Abstract
Background:
Heart failure (HF) is a frequent cause of morbidity and mortality of end-stage kidney disease (ESKD) patients on hemodialysis. It is not easy to distinguish HF from water overload. The traditional HF definition has low sensitivity and specificity in this population. Moreover, many patients on hemodialysis have exercise limitations unrelated to HF. Therefore, we postulated two new HF definitions ((1) Modified definition of the Acute Dialysis Quality Improvement working group; (2) Hemodynamic definition based on the calculation of the effective cardiac output). We hypothesize that the newer definitions will better identify patients with higher number of endpoints and with more advanced structural heart disease.
Methods:
Cohort, observational, longitudinal study with recording predefined endpoints. Patients (n = 300) treated by hemodialysis in six collaborating centers will be examined centrally in a tertiary cardiovascular center every 6–12 months lifelong or till kidney transplantation by detailed expert echocardiography with the calculation of cardiac output, arteriovenous dialysis fistula flow volume calculation, bio-impedance, and basic laboratory analysis including NTproBNP. Effective cardiac output will be measured as the difference between measured total cardiac output and arteriovenous fistula flow volume and systemic vascular resistance will be also assessed non-invasively. In case of water overload during examination, dry weight adjustment will be recommended, and the patient invited for another examination within 6 weeks. A composite major endpoint will consist of (1) Cardiovascular death; (2) HF worsening/new diagnosis of; (3) Non-fatal myocardial infarction or stroke. The two newer HF definitions will be compared with the traditional one in terms of time to major endpoint analysis.
Discussion:
This trial will differ from others by: (1) detailed repeated hemodynamic assessment including arteriovenous access flow and (2) by careful assessment of adequate hydration to avoid confusion between HF and water overload.
Background
Patients with chronic kidney disease (CKD) and particularly those with its advanced stage, suffer from and die of cardiovascular diseases and especially of heart failure (HF). Indeed, up to 36% of all end-stage kidney disease (ESKD) patients had HF at the initiation of dialysis, which is up to 36 times more than in the general population. 1
By the traditional patho-physiological view, HF is when the heart is unable to pump sufficient blood flow (cardiac output) to meet the body tissue’s needs for metabolism or is able to do it only at the cost of increased filling pressures. According to guidelines of the European Society of Cardiology (ESC), 2 HF is a clinical syndrome characterized by typical symptoms that may be accompanied by signs caused by structural and/or functional cardiac abnormality, resulting in reduced cardiac output and/or elevated intracardiac pressures at rest or during exercise. Three HF phenotypes are mentioned in the guidelines 2 according to left ventricular ejection fraction (EF): HF with preserved EF (HFpEF, EF > 50%), HF with reduced EF (HFrEF, EF < 40%), and HF with mildly reduced EF (HFmrEF, EF 40%–50%). Another phenotype is distinguished in some studies3,4: High-output heart failure (HOHF) defined by the presence of HF symptoms, elevated natriuretic peptides, and by the cardiac index above 3.9 L/min/m2. Furthermore, HOHF is linked with increased mortality according to the retrospective trial by Reddy et al. 4
The underlying causes of HF in patients with ESKD are similar to those in the general population such as advancing age, diabetes mellitus, and coronary artery disease. However, there are also other and more specific causes of HF in CKD patients, such as the retention of uremic toxins that may affect myocardial contractility, 5 very frequent arterial hypertension with increased arterial stiffness, 6 and mechanisms leading to hyperkinetic circulation and thus further increasing the demands on cardiac work.7,8 All heart structures can be affected by ESKD and contribute to HF development. They include anemia, the presence of arteriovenous dialysis access, and cyclic overhydration.
The optimal level of hydration at the end of a HD session is expressed as the dry weight. When the dry weight is too high, the patients have chronic organ congestion and significantly increased mortality. 9 Optimal setting of the dry weight in a patient is a challenge. High dry weight setting is usually better tolerated and could be asymptomatic or symptomatic (edema, hypertension, shortness of breath).
Arteriovenous fistula (AVF) is the preferred hemodialysis vascular access over catheters. 10 Usual flow volume values of AVFs reach 600–1500 mL/min, but sometimes reach up to 2–5 L/min. In some patients, AVF creation leads to manifest HF, 11 but in many others even high flow values are clinically asymptomatic. Some recent observations documented improved kidney graft perfusion or brain oxygenation during a short-term manual AVF compression.12,13 Nevertheless, AVF flow was not considered in the majority of trials investigating structural heart changes in ESKD patients.
HF diagnosis is tricky in ESKD patients on hemodialysis. Water overload due to incorrect dry weight setting or low patient’s compliance with fluid restriction lead to similar clinical and echocardiographic changes as HF. The K/DOQI (Kidney Disease Outcomes Quality Initiative) guidelines recommend to diagnose HF in hemodialysis patients similarly as the ESC guidelines, 2 that is by the presence of symptoms (at least temporary—during decompensation) and documentation of structural heart disease. A more advanced and recent approach was offered by the Acute Dialysis Quality Initiative (ADQI) XI workgroup, which proposed a new classification of HF in ESKD patients. 14 The ADQI definition is based on three elements: (1) Echocardiographic evidence of structural or functional heart abnormalities; (2) Shortness of breath occurring in the absence of pulmonary disease, and (3) The response of congestive symptoms to ultrafiltration. However, also ADQI recommendations was criticized 15 : (1) Shortness of breath, but also volume overload could be multifactorial and may not be related to structural heart disease; (2) Some patients have no HF symptoms due to other exercise limitations, such as orthopedic, neuropathy, or frailty, but still might have advanced structural heart changes; (3) Pulmonary hypertension and coronary artery disease were not incorporated into the criteria; (4) The primary ADQI study was retrospective; (5) Echocardiographic finding of >1 sign of the structural heart disease was observed in as many as 87% hemodialysis patients, which decreases their usefulness; and (6) The predictive power of various echocardiographic criteria (such as low ejection fraction vs left atrial dilatation) differs. 15 Moreover, the echocardiographic cut-off values used in the ADQI study were different from those in the cardiological guidelines. 16
The classical HF definition, stresses out (low) cardiac output (CO) and (high) filling pressures at rest or during exercise. 2 At rest, less than 10% of HF patients have low CO in the general population. 17 In patients with dialysis AVF, systemic CO is divided between the supply of organs and feeding of AVF. In other words, measured CO value by any available method overestimates the real amount of blood per time dedicated to organ perfusion. From the hemodynamic point of view, AVF represents a shortcut between the systemic arteries and veins. Thus, part of the cardiac output (CO) is stolen by the AVF. Usual AVF flow volume values (500–1500 mL/min) represent 10%–30% of the usual resting CO (4–6 L/min), considering only usual CO would be misleading. To solve this problem, the term effective cardiac output (COef.) has been introduced and could be calculated as the difference between the total CO and AVF flow volume. 18
Altogether, HF diagnosis is difficult in patients on chronic hemodialysis and both ESC and ADQI definitions possess various limitations. To overcome these issues, we modified the ADQI HF definition and added another—hemodynamic HF definition. We designed a prospective longitudinal single center observational cohort study on the diagnosis of HF in end-stage CKD patients treated by chronic hemodialysis (CZecking HF-CKD study). This study plans to prospectively collect detailed repeated data concentrated on cardiac and volume status from at least 300 patients during a 3-year period. Moreover, specific endpoints will be collected from all included subjects. We believe that this study will bring important real-world experience and will improve the diagnostic criteria of HF in ESKD patients.
Project hypotheses
Newer HF definitions (2 + 3, see below) will better identify patients with higher risk of major cardiovascular events (cardiovascular death, HF development or worsening, non-fatal myocardial infarction, and stroke).
In patients with non-cardiac exercise limitation (orthopedical and neurological problems, myopathy, frailty, etc.), the use of a combination of advanced structural heart changes (as defined in Table 1) and of elevated NTproBNP level (above determined cut-off) would identify patients with a similar risk of endpoints as in the symptomatic HF subjects.
Careful assessment of dry weight setting will enable us to estimate real world HF prevalence by excluding simple water overload mimicking HF.
Modified ADQI echocardiographic criteria of heart failure.
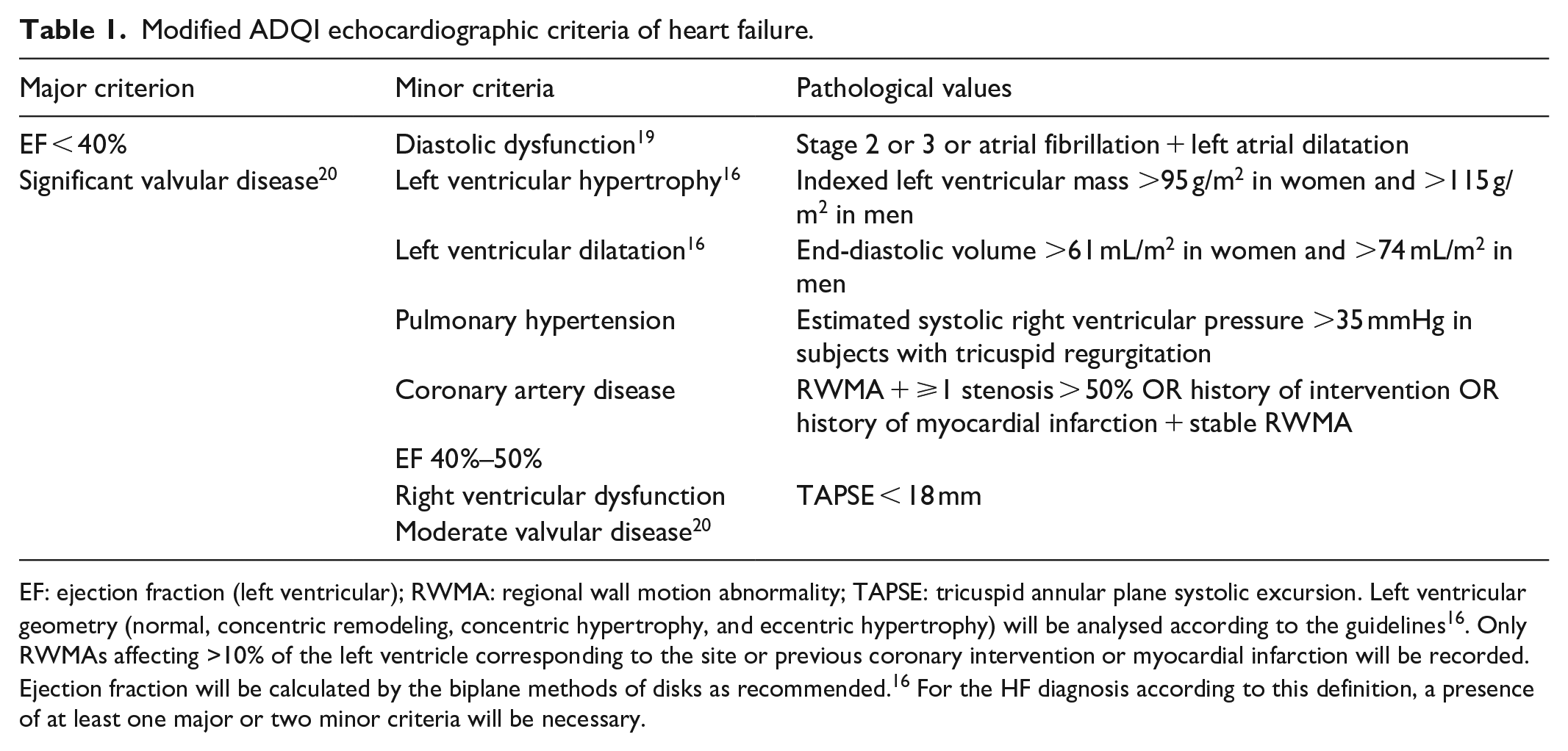
EF: ejection fraction (left ventricular); RWMA: regional wall motion abnormality; TAPSE: tricuspid annular plane systolic excursion. Left ventricular geometry (normal, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy) will be analysed according to the guidelines 16 . Only RWMAs affecting >10% of the left ventricle corresponding to the site or previous coronary intervention or myocardial infarction will be recorded. Ejection fraction will be calculated by the biplane methods of disks as recommended. 16 For the HF diagnosis according to this definition, a presence of at least one major or two minor criteria will be necessary.
The following three HF definitions will be used for the purpose of this study separately:
Traditional definition by the K/DOQI and ESC 2 (clinical syndrome defined by typical symptoms and signs caused by a structural and/or functional cardiac abnormality). The frequency of HF phenotypes (including HO-HF) will be analyzed.
Modified ADQI XI Workgroup definition: see Figure 1 and Table 1 for explanation.
Haemodynamic definition: Low effective cardiac index (<2.5 L/min/m2) in patients with at least one of the following abnormalities: (a) Left ventricular hypertrophy; (b) Left ventricular EF< 40%; (c) AVF flow volume >1500 mL/min, but in the absence of significant valvular/pericardial disease; (d) Diastolic dysfunction grade 2 or 3; (e) Moderate or severe left atrial dilatation (LAVi > 40 mL/m2); (f) Estimated pulmonary artery systolic pressure >40 mmHg.
Our two HF definitions (Nos. 2 and 3) will be compared with the ESC/KDOQI HF definition regarding the frequency of HF, symptoms, echocardiographic findings, and prognosis. Subjects not having HF according to the particular definition will create control groups.
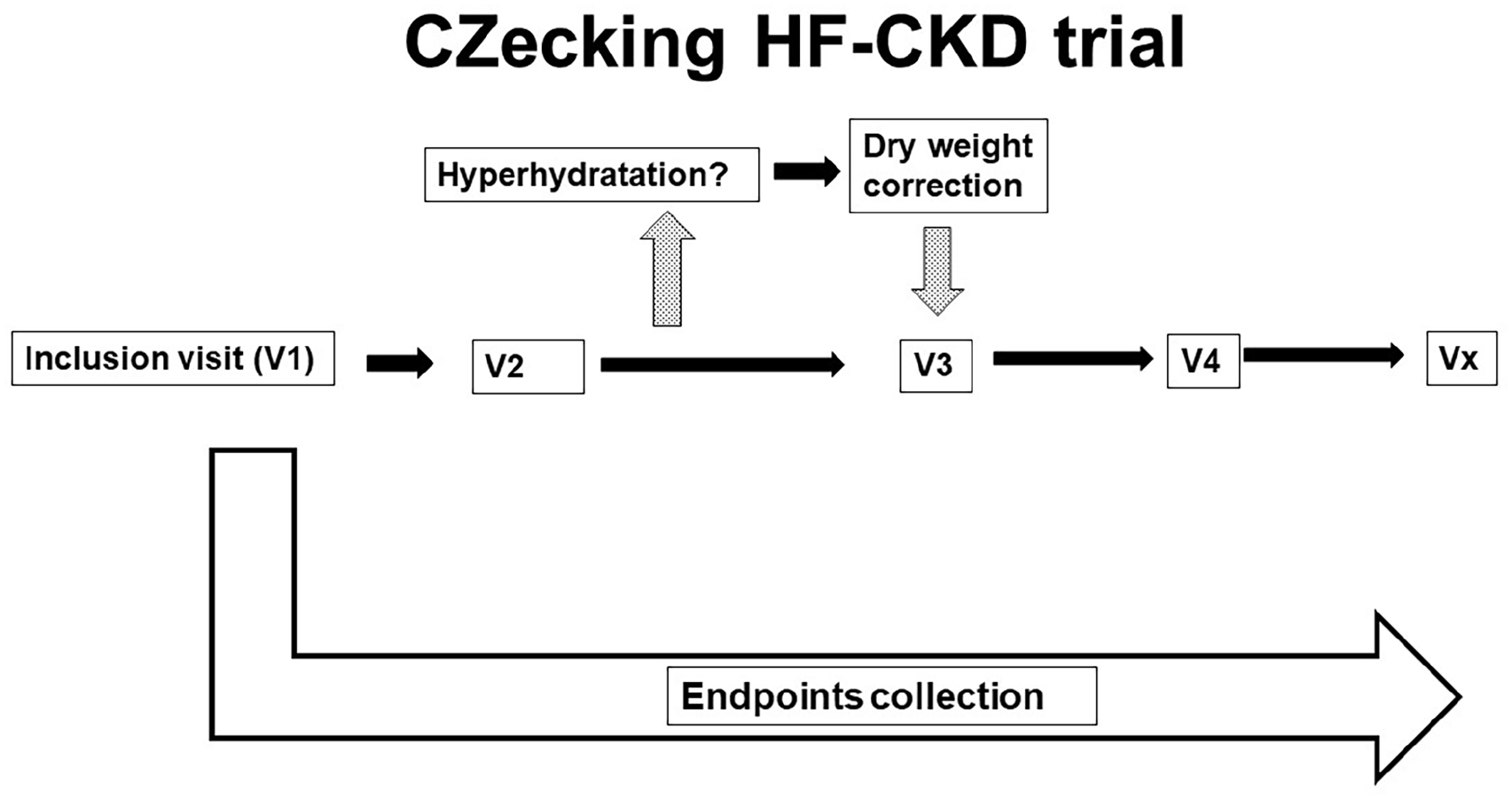
Study scheme.
Methods and design
Project scope and aims
Main aims of this project
To improve in-time diagnosis of HF (and its differentiation from water overload) in ESKD patients by close cooperation between nephrologists and cardiologists with use of symptom analysis, NTproBNP levels, expert echocardiography with non-invasive hemodynamic assessment, and other examinations.
Specifically:
To compare two newer HF definitions (Modified ADQI and Hemodynamic, see above) with ESC/KDOQI definition regarding to predefined endpoints by the time-to-event analysis
To improve HF diagnosis in subjects with non-cardiac exercise limitations by the combination of echocardiographic changes and NTproBNP level.
Secondary aims of the project
To determine specific risk factors (echocardiographic parameters, NTproBNP, AVF flow volume) of the defined major endpoints by a complex of examinations (see Methods).
To describe water overload-associated changes and to differentiate them from HF changes (a subgroup of patients with incorrectly high dry weight will be analyzed for this aim).
To elucidate the role of arteriovenous fistula on HF development by analysis of COef. Specifically, we will describe the frequency of high flow fistulas (AVF flow >2000 mL/min) and analyze the relation between CO and AVF flow (see Hemodynamic definition)
To estimate most frequent cardiovascular pathologies and prevalence of HF in the Czech hemodialysis population.
Design of the CZecking HF-CKD trial
This study is designed as a longitudinal, prospective, observational, single-centre cohort study with regular assessment of endpoints. Inclusion of first patients started in 2019 and the planned end-date of the study is 31st December, 2025. The study is being conducted under the collaboration of six hemodialysis units and one complex cardiovascular center of a university-based hospital. All prevalent patients that fulfil the broad eligibility criteria will come for the index examination (see below) from the collaborating hemodialysis units and then be invited to re-examination every 6 or 12 months (depending on the findings). The participants will be followed life-long or till kidney transplantation (kidney transplantation and subsequent care is provided in another institution). Endpoints will be collected every 6 months since the index visit—see Figure 2 for details. In case of pathological findings or suspicion on a heart disease, all necessary procedures will be suggested and performed in the tertiary university-based hospital. Best therapy according to the current knowledge will be offered to the patients, including coronary revascularization, correction/replacement of valvular disease, arrhythmia control, recommended prescription, etc. 21
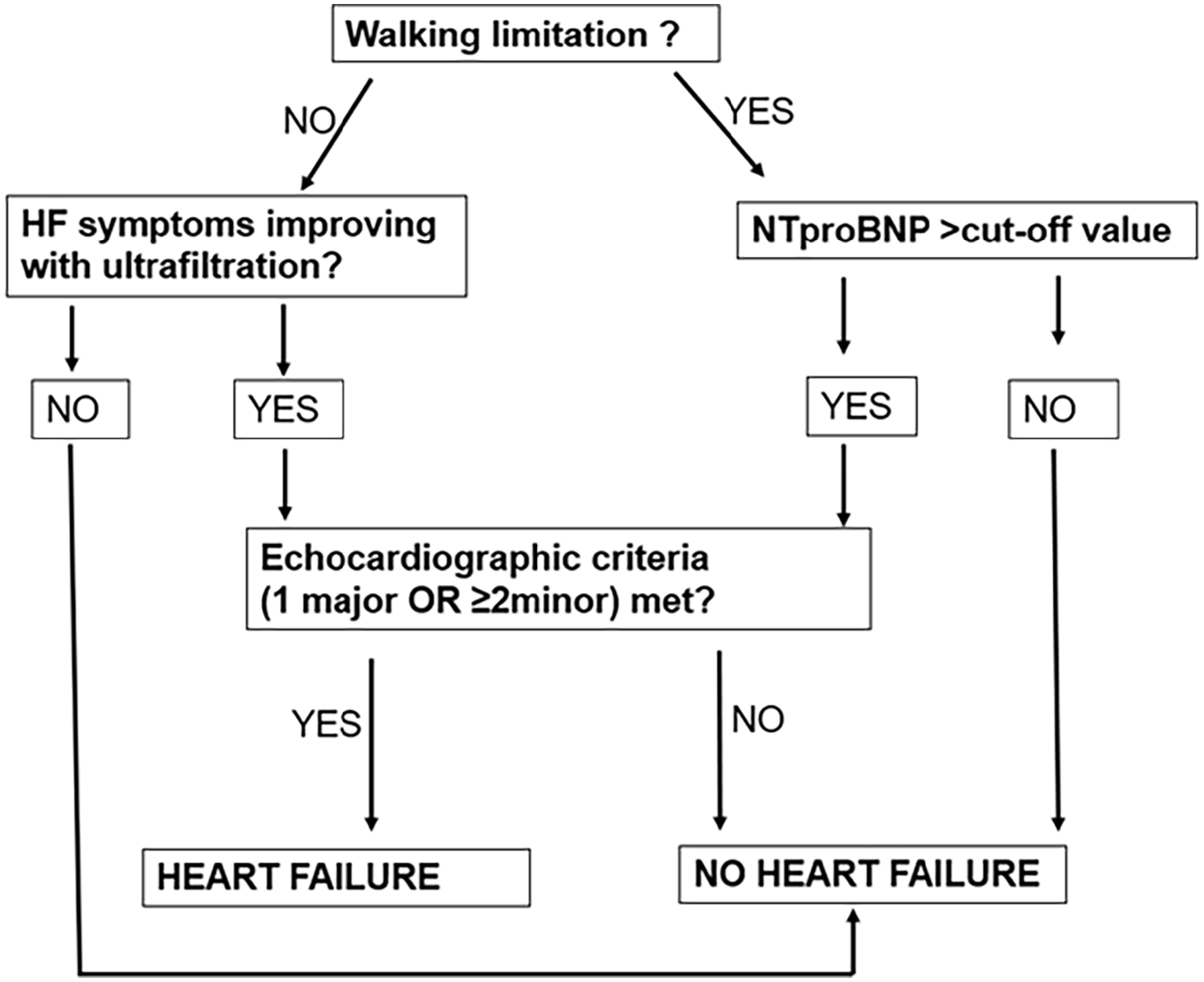
Modified ADQI heart failure definition in this study.
In case of patients over- or underhydration, dry weight adjustment will be suggested and the patient invited for another examination within 1 month. In this situation, only data from the visit with the correct dry weight setting will be recorded for the main purpose of this study, while the comparison of these two examinations will help us to describe water overload changes. See Figure 1 for the study scheme.
Eligibility criteria
Participation in this study will be offered to all patients treated by chronic haemodialysis in the collaborating centers supposing that they will be clinically stable and willing to participate. Specific inclusion criteria are as follows: ESKD treated by hemodialysis for at least 3 months, age >18 years and absence of dementia.
Exclusion criteria
It include: (1) Planned kidney transplantation from a living donor within 3 months of the index visit; (2) Life expectancy <6 months at the index (first) visit of any reason.
Data collection
We will collect basic history, medication, hemodialysis data (dialysis frequency and duration, dialysis type—hemodialysis/hemodiafiltration, dry weight, ultrafiltration, residual diuresis, presence of intradialytic hypotension), vital signs (systemic blood pressure (BP) and heart rate) and the following specific examinations:
HF symptoms: standard clinical assessment and using “Prague CKD-HF questionnaire”—see Supplemental File 1.
Blood samples (full blood count, albumin, total protein, NT-pro brain natriuretic peptide). In the hospital-linked hemodialysis unit, another two blood samples will be drawn and stored frozen for further analysis. Blood samples will be collected from the hemodialysis set before the initiation of the hemodialysis.
AVF flow volume (flow measured in the feeding brachial artery by duplex ultrasound as described earlier). 12
Appropriate dry weight setting will be assessed by an experienced nephrologist with the use of clinical examination (lack of effusions, edema), estimation of central venous pressure (CVP) by inferior vena cava diameter and collapsibility 22 and by electric bioimpedance (Body composition monitor, FMC). Moreover, the presence or absence of pericardial/pleural effusion or ascites will be actively searched during echocardiography.
Rhythm analysis—electrocardiography—sinus rhythm versus atrial fibrillation.
Expert echocardiography—detailed analysis of the volumes of heart cavities, quantification of valvular disease, diastolic dysfunction according to the recent guidelines, calcification extent, and cardiac output calculation (using the left ventricular outflow tract diameter and velocity time interval) as described earlier. 23 Echocardiography and all other examinations will be performed in a non-dialysis day together within 1 h. Structural heart disease definition is presented in Table 1.
Calculated variables
Effective cardiac output: the difference of CO a AVF flow volume
Mean arterial pressure (MAP = 2/3 of diastolic pressure + 1/3 of systolic pressure)
Systemic vascular resistance: (MAP-estimated CVP)/COef.
Total vascular resistance: (MAP-estimated CVP)/CO
Access resistance: (MAP-estimated CVP)/AVF flow volume.
Endpoints
Major endpoint
A composite consisting of at least one of the following:
Cardiovascular death
HF: worsening: by one class of NYHA; hospitalization for de-compensation; ejection fraction decrease by ⩾10%; or new diagnosis of HF
Non-fatal myocardial infarction or stroke.
Minor endpoints
Components of the primary endpoint
Pacemaker/defibrillator implantation
The necessity for dry weight decrease by >3% of body weight because of symptomatic overhydration
Vascular access intervention
The need for elective coronary or valvular intervention (percutaneous or surgical)
Other cardiovascular morbidity non-defined above (acute leg ischemia, pulmonary embolism, etc.)
Non-cardiac death.
Statistical considerations
Considering the explorative and observational character of this study, no formal sample size will be calculated. Nevertheless, considering the high morbidity and mortality of ESKD patients on chronic hemodialysis therapy, including >300 subjects and following them for >3 years will give us sufficient data. When designing this study, we studied literature data of similar populations: (1) An Austrian study 24 investigated the mortality of ESKD patients and the sample size was 153 subjects, mean age 67 years. Within 2.6 years there was significantly higher mortality in patients suffering from diastolic dysfunction. (2) In an American study 25 of 621 older subjects, 1-year survival was 65% and the presence of HF was identified as a risk factor of mortality. The recent annual USRDS data report demonstrates that 41.3% of patients suffering from CKD 4–5 have the diagnosis of HF. All continuous variables will be tested for the data distribution (Gaussian vs non-Gaussian) and expressed as mean ± SD or median (interquartile range) as appropriate. Categorical variables will be presented as percentages. We will use the repeated measure ANOVA test or Friedman’s test and paired t-test or Wilcoxon paired test as appropriate for testing the significance of observed changes in time. The relation between variables will be tested using the Pearson or Spearman analysis and by analysis of co-variance. Specificity, sensitivity, positive, and negative predictive values for HF diagnosis of all questions in the Questionnaire will be calculated as well as the relative weight of all individual questions for the HF diagnosis. ROC (Receiver-Operating Characteristics) curves will be calculated for HF detection according to the NTproBNP level, an ejection fraction of the left ventricle, diastolic dysfunction grade, and other parameters. The effect of the HF diagnosis on patients’ survival will be analyzed using Kaplan-Meier survival analysis (log-rank test) and Cox Proportional-Hazards regression (risk factors) and depicted graphically. The significance (p-value) levels in case of multiple testing will be re-set according to Bonferroni correction or a correction appropriate post hoc analysis for a given test. The p-value lower than 0.05 will be considered as significant. For the statistical analysis, the appropriate statistical software will be used. Some missing data are expected in echocardiography due to non-ideal acoustic window; they will be treated as “missing at random.” In case of impossibility of performing echocardiography due to acoustic limitation, the case will be excluded from analysis. Loss to follow ups are not expected because the patients are vitally connected to their hemodialysis units. In case of (very rare) loss to follow up, the survival status will be recorded from the national database.
Discussion
Available literature data document the high burden of HF in ESKD patients. According to the study by Harnett et al. 26 up to 31% of all ESRD patients suffer from HF at the time of hemodialysis therapy initiation. In this prospective study carried in three centers and published 26 years ago, the median survival of patients suffering also from HF at the initiation of hemodialysis was 36 months in comparison to 62 months in patients without HF. In a recent prospective study, Antlanger et al. 27 analyzed 105 patients on maintenance hemodialysis with regards to HFpEF and HFrEF and with the use of comprehensive echocardiography. They proved that both these phenotypes were associated with higher mortality than no HF and HFrEF was stronger predictor. The K/DOQI/ESC definition of HF was used in these studies.
Further studies confirmed high prevalence of HF in patients on hemodialysis and higher associated mortality,28,29 but they were large epidemiological trials, analyzing diagnoses only from the hospital discharge letters. Although such trials were robust and analyzed tens of thousands of subjects, they had the usual weakness of this type of trials, and, moreover, it had to be difficult to distinguish HF from water overload in them. In other words, our knowledge of HF in ESKD patients on hemodialysis is less firm than generally presumed. This is also influenced by changing diagnostic criteria of HF in ESKD patients. The older definition (K/DOQI) is the same as in the general population (ESC). A more complex newer definition by the ADQI XI work group is more specific and based on a combination of symptoms, structural heart disease, and symptom improvement after ultrafiltration. 15 However, the trial that brought the core data for the ADQI definition also came from a retrospective trial and has further limitations mentioned above. We therefore modified the ADQI definition for the purpose of this study. Moreover, we added also the hemodynamic definition of HF based on the assessment of the effective cardiac output. This is a concept of AVF as a systemic steal introduced by Amerling et al. 18 and further extended by Basile et al. 30 Although this is an interesting concept, it has not been analyzed prospectively in a clinical trial. Low effective CO, but normal total CO could occur in patients with too wide arteriovenous anastomosis, where the percentage of CO running through AVF exceeds 20%–30%. Interestingly, in the general population, less than 10% of HF patients have low CO at rest. 17 From our clinical experience, some of these patients have non-dilated stiff left ventricle that is not able to increase CO in response to increased filling pressures.
The pathological cut-off values of the echocardiographical findings used in the ADQI definition 15 of HF differed from the values mentioned in the cardiological guidelines. 2 The argument for such modification was that hemodialysis patients commonly have advanced structural heart changes and that the use of the “cardiological” cut-offs would not distinguish low- and high-risk patients. Nevertheless, up to 87% of included patients had at least one echocardiographic pathology in the ADQI study. 15 We rather believe that the structural changes that would make the diagnosis of HF (Table 1) should be used in patho-physiological contexts. While the diagnosis of HFrEF is usually easy and is based on the symptoms and 1 value (ejection fraction) this is more difficult in the case of HFpEF (and to some extent of HFmrEF). 2 Left ventricular hypertrophy and diastolic dysfunction (that includes by definition also left atrial dilatation and pulmonary hypertension) create a complex of mechanisms responsible for HFpEF. 31 Similarly, mitral valve disease is connected with left atrial dilatation, 32 etc. Therefore, the individual adding of these parameters probably could not establish HF diagnosis. Only low ejection fraction and significant valvular disease are clearly related to the symptoms, so we established these two criteria as “major,” while the other as “minor.” Although the echocardiographic 19 and clinical 31 diagnosis of HFpEF underwent significant progress in the general population, appropriate echocardiographical findings are unstable in hemodialysis patients. 33 Therefore, diastolic dysfunction is set as a minor criterion in this study. Not only high AVF flow can contribute to the development of HOHF: other factors include anemia, water overload, subclinical inflammation, etc. 4 However, the prevalence of HOHF or even of high cardiac output is not known in the ESKD population, so it is one of the goals of this project.
The current ESC definition of HF restricts itself to stages at which clinical symptoms are apparent. 2 Before clinical symptoms develop, patients might have structural or functional heart abnormalities that are so-called precursors of HF and are linked with increased mortality. Unfortunately, 42% of patients on chronic hemodialysis had severe limitations in moderate physical activities. 34 Therefore, HF diagnosis is probably late in these patients. In this project, we plan to create a complex criterion of HF that includes the combination of structural heart disease and high NTproBNP level. The level of NTproBNP is not specific for HF in ESKD patients and could also mirror volume overload. 35 In our study, we will ensure adequate hydration of the patients at the time of the examination by both central venous pressure estimation and by electric bioimpedance, so we believe that the combination of high NTproBNP and structural/functional changes by echocardiography could select patients with (silent) HF. Regular lung ultrasound according to the LUST study 36 would be more appropriate, but it is logically achievable only in patients coming for hemodialysis to our hospital (ca 20% of all included subjects).
Lower plasma protein and albumin are also a frequent HF consequence due to complex mechanisms and is associated with higher mortality. 37 They will be also recorded in this project.
Limitations of this project include general drawbacks of observational trials, such as the inability to control different variables. Many mechanisms are involved in the development of both ESKD and of HF in this population. However, similar proportion of associated states are frequent also in other hemodialysis units at least in the developed countries.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298221099843 – Supplemental material for CZecking heart failure in patients with advanced chronic kidney disease (Czech HF-CKD): Study protocol
Supplemental material, sj-pdf-1-jva-10.1177_11297298221099843 for CZecking heart failure in patients with advanced chronic kidney disease (Czech HF-CKD): Study protocol by Jan Malik, Anna Valerianova, Satu Sinikka Pesickova, Zdenka Hruskova, Vladimira Bednarova, Pavel Michalek, Vladimir Polakovic and Vladimir Tesar in The Journal of Vascular Access
Footnotes
Author’s note
The authors are members of the Vascular Access Society.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Ministry of Health, Czech Republic-DRO (General University Hospital in Prague-VFN), 00064165.
Ethics approval and consent to participate
The study complies with the Declaration of Helsinki. This study was approved by the Institutional Ethical Committee on 18th February 2021 (reference number 1/21). We will present obtained and analysed data at domestic and international medical conferences and will publish them as scientific papers in various dedicated peer-reviewed journals.
Trial registration
The study is registered by the International Standard Randomised Controlled Trials Number – ISRCTN 18275480. It was assigned on 16 March 2021 and updated on 07 July 2021.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
