Abstract
Background:
Haemodialysis remains the most common modality of renal replacement therapy. National and international guidelines continue to promote arteriovenous fistulas or grafts as the preferred vascular access for haemodialysis, given the increased risks associated with use of central venous catheters (CVCs). Our renal centre pursues a ‘fistula first’ culture and uses root cause analysis and a patient safety incident based approach to meet the recommended standards of minimal CVC use in dialysis patients.
Methods:
We undertook a retrospective observational review looking at patterns of CVC use amongst our patients to identify themes and changes over time. Using data collected over a 5 year period, we examined 100 patient safety incidents involving CVC use in planned haemodialysis patients. We used a contributory factors framework to identify systemic contributors to each incident.
Results:
During the study period our centre achieved the national standard of at least 60% of incident dialysis patients commencing planned haemodialysis via arteriovenous access. About 26% of cases of CVC use in incident dialysis patients were deemed potentially avoidable. The most common contributory factor identified in these cases was poor communication.
Conclusions:
Using a root cause analysis based methodology to examine CVC use in haemodialysis is a novel approach to quality improvement in this area. Our methodology can be used as a framework by other centres to optimise the provision of safe, effective, and timely vascular access for dialysis, with multiple benefits for both renal services and individual patients.
Introduction
Patients with advanced chronic kidney disease (CKD) with continually declining renal function will, if appropriate, receive counselling on dialysis modalities to consider starting renal replacement therapy (RRT). Following dialysis education the majority of patients still opt for, or are limited to, haemodialysis (HD) as their modality of choice, which is most commonly undertaken in a hospital or satellite dialysis unit. 1
When HD is selected as the preferred choice of RRT, planning takes place to establish safe and appropriate vascular access through which patients can dialyse. Arteriovenous fistulas (AVFs) are considered to be the gold standard for vascular access, followed by arteriovenous grafts (AVGs). 2 In our centre, like many others, central venous catheters (CVCs) are only used in exceptional circumstances. 2 Examples of such circumstances are when multiple prior attempts to establish arteriovenous access have failed, or when HD is expected to be required for a short duration, perhaps due to impending transplantation or a temporary peritoneal dialysis (PD) sojourn with a return to PD planned in due course. 3
There are many possible CVC associated complications, of which the most feared is an increased risk of bacterial infection compared with AVFs or AVGs, 4 potentially resulting in life-threatening septicaemia. CVCs are also more likely to lose patency, require frequent replacement or intervention to restore patency, and overall have increased morbidity and mortality compared to arteriovenous access. 2 Work done previously has found up to a 3.4-fold increase in relative risk of death with CVC use versus AVF. 5
The ‘fistula first’ approach continues to be advocated both in the UK and internationally,2,3 including in the latest iteration of the Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines. Although the guidelines acknowledge that clinician judgement and patient characteristics are both important when determining the modality of vascular access, AVFs are still considered to be the safest and therefore best choice for most patients overall. 3 KDOQI go on to suggest that incident dialysis patients with CVCs should convert to an AVF or AVG ‘if possible, to reduce their risk of. . .adverse consequences’. Whilst opinions on the ‘fistula first’ approach have varied in recent years, our centre believes that an AVF should still be the access of choice in the vast majority of HD patients in line with KDOQI guidance.
To mitigate the potential harmful effects of unnecessary CVC use for HD, it is imperative that renal units monitor and attempt to reduce the number of patients dialysing via CVC in line with national guidance. In their clinical practice guideline for vascular access, the UK Renal Association advises ‘60% of all incident patients with established end stage kidney disease commencing planned HD should receive dialysis via a functioning AVF or AVG 2 ’. There are several reasons why dialysis centres may struggle to meet this target, ranging from patient and staff behaviours and attitudes towards dialysis access, to systems factors including access to surgeons trained in AVF and AVG formation. Not all of these factors are modifiable; however in many circumstances the use of a CVC for chronic RRT may be avoidable.
In line with expert consensus and for all of the reasons discussed above, our centre continues to pursue a ‘fistula first’ culture. Our tertiary renal unit in Birmingham, England, takes a forthright approach to this issue by utilising a Patient Safety Incidents (PSI) approach as a monitoring and improvement tool for dialysis access. PSIs occur in all areas of healthcare, and at their most serious can lead to fatal outcomes. 6 Effective investigation of PSIs is essential to optimise learning and to take action to prevent further similar incidents, with root cause analysis (RCA) being one method of doing this. 7 An RCA can be described as ‘a problem-solving tool used to identify how and why patient safety becomes compromised by a specific incident’. 7 Several events or factors, either at an individual or organisational level, often contribute to each incident, all of which should be examined within an RCA.
Our centre considers a vascular access PSI to have occurred if a patient who has been known to the pre-dialysis service (also referred to as the advanced kidney care clinic or low-clearance clinic) for 3 months or more commences HD without permanent vascular access (an AVF or AVG). We conduct RCAs on every new dialysis access PSI on a monthly basis using a ‘Contributory Factors Framework’ (CFF) which captures information on systemic contributors to incidents. 8 This helps to identify and apply learning points from every dialysis access PSI which occurs, and to pursue our philosophy of ‘fistula first’ at a local level.
Methods
As a product of monthly RCA meetings we have collated a significant pool of data relating to the number of incident patients commencing HD by each vascular access type. We undertook a retrospective observational review looking at patterns of CVC use amongst our patients to identify themes and changes over time. Our renal unit is one of two centres based in the large city of Birmingham, with a catchment area of 5 million people and looking after an average of 450 HD patients per month. In this study we looked at monthly data from April 2014 to March 2019, choosing to exclude the more recent period affected by COVID as there were several confounding factors affecting service delivery for dialysis patients at that time.
Within this time period there were 100 PSIs as defined above involving vascular access for HD. To meet inclusion criteria to feature in the database and analysis, patients must have been known to the pre-dialysis service within our centre for at least 3 months prior to starting dialysis, had not previously had HD, and had commenced HD via a CVC. There were no exclusion criteria, and patients were included in the analysis even if HD was not their first modality of RRT (e.g. patients who had previously had PD).
The RCA meetings are attended by the lead nurse and consultant for the pre-dialysis and vascular access service, as well as any other interested trainees and staff members from the dialysis units. On a monthly basis all patients who have commenced HD during the previous month via a CVC are discussed, and case notes are entered into a cumulative spreadsheet. For every CVC PSI it is considered whether any of a number of defined factors contributed to CVC use. These are: deviation from current systems, staff actions, inadequate staff training or skill, inadequate resources, poor communication, equipment failure, controllable environmental factors, uncontrollable external factors, or ‘other’ factors. A judgement is then made by those in attendance as to whether an action plan is required for potentially modifiable factors.
At the RCA meetings there is a ‘no blame’ culture, with the focus being on overcoming any barriers to early AVF/AVG formation and utilisation. In addition we foster an attitude of learning for the prevention of future events. The outcomes of each meeting are actioned by the lead nurse or consultant, including updating local policies, organising additional targeted training, and communicating learning points directly to relevant members of staff.
Results
The number of incident patients commencing planned HD via CVC ranged from 14.3% to 27.8% of total incident HD patients at our unit in each 12 month period. During our study period this always remained within the national standard set by the UK Renal Association of 60% of incident patients with end stage kidney disease commencing planned HD via an AVF or AVG.
We broke this down further into avoidable and unavoidable CVC use (see Figure 1). 100 PSIs involving vascular access for dialysis were reported during the study period. Of these, 26% (n = 26) were considered to have been avoidable. The most commonly cited contributing factor in the cases deemed to be avoidable was poor communication (42.3%, n = 11), which refers to communication within or between clinical teams, or between healthcare professionals and patients. This was followed by staff actions, for example late referral for vascular access planning (30.8%, n = 8), then controllable environmental factors, such as patient reviews happening outside of formal clinic pathways and causing delays (23.1%, n = 6). Inadequate staff resources (19.2%, n = 5), which largely referred to a lack of surgical capacity, and inadequate staff training or skill (7.7%, n = 2) also contributed in a smaller number of cases. In some cases multiple contributing factors were recognised. Of note, in a small number of cases deemed avoidable it was documented that the patient may have benefitted from formal support by a psychologist during dialysis planning.
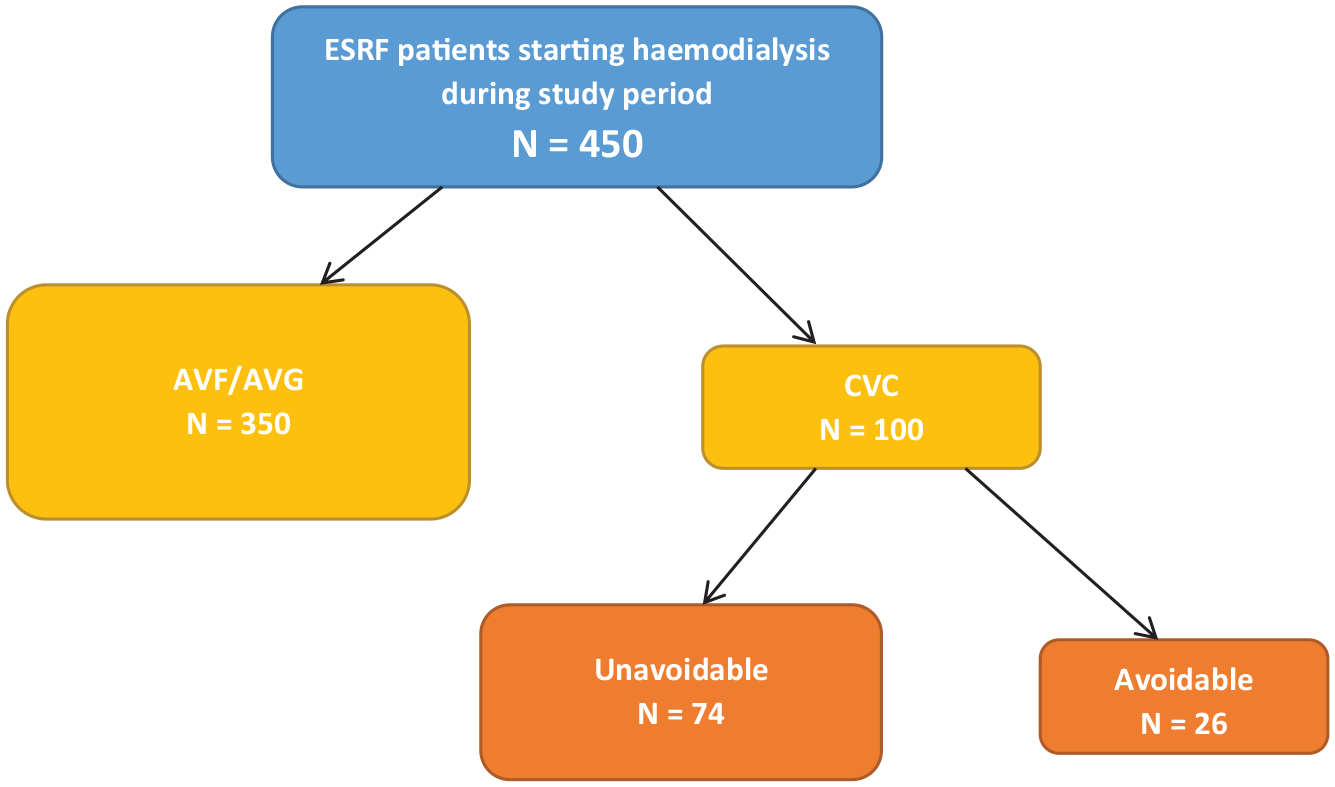
flow chart demonstrating type of vascular access used for planned haemodialysis during the study period, and the classification of CVCs as avoidable or unavoidable by the vascular access team.
Amongst the cases examined as a PSI that were deemed unavoidable, there were multiple factors which led to patients commencing HD via CVC. These included patients becoming acutely unwell with a rapid and unpredictable deterioration in renal function, issues with PD catheters or infections, and many cases where patients declined early dialysis planning. During the study period there were also a number of new HD patients who were not known to the pre-dialysis service, therefore these were not considered as vascular access PSIs.
Discussion
Using an RCA based methodology to examine CVC use in planned HD is a novel approach to quality improvement in this area. As far as we are aware this is not undertaken in a similar timetabled, comprehensive format in any other centre in the UK or worldwide. Our methodology can be used as a framework to optimise a dialysis centre’s approach to providing safe, effective, and timely vascular access, with the potential implications for clinical practice being wide ranging. At the level of each trust or dialysis centre, accurately monitoring and proactively discussing cases of dialysis initiation with a CVC promotes a continuous learning culture within the renal team and wider governance and quality assurance frameworks. It may also stimulate increased collaboration with other colleagues such as vascular surgeons based on the outcomes of these discussions.
As this methodology helps to identify problems at a systems level at the earliest opportunity, it enables prompt action to be taken to improve clinical pathways, provide further training to staff, or acquire extra resources as needed. Overall this approach could enable more centres to reach national targets for reducing inappropriate CVC use, with the related financial incentives and the positive reputation associated with the provision of a high standard of care as defined by professional bodies. It is possible to initiate this approach with existing staff and resources, without extra cost, and it has the additional advantage of being replicable across multiple sites within a trust.
There are also many potential benefits to reducing CVC use at an individual patient level. The evidence for increased risk of bacterial infection with an indwelling CVC is compelling.4,9 Fewer events relating to access complications,1,2 and therefore fewer hospital attendances, will positively affect a patient’s quality of life as well as their health. Patients benefit from strengthened systems and more streamlined care pathways put in place during the continued learning from RCA meetings. They will also benefit from care delivered by an informed, collaborative team in whom they will be able to place their trust.
Multiple professional bodies including the UK Renal Association and others worldwide support a reduction in the use of CVCs for HD, therefore renal centres must strive to meet national targets and standards. However, it is also important to acknowledge that there are cases where an AVF or AVG may not be the best modality of vascular access for a patient. In particular there is evolving discussion in the literature regarding vascular access for dialysis in older patients. Elderly patients with advanced CKD are often frail, and if dialysis is initiated in this group the mortality risk is high. 10 The procedural burden of fistula formation in particular may be off-putting in this patient group, 11 and several papers have argued that an AVG specifically should be strongly considered as the vascular access modality of choice in older patients.12,13 There are also patients in whom upper limb vasculature is not suitable for AVF or AVG formation, including those with diabetes or significant atheromatous disease. 11 In circumstances such as these, it may not be possible or even desirable to avoid CVC use. Therefore, whilst the arguments for reducing CVC use in HD are robust, clinicians and patients must collaborate to make the best choice for each individual.
We acknowledge that this study has some limitations. The assessment of whether a vascular access incident is avoidable or unavoidable is in part subjective; whilst in some cases it is immediately apparent that an action or inaction caused a delay in AVF or AVG formation, many cases are less clear cut. There are also likely to be confounding factors affecting CVC use, or non-use, in HD, for which it is not possible to adjust.
With this work we demonstrate that there is a correlation between our centre’s RCA based approach to reducing CVC use in incident HD patients, and meeting the gold standard national recommendations for vascular access in this group. This approach can in turn improve clinical practice and reduce the risks associated with CVCs. We would recommend that all dialysis centres adopt this approach in order to audit and quality assure their vascular access practice on a regular basis.
Footnotes
Acknowledgements
The authors gratefully acknowledge the contribution of the Vascular Access Team and Kidney Failure Support Team at Birmingham Heartlands Hospital for their help with data collection for this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
We confirm that Ethical Committee approval was not required for this work as all data was fully anonymised. Verbal consent was obtained from patients at the point of clinical care.
