Abstract
Background:
Placement of peripheral intra-venous cannulas and epicutaneo-caval catheters is routinely performed in in Neonatal Intensive Care Unit (NICU), and both devices require visible superficial veins easy to cannulate. NICU patients are intrinsically characterized by poor and fragile vein asset, so that puncture and cannulation of superficial veins is often a challenge even for trained clinicians and cannulation frequently results in a stressful, painful, difficult procedure.
Methods and results:
Rapid Superficial Vein Assessment is meant to offer a systematic pre-procedural evaluation of all superficial veins of the newborn, so to allow a rational choice of the best insertion site, tailored on the single patient, and optimized for the specific type of venous access device. The superficial veins are examined systematically, both with and without NIR technology, exploring seven skin areas in the following order: (1) medial malleolus, (2) lateral malleolus, (3) retro-popliteal fossa, (4) back of the hand and wrist, (5) antecubital fossa, (6) anterior scalp surface, and (7) posterior scalp surface.
Conclusions:
The aim of the protocol is to increase the first attempt success rate and reduce the duration of the procedure, the number of attempts for single patient and possibly to limit complications, stress, and pain in neonates.
Keywords
Background
A reliable venous access is indispensable in premature and critically ill patients in Neonatal Intensive Care Unit (NICU) for infusion of drugs, fluids, parenteral nutrition, and blood products. 1 Therefore, placement of Peripheral Intra-Venous Cannulas (PIVC) and of Epicutaneo-Caval Catheters (ECC) are routine procedures in NICU. Both devices require a properly visible vein in an appropriate site. As regards PIVC, access to an unreliable or difficult superficial vein increases the difficulty of the procedure and reduces the durability of the cannula. This implies stressful and painful frequent repositioning of cannulas in neonates already characterized by vein fragility and clinical instability. Danski et al. 2 reported that 99.6% of NICU patients need a venous access and in 49.2% the chosen device is a PIVC; though, 63.15% of PIVC will have complications requiring early removal ad repositioning. According to Legemaat et al., 1 62% of PIVCs are placed because of unscheduled removal of a previous PIVC. Infiltration (extravascular leakage of non-vesicant solutions, that can be associated with skin, muscle, and nerve damage) is considered the most common PIVC-related complication (23%–78%), 3 followed by extravasation (extravascular leakage of a vesicant solution) (11%–23%). 4 Infiltration and extravasation represent all-together a leading cause of iatrogenic injury in NICU-admitted newborns (45.6%–69.89% of neonates with a PIVC),2,5 and the elective repositioning of PIVC doesn’t seems to reduce the risk of future infiltration/extravasation. Less frequent complications of PIVC are phlebitis (consequence of mechanical, chemical, or infective injury to the vein wall), ranging from 3.5% to 17.84%, and catheter obstruction (12.27%). 4 Though this issue has never been specifically addressed in the literature, it is likely that many PIVC-related complications are secondary to an initial choice of a vein inappropriate for its location or prone to early rupture during cannulation due to a winding path. ECC is recommended by US Centers for Disease Control and Prevention Healthcare Infection Control Practices Advisory Committee guidelines for an expected intravenous treatment exceeding 6 days. 6 These intravenous devices are inserted through a superficial vein and advanced to reach the central circulation. This implies not only the puncture and cannulation of the superficial vein, but also the progression of the catheter into the vasculature (so-called “tip navigation” and “tip location”); an inappropriate choice of the vein may be associated with difficult or impossible cannulation and/or progression of the catheter, or with late complications such as local thrombophlebitis or dislodgment. The ECC used in neonates are a completely different device if compared with Peripherally Inserted Central Catheters (PICC) of children and adults. In fact, the diameter of brachial, basilic, and axillary veins in patients older than 2 years is usually >3 mm, thus allowing ultrasound-guided cannulation with 3 Fr or even larger PICCs. Ultrasound has a small role in visualization and puncture of superficial veins in the neonate, but in a recent study the authors showed that USG cannulation is superior for neonatal ECC placement to the standard single-wall puncture method, with a higher success rate, and decreases the total procedural time. 7 In literature, unscheduled removal of ECC ranges between 2.8% and 20.9%. 8 In a recent study on a cohort of 524 ECCs, 9 the rate of unscheduled removal was even higher (37.2%). Infective complications of ECC are close to 11.5 per 1000 catheter-days. The most frequent mechanical complications are lumen occlusion (5.0 per 1000 catheter-days), dislodgement (4.4), and rupture (4.0); other complications—extravasation, edema of the limb, phlebitis, and thrombosis are less common. Well-known risk factors for an unplanned removal are a previous ECC insertion, and the non-central tip position. 10 Therefore, the optimization of the number and quality of cannulations appears even more necessary. Both PIVC-related and ECC-related complications are associated with waste of time and resources, increased morbidity, and prolonged NICU-stay. Also, they increase pain and discomfort of the neonates, which is something that should be avoided. A new interesting strategy which may be helpful in optimizing visualization, puncture and cannulation of superficial veins is Near-Infra-Red technology (NIR). NIR technology is based on the selective adsorption of a specific wavelength of NIR rays (760 nm) by desaturated hemoglobin. As desaturated hemoglobin is located exclusively in venous blood, NIR devices offer an accurate visualization of superficial veins (within 7 mm from the skin surface, i.e. the maximal depth reached by the NIR rays). As opposed to ultrasound devices, NIR devices have no probe since the images are shown directly on patient’s skin or on a helmet-viewer so that both hands of the clinician are free to perform the puncture. The first clinical investigations of this technique did not find significant advantages of NIR when applied to unselected patients. More recently, the efficacy of NIR—as evaluated by the number of attempts per patient, duration of the procedure, and success rate at first attempt—was demonstrated in cases where an intermediate- to high-difficulty access is anticipated. A randomized clinical trial demonstrated the efficacy of NIR in preterm neonates, in terms of time to successful cannulation, success of the first attempt, dwell time of the device, and procedure-related pain. 11 In another randomized clinical trial, NIR was associated with a trend toward more successful ECC placement in neonates, with clear benefit also in infants with difficult vessels visualization. 12 A recent Systematic Review demonstrated no evidence of an effect of near-infrared compared with landmark on first-time insertion success (RR, 1.21; 95% CI, 0.91–1.59) or number of attempts (MD, −0.65; 95% CI, −1.59 to 0.29); however, it significantly reduced PIVC insertion time (MD, −132.47; 95% CI, −166.68 to −98.26) and increased first-time insertion success in subgroup analysis of patients with difficult intravenous access (RR, 2.72; 95% CI, 1.02–7.24). The population’s studies included a wide range of children (0–17 years) not only newborns so the conclusion could be afflicted by this bias. 13 The other authors showed the infrared device reduces the procedure time, decreases the number of attempts, increases success rate of first attempts, and lowers the pain felt during PIVC placement procedures. 14 Considering that prevention of complications is a mandatory goal of any modern NICU, we must optimize the strategy of placement of PIVC and ECC, so to avoid multiple painful attempts, damage, or rupture of inappropriately chosen veins, and impoverishment of an already poor and fragile vein asset. With this goal in mind, we designed a structured, systematic protocol for the pre-procedural evaluation of all superficial veins of the neonate, adopting both direct and NIR-based visualization.
The RaSuVA protocol
The Rapid Superficial Vein Assessment (RaSuVA) protocol represents a systematic full anatomic evaluation to choose the best insertion site tailored on the specific patient, optimized for the specific kind of device required.
The RaSuVa implies a complete assessment of the neonate, from foot to head (Figure 1). The superficial veins are examined systematically, both with and without NIR technology, exploring seven skin areas in the following order:
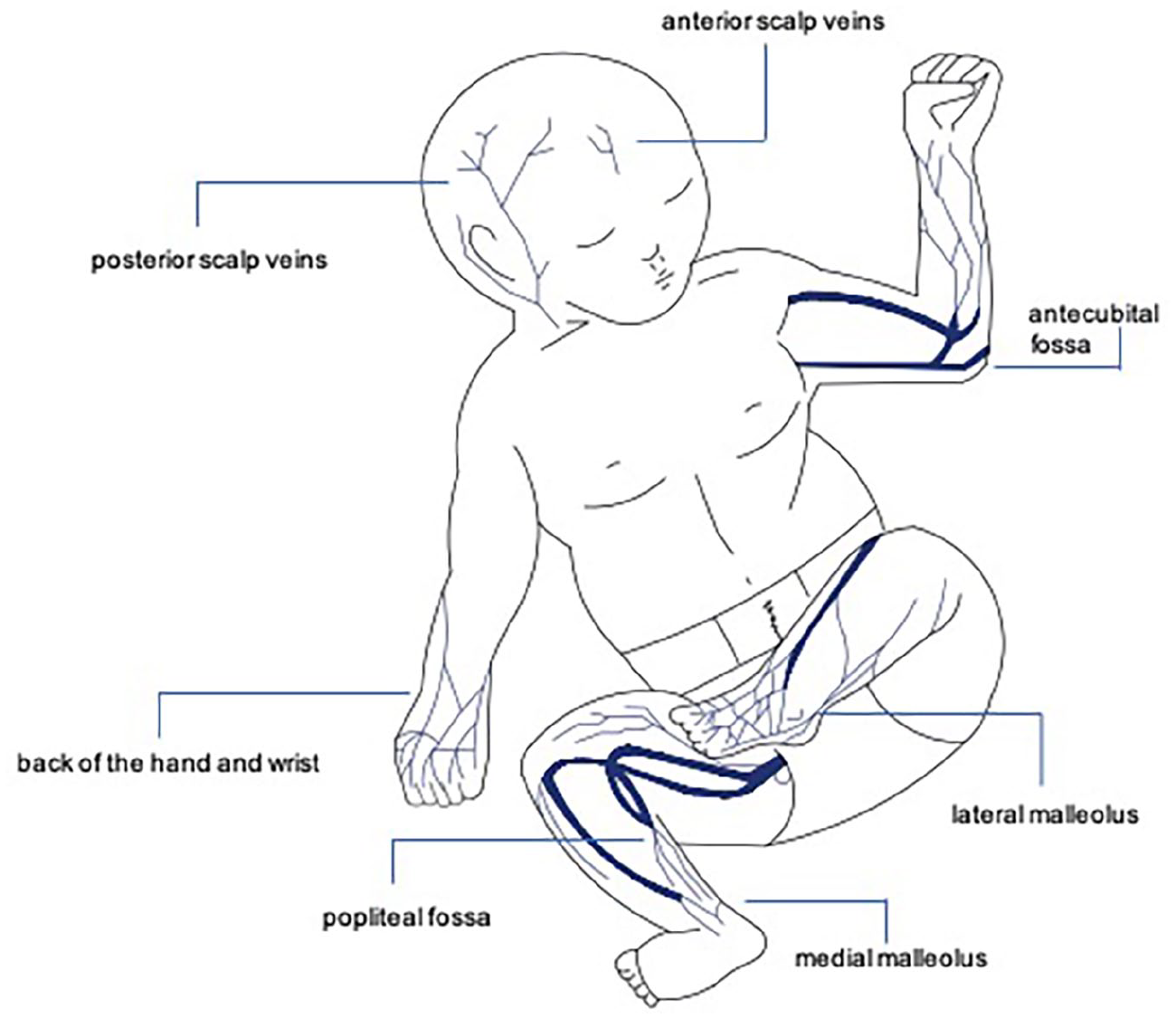
Seven steps to exploring superficial veins.
Medial malleolus
Lateral malleolus
Retro-popliteal fossa
Back of the hand and wrist
Antecubital fossa
Anterior scalp surface
Posterior scalp surface
Each potential venipuncture site is examined bilaterally, first by direct visualization, then by NIR technology. The latter is important for different reasons. First, it allows the visualization of veins which may not be visible with naked eye because of skin pigmentation and/or skin edema, and/or poor light in the environment and/or limited visual acuity of the operator. Also, it allows to evaluate the trajectory of the vein; in fact, an apparently straight segment of the vein may be followed a winding path, completely hidden to the naked eye, but evident with NIR: these veins are prone to early rupture during cannulation, or to parietal damages that can result in leakage and phlebitis. Moreover, NIR technology can unveil the patency of a vein laying inside or under a previous hematoma and it can discriminate between vein and artery, as well as between patent vein and obstructed vein. Such elements are also particularly important to predict the chance of ECC to reach a central position.
In steps (a) to (e), both direct and NIR-based visualization should be performed with and without tourniquet: the superficial veins will be more easily visible with tourniquet, even though the augmented parietal pressure may eventually damage the fragile vasculature. For this reason, tourniquet should be applied during cannulation only if proven to be useful at the moment of the pre-procedural evaluation.
After this complete overview of the superficial veins, on both sides and foot-to-head, the most appropriate device for the patient should be selected, the main choice being between a peripheral (PIVC) versus a central venous access device (ECC). Critically ill and/or premature newborns usually require central venous access for administration of parenteral nutrition, blood products, and/or irritant/vesicant infusions. Central vein access may be obtained in neonates with three different methods: (a) by umbilical venous catheterization (in the first 24–48 h after birth); (b) by direct ultrasound-guided access to a central vein, either in the neck/chest area (Centrally Inserted Central Catheters = CICC) or in the groin area (Femorally Inserted Central Catheters = FICC); (c) by peripheral insertion of ECC (not to be confused with the PICC used in adults and children, which require the puncture of deep veins of the arm). ECCs are small silicone or polyurethane catheters (1–2.7 Fr), suitable for expected treatments inferior to 14 days 15 and they are inserted in superficial veins of the limbs or of the scalp using the split needle technique or the modified Seldinger technique the latter being better in terms of success at ECC insertion and reduction of the number of venipunctures. 16
Peripheral lines are not appropriate for delivery of parenteral nutrition and/or of irritant/vesicant infusion. Also PIVC should be preferably used for intravenous treatments expected to last less than 1 week, while treatments longer than 7 days but inferior to 14 days require ECC. On the other hand, a critically ill neonate candidate to surgical treatment or to prolonged stay in ICU (>14 days) would be the ideal patient for a CICC or a FICC.
If properly carried out, the RaSuVA protocol will offer an accurate overview of:
the patient’s superficial vein pattern;
the characteristics of each vein in terms of trajectory and flow;
the expected difficulty of cannulation for each vein.
After the individuation of appropriate superficial veins, the choice of the site also depends on the device selected (PIVC vs ECC).
A PIVC should be preferably placed at the lateral malleolus or on the dorsum of the hand and wrist. The large veins located in the antecubital fossa or at the medial malleolus (saphenous vein) should be preferably left as the first choice for placement of ECC. Scalp veins are a secondary option for both PIVC and ECC, due to many reasons: the need for shaving before cannulation, the intrinsic tortuosity of the veins, the potential risk of aesthetic damage in case of complications, as well as the generally low parental compliance.
Conclusion
The RaSuVA protocol represents a rational strategy to choose the best insertion site, tailored on the single patient, and optimized for the specific kind of intravenous device required. The aim of RaSuVA is to offer a complete knowledge of the superficial veins of the patient, so to lead to a rational choice of the vein to be cannulated, with the final goal of increasing the success rate at first attempt, the duration of the procedure, with puncture-related pain and stress for neonates, and possibly to limit complications: goals that we consider crucial for any NICU.
Footnotes
Author contributions
All authors contributed to the study conception and design. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
