Abstract
Introduction:
Over the past decades, significant efforts have been made to reduce early and late catheter-related complications in critically ill patients, using approaches based on bundles of evidence-based interventions.
Methods:
In this prospective clinical study, the authors evaluated the incidence of catheter-related complications in their neuro-intensive care unit during a 4-year period, adopting systematically the GAVeCeLT bundles for the insertion and management of all central venous access devices: centrally inserted central catheters (CICCs), peripherally inserted central catheters (PICCs) and femorally inserted central catheters (FICCs). All early/immediate and late complications were recorded.
Results:
On 486 central lines (328 CICCs, 149 PICCs and 9 FICCs), the only clinically relevant early/immediate complication was primary tip malposition (1%). In regards late infective complications, the authors did not record any case of catheter-related bloodstream infection; though, they observed one case of central line associated blood stream infection (one CICC; 0.14/1000 catheter days), and 15 cases of catheter colonization (12 CICCs and 3 PICCs; 2.09 episodes/1000 catheter days). Late non-infective complications were few: 14 accidental dislodgments (2.9%), 18 irreversible lumen occlusions (3.7%), and no episodes of symptomatic catheter-related thrombosis or tip migration.
Conclusion:
The systematic adoption of the GAVeCeLT bundles for CVAD insertion and maintenance was associated with a minimization of catheter-related complications. The strict adherence to the recommendations included in these bundles was the major determinant for clinical success.
Keywords
Introduction
Central venous access devices (CVADs) are an essential component in the management of critically ill patients; though, they are potentially associated with immediate or early complications (accidental arterial puncture, pneumothorax, primary malposition, etc.), infective complications, such as catheter-related bloodstream infections (CRBSI), and non-infective late complications (catheter-related thrombosis, catheter dislodgment, tip migration, irreversible lumen occlusion, etc.).
One of the most severe and expensive complication is nonetheless CRBSI, which is characterized by high morbidity and mortality. 1 A widely accepted method used for the diagnosis of CRBSI is the differential time to positivity (DTP) between blood cultures from the catheter and from a peripheral vein.2,3 If both cultures are positive for the same germ, but catheter blood culture becomes positive at least 120 minutes before the peripheral blood culture, the diagnosis of CRBSI is established. A simultaneous positivity – or an early positivity of peripheral blood over catheter blood – excludes the diagnosis of CRBSI. The presence of a germ in the catheter blood culture only indicates a catheter colonization but not a catheter-related infection. Catheter-related bloodstream infections (CRBSI) should not be confused with Central Line Associated Blood-Stream Infection (CLABSI), which is defined as a primary bloodstream infection in patients with a CVAD placed within the 48-h period before, not related to another site infection. Since in Intensive Care Unit (ICU) many bloodstream infections may be secondary to sources other than the CVAD (pneumonia, urinary tract infections, surgical infection, etc.) the incidence of CLABSI overestimates the true incidence of CRBSI. 2
Over the past two decades, significant efforts have been made to reduce CRBSI, using approaches based on bundles of evidence-based interventions.4,5 Accordingly, current guidelines recommend standardized protocols during CVADs insertion and management, with the purpose of reducing the risk of infection.6,7 Such recommendations cover all CVADs utilized in ICU, that is, Centrally Inserted Central Catheters (CICCs), Peripherally Inserted Central Catheters (PICCs), and Femorally Inserted Central Catheters (FICCs). Though the relative risk of infection associated with each type of CVAD is still controversial, it is widely accepted that any CVAD, either PICC or CICC or FICC, should be inserted using a proper insertion bundle designed to minimize the risk of infection, as well as the risk of other catheter-related complications (puncture-related complications, CRT, etc.).6 –10
In 2010, the Italian Group for the Study of Long-Term Central Venous Access Devices (GAVeCeLT) developed the SIP protocol (Safe Insertion of PICCs) with the aim of minimizing all risks associated with PICC insertion.10,11 This protocol provided eight evidence-based recommendations, easy to use, inexpensive, cost-effective and able to reduce not only early complications but also late complications related to insertion, including infection and catheter-related thrombosis (CRT). Two years later, the same group developed another protocol for the safe Insertion of CVADs, that was integrated in the policies of various Italian hospitals; 12 this protocol consists of few and clear evidence-based recommendations meant to minimize all complications related to the insertion procedure (puncture-related complications, malposition, arrhythmia, CRT and infection), and it has been recently published on international journals, split in two different protocols for the Safe Insertion of CICCs (SIC) 8 and for the Safe Insertion of FICCs (SIF). 9
For many years now, the Neuro-Intensive Care Unit has endorsed the GAVeCeLT systematic bundles for the insertion and management of all CVADs (PICCs, CICC and FICCs); in this prospective clinical study, the authors evaluated the incidence of CVAD-related complications during a 4-year period, so to verify the effectiveness of these bundles in minimizing complications.
Methods
This single-center prospective study was conducted, after the approval of Ethics Committee, in the neuro-ICU of ‘A. Manzoni’ hospital in Lecco, Italy. This ICU has six beds staffed by trained nurses with a 1:2 nurse-patient ratio.
Since 2015 the authors have implemented a comprehensive CRBSI prevention program, adopting the GAVeCeLT recommendations for insertion and management of CVADs. The program included: (1) intensive education of ICU staff about the GAVeCeLT recommendations for both insertion and maintenance of CVADs; (2) systematic application of these bundles, (3) regular auditing of the clinical practice by dedicated ICU nurse staff and (4) close monitoring of CRBSI rate, with periodic feedback with the ICU medical staff.
The primary objective of this study was to investigate the actual rate of infective complications, differentiating among CRBSI, CLABSI and catheter colonization, using DTP as diagnostic tool. The secondary objective was to evaluate the incidence of all relevant non-infective catheter-related complications, both early and late, so to assess the feasibility and the effectiveness of the GAVeCeLT bundles.
Inclusion criteria
Over a period of 47 months (Jul 2015–May 2019), all consecutive adult patients (⩾18 years old) requiring a CVAD during their ICU stay were considered for inclusion. Were included all patients with CICCs, PICCs and FICCs left in place for more than 72 h. Catheters in place in ICU for less than 72 h (either because the device was removed for clinical reasons, or because the patient was moved to another ward) were not considered in this analysis.
Criteria for the choice of the CVAD
As suggested by current guidelines,6,7 central lines were inserted in patients requiring multiple infusions lines, and/or infusion of parenteral nutrition, and/or solution potentially associated with damage to the endothelium, and/or hemodynamic monitoring, and/or frequent daily blood sampling. Most CVADs inserted in the acute phase were CICCs; after the acute phase, PICCs were considered in patients needing prolonged intravenous treatment. When the CICC was inserted by a supraclavicular approach (internal jugular vein, subclavian vein, brachio-cephalic vein), the exit site was either located in the supraclavicular fossa or – tunnelling the catheter – in the infra-clavicular area. The presence of a tracheostomy was considered a strong indication for a PICC or for a CICC inserted by an infraclavicular approach (axillary vein). Hemiplegia or arm paralysis were considered as a contraindication to PICC insertion. FICCs were inserted as a secondary option, only in case of contraindication to both CICCs and PICCs.
The GAVeCeLT bundle for safe insertion of central venous catheters
All CVADs were inserted according to the GAVeCeLT insertion bundles (SIC, SIP and SIF), that are based on the following strategies: (1) pre-procedural ultrasound examination of the deep veins to be cannulated, according to the protocols of Rapid Peripheral Vein Assessment (RaPeVA)10,11 Rapid Central Vein Assessment (RaCeVA) 13 and Rapid Femoral Vein Assessment (RaFeVA) 14 ; (2) proper hand hygiene before insertion, maximal barrier precautions at insertion (full sterile body drape over the patient; cap, mask, sterile gown, sterile gloves for the operator; long sterile cover for the ultrasound probe), and skin antisepsis with 2% chlorhexidine in alcohol; (3) choice of the most appropriate site of venipuncture (the caliber of the vein is considered optimal when at least three times the external caliber of the catheter), considering the option of tunneling the catheter when the puncture site is not judged to be an optimal exit site; (4) real time, ultrasound-guided venipuncture; (5) ultrasound-based tip navigation (i.e., ultrasound assessment of the proper direction of the guide wire and/or the catheter); (6) intra-procedural tip location using intracavitary ECG method and/or echocardiography; (7) securement of the catheter using sutureless devices; (8) protection of the exit site using cyanoacrylate glue and semipermeable transparent dressing.
The GAVeCeLT bundle for maintenance of central venous catheters
After insertion, all CVADs (CICCs, PICCs and FICCs) were handled according to the following maintenance bundle developed by GAVeCeLT: (1) optimized policy of hand hygiene; (2) dressing change once a week, or more frequently in case of need (soaked, detached or soiled dressing); (3) skin antisepsis with 2% chlorhexidine in alcohol; (4) consistent use of sutureless devices; (5) consistent use of transparent semipermeable dressings; (6) use of needle-free connectors with neutral displacement; (7) disinfection of hubs by scrubbing or using port protectors; (8) change of infusion lines every 72 h if used continuously, with exception for propofol (every 6–12 h) and lipid emulsion (every 24 h); (9) flushing with saline before and after each infusion.7,11
Data collection
The data were collected prospectively by the ICU nurses. Each day a dedicated surveillance form – including age, gender, date of ICU admission, clinical diagnosis – was updated for each patient, recording all relevant data concerning the insertion of the CVAD (date, type, site, tip location method, type of sutureless device, occurrence of immediate/early complications, such as pneumothorax, accidental arterial puncture, hematoma, injury of the brachial plexus or phrenic nerve and catheter malposition) and the maintenance of the CVAD (suspected infection, CRBSI, abnormalities of the exit site, results of blood cultures, days in place of the CVAD, reasons for removal, symptomatic CRT, secondary malposition, catheter occlusion, catheter dislodgment).
In case of suspected catheter-related infection, peripheral blood cultures and catheter blood cultures were simultaneously carried out. All such cases were evaluated by an infection control team which included a clinical microbiologist and infectious diseases specialist. According to the DTP, the diagnosis of CRBSI was established when both cultures were positive for the same germ, with the catheter blood positivity at least 120 min earlier than peripheral blood positivity.2,3 A positive culture from peripheral blood only was considered as a false positive due to accidental contamination; a positive culture from catheter blood only was considered as a colonization of the catheter.
In regards non-infective late complications: (a) ultrasound venous scan was performed only in case of symptoms suggesting CRT; no effort was done for assessing the incidence of asymptomatic CRT; (b) dislodgment of the external tract of the catheter and secondary malposition (tip migration, without movement of the external tract) were considered clinically relevant only if the catheter tip had moved 2 cm or more from its ideal location (junction between superior vena cava and right atrium); (c) lumen occlusion was considered clinically relevant only if irreversible.
Statistical Analysis
All data was included in Microsoft Excel, version 2016 for data recording and analysis. Continuous data were expressed as mean and standard deviation (SD), or median (IQR). Categorical data were reported as absolute numbers and percentages (%).
To calculate the incidence rate of CRBSI as events per 1000 catheter-day, the authors divided the total number of episodes of CRBSI/total number of catheter days during the year of the study and then multiply by 1000.
Results
During the study period, were enrolled 467 patients (mean age 63.7 ± 17.5 years, males 61%) for a total of 609 CVAD insertions. Of these, 123 catheters were excluded because in place less than 72 h, so that 486 CVADs were considered in the final analysis (328 CICCs, 149 PICCs and 9 FICCs). The reasons for ICU admission were neurological or neurosurgical diseases in most cases (78%), followed by respiratory failure (7%) and major trauma (5%). CVADs had a median (IQR) of days in place of 10 [6-17] days.
CICCs (
PICCs (
Table 1 summarize the characteristics of the patients and catheters enrolled in the study.
Characteristics of patients and central venous catheters.

In all CVADs, ultrasound was used for different purposes (for choosing the most appropriate vein, for venipuncture, for ruling out puncture-related complications, for tip navigation and tip location, for the diagnosis/monitoring of non-infective late complications). All CVADs were secured using a sutureless device, either adhesive to the skin (StatLockTM, Becton, Dickinson and Company, Franklin Lakes, NJ, USA.;
In regards the incidence of immediate/early complications, the authors did not record any case of pneumothorax, hematoma, primary malposition, arterial puncture or injury of the brachial plexus or phrenic nerve (Table 2). Primary malposition occurred only in 5 cases (1%); in these 5 patients, all with atrial fibrillation, the location of the catheter tip could not be verified during the procedure; after a post-procedural chest x-ray, the tips appeared to be too deep and were withdrawn by 1–2 cm.
Catheter-related complications.
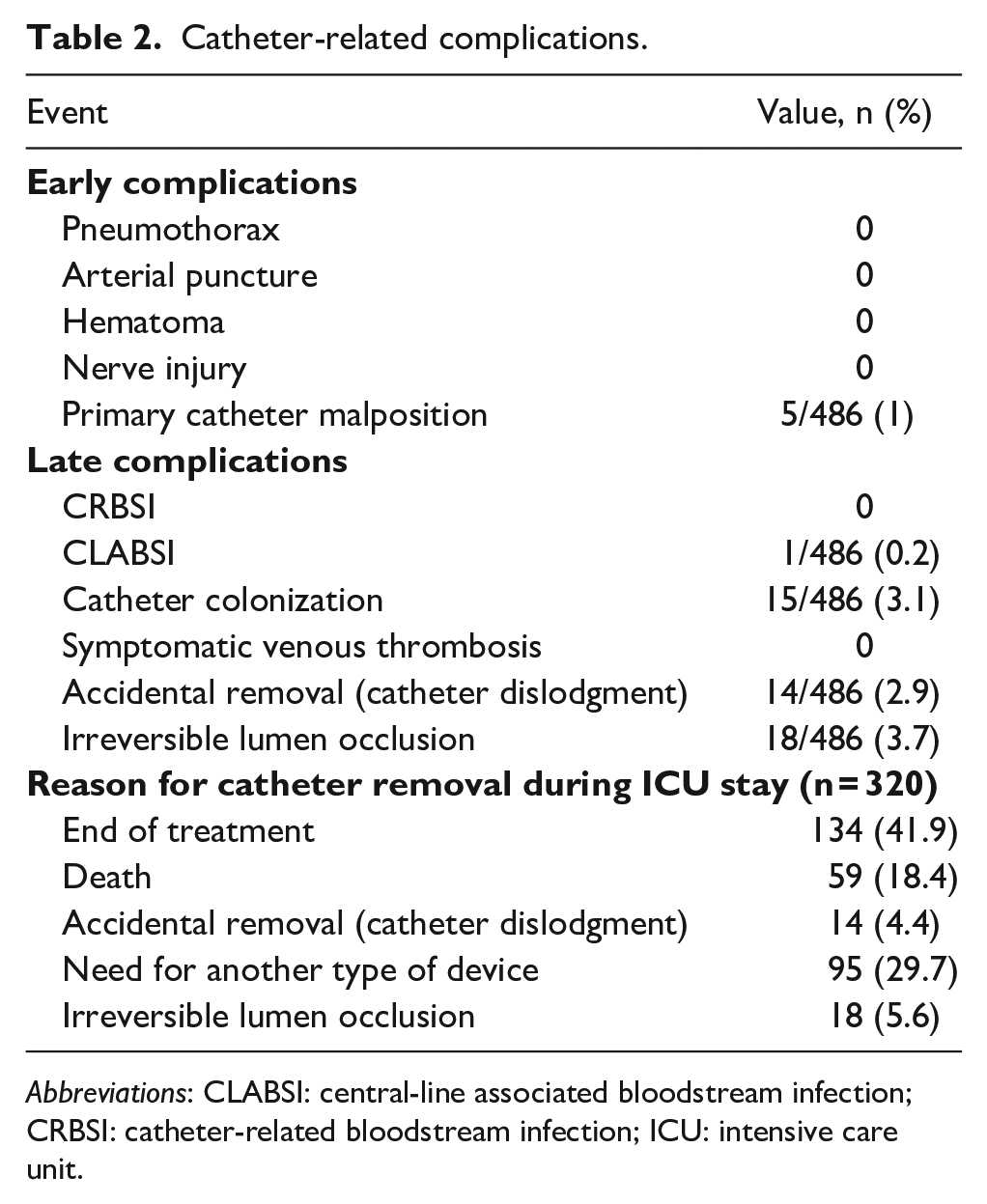
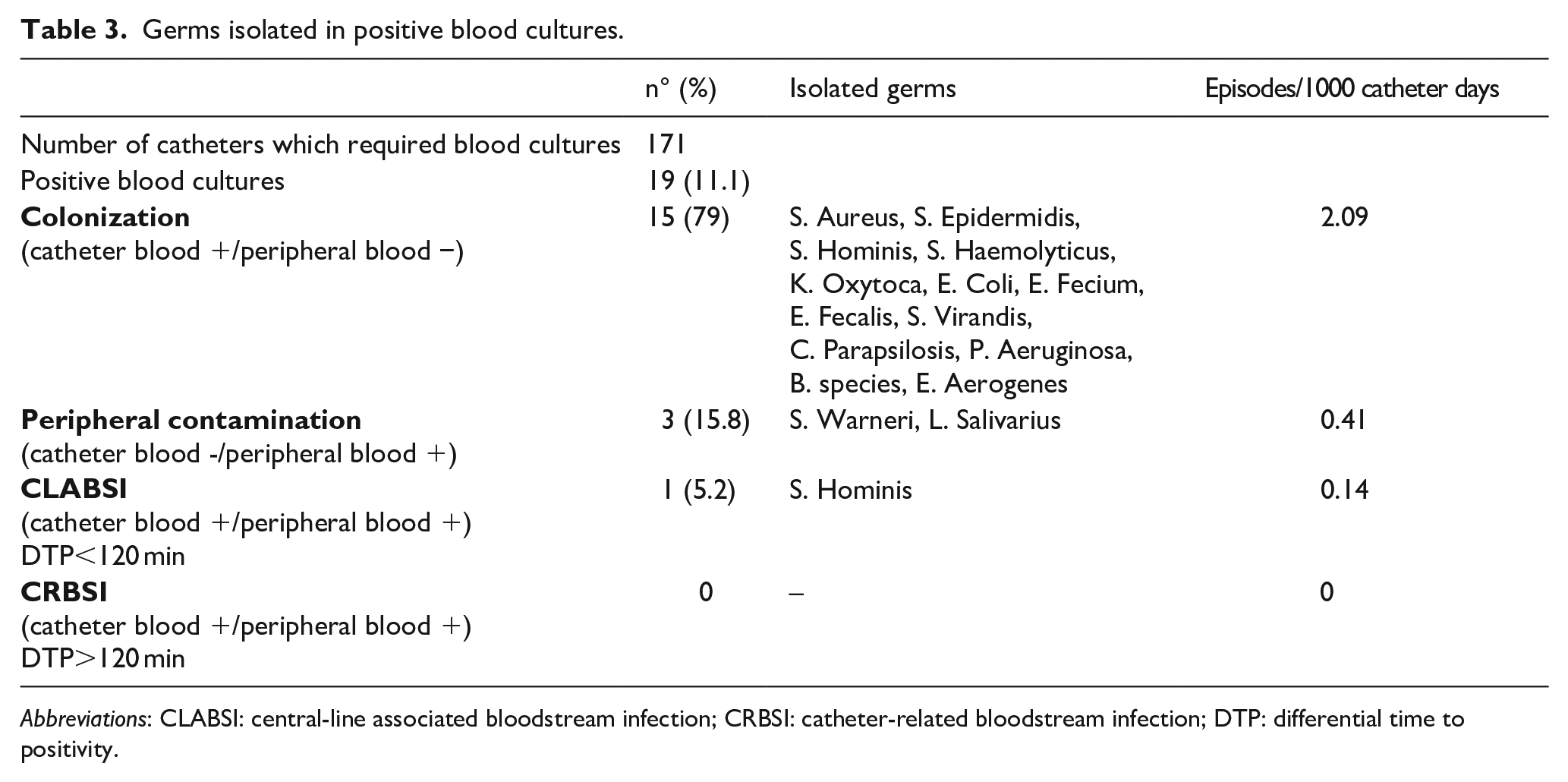
In regards the incidence of late infective complications, the authors did not observe any case of CRBSI. They documented only one case of central line associated blood stream infection (CLABSI) (0.14/1000 catheter days). This occurred in a 65-year-old woman, admitted to the ICU for a severe neurological problem. She had a tracheostomy and a triple-lumen CICC placed by a supraclavicular approach. Nine days after CICC insertion, S. hominis was cultured from both peripheral and catheter blood, with DTP < 120 min (34 h for peripheral blood vs. 46 h for catheter blood cultures); thus, the event was classified as a CLABSI (central line associated blood stream infection) and not as a CRBSI (catheter-related blood stream infection). The catheter was left in place and an appropriate antibiotic treatment was started. In 15 patients (12 CICCs and 3 PICCs), after paired peripheral/catheter blood cultures, a diagnosis of catheter colonization was established (positive culture from the catheter blood but not from peripheral blood), for a total incidence of 2.09 episodes/1000 catheter days (Table 3). All colonized catheters were removed and replaced, and no antibiotic treatment was started.
Germs isolated in positive blood cultures.
In regards late non-infective complications, they did not observe any case of symptomatic CRT; in one case with symptoms suggesting venous thrombosis, a fibroblastic sleeve was visualized by ultrasound. They recorded 14 accidental dislodgments (2.9%), 18 irreversible lumen occlusions (3.7%), and no tip migration (Table 2).
Discussion
These data confirm that the incidence of complications related to CVADs can be minimized by a strict adherence to evidence-based recommendations; this is best accomplished by using appropriate insertion bundles and maintenance bundles.
Today, catheter-related complications should be regarded as a preventable event. Several different strategies have proposed for reducing the incidence of early and late complications. After the adoption of the GAVeCeLT bundles for insertion and maintenance of CVADs, this neuro-ICU has been able to achieve a zero incidence of CRBSI for a 4-year period. In a national survey of adult ICUs in the USA, a low incidence of CLABSI was significantly associated with a high (>95%) overall compliance to the bundles. 15 Suboptimal compliance rate, particularly for maximal sterile barrier precaution, might partly explain why in some studies 16 the reduction of CLABSI was considerably lower than in other studies.17,18 Suboptimal compliance may be due to inappropriate education of the staff, or by the perception that some strategies may not be relevant, For example, in clinical practice, maximal barrier precautions are often perceived as a time-consuming strategy, if compared with other elements of the bundle; though, nowadays, the availability of all-inclusive insertion packs has facilitated the adoption of barrier precautions, motivating behavioural changes and the staff adherence to the bundles.
This data suggest that infective complications are not related to the type of central venous catheter (CICC vs. PICC) but to the systematic use of proper bundles for CVAD insertion and management. Interestingly, the authors recorded a higher rate of colonization for CICCs compared to PICCs. This might be explained by the higher bacterial concentration in the skin of the cervico-thoracic region compared to the middle third of the arm. Though, such catheter colonizations did not result in CRBSI.
Many factors are related to an increased risk of CLABSI in ICU: use of femoral access; 19 prolonged ICU stay before CVAD insertion; nurse to patient ratio; previous CVAD insertion in the Emergency Room and not in ICU; 19 CVAD left in place for more than 8 days.19,20
The authors avoided FICCs as much as possible. Also, they removed all types of CVAD when no longer needed. Considering that the choice of exit site is crucial to reduce risk of infection, they consistently avoided an exit site at the neck, preferring an exit site in the supraclavicular fossa or – in case of tracheostomy – in the infraclavicular area or at midarm. Also, nurse-to-patient ratio was 1:2 (close to optimal).
They adopted the standardized GAVeCeLT protocols of preprocedural venous assessment – RaCeVA, 13 RaPeVA, 10 and RaFeVA 14 – to establish the ideal puncture site of CICCs, PICCs and FICCs, respectively. In regards the choice of the ideal exit site, they have adopted – when necessary – the strategy of tunnelling the catheter. Tunnelling is an easy and effective tool to obtain an optimal puncture site and, at the same time, an optimal exit site; it was originally considered as a technique mainly related to long term cuffed-tunnelled catheters, but recently it has been extended also to non-cuffed catheters, because of its clinical benefits (easier choice of the puncture site, improved catheter stabilization, reduced risk of infection, longer life of the device). Though tunnelling of non-cuffed catheters was already considered as an option by GAVeCeLT 7 since 2015, only recently it has been described in detail in the RAVESTO protocol (RAVESTO = Rapid Assessment of Vascular Exit Site and Tunneling Options), 21 that describes the main options of subcutaneous tunnelling and their indications in different clinical situations in patients with complex vascular access. In this study, tunnelling was adopted in 20% of PICCs: that is, in all 6Fr triple-lumen PICCs, so to be able to cannulate a vein of at least 6 mm in diameter, and still achieve an exit site at the middle third of the arm. Some CICCs were also tunnelled when the puncture site was too close to the tracheostomy.
After implementing GAVeCeLT bundles for CVAD insertion, which include the consistent use of real time ultrasound guided venipuncture, the authors did not record any relevant puncture-related complication (pneumothorax, arterial puncture, nerve injury and hematoma). Other late non-infective complications (symptomatic CRT, lumen occlusion, catheter dislodgment) were absent or infrequent, because of the consistent adoption of the GAVeCeLT bundles for CVAD insertion and maintenance, as described above. Intensive education of the ICU staff about the GAVeCeLT bundles, periodic audit of the clinical practice by dedicated nurses, as well as close monitoring of all complications, with periodic feedback with the ICU medical staff, were crucial to obtain these results.
Strengths and limitations
The strengths of this study are its prospective design, the long period of observation (47 months), and the consistent adoption of well-defined insertion and maintenance bundles. However, there are also some limitations. First, the study was carried out in a neuro-ICU, so that the results cannot be automatically extended to other hospital wards, such as a surgical ICU, or to other type of patients; though, the authors believe that the systematic application of bundles is expected to yield favourable results in many clinical settings. Also, they did not stratify the patients according to their clinical conditions, some of which might have been a critical risk factor for development of infection. Last, the CVADs they studied stayed in place for a limited period (average 10 days); longer dwelling is expected to be associated with an increased risk of infection, CRT and other late complications.
Conclusion
This prospective clinical study showed that – in a neurological ICU - the consistent adoption of the GAVeCeLT bundles for CVAD insertion and maintenance is associated with a minimization of catheter-related complications, for a prolonged period (4 years). The authors had no early complications, no CRBSI and a very low rate of CLABSI (0.14/1000 catheter days) and of catheter colonization (2.09/1000 catheter days), a higher incidence of catheter colonization being observed in CICCs compared to PICCs. The rate of non-infective late complications was also very low. The strict adherence to the recommendations included in these bundles was the major determinant for clinical success.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
