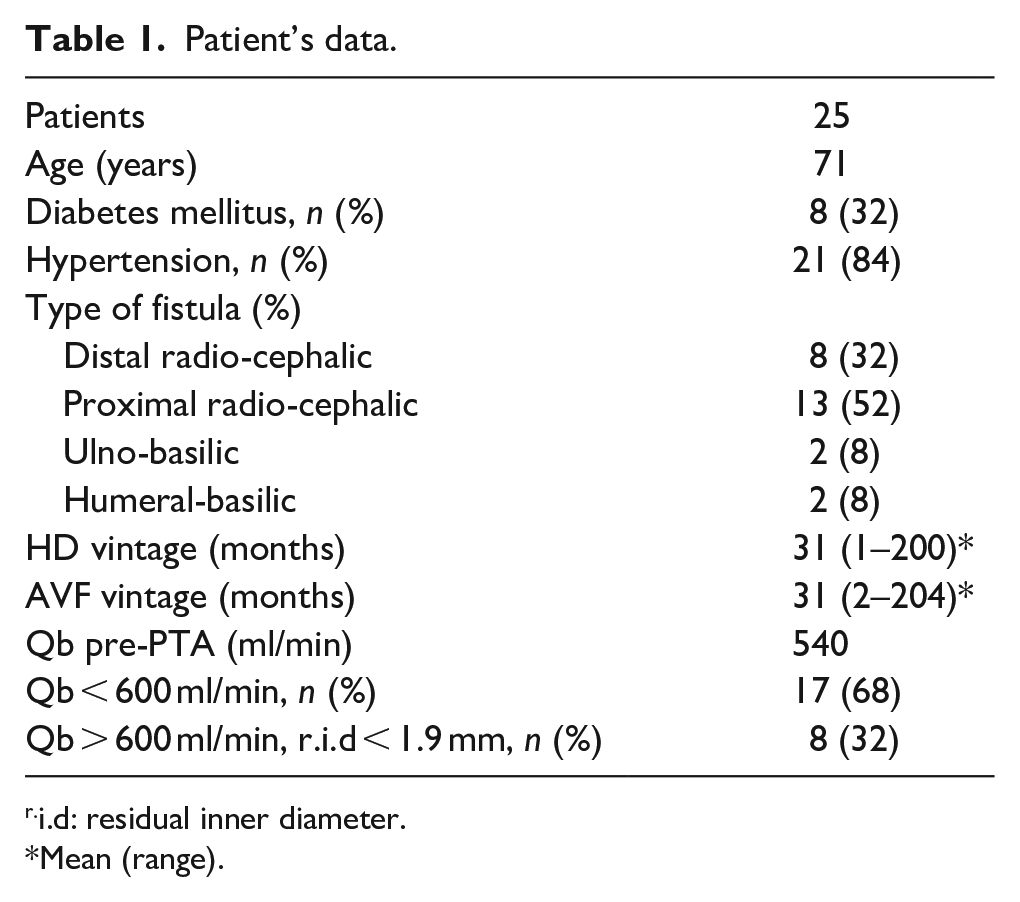
i.d: residual inner diameter.
Mean (range).
All patients were treated with PTA alone, without stenting, by DGT. Mean diameter of angioplasty balloons used was 6.7 mm (DS 0.55), in the venous tract (ultra-high pressure non-compliant), and 4.1 mm (DS 0.33) in the arterial tract (high pressure semi-compliant). Follow up was based on clinical surveillance and on parameters revealed by CDU examination scheduled within the first 1 month after intervention and repeated at 3, 6, and 12 months. All patients submitted informed consent for the required procedure; the Internal Review Board approved the analysis of data.
Pre-interventional evaluation
Before the intervention, all patient were evaluated by clinical and CDU examination performed in standardised environmental conditions (patient position, room temperature and ultrasound equipment) by the same investigator. Access flow rate (Qb) was measured by CDU, followed by further haemodynamic and morphologic exploration of the access. The Qb was automatically calculated by the software by the following formula: Blood flow (ml/min) = TAV (Time Averaged Velocity) × Area × 60, where TAV is the mean velocity referred to volume sample. 16 The site of volume was the brachial artery at the mid/proximal third of the arm. 17
Double Guide Technique (DGT)
All procedure were performed under local anaesthesia along with sedation. The technique is as described by Turmel-Rodrigues. In short, by means of retrograde access through the outflow vein by a single 6 F introducer, two guide wire were navigated (Figure 1): the first one into proximal radial artery (GW1), the other into the distal artery (GW2). The introducer was removed and repositioned over the GW2, the GW1 left aside in non-coaxial fashion. An ultra-high pressure non-compliant balloon 6 or 7×40 mm were inserted through GW2, the distal mark pushed at the curve drowned trans-anastomotically by GW1 (Figure 2). Inflations at 30 atm were performed. After a check for spasm, recoil or rupture, balloon, GW2 and the introducer were removed, the GW1 being left in place. The introducer was repositioned over the GW1 and a semi-compliant 4 or 5×40 mm balloon was pushed trans-anastomotically and inflated at 18 atm (Figure 3). The final angiographic control was performed and the GW1 and balloon removed after check for elastic recoil or rupture.
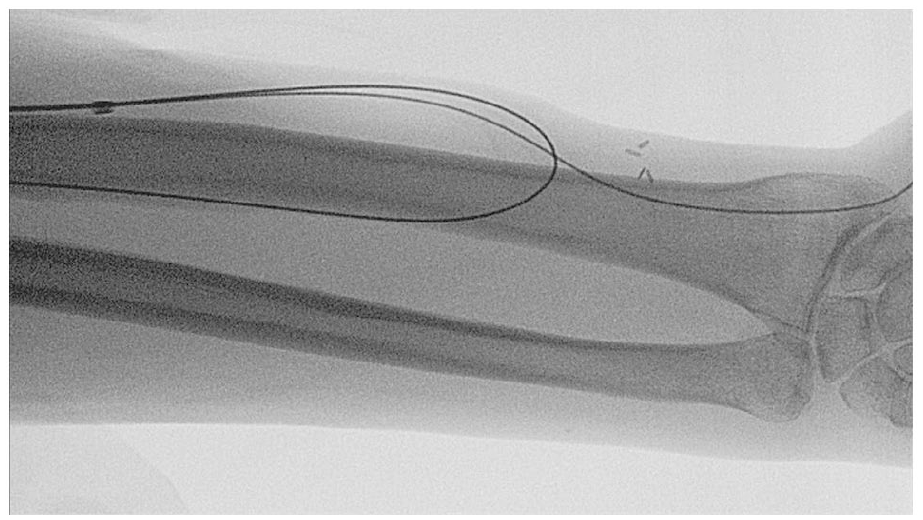
Two guidewire are navigated, one into proximal radial artery (GW1), the other into distal artery (GW2).

juxta-Anastomotic area dilated first with 6 mm high pressure balloon through GW2.

juxta-Anastomotic artery dilated finally with 4 mm semi-compliant balloon through GW1.
Anticoagulation with unfractionated heparin was routinely administrated (2000 UI). After manual compression for 10 min, a sterile dressing, non-circumferential but slightly compressive, was applied for 24 h.
Statistical analysis
Data were analysed using version 16.2.0 of MedCalc software. Categorical variables are expressed as percentage values, continuous variables are summarised as mean ± standard deviation (SD). Survival analysis was estimated by Kaplan-Meier.
Results
In total, 25 patients were treated by DGT because of JAS during the study period. Immediate procedural success was obtained in all cases. The only early complication occurred was mild extravasation in four patients (16%), treated successfully by endovascular haemostasis by low pressure prolonged inflation balloon tamponade. The length of the follow-up period was 12 months: 21 patients fully completed the observation, 1 death occurred from independent reason, 3 patients were lost to follow up. Primary patency at 1-year was 52%; meanwhile eight patients (32%) needed 1, one patient (4%) 2 and one patient (4%) 4 more procedures on the target lesion within the first 12 months because of stenosis recurrence. The recurrence rate in this series was therefore 0.56 procedure/pt/year. In one patient (4%) it was necessary to shift to Central Venous Catheter due to untreatable thrombosis occurred in intensive care unit. Secondary patency at 12 months was 95%. Mean access flow, as measured by CDU, was 830 ml/min at 12 months after treatment (Table 2). None of the patients had distal ischaemia (HAIDI syndrome) nor high flux associated with the treated AVF.
Results of endovascular treatment by Double Guidewire Technique.

Discussion
Arteriovenous fistulas are the recommended form of vascular access for patients with end-stage renal disease treated by haemodialysis. These fistulas are however susceptible to dysfunction due to stenosis, particularly in the juxta-anastomotic site. Treatment of JAS is controversial, with surgical and endovascular approaches each having advantage and disadvantages.
The endovascular approach is minimally invasive, allow venous length preservation, virtually no scars is left in place and the risk of infections is very low. Venous and arterial lesions are treated in situ and the normal tract immediately upstream or downstream is preserved. On the contrary, with surgery approach stenosis is to be treated creating a new anastomosis immediately above the area of stenosis, implicating loss of vascular heritage 18 and the relative risk of high flow. By surgery however the pathologic vein is bypassed and new stenosis occurrence is rare. The main concern of endovascular treatment is therefore recurrence of stenosis, and several reports have been focussed on that aspect. Clark et al. 19 reported a 1-year primary patency rate of 26% in fistulas located in the forearm and in the upper arm, stressing the role of length’s lesion and comorbidity as diabetes, peripheral and coronary vascular disease. Manninen et al. 20 reported primary patency rate of 20% at 1-year for arteriovenous anastomosis versus 52% in other anatomical locations.
The dilatation technique described in some reports would suggest however that sometimes stenosis remained under dilated,19,20 the reason being that the small diameters of the forearm arteries precluded dilatation of the juxta-anastomotic vein with large balloon. It is debatable whether in these cases we observe an early recurrence or a treatment failure. As the dilatation technique used has an important influence on success rate and 1-year primary and secondary patency rates, Turmel-Rodrigues et al.13,14 described the Double Guide Technique with rather satisfactorily results. His technique is based on the rationale of using the right sized balloon tailored on the three different juxta-anastomotic zones (artery – anastomosis – vein). We herein reported our single centre experience on dilatation of the juxtanastomotic stenosis by DGT. In order to recap the salient part of the technique we remind that the guidewire advanced into the distal artery allow for dilatation with high pressure 6–7 mm balloon of the juxta anastomotic vein and anastomosis surface area; whereas the guidewire into the proximal artery is used to dilate the juxta anastomotic artery with semi-compliant 4–5 mm balloon.
As a matter of fact, the venous tract requires dilatation with a larger diameter (6–7 mm) that fully engage the anastomotic chamber (possible with sliding on guide positioned in the distal artery). Arterialised, hyperplastic, and fibrous vein are often quite resistant to dilatation and high-pressure balloons with burst pressures over 25 atm must be used to efface completely the stenosis. The arterial tract, on the other hand, requires treatment with smaller diameter balloon of 4–5 mm (which would not be therapeutic for a venous segment), and are rather rarely resistant up to 15 atm. Therefore, a semi-compliant balloon – suitable for curvilinear transanastomotic tract – is enough. In summary the DGT made possible, by a relatively simple approach, to use specific therapeutic diameters for each anatomical tract (including anastomotic chamber), thus preventing relapses by avoiding under dilation. The final dilatation of the proximal artery is also beneficial by resolving any possibly spasm occurred at the inflow.
Our results are even better from those presented by the seminal description from Turmel-Rodrigues, but some difference between the series must be ascertained. In our series we selected just pure, first occurring juxta-anastomotic stenosis in mature fistulas, whereas in Turmel-Rodrigues experience also the most challenging immature and thrombosed accesses were treated. Therefore, a higher risk of recurrence is introduced. It must also be outlined that the selection of JAS occurring in previously mature fistulas exclude all the angioaccess failing because of surgical pitfalls (including twisting and coiling). In our view this is not a limit of the study, but a point of strength, as we tested the technique and its rationale on pure JAS lesions without the negative bias of trying rescuing “‘bad surgery’.
It is also worth to outline that given the moderate flow resulted at 12 months of follow up, any HAIDI syndrome has complicated the procedure in the short and mid-term.
The study of course has some limitations given by the retrospective analysis of a single centre experience Data however were collected prospectively and the single centre introduce the positive bias of an accurate test of the DGT.
In our view the endovascular approach is rather justified, by the use of DGT, especially in patient affected by JAS who are also at risk of HAIDI, with a limited length of the ponctionable AVF.
Given the very promising results we have expanded in our daily practice the indication for DGT for JAS to all the pure and de novo occurring lesion. The early recurrent patients – after first dilatation – being shifted towards surgical proximalisation of the arteriovenous anastomosis.
Conclusion
We described how, in our practice, endovascular treatments performed with Double Guide-wire Technique (DGT) have been effective for treatment of JAS in arteriovenous fistulas with a notable low recurrence rate. Using DGT is possible to avoid under-dilation of the juxta anastomotic venous tract, a fundamental premise for the prevention of relapses. DGT seem therefore to represent a minimally invasive, effective and safe treatment option for JAS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
