Abstract
Background:
The growing size of the end stage renal disease (ESRD) population highlights the need for effective dialysis access. Exhausted native vascular access options have led to increased use of catheters and prosthetic shunts, which are both associated with high risks of access failure and infection. Emerging alternatives include tissue-engineered vascular grafts (TEVG). Here we present the endpoint results for 10 ESRD patients with the scaffold-free tissue-engineered vascular access produced from sheets of extracellular matrix produced in vitro by human cells in culture.
Methods:
Grafts were implanted as arteriovenous shunts in 10 ESRD patients with a complex history of access failure. Follow-up included ultrasound control of graft morphology and function, dialysis efficiency, access failure, intervention rate, as well as immunohistochemical analysis of graft structure.
Results:
One patient died of unrelated causes and three shunts failed to become useable access grafts during the 3-month maturation phase. The 12-month primary and secondary patency for the other six shunts was 86%. Survival of six shunts functioning as the vascular access was 22 ± 12 months with longest primary patency of 38.6 months. The dialysis event rate of 3.34 per patient-year decreased significantly with the use of this TEVG to 0.67.
Conclusions:
This living autologous tissue-engineered vascular graft seems to be an alternative to synthetic vascular access options, exhibiting advantages of native arteriovenous fistula.
Keywords
Introduction
In only a decade, the number of patients on hemodialysis has increased by 35% (from 365k in 2009 to over 492k in 2019) despite a simultaneous 40% increase in the number of functioning renal transplants. 1 One of the major factors that increased the demand for renal replacement therapy over the same period was a 19% decrease of mortality rate in the ESRD population. 1 Nonetheless, improved vascular access (VA) options could further increase the survival of ESRD patients and would significantly improve the quality of life of this growing patient population.
VA complications are the predominant cause of morbidity and hospitalization of hemodialysis patients. 1 The native arteriovenous fistula (AVF) is recognized as the current gold standard for VA by clinical practice guidelines and programs like the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI). 1 In the U.S., the use of AVF has increased significantly over the last decade under the influence of the Fistula First Breakthrough Initiative. 2 Nevertheless, synthetic grafts are still commonly implanted for vascular access, despite well-known complication risks. 1 In addition, a central venous catheter (CVC), without maturing fistula or shunt, was the initial VA in over 60% of chronically hemodialysed patients, while it was the ultimate access in over 18% of them. Initial lack of suitable native vessels for arteriovenous shunting and exhausted AVF sites may be considered the main causes of these troubling numbers. While the CVC and synthetic arteriovenous graft (sAVG) can provide sufficient dialysis efficiency, the poor outcomes with lower patency rates of these vascular accesses compared to AVF are well documented.1,3 Furthermore, AVF has a 4 to 12-fold lower access-related infection rate, compared to synthetic options.1,4 This represents to a significant economic advantage, as shown by the overall maintenance costs reported by Medicare, of $58,588 per patient/year for AVF versus $72,729 and $79,364 for sAVG and CVC, respectively. 5
In light of the clearly superior outcomes of fistula compared to synthetic VA options, the development of a completely biological tissue-engineered access graft has been an important research goal in both the academia and the industry. Previously, we have reported preliminary to mid-term results of the nine autologous, scaffold-free, tissue-engineered vascular graft (TEVG, LifeLine™ from Cytograft Tissue Engineering) used for VA. 6 Here we present the comprehensive ultimate results for a group of 10 patients, including dialysis efficiency and structure characteristics of the grafts during their chronic use for hemodialysis. The cumulative follow-up period was 11.3 patient-years.
Material and methods
Study design
We analyzed long-term outcomes of the scaffold-free TEVG for hemodialysis access in 10 elderly ESRD patients (60 ± 14 yo) with a complex history of access failure.
Study was carried out according to The Code of Ethics of the World Medical Association (Declaration of Helsinki) and was approved by
Patient demographics and medical history
Study included 10 (seven from Argentina and three from Poland) ESRD patients, who underwent the autologous LifeLine™ graft implantation as an upper limb arteriovenous fistula, between 2005 and 2009. At the moment of enrollment patients were on hemodialysis for 49 ± 44 months (range: 4.4–147.5). A high risk of failure of the current vascular access within 12 months, with exhausted options for a new upper limb native vein fistula was the major inclusion criterion (Supplemental Table S1).
Graft production and implantation
Vessels were built, as previously described, using the technique that relies on the ability of human skin fibroblast to assemble robust sheets of extracellular matrix when cultured in the right environment. 6 Briefly, skin and superficial vein samples were biopsied under local anesthesia. Dermal fibroblasts were isolated following epidermal/dermal separation using thermolysin followed by collagenase digestion. The resulting cells were grown in media supplemented with 20% fetal bovine serum and ascorbic acid (500 µm) to promote the production of collagen. After 8–9 weeks in culture, cells became embedded in their own cell-assembled extracellular matrix and could be detached from the cell culture substrate as a cohesive sheet. Sheets of living fibroblasts were then wrapped around a temporary support mandrel (4.8 mm diameter) and placed in a bioreactor for a maturation phase to fuse the individual layers of 4-ply laminate role. After a maturation phase of 10–12 weeks, the construct was air dried, and then wrapped with a second living sheet (4 plies). After a second maturation phase in the bioreactor, the entire construct was well-fused and the lumen of vessels was seeded with living autologous endothelial cells that were allowed to grow to confluency over 1 week. For each patient, 10–16, 18 cm-long conduits were fabricated. Two endothelialized vessels were transported to the clinic for implantation while others were used for final release testing (mechanical properties, sterility, mycoplasma, endotoxin, endothelial coverage, and histological examination).
Grafts were implanted as the brachial-axillary or brachial-basilica arteriovenous shunt in nine patients. In one case the forearm access was created. For the first two patients, a short graft was used (14–18 cm). In latter cases implanted graft consisted of two conduits, anastomosed end-to-end at the moment of implantation. Procedures did not require special handling or instrumentation.
Access function and dialysis efficiency
A minimum 3-month safety and maturation phase prior to graft puncture was a mandatory part of the clinical trial protocol. Apart from the prospective analysis of the LifeLine™ shunt functionality, the retrospective analysis of previous vascular access efficiency, as well as general population data published by the United States Renal Data System (USRDS) were used for comparison. The single pool Kt/V (spKt/V) was calculated with Daugirdas formula. 4 Analysis included also dialysis blood flow, post-dialysis hemostasis and antithrombotic treatment. Dialysis flow rate was individually adjusted by an attending nephrologist according to his experience and hemodialysis unit internal guidelines.
Graft morphology
Graft morphology was assessed with an ultrasound examination on day 2, 4, week 1, 2, 4 and monthly thereafter. Multipoint measurements included feeding artery, outgoing vein and graft diameters, with particular analysis of the anastomoses, as well as sites of puncture. In addition, the minimal and maximal graft diameters were recorded. Doppler assessment included flow volume and flow velocity at anastomoses.
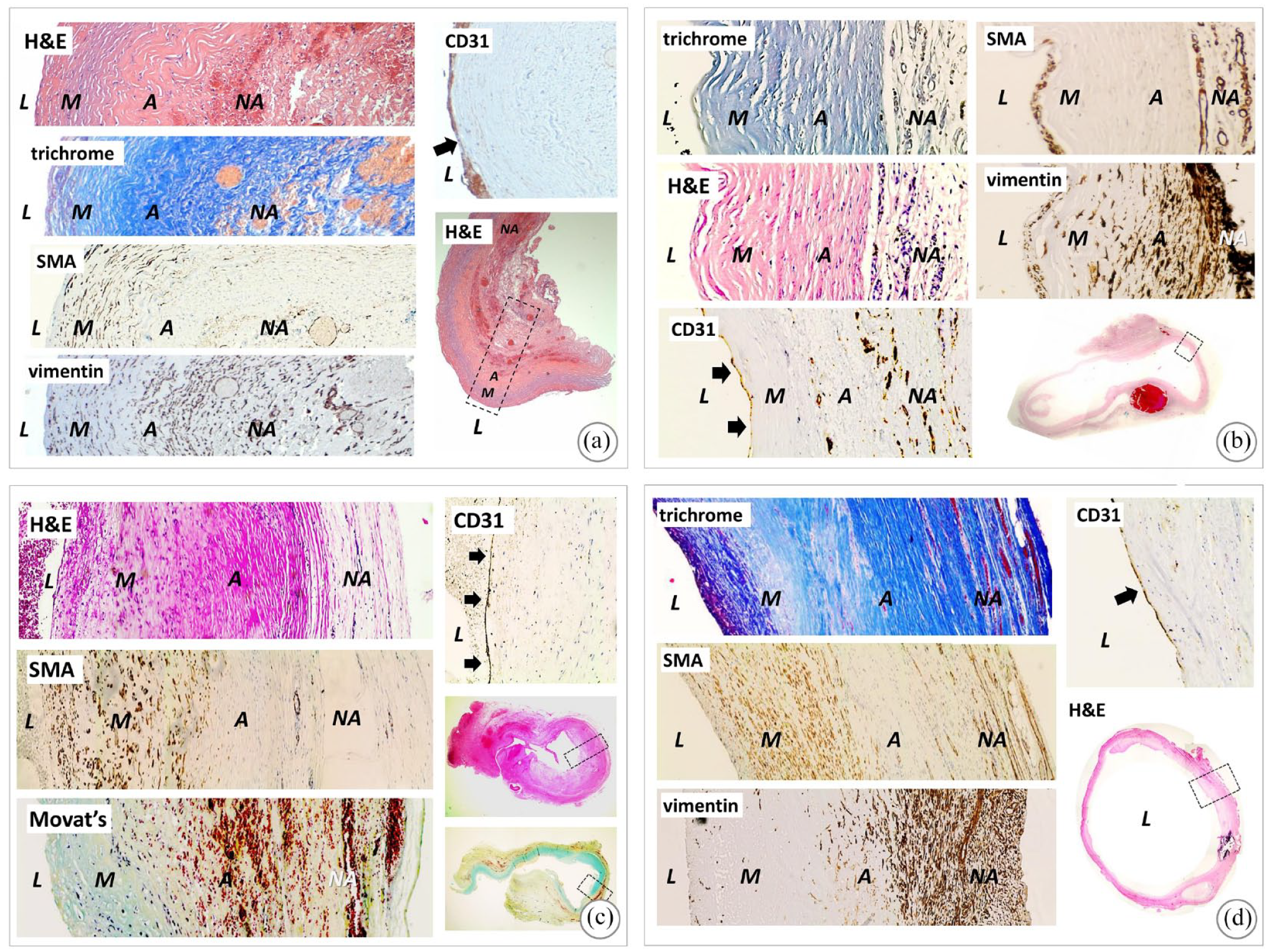
Four of six grafts functioning as the hemodialysis access were excised (in whole or in part) during surgical intervention or after cessation of the access use and were subjected to the histological analysis. This included H&E, Movat’s and Masson’s trichrome, as well as the immunohistochemical staining of endothelial cells (CD31) and smooth muscle cells/myofibroblasts (SMA—alpha-smooth muscle actin and vimentin).
Statistical analysis
Data are shown as means ± SD. Paired statistical analyses were performed using Student’s
To account for the variation of time intervals between graft assessments, the average over time was calculated for chosen parameters as the mean of a function over the entire observation interval (integral over the analyzed time period).
Results
Graft survival
Previously we have reported early outcomes of this study. 6 Briefly, following strict safety rules, two patent grafts were removed within 1 month after implantation (one because of immune reaction, one due to a surgical wound complication) and one patient died of unrelated causes 5 weeks after implantation. During the entire observation period two shunts (20%) were abandoned due to graft thrombosis: graft 3 thrombosed after 10 weeks and was successfully restored but ultimate patency was lost 3 weeks later; and graft 4 thrombosed after 29 months. Overall, 12-month death-censored access survival was 56%. Apart from two shunts removed for safety reasons early after implantation and one graft which thrombosed, the other access was removed due to a steal syndrome after 9.8 months. Considering only the six grafts functioning beyond the “maturation period,” the 12-month graft survival was 83%. The mean graft survival time for these grafts was 22 ± 12 months (Table 1).
Including only five fully LifeLine™-dependent patients, the event rate before TEVG application was 3.34 per patient-year. Application of the LifeLine™ access significantly reduced this rate to 0.67. While this is a small study, the LifeLine™ results obtained with a very challenging patient cohort compare favorably to that of AVF (Figure 1). 7
Analysis of the LifeLine™ graft patency and survival. Patient numbering in accordance with previous report 6 (patient 7 was withdrawn from the study prior to implantation).
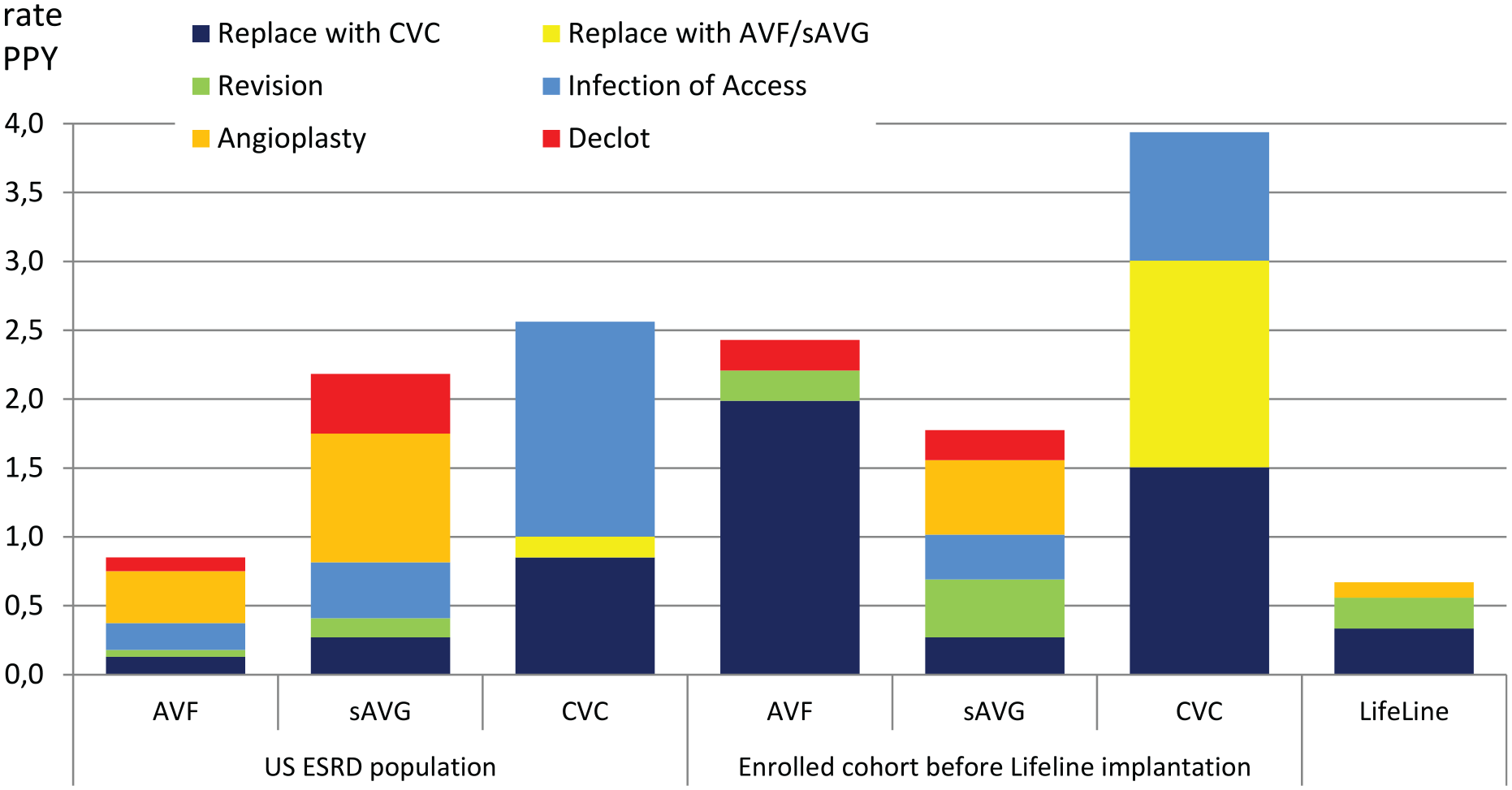
Access related interventions/events rate per patient year (PPY). Five LifeLineTM dependent patients in comparison to the results of the preimplantation standard of care for enrolled cohort and the US ESRD population statistics. 7
Graft properties
All graft lots met or exceeded release criteria (Supplemental Table S2).
No significant stenosis was observed at the arterial (AA) and venous (VeA) anastomosis. Mean diameter of VeA and outflow vein diameter, measured 2 cm from VeA was 5.2 ± 0.7 mm and 8 ± 3 mm, respectively. A clinically significant (>80%) downstream axillary vein narrowing was observed in one case (patient 11) as the complication of the previous thoracic axillo-axillary ePTFE shunt (Supplemental Table S3 and Figures S1–S9). The unpunctured arterial and venous side of graft showed a slight diameter change rate of 0.1 ± 0.1 mm/month and 0.3 ± 0.2 mm/month, respectively. Larger graft dilation was observed at sites of recurrent punctures (0.6 ± 0.6 mm/month, range: 0.08–1.6 mm/month) in the same five LifeLine™-dependent patients (
The pre-implantation wall structure was bilayered with an internal acellular layer and an external layer with living fibroblasts (Supplemental Figure S12). Immunohistochemical analysis revealed that the LifeLine™ underwent a progressive remodeling process in vivo (Figure 3).
Dialysis efficiency
The LifeLine™ shunt was used as the exclusive hemodialysis vascular access in five patients (#1, 4, 9, 10, and 11) for a cumulative period of 84.6 months (mean 17 ± 13 months, range: 4.4–35.1 months). Grafts were punctured for the first time at 128 ± 20 days (range: 67–149 days) after implantation. The clinical trial protocol was conservative regarding the first hemodialysis sessions and involved the use of access initially once per week with increasing frequency to achieve full graft utilization within 2–10 weeks. Following this, all patients were dialyzed for 4 h, 3 times per week, with 16 G needles. Hemodialysis efficiency assessed with spKt/V was 1.5 ± 0.2 at mean dialysis blood flows of 320 ± 66 ml/min. These results were insignificantly better than achieved using central venous catheters in the preceding year (Figure 4).
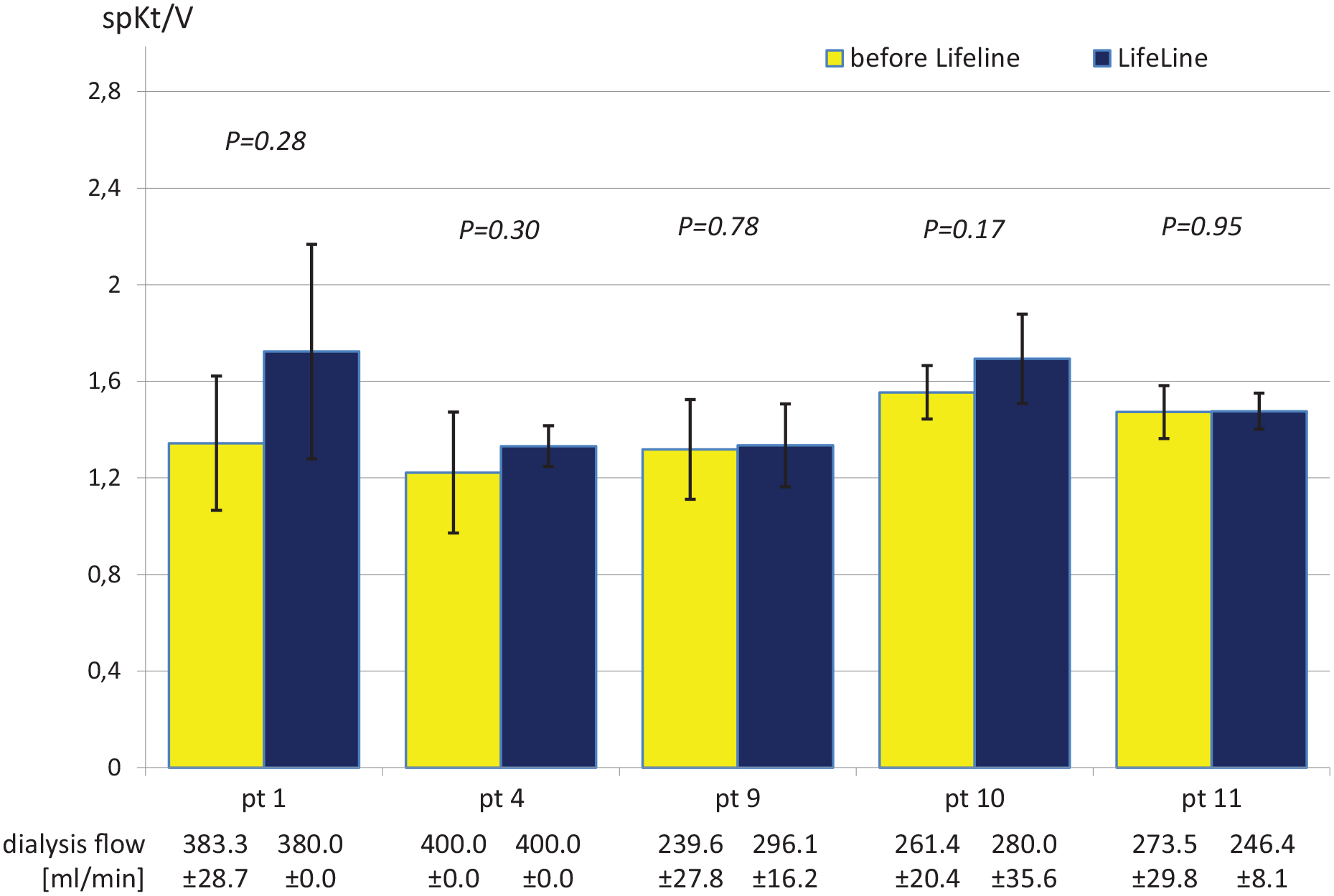
The mean flow after hemodialysis onset was 1317 ± 696 ml/min (range: 839–2837 ml/min), allowing proper access functionality. Unsurprisingly, there was a significant correlation between flow volume and the minimal flow lumen identified along the feeding artery, shunt, and outgoing vein (R Spearman = 0.69,
In all dialysis patients, hemostasis time after needle removal was below 10 min with mild compression. In only one case (patient 9), and only at the first hemodialysis session, LifeLine™ access was complicated with a 2-cm false aneurysm at the puncture site. This incident was caused by an overdose of acenocoumarol (INR at 3.4). Conservative management brought a full
Antithrombotic treatment and graft thrombosis
Five patients with symptomatic cardiovascular diseases (#2, 6, 9, 10, 11) were on oral antiplatelet therapy. Two patients with atrial fibrillation (#9 and 10) and three patients with CVC (#5, 11) were treated with low molecular weight heparin or vitamin K antagonist (acenocoumarol). In the CVC group this treatment had been continued until the LifeLine™ became the exclusive vascular access. Two patients (#3, 4) whose graft thrombosed were not treated with any antithrombotic/antiplatelet agent. However, in first case computed tomography angiography revealed critical stenosis of the subclavian vein after previous catheter implantations. In the second one, the Doppler surveillance revealed a progressive non-critical narrowing at the VeA (up to 40%).
Interventions
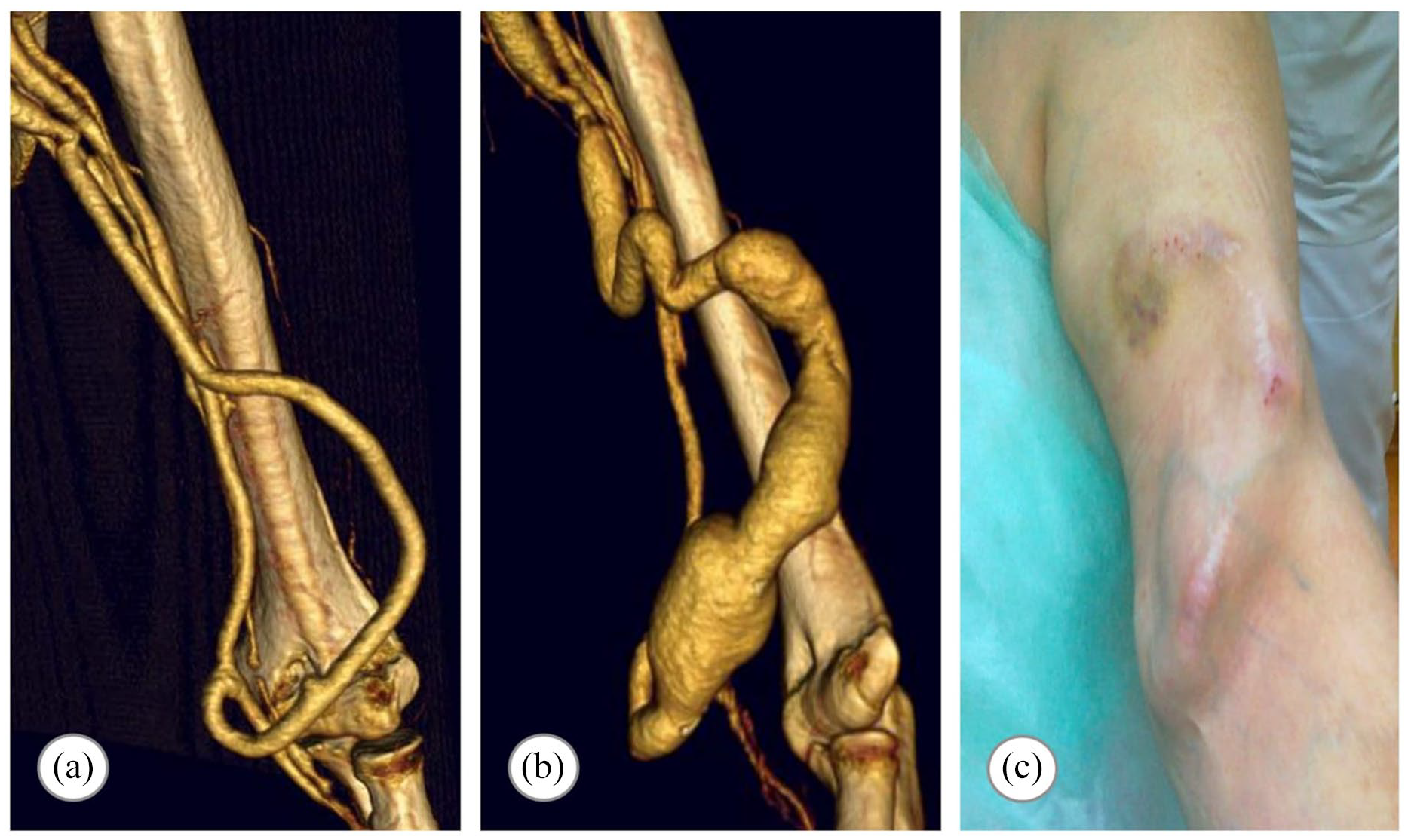
First implanted graft demonstrated a short graft “camel hump” dilation up to 2.5 cm (type 2a according to the Valenti et al. 8 classification) at the spot of recurrent punctures, 11 months after implantation (Supplemental Figure S1 H). A successful aneurysmectomy allowed further access usage. A different management had been applied in patient 9, with the observed slow graft dilation (type 2a) up to 3 cm (Figure 2, Supplemental Figure S7 G–N). The chronicity of the process, long fusiform dilation strictly related to the graft needling allowed the successful conservative treatment. Entire graft slow dilation up to 1.9 cm (type 1a) was observed in two cases (patients 3 and 6) and did not require any intervention (Supplemental Figures S3 and S6).
The uncomplicated subclavian vein angioplasty with stent implantation was performed in one patient (#11) suffering from an increased upper limb edema after the shunt creation, allowing access usage for the following 13 months, till patient’s death. It is worth emphasizing that 7 F (2.4 mm in outer diameter) sheath was placed percutaneously in the LifeLine™ graft providing delivery access for this procedure. Hemostasis after system removal was achieved within 10 min of mild compression with one safety skin suture.
Graft banding procedure was applied in one diabetic patient with symptoms of the ischemic steal syndrome (#10, Supplemental Figure S8 I). Due to the lack of sustained clinical improvement, shunt was ultimately ligated after 9.8 months of primary patency.
There was no report of access-related infection.
Cost analysis
The cost of graft-related treatments was estimated before and after TEVG implantation for five patients dependent on the LifeLine™ graft. The costs per event were based on the average Medicare hospital outpatient rates (2015/2016 National Average Medicare Physician Fee Schedule and Ambulatory Payment Category Reimbursement) and access infection costs. 9 Based on these estimated costs, the average LifeLine™ access maintenance cost was $293 ± 250 per patient per month (PPPM) compared to the $2018 ± 1250 PPPM for preimplantation expenditures (Figure 5). This calculation does not include the cost of an autologous LifeLine™ graft at $15k, approximately. This expense is only an approximation since it is very difficult to evaluate production costs in a commercial setting (large scale, automation, GMP, and various overheads).
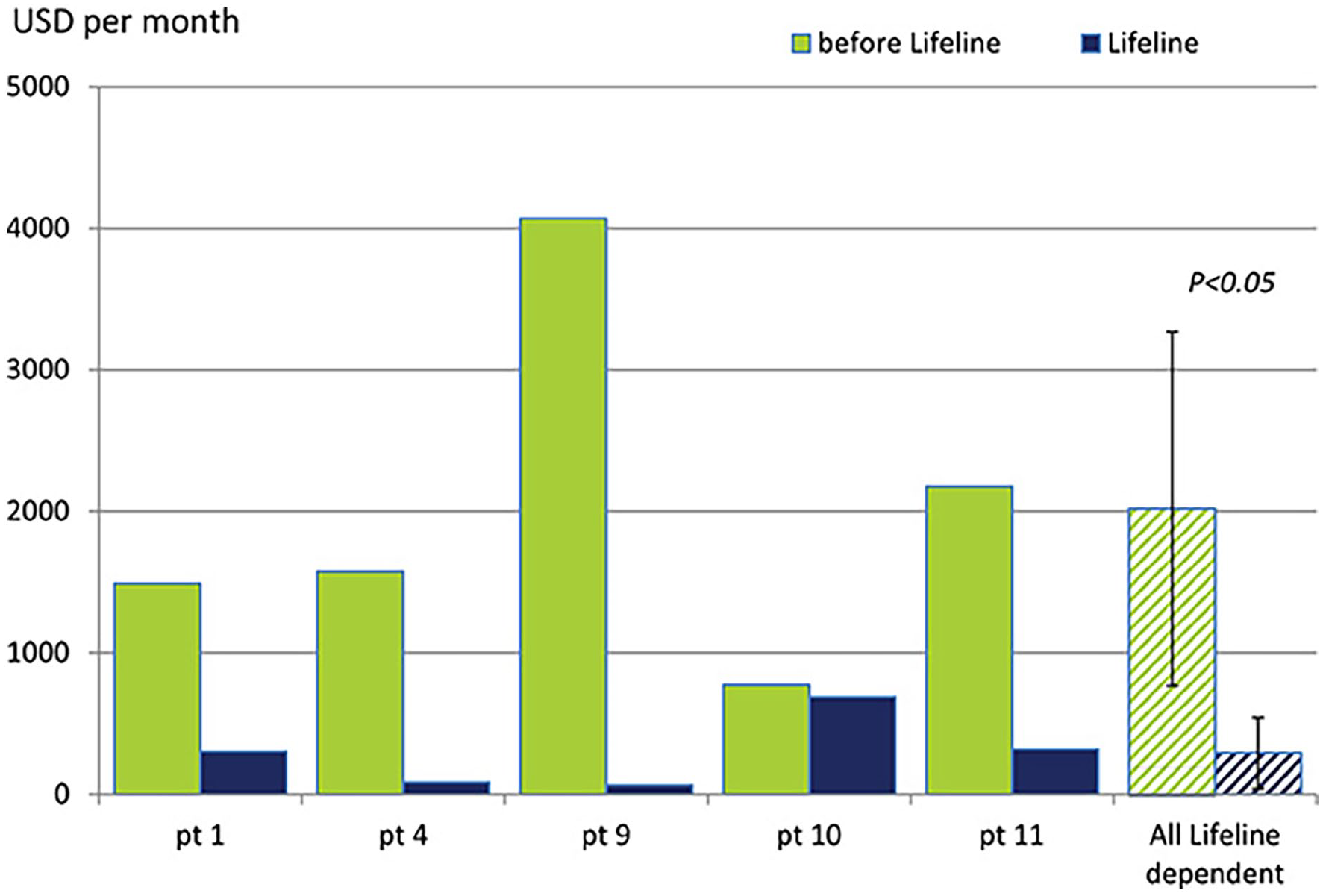
Cost analysis. Comparison of the estimated event/intervention related costs (average expenditures per month in USD) before and after TEVG application in the LifeLineTM dependent patients. (mean±SD, Student T test). Procedure cost estimation according to the 2015/2016 National Average Medicare Physician Fee Schedule and Ambulatory Payment Category Reimbursement. Analysis included cost of the CVC implantation at $2550 and its removal at $400, shunt revision/declot charged with $4150, costs of the PTA estimated at $5100 and cost of AVF/sAVG creation set at $3500. Since infection treatment depends on the VA type, costs of 26 k$, 10 k$, and 6 k$ were used for CVC, sAVG and AVF dependent patients, respectively. 9
Discussion
We present the final results for 10 patients implanted with a scaffold-free TEVG built exclusively from autologous fibroblasts and endothelial cells. The overall observation was 136 patient-months with 85 patient-months of continuous graft use as the exclusive vascular access. In this study, we followed the NKF KDOQI recommendation set forth for AVF that suggests a 3–4 months maturation prior to the first puncture. 4 However, the LifeLine™ graft likely does not require this maturation time since its strength and diameter do not require vascular remodeling to be effective access shunt. In addition, the TEVG was shown to seal well after puncture in vitro under arterial pressure, unlike a standard ePTFE graft (Supplemental Video 1). 10 However, it may still be clinically more cautious to allow some “maturation” (2 weeks) to achieve ingrowth eliminating perigraft void space.
With a 33% failure before usage, LifeLine™ may be considered equal to AVF that has shown rates of failure-to-mature as high as 50%.11,12 It should be noted that the cohort of patients enrolled to this study represents a very challenging patient population with high levels of access failure risk factors. 13
Increase of the entire graft diameter, as well as significant graft dilation at puncture sites, was observed. These are the remodeling processes typical for the AVF chronic response to high blood pressures, high volume flows, and access needling 14 affecting up to 60% of AVFs. 15 As the processes responsible for the native AVF aneurysmal dilatation remain unclear, it is hard to point to factors other than recurrent punctures as the cause of changes observed in the LifeLine™ graft. According to the NKF KDOQI guidelines an asymptomatic fistula aneurysm does not require intervention regardless of its size.4,16 Our approach in this safety-study was more cautious and consequently one patient with localized asymptomatic type 2a aneurysm was qualified for the surgical intervention due to dynamics of dilation.
Apart from the initial vascular abnormalities, no de novo critical stenosis of the downstream vein was observed. Observation of the low incidences of the LifeLine™ venous anastomosis and juxta-anastomotic stenosis is consistent with a comparative analysis of the venous intimal hyperplasia presented by Prichard et al. in the experimental models of TEVG and PTFE shunts. 17 The highest level of venous anastomosis stenosis observed in this study was 40% and it may have been a contributor to the graft thrombosis at 27th month. However, it had not affected graft functionality over the previous 2 years of chronic use for dialysis access. These stenotic complications are common for sAVGs and are largely responsible for their low primary patency rates. One of the proposed mechanisms responsible for this phenomenon is the compliance mismatch between prosthetic graft and native vein. 18 The resulting wall stress, turbulent flow, and lower shear stress zones, are thought to cause intimal hyperplasia, leading to the critical outgoing vein stenosis and subsequent graft thrombosis. Additional factor aggravating this process is a foreign body reaction with an inflammatory response. 19 Previously, we have shown that the fully biological structure of the LifeLine™ graft, after 5–6 moths of in vivo remodeling, appears to develop artery-like compliance. 20 Combined with the lack of a pro-inflammatory synthetic scaffold and the presence of an endothelial lining, these key characteristics may explain better outcomes in comparison to the sAVG.
Surgical interventions revealed a proper graft wall integration, robustness and wall structural integrity. The histological analyses of the explanted grafts confirmed a preserved, well organized, concentric multilayer architecture, even after the long-term use. The immunohistochemical staining revealed the cellularization of the graft with the appearance of a confluent endothelial lining, a media-like layer rich of SMA-positive cells, and an adventitial layer of living fibroblasts surrounded by a highly vascularized peripheral neoadventitia. This wall remodeling was consistent with the study on the LifeLine™ graft in the animal model. 21 A similar adaptive process of arterialization with increasing proliferating activity and SMA expression in the media is also typical for the maturing arteriovenous fistula.22–24 Analysis of grafts structure at different time points showed a slow remodeling process with increasing medial cell density and differentiation from a myofibroblastic to a more SMC-like phenotype. While it is not possible to conclude definitively, the histology of the graft at longest time point (38.6 months) suggests that the original extracellular matrix (ECM) was still largely present. These data suggest that the LifeLine™ fully integrates in the host and undergoes a slow remolding process that may be still ongoing after 3 years.
Apart from the well-recognized antithrombotic effect, endothelial lining influences graft wall repopulation and remodeling, and suppresses intimal hyperplasia. 25 Previous studies on the biodegradable synthetic polymer scaffold-based TEVG pointed to the early graft macrophage infiltration as a result of the foreign body inflammatory reaction with subsequent fibroblast and myofibroblast proliferation. Matsuzaki et al. 26 confirmed the necessity of this process for the formation of the neotissue in a biodegradable scaffold, but also demonstrated its detrimental role in the graft stenosis. Hence, a balance between inflammatory reaction and its suppression seems to be crucial for scaffold-based vascular conduits to avoid intimal hyperplasia and stenosis. Moreover, polymer breakdown products were recognized as promoters of SMC de-differentiation, migration and proliferation, accompanied by extensive ECM synthesis and fibrosis. 27 Therefore, the LifeLine™, which is devoid of any synthetic materials, seems to undergo a remodeling mechanism that is not based on the inflammation-associated fibrosis and may be less prone to graft narrowing, as suggested by our clinical results. Repopulation of an ECM-based graft with functional SMCs could be critical for long-term in vivo functionality. 28 While SMCs are essential in regulating vessel wall contractility, tone and compliance, proliferation of SMA-positive myofibroblasts may exert a negative effect with vessel graft stenosis and consequently shunt thrombosis. Tillman et al. observed extrinsic (from surrounding tissues) infiltration and repopulation of the decellularized arterial scaffolds in arteriovenous fistula in sheep. 29 Of high significance are results presented by Lawson et al. who observed extrinsic migration of the SMA-positive cells into the biodegradable scaffold-based acellular TEVG (Humacyte Inc.) used as arteriovenous access in hemodialysis patients. 30 Recently published post-implantation histological analysis of these grafts revealed increasing contribution of the SMA-positive cells in the media over time. 31 While the same authors did not observe any true-aneurysmal graft dilation, they noted a low 12-month primary patency rate (28%) despite antiplatelet treatment and graft in vivo endothelialization. This, unlike our observations suggests a high rate of the stenotic complications, albeit detailed data were not presented. 30 Our observations of the LifeLine™ internal layer repopulation with SMCs, in line with their absence in adventitia may suggest that repopulation with SMC progenitor cells from the vessel lumen had taken place. It would be consistent with work by Enomoto et al. who proved repopulation of scaffold-free fibroin TEVGs with bone marrow-derived progenitor cells that organized into a media-like smooth muscle layer. 32 Nevertheless, we cannot completely exclude migration and differentiation of the adventitial fibroblasts toward myofibrablasts and myocytes. Overall, different clinical outcomes of the scaffold free autologous LifeLine™ hemodialysis access and scaffold based acellular TEVG shunts (like Humacyte graft) may be affected by a different process of graft remodeling and repopulation in vivo. In the latter case it may be highly influenced by presence of the synthetic scaffold and induced detrimental foreign body inflammatory reaction, whereas our results suggest that the remodeling of LifeLine™ graft resembles that of the native vein used as an arteriovenous fistula, with similar response to hemodynamic conditions and recurrent injuries.
The AVF application is undoubtedly a very demanding one for a TEVG because of its non-physiological blood flow and repeated mechanical injuries. Our results prove that this TEVG not only can show long-term functionality as a basic vascular conduit, but it can also serve as an efficient sole vascular access for hemodialysis without any infectious events. The LifeLine™ intervention and event rate was comparable to AVF and less than half that of sAVG- or CVC-related incidences (US ESRD population). Among five patients chronically dialysed with the LifeLine™ three were treated with low dose aspirin without thrombotic complications, whereas one patient whose shunt thrombosed was untreated. It may point to the beneficial effect of antiplatelet prophylaxis in LifeLine™ dependent ESRD patients. This treatment was already confirmed to significantly reduce the AVF thrombosis. 11 Consequently, general maintenance costs of the LifeLine™ access and matured AVF seem to be comparable. Medicare outlays of the ESRD treatment in case of the sAVG and CVC are 24% and 35% higher than AVF-related outlays. 5 Thus, the $15k approximate cost of an autologous LifeLine™ graft treatment could be balanced within first 1 year of treatment. This is more pronounced with presented comparison of event rate and related expenditures before and during LifeLine™ application. Moreover, analysis presented by Rosas and Feldman shows that the “fistula first” strategy is associated with higher overall costs as compared to application of the sAVG as the first access. 33 It is highly influenced by the AVF long and uncertain maturation process with high risk of failure, what also influences adversely long term access related costs. 34 Similarly, costs for the functioning LifeLine™ shunts should also be included for patients who lost access within the “maturation period” due to access-related reasons, which increases global expenditures of TEVG-based treatment by approximately 30%. Results from a US national study revealed that only 55% of arteriovenous fistulas were used within 4 months of placement. 35 In our cohort, this number reached 66% and only one graft thrombosed prior to canulations, whereas two other patent shunts were abandoned for safety reasons (immune reaction and surgical wound complication). In this light, a TEVG, which does not need a long maturation period combines advantages of the AVF (no infections and low rate of thrombosis) and sAVG (initial diameter and volumetric flow appropriate for immediate hemodialyses), without their main drawbacks. 36
Promising results from Shin’oka et al. 37 confirmed successful application of an autologous living scaffold-based TEVG as the cavopulmonary bypass. These patients were the first humans to receive a TEVG and this is the only other living strategy to date, although this was in the low-pressure vascular environment of the pulmonary circulation. Despite our preliminary results having been already published 10 years ago only one other—non-living, scaffold-based approach (Humacyte trial) has reached clinical trial in the arterial circulation.30,38
The 6-month fabrication process, as well as the need for skin and vein biopsy, are significant limitations to the clinical application of the autologous LifeLine™ graft. However, good clinical outcomes and preliminary cost estimations support the use of this autologous TEVG for advanced ESRD patients after multiple vascular access failures and infection events. This approach could be integrated in a comprehensive patient treatment plan in healthcare systems that promote a more holistic approach. Finally, an allogenic approach was shown to be feasible and would offer a more commercially feasible route. 39
Supplemental Material
sj-docx-10-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-10-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-11-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-11-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-12-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-12-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-13-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-13-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-14-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-14-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-15-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-15-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-16-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-16-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-5-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-5-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-6-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-6-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-7-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-7-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-8-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-8-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-docx-9-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-docx-9-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-pdf-1-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-pdf-1-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-pdf-2-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-pdf-2-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-pdf-3-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-pdf-3-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Supplemental Material
sj-pdf-4-jva-10.1177_11297298221095994 – Supplemental material for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access
Supplemental material, sj-pdf-4-jva-10.1177_11297298221095994 for Long-term results of autologous scaffold-free tissue-engineered vascular graft for hemodialysis access by Wojciech Wystrychowski, Sergio A Garrido, Alicia Marini, Nathalie Dusserre, Sam Radochonski, Krzysztof Zagalski, Jorge Antonelli, Manuel Canalis, Andrea Sammartino, Zbigniew Darocha, Ryszard Baczyński, Tomasz Cierniak, Heinz Regele, Luis M de la Fuente, Lech Cierpka, Todd N McAllister and Nicolas L’Heureux in The Journal of Vascular Access
Footnotes
Author contributions
WW analyzed and interpreted data, wrote the manuscript; ND managed grafts production; SR collected data, HR performed histology; TNM managed all clinical trials, critically revised manuscript; NLH invented sheet-based tissue engineering, manufactured grafts, interpreted data, substantially revised manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have disclosed potential conflicts of interest. TNM was CEO, NLH was CSO and both were cofounders and significant stockholders of Cytograft Tissue Engineering (the company no longer exists). ND and SR were employees of Cytograft.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Cytograft Tissue Engineering
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
