Abstract
Background:
Creation and maintenance of dialysis vascular access (VA) is a major component of healthcare resource utilization and cost for patients newly started on hemodialysis (HD). Different VA format arises due to patient acceptance of anticipatory care versus late preparation, and clinical characteristics. This study reviews the clinical journey and resource utilization required for different VA formats in the first year of HD.
Method:
Data of patients newly commenced on HD between July 2015 and June 2016 were reviewed. Patients were grouped by their VA format: (A) pre-emptive surgically created VA (SCVA), (B) tunneled central venous catheter (CVC) followed by SCVA creation, (C) long-term tunneled CVC only. Clinical events, number of investigations and procedures, hospital admissions, and incurred costs of the three groups were compared.
Results:
In the multivariable analysis, the cost incurred by the group A patients had no significant difference to that incurred in the group B patients (p = 0.08), while the cost of group C is significantly lower (p < 0.001). Both the 62.7% of group A with successful SCVA who avoided tunneled CVC usage, and those with a functionally matured SCVA in group B (66.1%), used fewer healthcare resources and incurred less cost for their access compared to those did not (p = 0.01, p = 0.02, respectively) during the first year of HD.
Conclusion:
With comparable cost, a pre-emptive approach enables avoidance of tunneled CVC. Tunneled CVC only access format incurred lower cost and is suitable for carefully selected patients. Successful maturation of SCVA greatly affects patients’ clinical journey and healthcare cost.
Introduction
The prevalence of end-stage kidney disease (ESKD) is increasing worldwide.1–3 The ESKD population requires long-term kidney replacement therapy (KRT). One of the KRT modalities is hemodialysis (HD). A functioning vascular access (VA) is essential for patients on HD. Vascular access can be in the form of a tunneled central venous catheter (CVC), or surgically created vascular access (SCVA)—arteriovenous fistula (AVF) and arteriovenous fistula graft (AVG). 4 Each type of VA has its benefits and risks. Different VA formats in the first year of HD arise due to a number of factors such as patient demographics, comorbidities, life expectancy, and urgency of HD treatment.4–7 The common formats of VA include: (i) pre-emptive SCVA before HD initiation, (ii) HD initiation with tunneled CVC followed by SCVA creation, and (iii) HD initiation with long-term tunneled CVC only.
Earlier studies reviewed the clinical course of patients on a specific VA type or format of HD vascular access.4,8–10 There are few studies comparing the clinical journey, resource utilization, and healthcare economic burden by various VA formats. 11 This information improves decision making as dialysis demand is increasing. In this study, we retrospectively reviewed consecutive newly commenced HD patients by the three different VA formats and compared VA-related outcomes, clinical journey together with healthcare resources consumption in the first year of HD.
Methods
Study population
Study data of consecutive ESKD patients who initiated long-term HD between 1 July 2015 and 30 June 2016 in a tertiary hospital in Singapore was extracted from the hospital electronic medical records, and collected using REDCap (Research Electronic Data Capture). 12 Long-term HD is defined as patients who have been undergoing regular HD for more than 90 days. Patients lost to follow-up within the first year of HD initiation were excluded (n = 3). The period of data collection was from the initiation of HD up to 1-year post-HD initiation, or till the date of conversion to other KRT modality, or till the date of death. For patients with pre-emptive SCVA before the initiation of HD, VA-related clinical data were collected from the date of SCVA creation. This study was approved by the institutional ethics committee and complies with the Human Biomedical Research Act (HBRA) regulations.
Three groups of patients were identified based on their VA format namely: (A) pre-emptive SCVA before commencement of HD; (B) HD initiated with tunneled CVC first, followed by SCVA creation; (C) HD with tunneled CVC only.
Study data
Data collected includes demography, ambulatory status, comorbidities, etiology of ESKD and all VA-related investigations (venous and arterial duplex mapping, AVF/AVG duplex studies), surgical and interventional procedures, VA-related complications and its related hospital admissions, and survival status. Surgical and interventional radiologic procedures include insertion or exchange of CVC, surgical creation of AVF or AVG, endovascular procedure or open surgery to assist maturation of SCVA, or salvage of failing or thrombosed accesses. VA-related complications and hospital admissions include infections or blockage of tunneled CVC, bleeding and hematoma, steal syndrome, thrombosis, and infection. Functionally mature SCVA was defined as tunneled CVC removal after HD performed using the AVF or AVG.
The costs of various investigations and procedures were estimated based on the mean cost incurred by the historical patients in the institution (Supplemental Table 1). The cost of in-patient hospital stay treating VA-related complications was calculated by multiplying days of hospital stay with the cost of the subsidized class. The total VA-related healthcare cost is calculated based on the sum of above-mentioned costs incurred by the patient in the first year after initiation of HD. All costs are expressed in Singapore dollars (S$). Cost incurred from inpatient hemodialysis, antibiotics and other medication, peritoneal dialysis (PD) catheter insertion or kidney transplantation were not included.
Outcomes
The aims of this study were: (i) to explore the clinical journey of patients in the three groups during the first year of hemodialysis; (ii) to compare resource utilization and VA-related costs in the first year of HD; (iii) to compare the VA-related costs within subgroups of Group A (successfully initiated HD with pre-emptive SCVA vs failed SCVA) and Group B (functionally mature SCVA vs failed first SCVA).
Statistical analysis
All statistical data were analyzed using R (version 4.1.2). Patient demographics, comorbidities, resource utilization, and costs were compared among the three VA format groups. Categorical outcomes are reported as frequency and proportions, and compared with the Fisher exact test. Skewed continuous variables are reported as median accompanied by first quartile and third quartile, and compared using Kruskal-Wallis test (among three groups) or Wilcoxon Rank sum test (among two groups). We used a multivariable generalized linear model with a gamma link function to assess the impact of VA formats on VA-related costs as the distribution of costs are positively skewed. Covariates were included if p < 0.05 in univariate analysis. Statistical significance was taken at p ⩽ 0.05.
Results
Over the study period, 145 ESKD patients initiated HD. Sixty-seven (46.2%) patients had pre-emptive SCVA (Group A). The median time of pre-emptive SCVA before HD initiation was 253 days (first–third quartile:101–514 days). Sixty-two (42.8%) patients started HD using tunneled CVC, and then had SCVA subsequently (Group B). Sixteen patients (11.0%) initiated and maintained on HD with only tunneled CVC (Group C).
The demographic data and clinical characteristics of the three groups of patients are in Table 1. The prevalence of diabetes (p = 0.03) and hypertension (p = 0.01) are significantly higher in Group A than the other two groups. Among the 16 patients in Group C, five patients had hypotension episodes during the first HD session (three of them had left ventricular ejection fraction <30%), five had active malignancies on palliative care, one refused SCVA because of needle phobia, two were considering PD, and three were considering kidney transplantation.
Patients’ demographics and clinical characteristics.
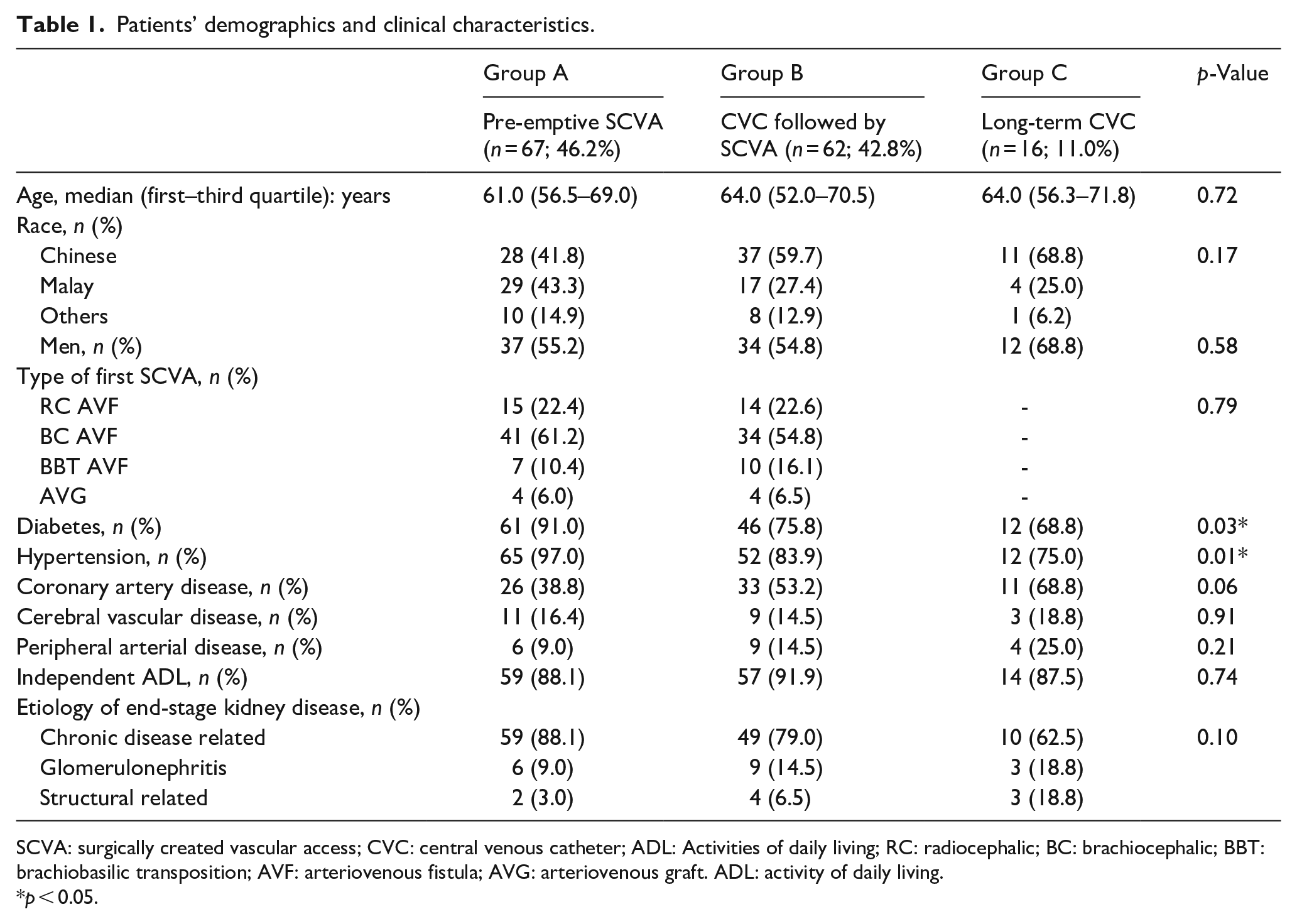
SCVA: surgically created vascular access; CVC: central venous catheter; ADL: Activities of daily living; RC: radiocephalic; BC: brachiocephalic; BBT: brachiobasilic transposition; AVF: arteriovenous fistula; AVG: arteriovenous graft. ADL: activity of daily living.
p < 0.05.
Table 2 shows the main outcomes and resource utilization. Group C had higher mortality (37.5%; p < 0.01) and conversion to other KRT modalities (31.2%; p < 0.001) compared to the other two groups. Between Group A and B, pre-emptive SCVA group had a higher percentage of functional successful SCVA in the year after HD initiation (p = 0.01), and a higher percentage needing VA salvage procedure(s) (p < 0.001). Group B had a higher percentage of patients (27.4% vs 11.9%) requiring multiple tunneled CVC insertions. Figure 1 illustrates the number of tunneled CVC procedures required for Groups A, B, and C. 62.7% of Group A with pre-emptive SCVA avoided tunneled CVC insertions completely. Between Groups A and B, there was no difference in the proportion of patients who underwent multiple VA creation surgeries.
Outcomes and healthcare resource utilization within 1 year of initiation of hemodialysis.
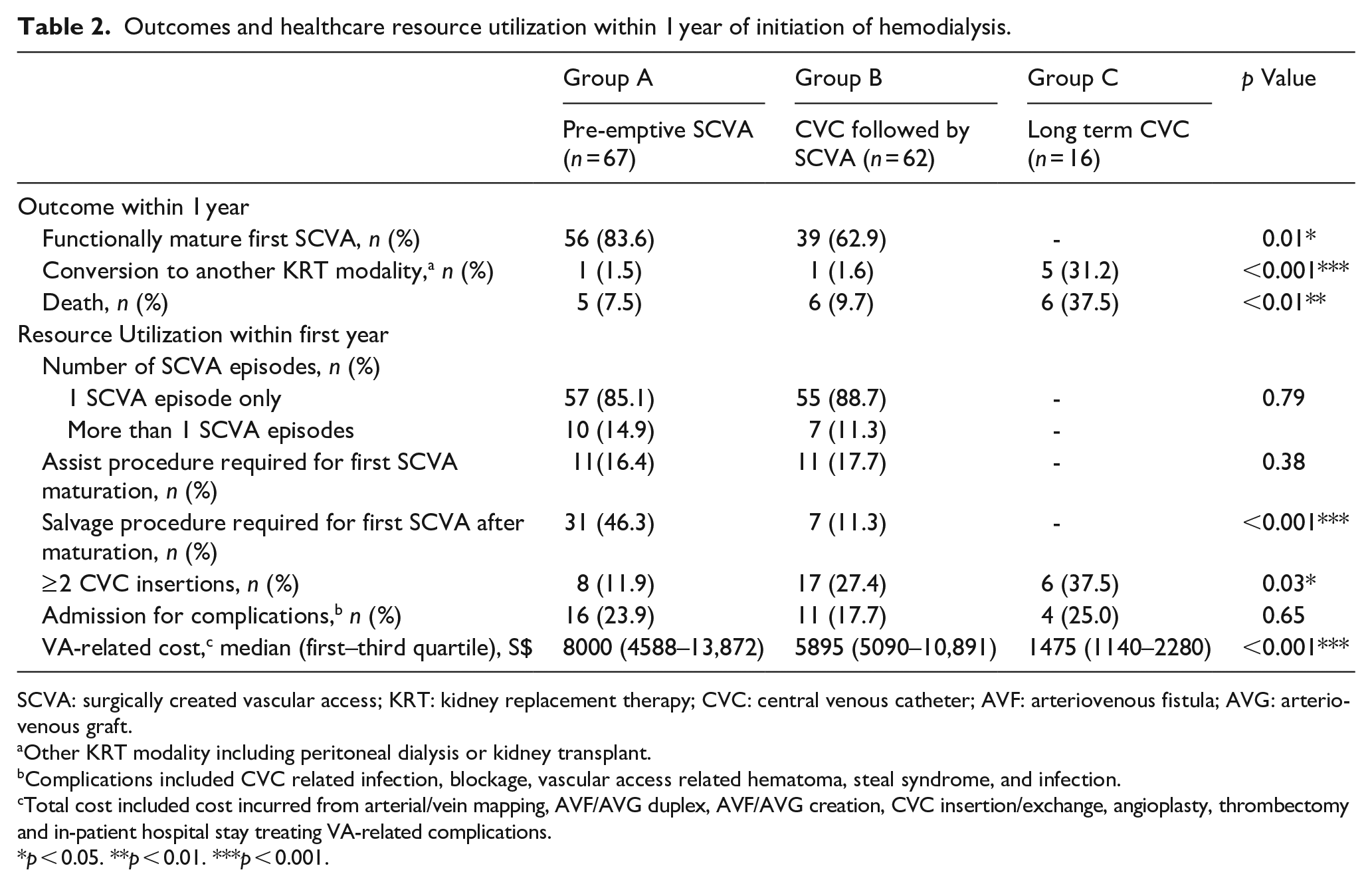
SCVA: surgically created vascular access; KRT: kidney replacement therapy; CVC: central venous catheter; AVF: arteriovenous fistula; AVG: arteriovenous graft.
Other KRT modality including peritoneal dialysis or kidney transplant.
Complications included CVC related infection, blockage, vascular access related hematoma, steal syndrome, and infection.
Total cost included cost incurred from arterial/vein mapping, AVF/AVG duplex, AVF/AVG creation, CVC insertion/exchange, angioplasty, thrombectomy and in-patient hospital stay treating VA-related complications.
p < 0.05. **p < 0.01. ***p < 0.001.
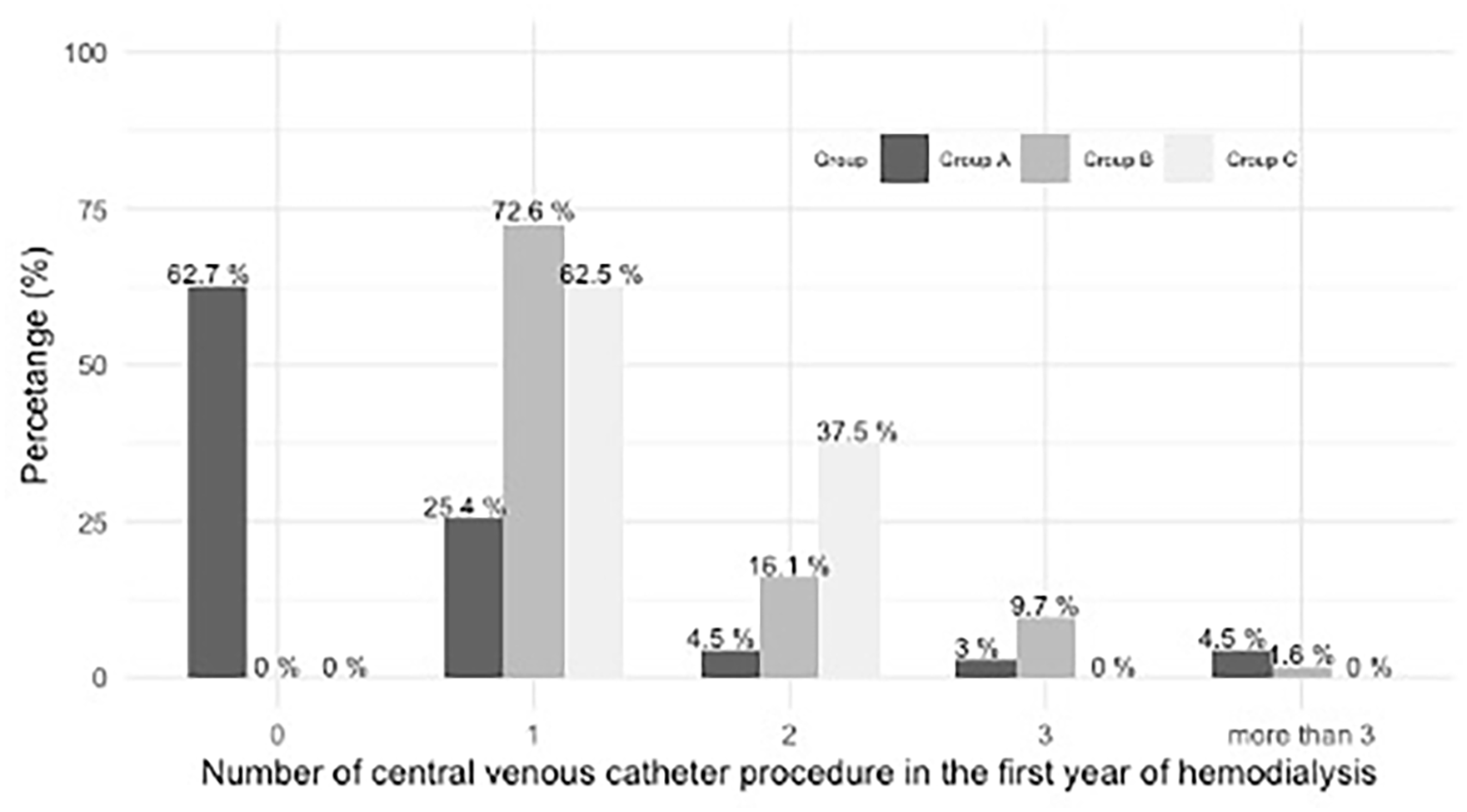
Percentage of patients underwent tunneled central venous catheter procedures in Group A, B, and C. Group A: pre-emptive vascular access creation before hemodialysis initiation; Group B: hemodialysis initiated with tunneled central venous catheter first, followed by vascular access creation; Group C: tunneled central venous catheter only for hemodialysis.
The median vascular access related healthcare cost for Group A was highest at S$8000 (first–third quartile: $4588–$13,872), followed by S$5895 (first–third quartile: $5090–$10,891) for Group B, and lowest for Group C at S$1475 (first–third quartile: $1140–$2280). In the multivariable analysis (details shown in Supplemental Table 2) using gender, race, type of vascular access, conversion to other KRT modality and mortality status as covariates for adjustment, with reference to Group A, the cost of Group B shows no significant difference (β = −0.18, 95% CI: −0.37 to 0.02; p = 0.08), while group C patients incurred lower cost (β = −1.55, 95% CI: −1.96 to −1.13; p < 0.001).
The detail clinical journey including the investigations, VA-related procedures, hospital stays, clinical outcomes and the corresponding costs of patients in Group A, B, and C are shown in Figures 2 to 4.
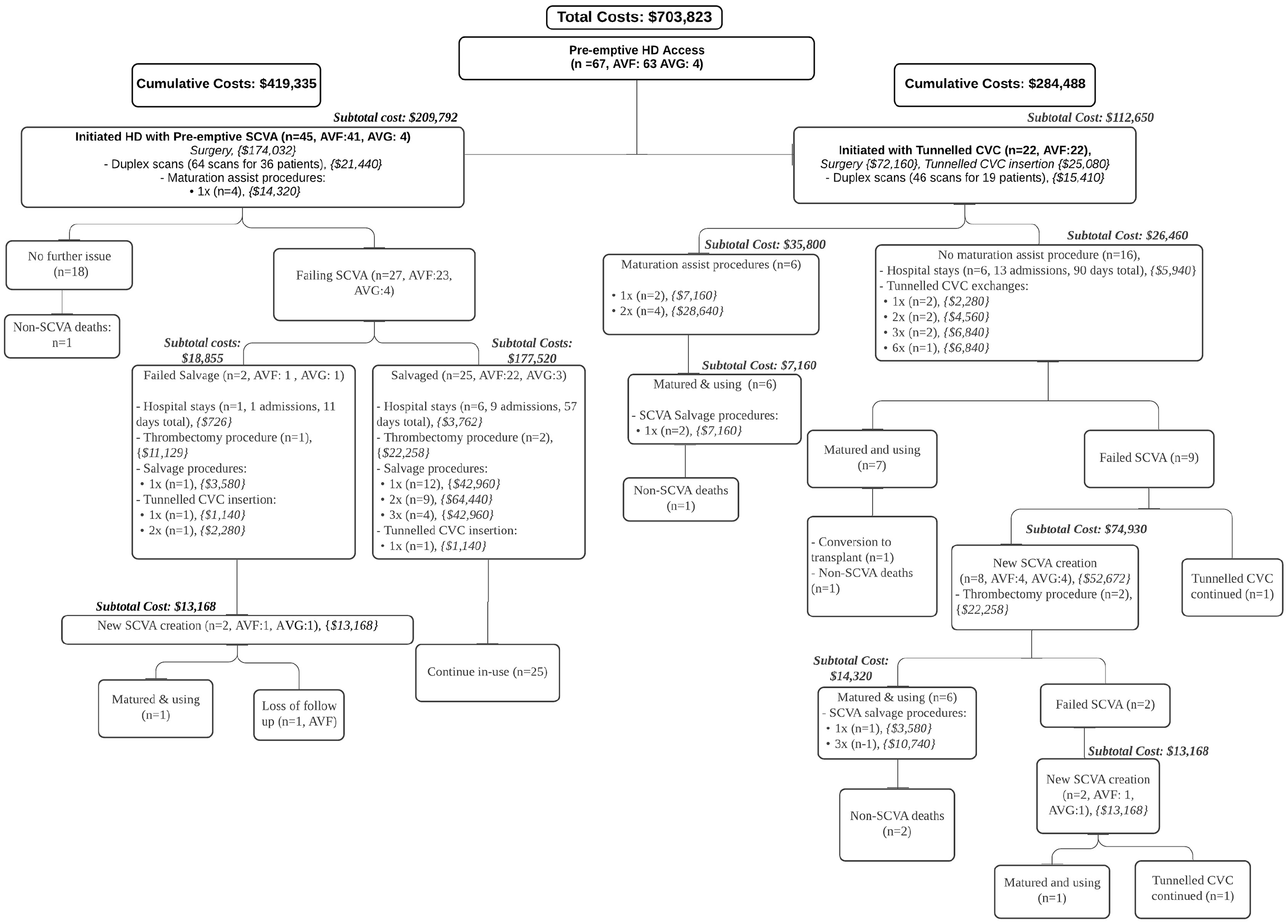
Clinical journey of Group A patients (pre-emptive vascular access).
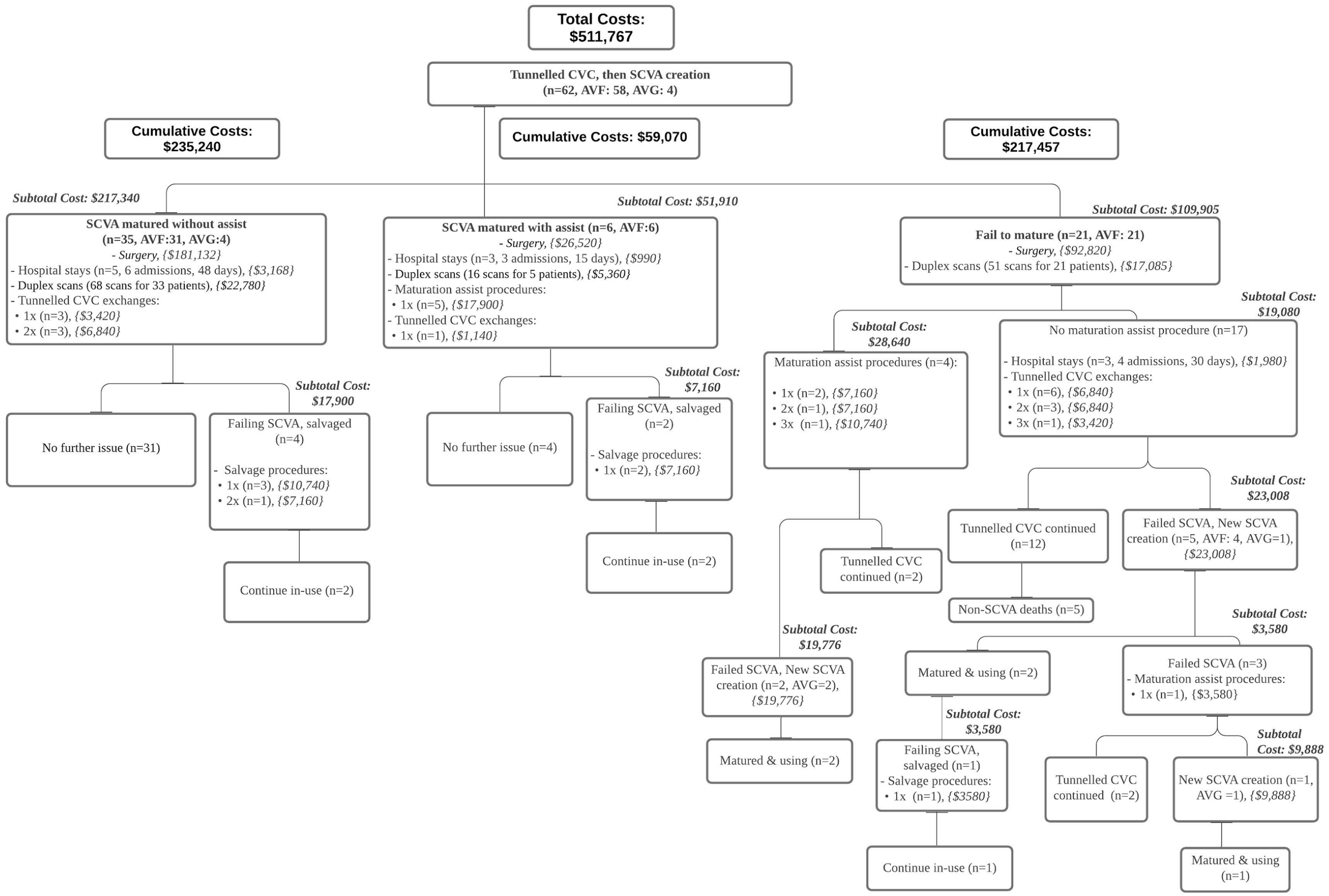
Clinical journey of Group B patients (initiated hemodialysis with central venous catheter).
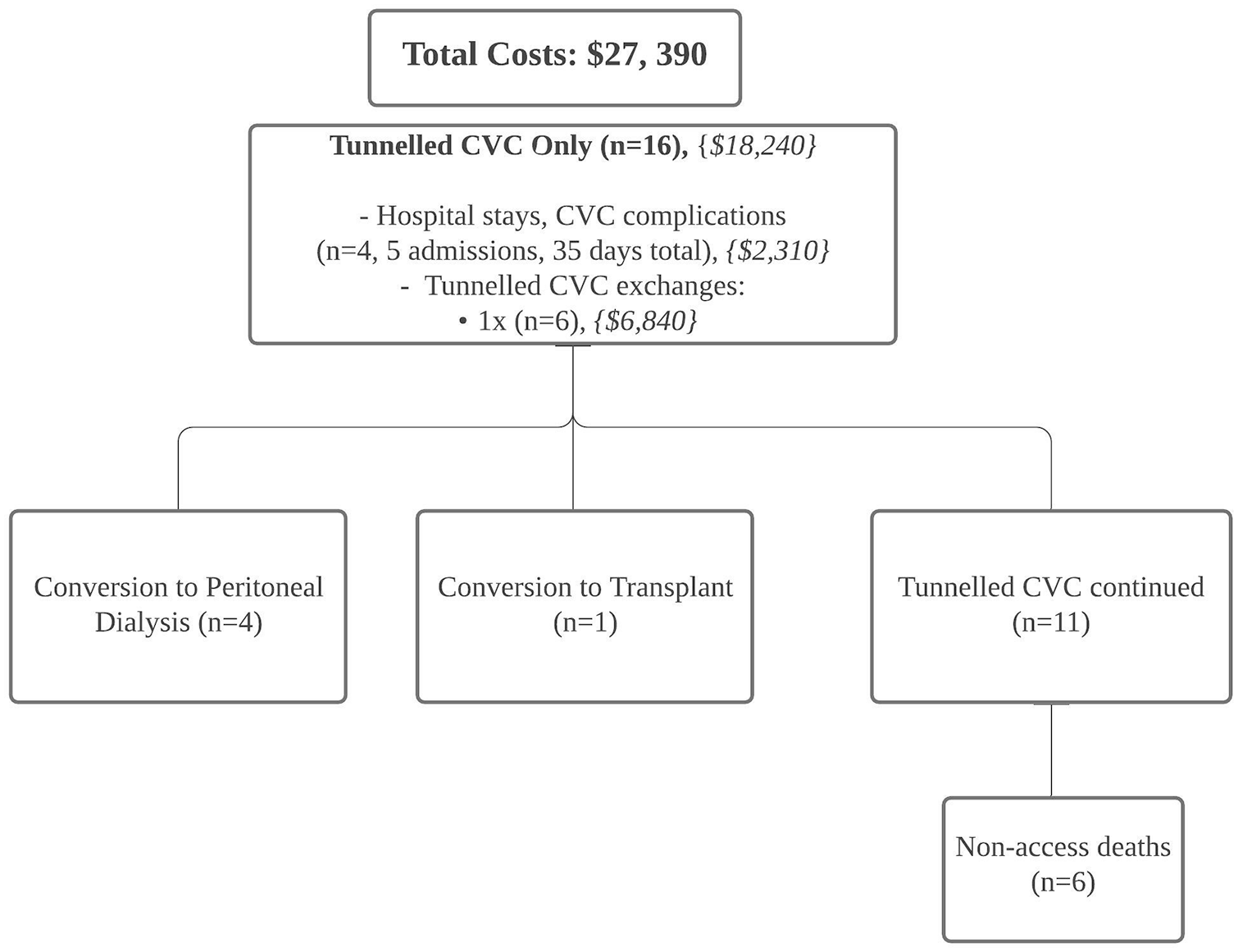
Clinical journey of Group C patients (hemodialysis with central venous catheter only).
Forty-five (67.2%) Group A patients initiated HD with a functionally successful pre-emptive SCVA (41 AVF and 4 AVG) (Figure 2). Table 3 compares subgroups in Group A: patients who successfully initiated HD with pre-emptive SCVA (Pre-emptive-S), and patients who failed to initiate HD with a pre-emptive SCVA (Pre-emptive-F). The Pre-emptive-S subgroup required fewer additional SCVA creation procedures (p < 0.01), had less assisted maturation procedures (p < 0.01), and incurred lower costs (p = 0.01) compared to those in the Pre-emptive-F subgroup (Figure 5).
Comparison of two subgroups in Group A: initiated hemodialysis with pre-emptive access (Pre-emptive-S) versus those with tunneled central venous catheter (Pre-emptive-F).
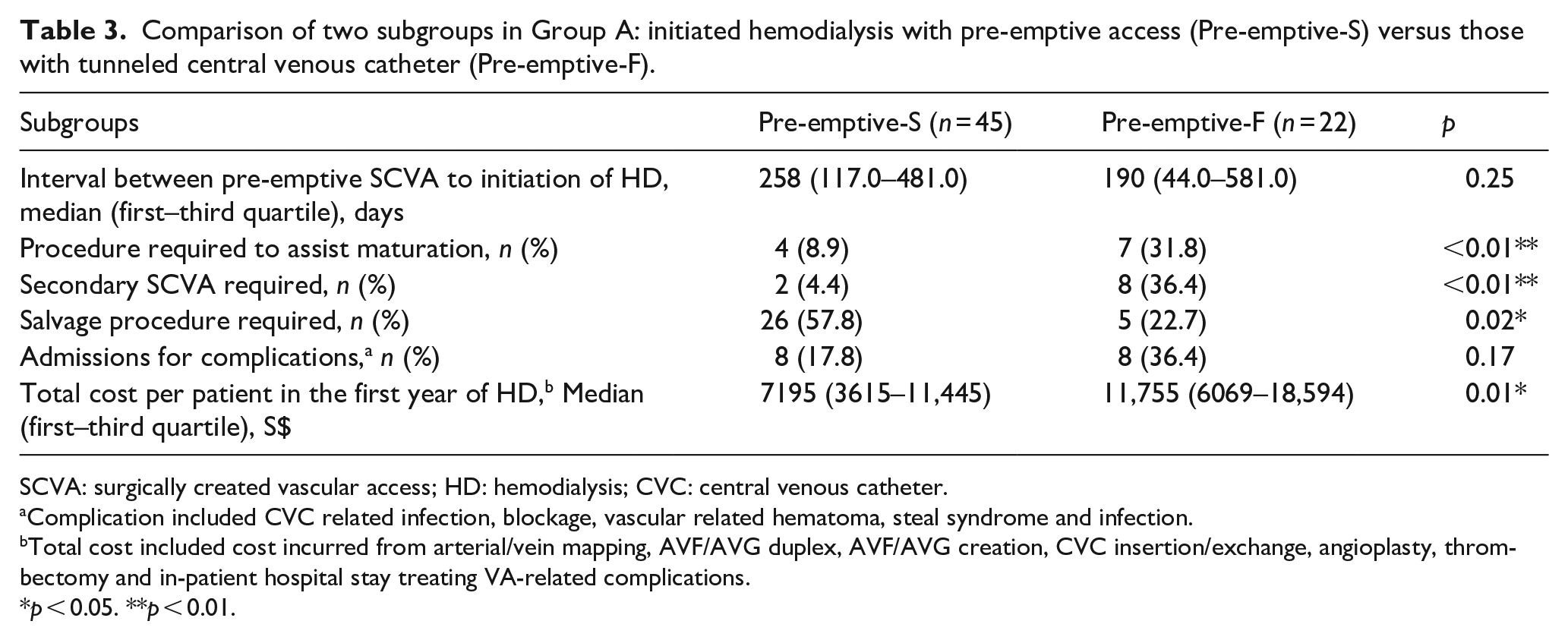
SCVA: surgically created vascular access; HD: hemodialysis; CVC: central venous catheter.
Complication included CVC related infection, blockage, vascular related hematoma, steal syndrome and infection.
Total cost included cost incurred from arterial/vein mapping, AVF/AVG duplex, AVF/AVG creation, CVC insertion/exchange, angioplasty, thrombectomy and in-patient hospital stay treating VA-related complications.
p < 0.05. **p < 0.01.
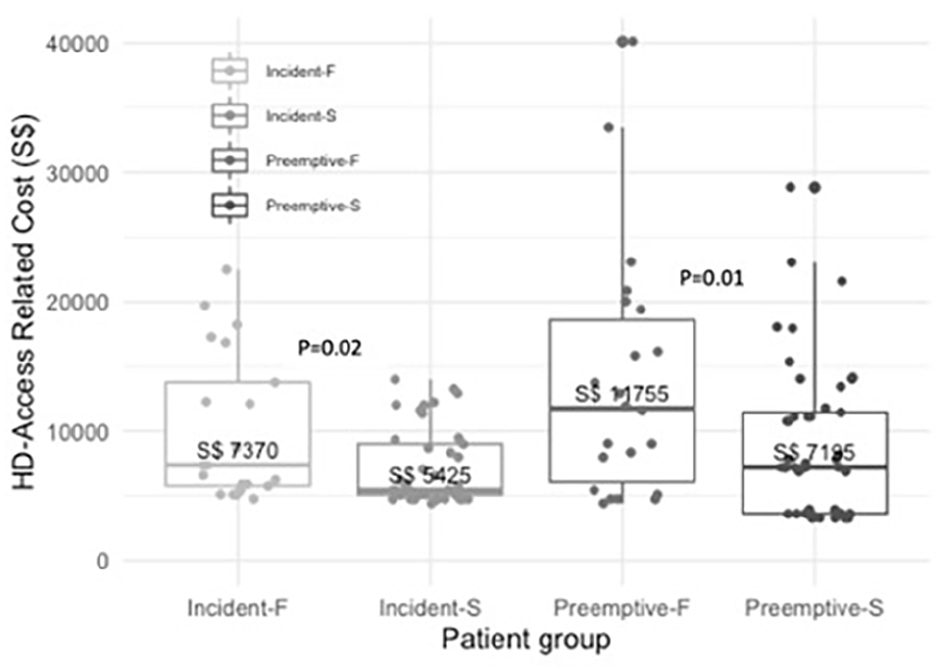
Hemodialysis vascular access-related healthcare costs among four groups (Incident-S; Incident-F; Pre-emptive-S; Pre-emptive-F). Wilcoxon Rank sum Test was used to compare HD access related cost (Incident-F vs Incident-S, Preemptive-F vs Preemptive-S).
Forty-one (66.1%) Group B patients achieved functional SCVA success, six of whom required procedures to assist in maturation (Figure 3). Twenty-one (33.9%) SCVA failed to mature by within the first year of HD initiation despite maturation assist procedures performed on four of them. Table 4 compares the two subgroups in Group B: 41 (66.1%) patients with functional successful primary SCVA with or without assist procedure (Incident-S), and 21 (33.9%) patients without functional successful primary SCVA with or without assist procedures (Incident-F). Incident-F subgroup incurred higher costs (p = 0.02, Figure 5), needed more tunneled CVC exchanges when compared to Incident-S (p = 0.04).
Comparison of two subgroups in Group B: first vascular access mature (Incident-S) versus first vascular access failed (Incident-F).

SCVA: surgically created vascular access; HD: hemodialysis; CVC: central venous catheter.
Complication included CVC related infection, blockage, vascular related hematoma, steal syndrome and infection.
Total cost included cost incurred from arterial/vein mapping, AVF/AVG duplex, AVF/AVG creation, CVC insertion/exchange, angioplasty, thrombectomy and in-patient hospital stay treating VA-related complications.
p < 0.05.
Discussion
The current study revealed the real-world clinical journal and healthcare resources consumptions of newly commenced HD patients on their dialysis access over the three VA formats, which was never being documented or reported in previous studies. The VA-relate cost may attribute to nearly one third 13 of the total healthcare cost for patients on long term hemodialysis. The current study presents the granularity of what modality of healthcare resources and how they were consumed along the clinical journey of incident hemodialysis patients. This detailed information provides insight for clinicians assigning, planning and performing VA for ESKD patients. Although the median cost of Group A was higher than Group B, the multivariable analysis shows that Group A and Group B incurred comparable VA-related costs. A significant proportion of Group A (62.7%) patients were able to totally avoid a tunneled CVC. Tunneled CVC insertion risks injury to the central veins and might induce long-term central vein obstruction.14,15 Pre-emptive VA strategy (Group A) has the beneficial effect of minimizing tunneled CVC usage compared to strategy of initial tunneled CVC followed by SCVA creation (Group B) without incurring extra costs.
The VA-related cost is lowest in Group C among this cohort. Nevertheless, Group C patients had higher mortality within the first year of HD initiation (37.5%) and highest conversion to other KRT modalities (31.2%). The higher mortality within first year of HD of Group C likely relating to poorer pre-morbid condition 16 where 5/16 patients in this group had hypotension during the first HD session, 5/16 had active malignancies on palliative care. This illustrates the clinical characteristics of patients who chose tunneled CVC only approach likely to be very different from those of Group A and B. On the other hand, because of the higher mortality and conversion to other KRT rate, the average time of Group C patients on hemodialysis within the study period was shorter than other two groups. The cost of conversion to other KRT was also not included in the study. This may induce bias to the VA-related cost calculation. In this study, Group C had five hospital admissions (35 hospital-days) occurred in four patients, and 37.5% of them required additional CVC exchange or re-insertion. The clinical and economic burden of CVC infections and exchanges may vary in different HD programs due to catheter care protocols. Prior studies suggested that the infectious complications of long-term tunneled CVC is higher than AVF and AVG,17–19 with wide variation in the reported incident rates (0.84–5.1 episodes per 1000 catheter-days) for catheter-related bloodstream infections.14,20,21 Furthermore, the longer-term adverse effects of tunneled CVC including infection and central vein obstruction may occur later.22–24 Therefore, even though Group C bore the lowest VA-related costs, a tunneled CVC only strategy should not be advocated to the majority ESKD patients. It is however, a cost-effective VA format with relatively simple clinical journey for patients with short life-expectancy or those likely to convert to PD or kidney transplant soon after dialysis initiation.
Many considered SCVA creation is a straight-forward clinical task, but it can be challenging and complex in reality. In the United States, up to one-third of AVF created were not used within the first year of creation. 25 The flowcharts (Figures 2 and 3) of the current study provides a detail insight into the clinical journey of the patients which varied from straight forward to very complex. In Group A, those with functionally successful pre-emptive SCVA (Pre-emptive-S) underwent significantly less maturation assist procedures and less secondary SCVA creation procedures than those not (Pre-emptive-F). It is congruent with Crooks et al.’s 26 finding suggesting that patients commenced HD with optimal vascular access had lower utilization, compared with those start with non-optimal access. In Group B, those with functionally successful SCVA within study period (Incident-S) underwent significantly less tunneled CVC exchange/reinsertion compares to those not (Incident-F). Furthermore, in both Groups A and B, patients with functionally unsuccessful first SCVA incurred higher costs (Figure 5) than those with successful ones. These illustrate that the success or failure of SCVA imposes drastic difference in the clinical journey of HD patients. In a United Kingdom cohort study, 11 a third of patients started HD with CVC had previously failed SCVA attempts, which contributed to higher VA-related costs. The authors suggested a well-functioning AVF is associated with the lowest economic burden. In contrast, failed AVF is associated with the highest costs. 11 Therefore, a high first SCVA success rate will result in substantial saving in healthcare resources and costs. In this study, 66% (66.1%) of Group B patients had a functionally mature SCVA within 12 months of starting dialysis. Based on our estimation, on average, one incident patient with a functionally matured first SCVA incurred S$2926 less compared to those who had a failed SCVA. In 2016, there were 1327 new patients who ever-started HD in Singapore. 1 Assuming the same percentage of patients required SCVA, and the maturation rate can be improved from current 66.1% to 90%, the potential VA-related economic savings in Singapore will be about S$0.83 million annually. With increasing prevalence of dialysis dependent population worldwide, and VA related cost contribute to nearly one third 13 of total hemodialysis related healthcare cost, a high first SCVA success rate will have great impact on the cost saving. In addition to higher costs, failed initial VA creation increases the duration of tunneled CVC use and the risk of central vein obstruction in longer term.
The challenge therefore is how to ensure a high SCVA success rate? The percentage of SCVA failure or prolonged failure-to-mature is high, ranges from 23% to 36.2%.25,27 Multiple risk factors affect AVF maturation, including gender, age, target vein size, target arterial size, diabetes, peripheral arterial disease,28–30and surgeons’ characteristics and experience. 31 Careful site selection, 32 pre-operative ultrasound assessment of vessels,33–35 intra-operative flow measurement, 36 ultrasound-guided cannulation in early matured SCVA,37,38 and SCVA maturation tracking system 39 are some of the ways to enhance SCVA usage and shorten CVC-dependent time and reduce healthcare expenditure. Appropriate use of AVG in patients with unfavorable venous asset might also minimize the percentage of failure-to-mature AVFs. Latest studies showed no definitive evidence of inferiority of AVGs when compared to AVFs.40–42 Effective training of access surgeons on clinical decision-making and skill should be a focus of the specialty.36,43 More effort should be injected into the healthcare system to monitor and enhance a high SCVA success rate.
The wide variation of vascular access clinical journeys might not be expected by the advance chronic kidney disease patients before their commencement of dialysis. A qualitative study revealed that patients often expressed ongoing and overwhelming fear over failure and complication of HD vascular access. 44 This study provides data for clinicians to explain the possible clinical journeys during the initial counseling sessions of pre-dialysis patients, and could potentially promote shared decision-making and avoid subsequent disappointment. A thorough counseling facilitates patients to make an informed choice, and be more ready to participate in monitoring and managing their SCVA.45,46 Early involvement of nephrologist to review and monitor advance chronic kidney disease patients provide better opportunity to make an optimal VA plan. 47
This study has its limitation. Firstly, the resource utilization and costs indirectly related to vascular access may be under-estimated. Cost on inpatient HD sessions, antibiotics for infections, dressing of wounds cannot be accurately retrieved. Secondly, the duration of hospital stay used in calculating the VA-related costs may also include hospitalization due to other medical problems concomitant to the VA-related issues. Thirdly, some of the healthcare costs like hospital stay is heavily subsidized by the government in the Singapore setting, which under-represent the actual financial burden of hospitalizations treating VA-related complications. Fourthly, this study included only patients commenced HD and through their first year of dialysis. Cost of pre-emptive SCVA created in patients who never commence HD and the associated VA maintenance cost was not captured. This may lead to under-estimate of the healthcare cost relating to the pre-emptive SCVA strategy. Fifthly, the current study only evaluated the VA-related costs in the first year of HD initiation. Some issues relating to HD vascular access may only occur after a longer period of time on HD. The retrospective nature, small sample size and short follow-up period may limit its generalizability. Lastly, although cost is worthy of attention, the overall format of HD VA for ESKD patients cannot be determined solely based on the short period cost.
In conclusion, among the three HD VA formats, pre-emptive SCVA creation shows benefits of reducing dependence on CVC, compared to the format of initial tunneled CVC followed by SCVA creation. Whereas a CVC only strategy is advisable in carefully selected patient groups with short life expectancy or planning to switch to other KRT modality. Importantly, the clinical implications are that, shared-decision should be made after patients have thorough understanding of the potential clinical journey they might go through with various HD VA format. Doctors should strive to establish a successful first SCVA. This will reduce the overall costs on HD VA, CVC usage duration, and the number of additional procedures. To achieve this, the whole process of vessel selection, SCVA surgery, and maturation monitoring have to be optimized.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298221095769 – Supplemental material for The clinical journey and healthcare resources required for dialysis access of end-stage kidney disease patients during their first year of hemodialysis
Supplemental material, sj-pdf-1-jva-10.1177_11297298221095769 for The clinical journey and healthcare resources required for dialysis access of end-stage kidney disease patients during their first year of hemodialysis by Pei Ho, Nur Nabila Farhana Binte Taufiq Chong Ah Hoo, Yi Xin Cheng, Lingyan Meng, Darryl Chai Min Shen, Boon Wee Teo, Valerie Ma and Carol Anne Hargreaves in The Journal of Vascular Access
Footnotes
Author contributions
P.H. and C.A.H. designed the present study. P.H., N.N.F.B.T., L.Y.M. drafted first copy of manuscript. V.M., D.M.S.C., N.N.F.B.T. were responsible for data acquisition. L.Y.M. and Y.X.C. were responsible for data analysis and/or interpretation. P.H., B.W.T., C.A.H. were responsible for supervision and mentorship. All authors approved the final copy of manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the institutional ethics committee (DSRB 2019/01003).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
