Abstract
Background:
CVCs are defined ‘complex’ when they are inserted through non-conventional accesses or positioned in non-usual sites or substituted by IR endovascular procedures. We report our experience in using diagnostic and interventional radiology techniques for complex CVC insertion and management; we recommend some precautions and techniques that could lead to long-term availability of central venous access and to avoid non-conventional sites CVC insertion.
Methods:
We retrospectively evaluated 617 patients, between January 2010 and December 2019, (mean age 71 ± 13; male 448/617), treated in our department for insertion of tunnelled CVC for haemodialysis.
Results:
Among 617 patients, 241 cases (39%) are considered ‘complex’ because they required either a PTA with or without stenting to restore/maintain venous access or had an unusual positioning site or required unconventional access. A direct correlation between CT angiography and PTA (r = 0.95; p-value <0.001) and an inverse correlation between CT angiography and unconventional ‘rescue’ access (r = −0.92; p-value <0.001) were found.
Conclusions:
Precise pre-operative planning of treatment in a multidisciplinary setting and diagnostic and interventional radiology procedures knowledge allows reducing complex catheterisms in haemodialysis patient.
Keywords
Introduction
Nowadays End Stage Renal Disease (ESRD) represents a very widespread condition in the world, affecting an increasing number of patients.
Despite the strong evidence that an autologous arteriovenous fistula (AVF) is superior to a haemodialysis catheter for long-term dialysis, there are certain scenarios in which catheter is the preferred method. These include the need for immediate dialysis or a bridge to further permanent haemodialysis access, comorbid conditions limiting life expectancy, patients in whom attempts to create/maintain a permanent access site have failed and those in whom all available sites for fistula are no longer feasible.1–3
About the 80% of patients with ESRD receive the insertion of a central vein catheter (CVC) during their pathology course4,5 and in almost half the countries participants to Dialysis Outcome and Practice Pattern Study (DOPPS), 50% of them started dialysis therapy with a CVC in a variable percentage between 23% and 73%.
This large number of patients has been observed despite 60%–79% of them was previously evaluate by a nephrologist more than 4 months before starting the therapy and 69%–88% of them 1 month before. For all patients evaluated less than 1 month before, the use of a CVC has a 50%–90% range. 6
Despite their higher rate of infection and failure than other types of access, CVC use remains very frequent. 7
Numerous and continuous revisions, repositioning and substitutions of CVC increase the risk of complications like thrombosis, stenosis, occlusions and infections which are responsible of almost 70% of CVC failure. 8
Malfunctioning caused by thrombosis and stenosis for AVF while thrombosis and dislocation for CVC, represent a clinical urgency as they do not allow dialysis.
In these clinical scenarios, Interventional Radiologist has a crucial role not only to resolve complications, but also to try to maintain a patent and working vascular pathway for a possible further CVC insertion.
CVCs are defined ‘complex’ when they must be inserted through non-conventional accesses or positioned in non-usual sites because of progressive depletion of the venous asset7,9; complex management of CVC is considered when IR endovascular procedures as angioplasty (PTA) with or without stent placement are necessary to their substitution or insertion. To date, in the literature there is no univocal definition, and there is a lack of this issue in recent KDOQI vascular access guidelines. 9
Non-conventional access sites are considered trans-lumbar, trans-renal, transhepatic access and trans-collateral veins. 4
In this study we report our experience in using Interventional Radiology techniques for complex CVC insertion. Moreover, we report recommended precautions and techniques for their management that, in our experience, could lead to long term availability of central venous access and to avoid non-conventional sites CVC insertion.
Methods
We retrospectively evaluated 617 patients, between January 2010 and December 2019, (mean age 71 ± 13; male 448/617), treated in our department for insertion of tunnelled CVC for haemodialysis.
All patients suffering from ESRD were sent by the Nephrology Department to our Department of Interventional Radiology for not recoverable malfunctioning AVF or for malfunctioning CVC.
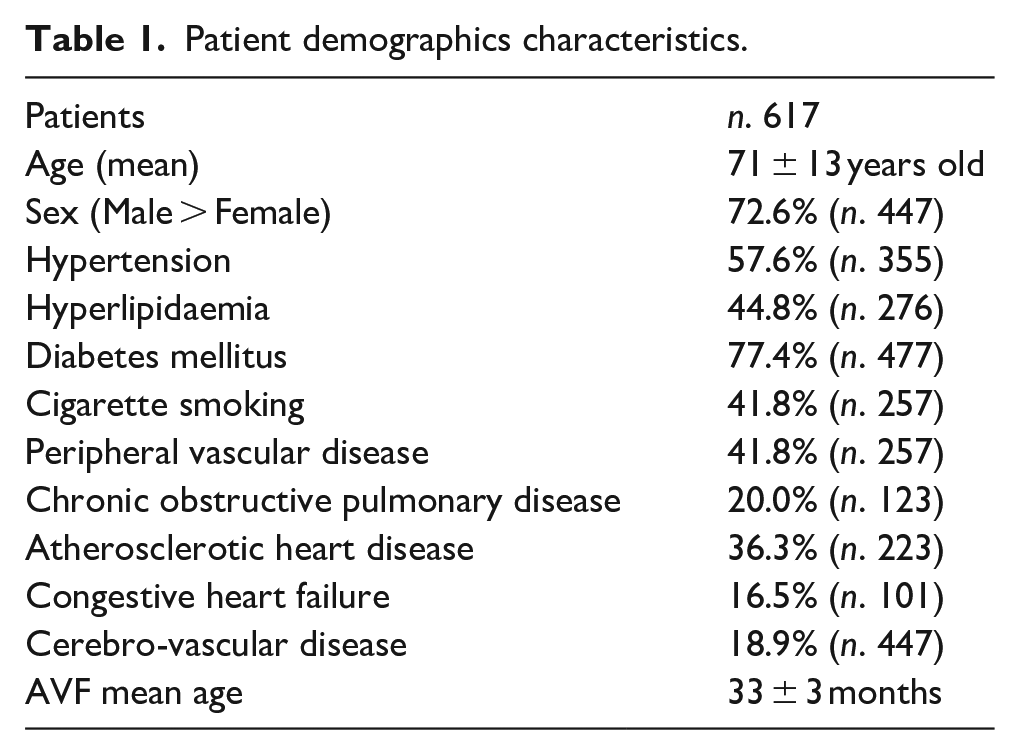
More detailed patient demographics characteristics are shown in Table 1.
Patient demographics characteristics.
Formal consent or IRB approval is not required for this type of study; informed consent was submitted to all patients before each procedure.
We evaluated how many patients underwent a ‘complex’ catheterisation and of these, how many underwent a catheterisation through an unconventional access.
We also assessed the number of performed PTAs (with or without stenting) necessary to maintain the patency of the venous way in the following cases: (1) correct ex-novo insertion of a CVC; (2) removal with subsequent replacement of a malfunctioning CVC (using the same access or a new access); (3) removal of malfunctioning CVC without insertion of a new catheter.
We considered the number of preliminary radiological investigations, in particular the number of Computed Tomography-angiography in venous phase (CT-Angio); then we evaluated how a preliminary CT-Angio can contribute to the management of complex and extreme catheterisations, observing the incidence trends for every year.
For each patient was performed a detailed anamnestic analysis of the vascular access’ history, including possible previous positioning of CVC, and was performed a careful physical examination (presence of superficial venous collaterals, oedema or local swelling); we also evaluated the venous anatomy and the possible presence of anatomical variants. Each patient underwent a preliminary imaging before the procedure to obtain a vascular map to plan the best possible treatment strategy. These were Eco-Colour-Doppler (ECD), phlebography and venous-CT-Angio, with a chronologically increasing trend for CT-Angio compared to phlebography.
In all cases, has been evaluated the central and peripheral venous availability (i.e. internal and external jugular veins, subclavian veins, anonymous left and right venous trunk, superior vena cava, common femoral veins, iliac veins, inferior vena cava and any collateral of satisfactory calibre).
ECD was performed before phlebographic or CT-Angio examination, to obtain further information and to indicate the execution of CT-Angio, to choose the access site for the interventional procedure and to get haemodynamic conditions.
All venous-CT-Angio were integrated by multiplanar reconstructions (MPR) and by specific vascular analysis using dedicated software, to evaluate the possible insertion site, the presence of central venous stenosis or obstructions related to previous AVF or CVC and to plan the procedure (PTA and/or stenting).
Then the patients were conducted in the angiography suite for the positioning/revision/removal of the CVC and for the treatment of any stenosis/thrombosis/obstructions.
When CVC must be removed or replaced (due to a stenosis or obstruction along the course of CVC, as demonstrated in preliminary radiological studies), PTA of the CVC route was performed because, in these patients with limited vascular reserves, it is important to guarantee patency of the vascular axis where CVC is placed, to allow possible future positioning of a further CVC and to avoid asymptomatic steno-occlusions.
Results
Between January 2010 and December 2019, 617 patients underwent the insertion of a tunnelled CVC at our Department: among these, 241 cases (39%) are considered with a ‘complex’ management because they required either a PTA with or without stenting to restore/maintain venous access or had an unusual positioning site or required unconventional access (‘rescue access’).
Among these, 222 patients (92.1% of complex CVCs, 35.9% of total CVCs) had a conventional access site, while 19 patients (7.8% of complex CVCs, 3% of total CVCs) had unconventional access or had an unusual CVC location: five in the azygos vein, four in the intercostal veins and nine in other collateral veins; in one case a trans-lumbar access to the inferior vena cava (for depletion of other possible vascular accesses) was performed. In our series there are no reported cases with trans-hepatic or trans-renal access.
In all patients a PTA was performed (241), and 11 self-expanding stents and eight covered stents were placed: six in right anonymous vein (three of which covered), three in left anonymous vein (one of which covered), three in right subclavian vein (one of which covered), one in left subclavian vein (covered), two in superior vena cava, three in right iliac vein (two of which covered) and one in left vein iliac. All the patients treated with PTA have a history of a previous CVC in the same vein or an ipsilateral AVF causing central vein stenosis.
A total of 185 (76%) preliminary CT-Angio were performed; analysing the number of exams performed for each year, we qualitatively noticed a progressive increase of CT-Angio exams corresponding to the increase of PTA performed; on the contrary, with the increase of CT-Angio performed, the number of catheters with unconventional ‘rescue’ access has decreased (Figure 1).
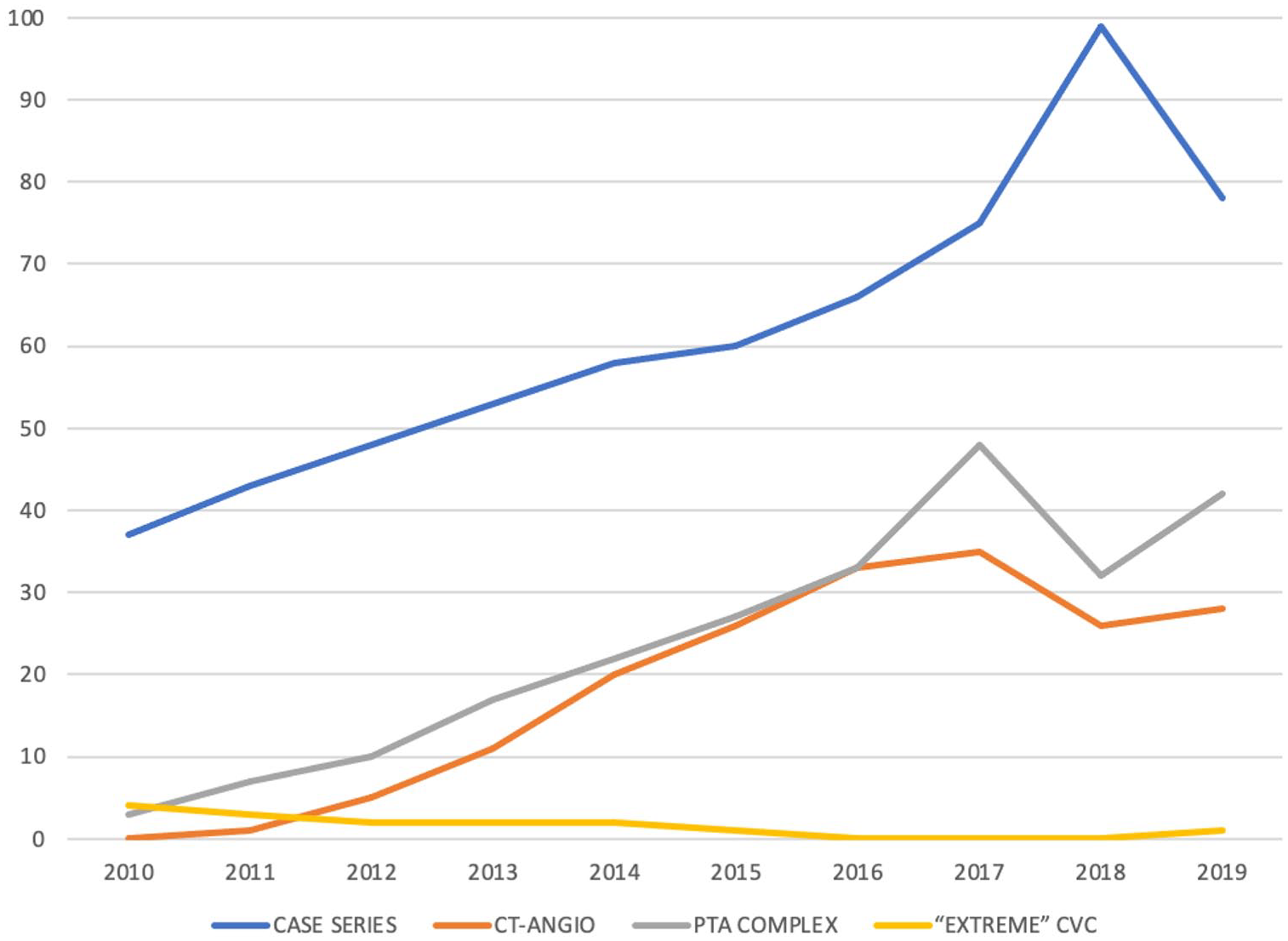
Comparison of frequencies sorted by year of: total number of cases, CT-angio exams, PTA performed in Complex CVC and extreme CVC.
To confirm our assumption, the Pearson correlation coefficient (r) was calculated to measure the linear association between two variables X and Y as:
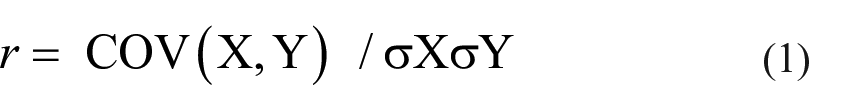
where COV is covariance, σX is the standard deviation of X and σY is the standard deviation of Y. The r coefficient ranges between −1 and 1 where: −1 indicates a perfect negative linear correlation, 0 indicates no linear correlation and 1 indicates a perfect positive linear correlation.
As result, a significant high positive correlation (i.e. direct correlation) between CT-Angio and PTA (r = 0.95; p-value <0.001) and a significant high negative linear correlation (i.e. inverse correlation) between CT-Angio and unconventional ‘rescue’ access (r = −0.92; p-value <0.001) were found confirming our assumption. Finally, correlation graphs showed the same results in graphical form, as shown in Figure 2(a) and (b).
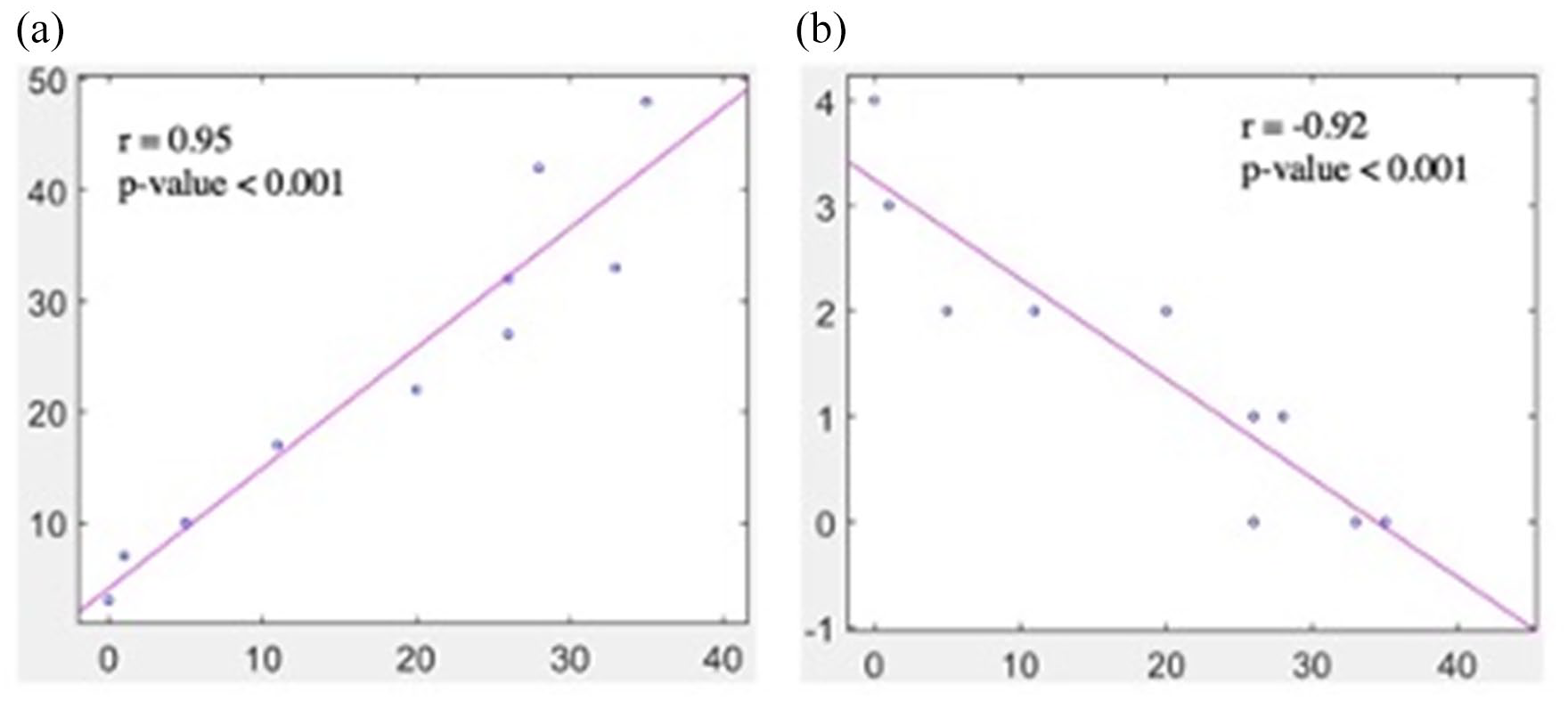
(a) and (b) Correlation graphs between CT-angio and PTA (r = 0.95; p-value <0.001) and between CT-angio and extreme CVC (r = −0.92; p-value <0.001).
Discussion
The presence of CVC in a central venous vessel can cause of multiple complications that might affect its vascular patency and therefore their future use (of both venous accesses and the same catheter for dialysis). The most frequent complications related to the CVC are stenosis, thrombosis and venous occlusion, as well as infections. The incidence of central venous stenosis in patients with CVC range between 3% and 50%, with the lowest rate between 3% and 10%, in case of access from the internal jugular vein. 10 The growth of a fibrin sheath around the tip of catheter, the continuous mechanical wall stress given by increased and turbulent blood flow, as well as the decubitus of CVC in the vascular wall, contributes to the onset of a stenosis and/or venous thrombosis. They also results in neointimal hyperplasia and stenosis. The presence or the creation of the AVF, especially if proximal and with high flow, accelerates and increase this flow anomaly.1,10–12
In a precarious condition of gradual development of stenosis and collateral venous branches, a new catheter insertion, in the absence of an appropriate pre-procedural planning, can further worsen the clinical setting and triggering a symptomatic set such as the big arm syndrome, the superior vena cava syndrome and the creation of collateral circles and neurological complications (Figures 3 and 4).13,14
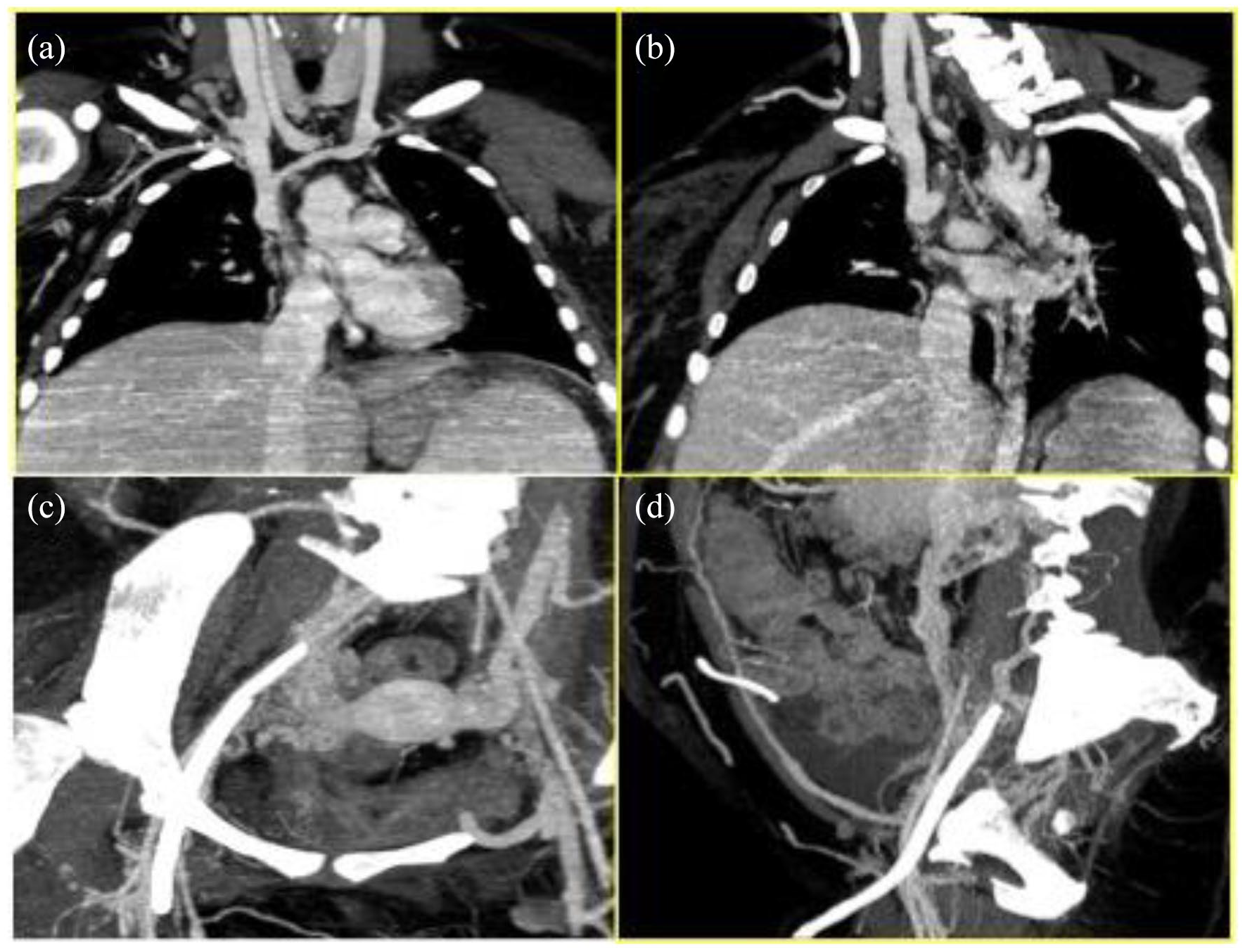
Patient 1: pre-procedural CTA (a and b) superior vena cava occlusion with hypertrophic azygos arch and (c and d) malfunctioning right femoral CVC with obstruction of homolateral iliac-femoral axis
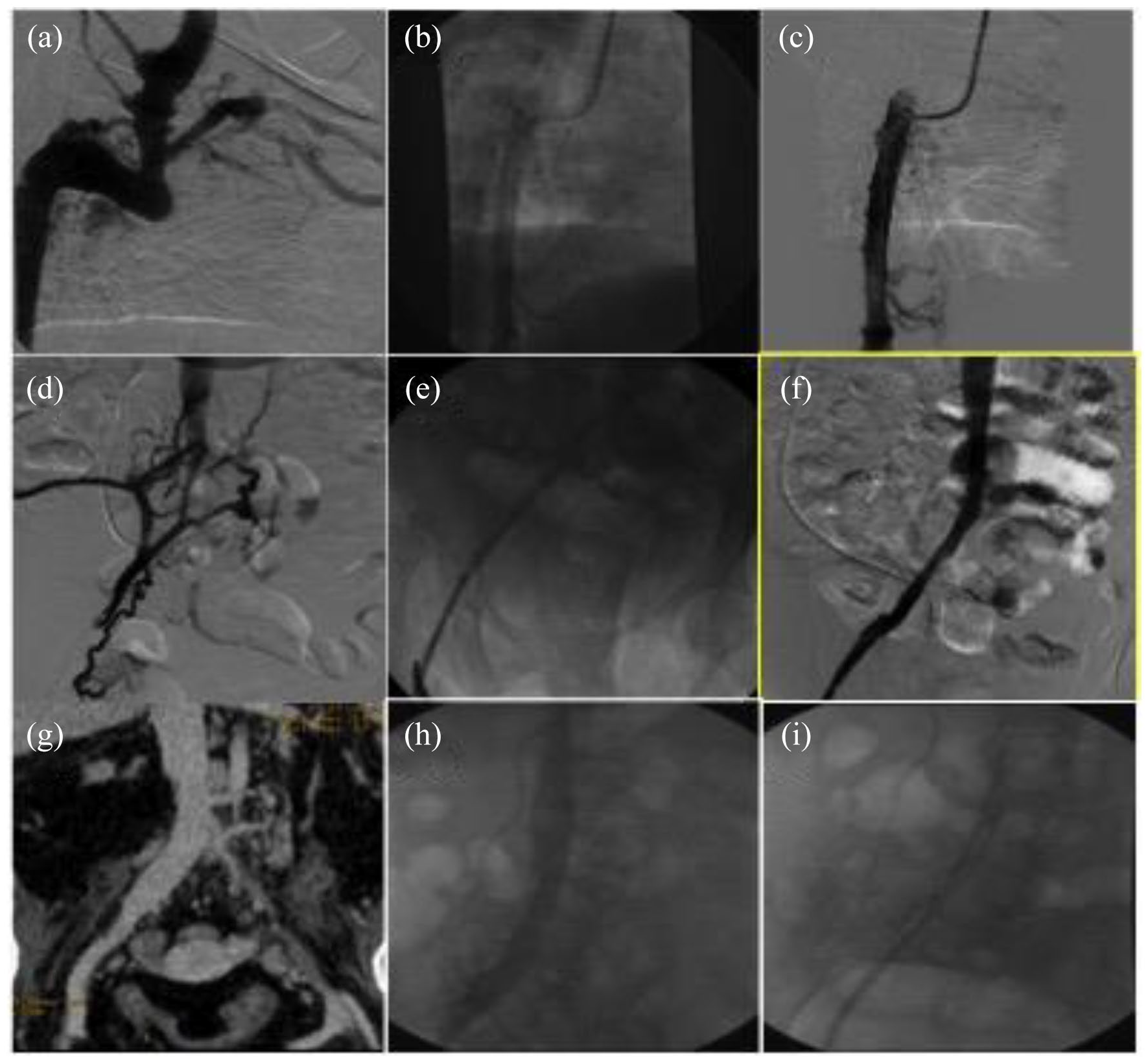
Patient 1: (a) angiography demonstrating patent and hypertrophic azygos vein, (b) CVC placement into the azygos vein, (c) phlebographic control, (d) angiography demonstrating the right iliac axis obstruction with the development of collaterals, (e) angioplasty of the right iliac vein, (f) post-angioplasty phlebographic control and CVC removal, (g) 8-months CTA confirming the patency of iliac-caval axis, (h) confirmation with phlebography and (i) fluoroscopic control of the CVC placement in the iliac axis.
According to our experience, therefore, we firmly believe that is mandatory to carry out a preoperative radiological planning with Venous-TC-Angio (including thorax and abdomen in the acquisition)15,16 in order to perform with multiplanar reconstructions and vessel analysis a correct intervention strategy; this allows first of all the maintenance of vascular patency, the treatment of complications, the insertion of a CVC that can remain functional as long as possible effectively reducing access to the angiography.15–19
It should be noted that performing a preliminary venous-CT-Angio, could leads to an increase in PTA and therefore to catheterisations with complex insertion, but could reduce the incidence of catheterisation with unconventional access with bailout techniques, which are certainly related to a lower patency over time.
In our opinion the management of these delicate patients should be focussed not only to settle the impending problem, but also to prevent a bad evolution, avoiding as much as possible future complications to easily manage the clinical course of the patients.
We use the ECD as preliminary diagnostic evaluation and it often leads to the execution of CT-Angio; it also provides information about the state of the fistula and the peripheral venous vessels, preliminarily assess whether these vessels are available for percutaneous puncture and for subsequent insertion of vascular introducers, finally it provides haemodynamic information to evaluate any flow turbulence.
The central venous compartment must be preserved as much as possible in haemodialysis patients as it represents a life-saving district for CVC insertion, indeed it often represents the last chance to create a good and long-lasting vascular access to continue dialysis once there are no more peripheral venous branches available.
For this purpose, it seems useful to choose as access site in the first step, one of those sites with known stenosis or occlusions, to preserve any other venous accesses intact; many authors suggest that longevity and complications of CVC inserted into occluded or collateral veins compared to conventional methods show no difference in terms of complications, dialysis flow rate, catheter bacteraemia or access longevity.2,20
It is to underline that our goal is to recanalize with a PTA the stenotic central venous vessels in which CVCs are already positioned, allowing both the positioning of a possible new CVC and its good functioning, as well as maintaining the patency of the vessel regardless of the CVC replacement or its removal; in the latter case, the aim would also be to preventively guarantee patency in order to allow any future use of this route for new CVC positioning (Figures 5 and 6).
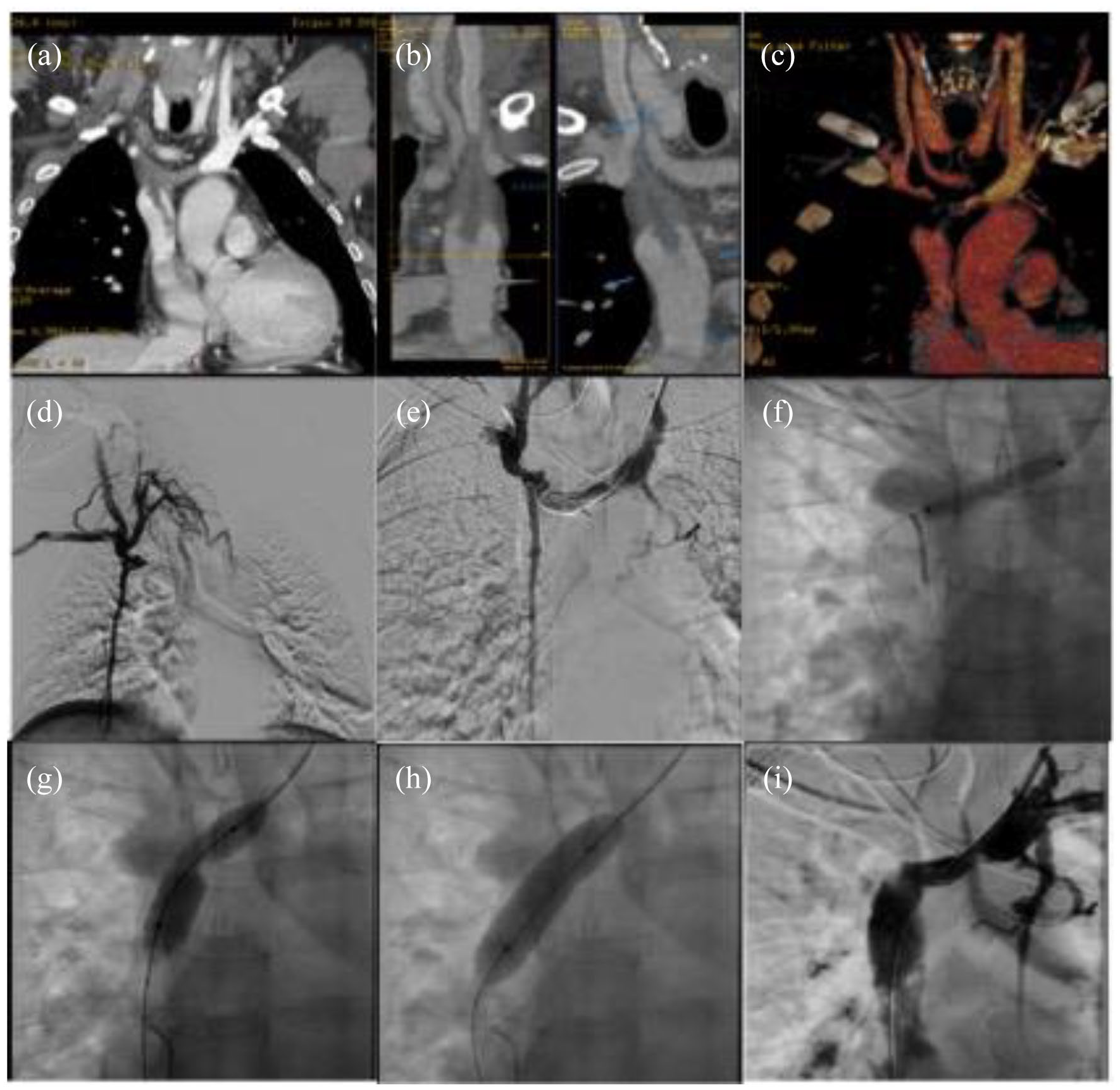
Patient 2: (a–c) CTA MPR e VR reconstructions demonstrating tight stenosis of both brachiocephalic veins, thrombosis of their confluence and floating thrombus into the superior vena cava, (d and e) phlebography confirming tight stenosis of brachiocephalic veins, the presence of collaterals and no opacification of the superior vena cava, (f–h) progressive angioplasty of the left brachiocephalic vein and (i) phlebographic result with good opacification of both left brachiocephalic vein and superior vena cava.
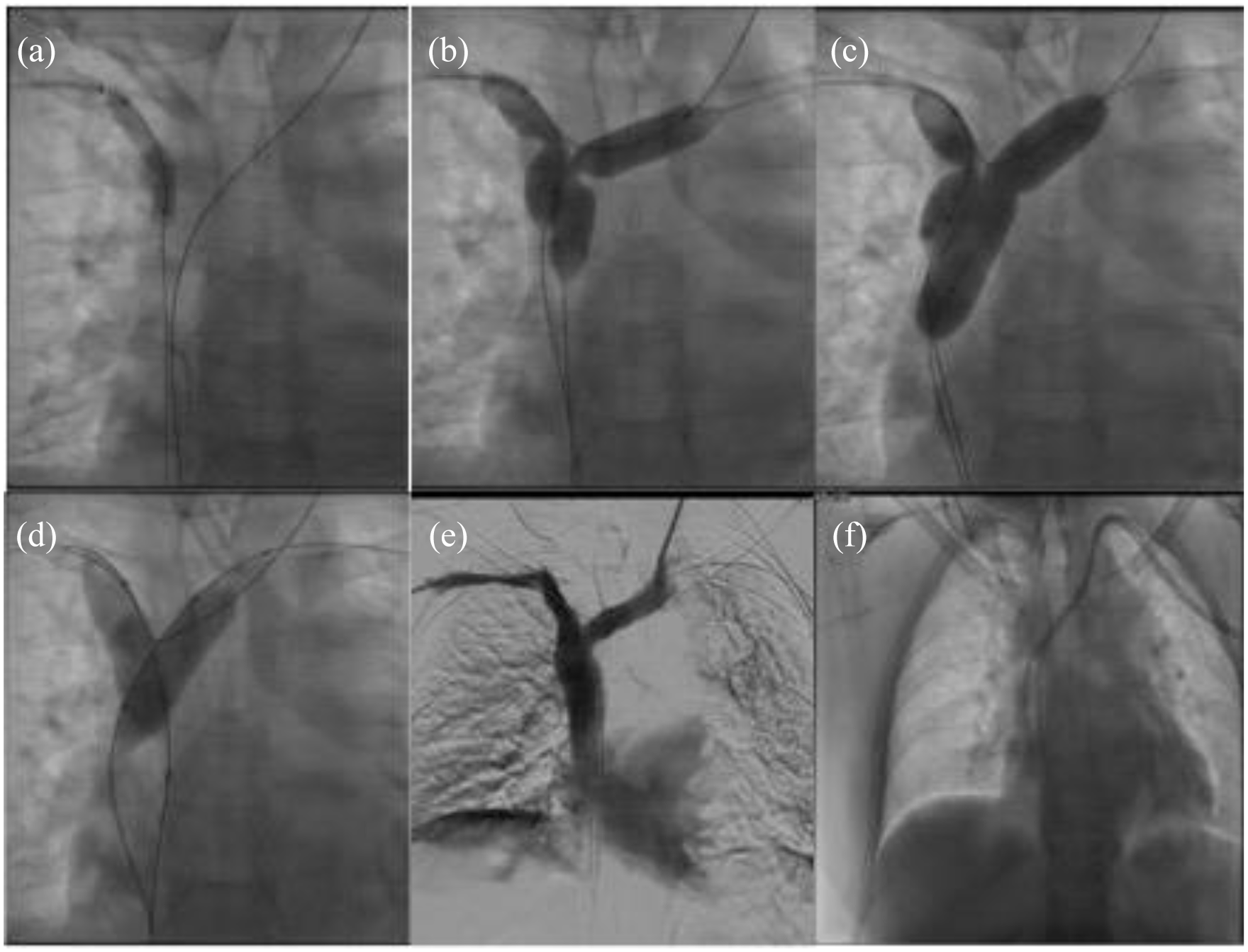
Patient 2: (a–d) progressive angioplasty of right brachiocephalic vein first and then of both brachiocephalic veins (kissing balloon), (e) phlebographic result with good opacification of brachiocephalic veins and superior vena cava and (f) tunnelled CVC placed into the superior vena cava with left internal jugular access.
It is very important to breakdown the fibrin sheath with progressive PTA 8 to preserve the venous axis for a possible future reuse of the access and, in our treatments, we generally avoided the use of a stent in the first instance, trying to obtain a satisfactory result only with the PTA, even using drug-eluting balloon – DEB (to ensure a longer lasting efficacy of the treatment and vascular patency), as also performed by other authors in the same district18,21,22 (Figure 5).
The use of metal stent in maintaining patency of central venous system has already been widely reported.2,23–26 Quaretti et al. 27 retrospectively observed a better primary patency for covered stent (stent-graft) and a lower rate of reoperations compared to uncoated stent (bare metal stent) and standard angioplasty (PTA).
In our series, in all cases in which we used a stent (to exclude the entire region of stenosis) we never reported complications such as migration, dislocation or vessel rupture, while some cases are reported in literature. 28
The rupture of a mediastinal vessel during PTA of the central venous vessels (in particular SVC or Cavo-atrial junction) is a fearsome complication, although very rare: we recommend progressive dilation without an overestimation of the balloon diameter. When we perform a procedure in superior vena cava under the azygos vein confluence or at the Cavo-atrial junction we always prefer a primary stenting with covered-stent due to the risk of rupture.
The CVC femoral route must be considered only after jugular accesses, due to the known frequent infectious complications, but can instead be usefully used as a bridge or ‘extreme’ solution and must therefore also be preserved with PTA, if necessary, for any future purposes. 29
The trans-collateral, trans-renal, trans-lumbar and transhepatic route have already been reported as viable alternatives in the absence of other vascular accesses6,30–33 and are to be reserved for truly ‘extreme’ conditions.
Conclusions
Our experience clearly demonstrates how nowadays the optimal use of all the diagnostic and interventional radiology techniques and procedures available already mentioned (CT-angiography, ultrasound-guided puncture of even thin or deep vessels, recanalisation and PTA of chronically obstructed vessels, maintain patency at the time of CVC removal, use of various types of stents) allows to manage ‘complex’ conditions avoiding the use of ‘extreme catheterisms’ always burdened by heavy complications. 32
Nowadays the imaging methods, such as CT-angiography, allows to plan an immediate and correct strategy and to plan further future CVC accesses in these patients, who now have limited vascular reserves.
Precise pre-operative knowledge of venous anatomy is mandatory for each patient; modern imaging techniques ensure a fine description of the vascular situation and a timely diagnosis of venous complications such as steno-occlusions or thrombosis, while endovascular interventional radiology procedures allow excellent minimally invasive treatment.
Furthermore, experience on the problems of vascular access, and the collaboration of the multidisciplinary team, especially with the nephrologist colleagues, are the basis for the management of the increasingly complex clinical-diagnostic-therapeutic situations represented by the haemodialysis patient.
Footnotes
Author contributions
The contribution of the Authors is to be divided into equal parts.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
