Abstract
Introduction:
Central venous occlusive disease (CVOD) is a complication that can occur in patients with end-stage renal disease who are receiving hemodialysis. When CVOD develops, patients often require multiple re-interventions to maintain their dialysis access. CVOD can be treated by various strategies such as balloon angioplasty, stenting, lower limb or extra-anatomical grafts, hybrid grafts or surgical bypasses such as right atrial (RA). In this systematic review, we aim to evaluate the indications, technical aspects, and outcomes after RA bypass grafting for the treatment of CVOD in hemodialysis patients.
Methods:
A systematic and comprehensive literature search was conducted using various electronic databases. We included articles that reported described and reported outcomes of RA bypass grafting for the treatment of CVOD in hemodialysis patients. A narrative review of the indications and technical aspects of RA bypass grafting was performed. We also pooled and reported the primary patency, secondary patency, postoperative complications, and 30-day mortality of RA bypass grafting.
Results:
A total of 21 studies with 55 patients who underwent RA bypass grafting were included in our systematic review. Follow-up period ranged from 0.5 to 84 months. The mean pooled primary patency and secondary patency of RA bypass grafting were 8.1 ± 4.9 and 21.7 ± 20.1 months, respectively. The incidence of early postoperative complications such as surgical site infection, bleeding, and access thrombosis was 0%, 4%, and 4%, respectively. The overall 30-day mortality was 4%.
Conclusions:
This systematic review summarizes the patient characteristics, technical features and outcomes of RA bypass grafting in the treatment of hemodialysis-related CVOD. RA bypass grafting may be a viable last-resort option when less invasive or conventional treatment options have been exhausted.
Keywords
Key findings
Right atrial bypass grafting for the treatment of hemodialysis-related central venous occlusive disease has a pooled mean primary and secondary patency of 8.1 ± 4.9 months and 21.7 ± 20.1 months, respectively. The incidence of early postoperative complications such as surgical site infection, postoperative bleeding, and access thrombosis was 0%, 4%, and 4% respectively. The overall 30-day mortality was 4%.
Take home message
RA bypass grafting is an option for the management of CVOD in hemodialysis patients. However, due to the magnitude of the procedure, RA bypass grafting should be reserved for selected patients who have failed less invasive therapy.
Table of contents summary
We systematically reviewed existing literature on the use of right atrial bypass grafting in the management of hemodialysis related central venous occlusive disease. We reported the pooled 12-month primary and secondary patency rates, 30-day mortality, and incidence of early postoperative complications after right atrial bypass grafting.
Background
The National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines recommend creating an autogenous arteriovenous fistula using a distal-to-proximal approach for patients who require vascular access for chronic hemodialysis. 1 However, in some patients who have exhausted all peripheral venous vasculature or have recurrent central vein stenosis, it can be extremely challenging to provide them with a reliable vascular access. Since existing KDOQI guidelines do not provide clear guidelines in the management of patients with recurrent central venous occlusive disease (CVOD), varying techniques have been described to maintain a functioning dialysis access. These techniques include balloon angioplasty, stenting, creation of lower extremity or extra-anatomic arteriovenous grafts, renal transplant, conversion to peritoneal dialysis, creation of hybrid grafts such as the Hemodialysis Reliable Outflow (HeRO) graft, or surgical bypasses such as right atrial (RA) bypass grafting.2–9
RA bypass grafting was first described by Duncan et al. 10 for two patients with subclavian vein thromboses, which threatened their upper extremity access grafts. The procedure entailed creating a bypass from the patent segment of the subclavian vein to the right atrium using a synthetic polytetrafluoroethylene graft via an infraclavicular incision and median sternotomy.
While initially devised to bypass central venous occlusion to maintain a threatened upper extremity access graft, this procedure has also been performed to create de novo vascular access in patients with known CVOD. Open surgical access and bypass to the RA can be considered to be of the most invasive procedures to create or maintain arteriovenous dialysis access in patients with end-stage renal disease (ESRD). Due to the magnitude of the procedure, significant morbidity and mortality can be expected. 11 Therefore, a decision to perform this procedure should only be made after careful consideration of other possible alternative methods of dialysis or renal replacement therapy and the patient’s comorbidities and risk factor profile. 12 However, despite the risks of performing a direct bypass to the RA, this procedure allows the maintenance of central venous drainage back to the heart even with extensive and severe CVOD involving the SVC to RA junction. 13
Due to the invasive nature of this procedure, almost all published literature assessing the outcomes of RA bypass grafts exist in the form of case reports or small case series, apart from a narrative review. 14 We performed a systematic review to synthesize the currently available evidence and report relevant clinical outcomes after RA bypass grafting for dialysis access.
Methods
We performed this systematic review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines. 15
Search strategy
We performed an electronic search using Medline (via PubMed), EMBASE Ovid, and the Cochrane Library from inception till 23 May 2021 with no language restriction. We used a combination of both MeSH and non-MeSH keywords: “atria,” “hemodialysis,” “hemodialysis,” “bypass,” and “graft” and modified the search syntax for compatibility. In MEDLINE, we used the following search strategy: (atria OR atrial OR atrium OR appendage) AND (bypass OR graft OR revascularization OR conduit OR shunt) AND (hemodialysis OR hemodialysis OR dialysis OR “renal replacement therapy”). We ensured that we included all possible relevant articles by performing backward reference searching of included articles and published review articles.
Eligibility criteria
We only included articles that reported outcomes in patients who had undergone RA bypass grafting to create or maintain hemodialysis access. We also included conference abstracts or letters if there were meaningful data that could be extracted. If there were articles with overlapping patient populations, we chose the publication with the larger sample size or the study that provided the most pertinent clinical information. We excluded articles that described the creation of RA bypass grafts for CVOD secondary to other etiologies such as fibrosing mediastinitis or malignancy. We also excluded animal studies, technical notes, and review articles.
Selection of studies and data extraction
Following a comprehensive search and deduplication, two authors (BP and ZHT) initially screened the titles and abstracts for relevance and subsequent full-text review. Next, a full-text review was conducted by the same authors for final inclusion into our systematic review. We adhered to the aforementioned inclusion and exclusion criteria. Conflicts were resolved by consensus or by appeal to a third author (JJN). After study selection, we utilized a standardized data sheet to extract data from the included articles. Variables that were relevant to this systematic review include study design (first author, year, study type, and follow-up period); patient characteristics (age, gender, comorbidities, and surgical indication); technical details (inflow vessel, conduit type, conduit size, and surgical incisions); and relevant outcome measures (primary patency, secondary patency, and postoperative complications).
Outcome measures
The outcome measures of interest in this study are primary patency, secondary patency, postoperative complications, and 30-day mortality. We adhered to the standard definitions for hemodialysis outcome measures defined by the Society for Vascular Surgery. 16 A considerable amount of heterogeneity existed in each article with regards to reporting of patency outcomes. Most articles did not specifically report primary and secondary patency but instead provided a timeline of the graft history. If the authors did not specifically define patency outcomes in their articles, we extrapolated them using the following modified definitions: (1) primary patency was defined as the time from RA bypass graft creation until the first intervention required to maintain or salvage the graft, completion of the follow-up period, or the patient’s death, whichever occurred first; and (2) secondary patency was defined as the time from graft creation until graft abandonment, completion of the follow-up period, or the patient’s death, whichever occurred first, regardless of any graft interventions performed. Postoperative complications relevant to the study are surgical site infection, postoperative bleeding, and early access thrombosis. We defined surgical site infection in accordance to the Centers for Disease Control and Prevention (CDC) guidelines. 17 Surgical site infection was defined as infection of the incision or deeper tissues within 30 days if a non-prosthetic conduit was used for RA bypass graft creation or up to 90 days if a prosthetic conduit was used. Postoperative bleeding is defined as any hemorrhagic event within 30 days of RA bypass graft creation requiring blood transfusion, radiological intervention, or surgery. Early access thrombosis is defined as complete thrombosis of the RA bypass graft within 30 days of creation. Lastly, 30-day mortality is defined as death occurring within 30 days of RA bypass graft creation. We assumed that there were no postoperative complications only when the article specifically stated that was the case.
Data analysis
Since the included articles largely consisted of case reports and series, we did not perform any formal weighted statistical pooling of outcome data using meta-analysis of proportions due to possible risk of bias caused by sampling error and small sample size. For continuous outcomes such as primary or secondary patency, we calculated and reported the overall mean and standard deviation using Cochrane’s formula. 18 For studies that reported other median and range, we converted them to mean and standard deviation for pooling purposes using a validated statistical method. 19 For categorical outcomes such as postoperative complications, we calculated and reported the overall proportion.
Quality assessment
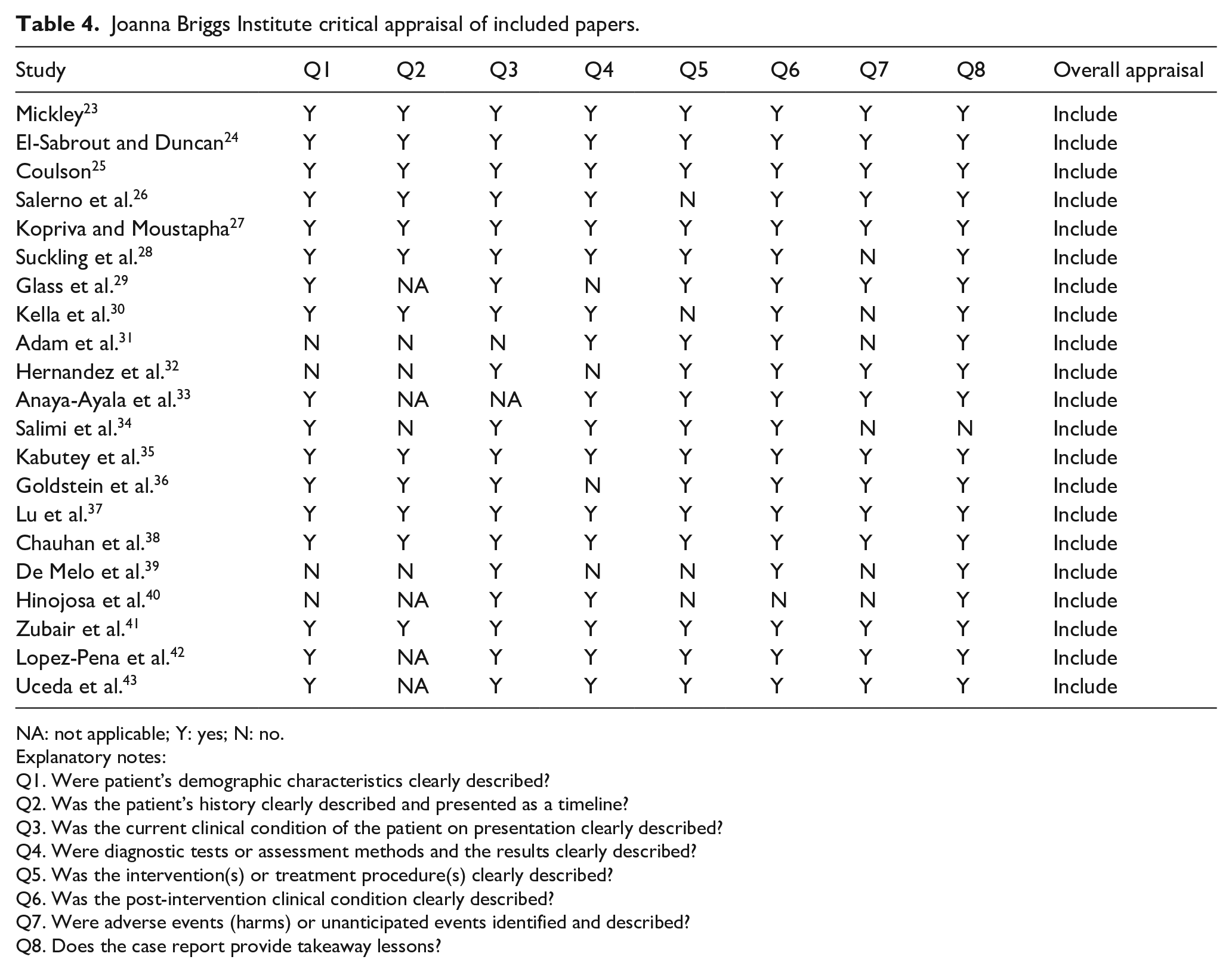
Included publications were subjected to a quality assessment using the Joanna Briggs Institute (JBI) critical appraisal tool, 20 which comprises eight domains assessing the internal validity and risk of bias of each included publication. As the critical appraisal questions in the JBI tool do not carry equal weightage, we based the quality assessment on the significance and relevance of each question based on the individual publication.
Results
Study selection
Our initial search revealed a total of 435 articles for possible inclusion. After elimination of duplicate articles and initial title and abstract screening, 41 articles remained for full-text screening. After the full-text screening, 18 articles were excluded, and 23 were considered for final inclusion in the systematic review. From the remaining 23 articles, two case series were further excluded from our systematic review because data about patients who had undergone RA bypass graft creation could not be separated from other irrelevant patient subgroups.21,22 Hence, a total of 21 articles were included in our systematic review. The article selection process is summarized in the PRISMA diagram (Figure 1).
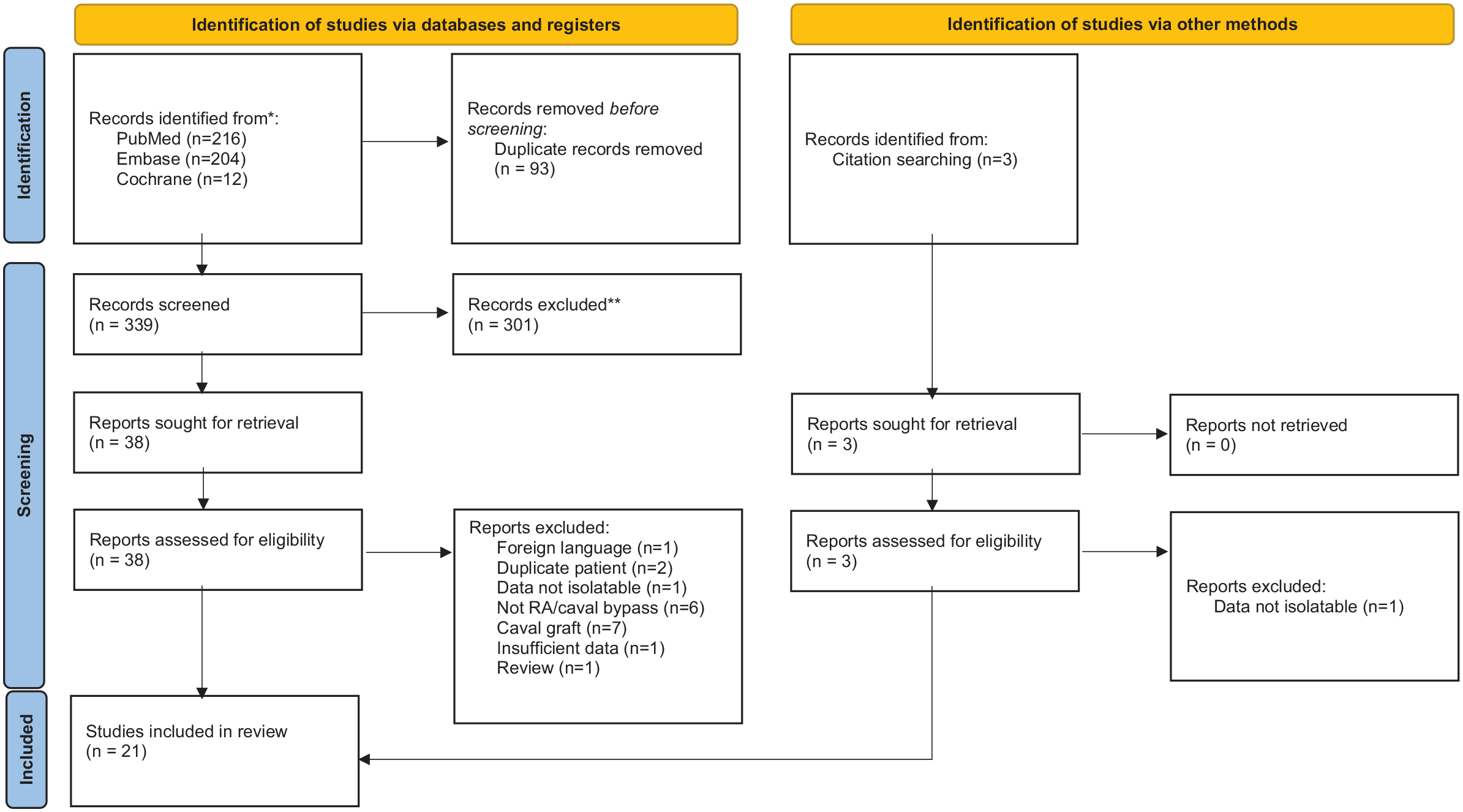
PRISMA diagram of included studies.
Characteristics of included studies
Out of the 21 included articles, 8 were case series, 12 were individual case reports, and 1 was a case-control study that compared axillo-atrial to axillo-iliac arteriovenous grafts for hemodialysis access. From the included articles, a total of 55 patients underwent RA bypass graft creation secondary to CVOD. We further subdivided these patients into two subgroups—de novo and revisional. Patients classified under the de novo subgroup underwent RA bypass graft creation as part of new hemodialysis access. This meant that either the graft could be needled directly, or the patient would undergo concomitant creation of upper extremity access. On the other hand, patients classified under the revisional subgroup underwent RA bypass graft creation to salvage or maintain an existing hemodialysis access functionality. More than half of the patients (60.5%) underwent a de novo procedure, whereas 39.4% underwent a revisional procedure. We could not ascertain the nature of RA bypass graft creation from Glass et al. or Anaya-Ayala et al. Characteristics of included studies are summarized in Table 1.
Baseline characteristics of studies included in the systematic review.
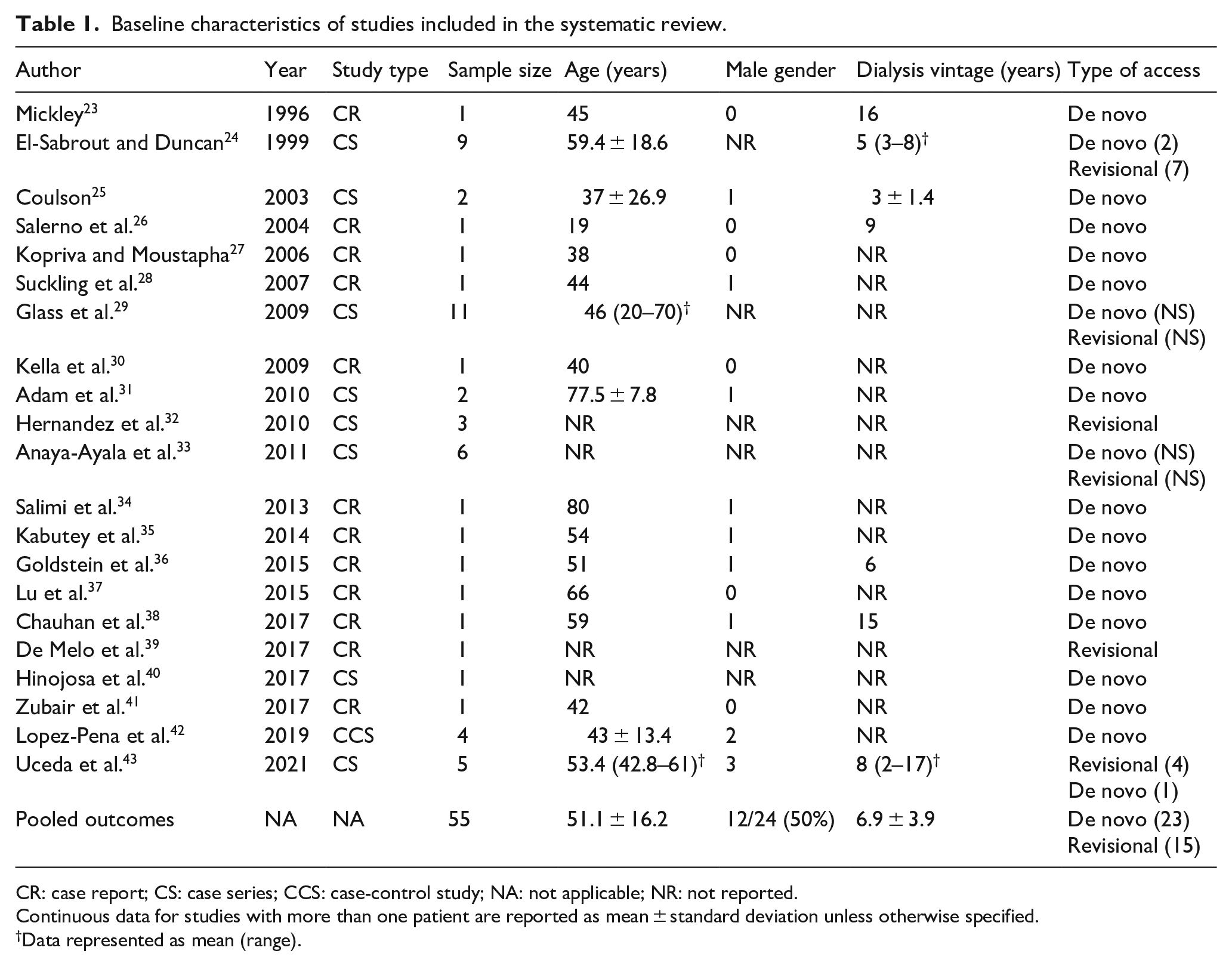
CR: case report; CS: case series; CCS: case-control study; NA: not applicable; NR: not reported.
Continuous data for studies with more than one patient are reported as mean ± standard deviation unless otherwise specified.
Data represented as mean (range).
Patient demographics
Seventeen articles reported the age of the patients who underwent RA bypass graft creation, ranging from 19 to 86 years, with an overall mean of 51.1 ± 16.2 years. Gender distribution between patients was equal. Only seven articles had reported the duration of dialysis dependence, which ranged from 2 to 16 years with an overall mean of 6.9 ± 3.9 years. We did not report patient comorbidities due to missing data and inconsistent reporting. Patient demographics are summarized in Table 1.
Technical details
All included articles described the technical aspects of RA bypass graft creation which varied widely. A comprehensive summary of technical details is found in Table 2. For patients that underwent de novo RA bypass graft creation, graft inflow was mainly derived from either the left or right axillary artery. In other patients, graft inflow was derived from the subclavian artery, brachial artery, ascending aorta, superficial femoral artery, innominate vein, and cephalic vein. For patients with arterial inflow, the RA bypass graft was usually needled directly for hemodialysis. For patients with venous inflow, a new ipsilateral upper extremity arteriovenous access was created concurrently. On the contrary, graft inflow was mainly derived from the subclavian or innominate vein for patients who underwent revisional RA bypass graft creation. These patients already have a functioning pre-existing upper extremity arteriovenous access. Most of the included articles did not specify the site of the outflow anastomosis on the RA. However, six articles used the RA appendage as the site of the outflow anastomosis, while two used the lateral wall of the RA. One article described the junction between the superior vena cava and RA as the site of the outflow anastomosis. None of the included articles reported the use of cardiopulmonary bypass during RA bypass grafting.
Technical aspects of right atrial bypass grafting.
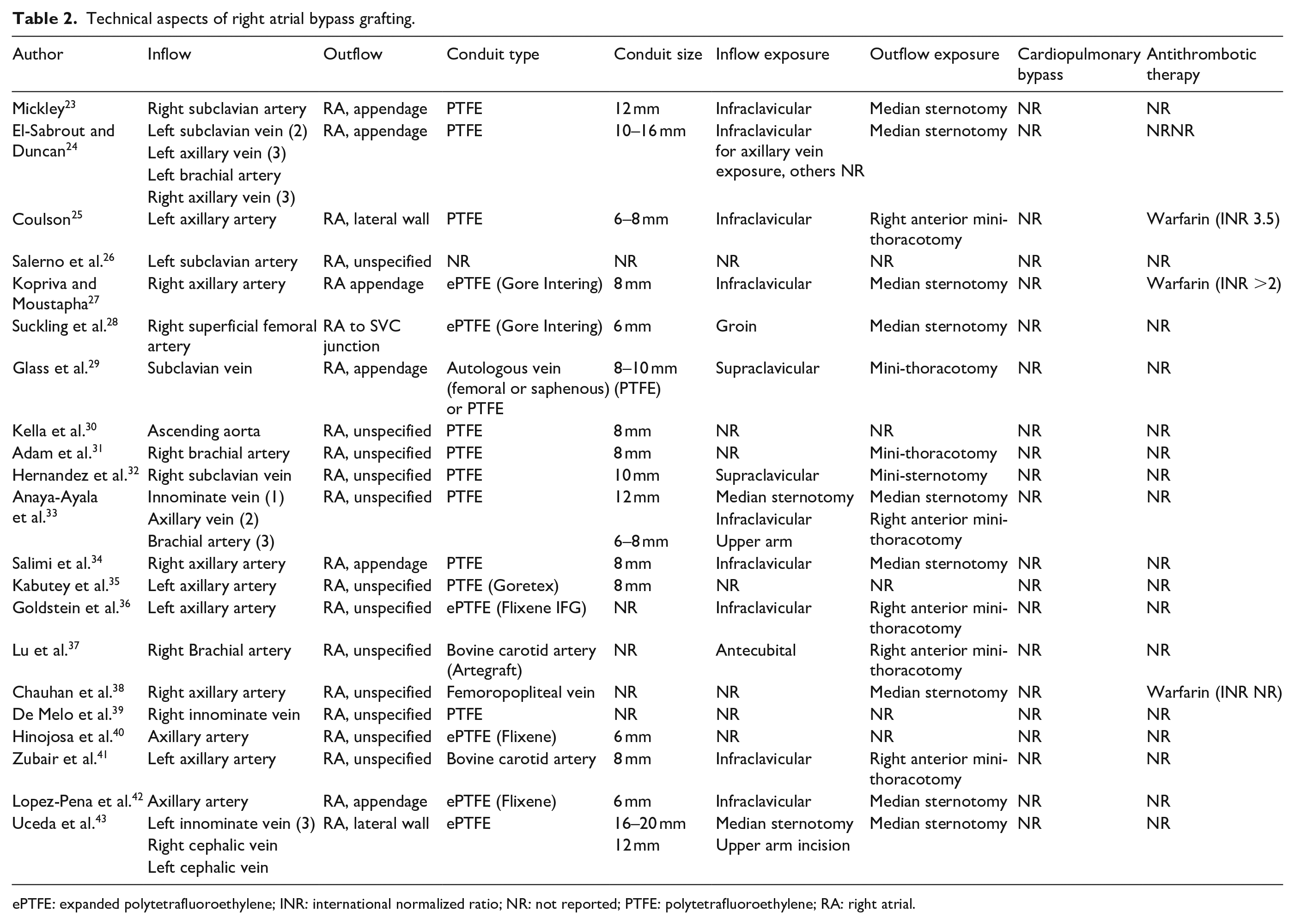
ePTFE: expanded polytetrafluoroethylene; INR: international normalized ratio; NR: not reported; PTFE: polytetrafluoroethylene; RA: right atrial.
Most RA bypass grafts were constructed using synthetic grafts made from polytetrafluoroethylene (PTFE) or expanded polytetrafluoroethylene (ePTFE). Only one article described the use of autologous vein, while two other articles described the use of bovine carotid artery as a bypass conduit. Bypass conduits ranged in size from 6 to 20 mm, but most were at least 8 mm in diameter. The choice of surgical incision for RA bypass grafting varied widely in the different articles. Usually, an infraclavicular or supraclavicular incision was used to expose inflow vessels such as the axillary or subclavian vessels. A median sternotomy was used to expose more central inflow vessels such as the innominate vein. For exposure of the RA, however, some articles described the use of a median sternotomy, while others described the use of a mini-thoracotomy incision. Following the creation of the RA bypass graft, the use of warfarin as antithrombotic therapy was reported in only three articles.
Patency outcomes
Thirteen articles presented relevant data which could be interpreted as primary patency. A summary of these results is provided in Table 3. The reported primary patency of the RA bypass grafts ranged from 0 to 53 months with a mean pooled primary patency of 8.1 ± 4.9 months. Sixteen articles presented relevant data which could be interpreted as secondary patency. The reported secondary patency ranged from 0.5 to 84 months with mean secondary patency of 21.7 ± 20.1 months. Except for one article, all included articles reported their length of follow-up. The reported length of follow-up for all articles ranged from 0.5 to 84 months with a mean follow-up period of 18.5 ± 16.0 months.
Outcomes of right atrial bypass grafting.
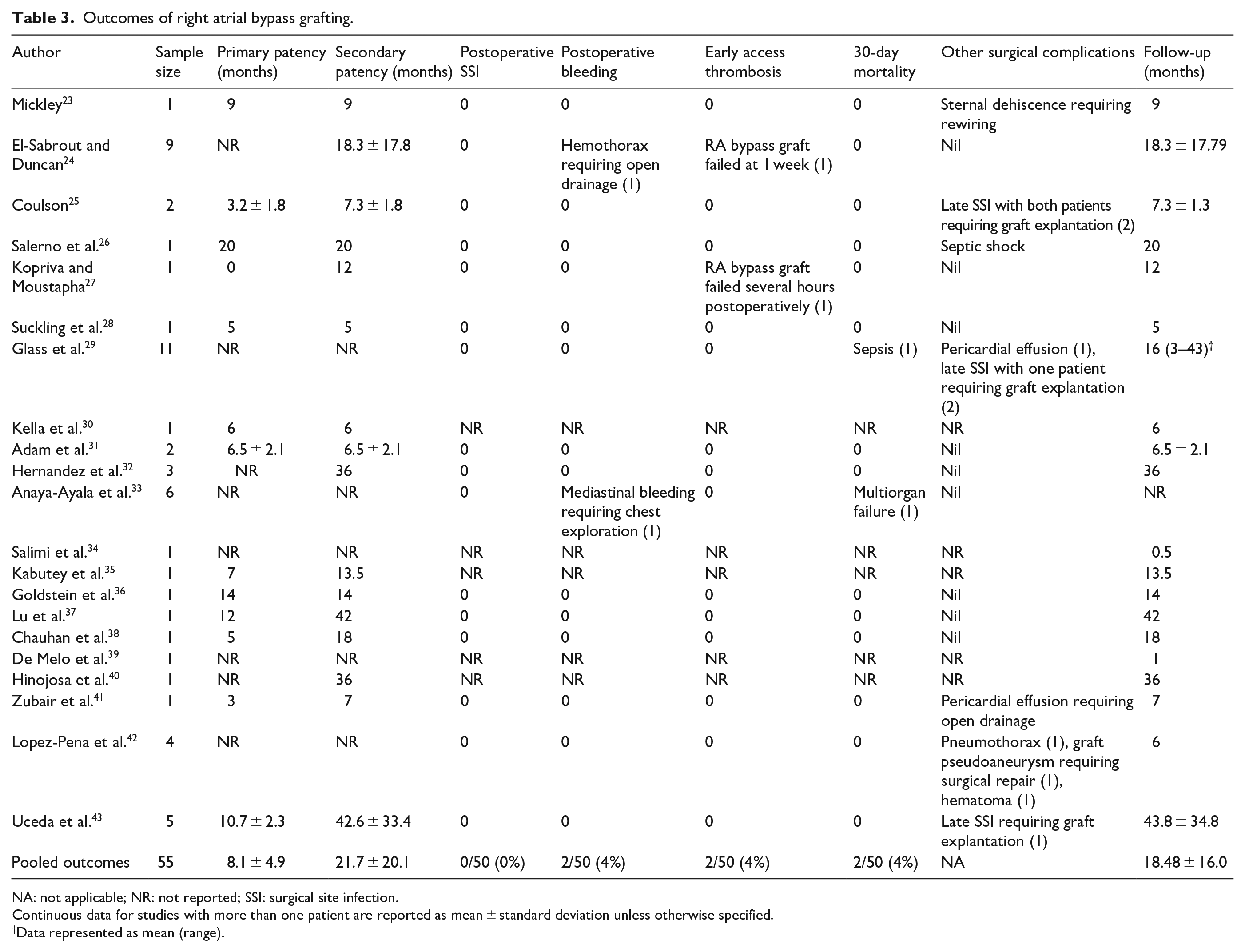
NA: not applicable; NR: not reported; SSI: surgical site infection.
Continuous data for studies with more than one patient are reported as mean ± standard deviation unless otherwise specified.
Data represented as mean (range).
Postoperative complications
Sixteen out of 21 included articles had reported relevant data regarding postoperative complications after RA bypass graft creation, as delineated in Table 3. The definitions of the postoperative complications of interest had been defined in the methods section. No patients developed postoperative surgical site infection, although five (9%) had developed late prosthetic graft infection requiring graft explantation, one of which was infected after 6 years.25,29,43 The incidence of postoperative bleeding was 4% (two out of 50 patients), with one patient developing a hemothorax that required open drainage on postoperative day 1. 24 Another patient developed a mediastinal bleed in the immediate postoperative period, which necessitated chest exploration. 33 The incidence of early access thrombosis was also 4% (two out of 50 patients), of which one patient’s graft failed several hours after surgery due to graft kinking, and another patient’s graft failed at 1 week.24,27 Other postoperative complications that were reported included sternal dehiscence requiring rewiring (one patient), pericardial effusions (two patients), pneumothorax (one patient), postoperative hematoma (one patient), and graft pseudoaneurysm (one patient).23,29,41,42 The overall 30-day mortality was 4% (two out of 50 patients), for which the first patient died secondary to sepsis, and the second patient died due to multi-organ failure.29,33
Quality assessment
All studies included in our analysis were assessed to be of either moderate or high study quality via the JBI Critical Appraisal Tool, signifying a low risk of bias, as shown in Table 4.
Joanna Briggs Institute critical appraisal of included papers.
NA: not applicable; Y: yes; N: no.
Explanatory notes:
Q1. Were patient’s demographic characteristics clearly described?
Q2. Was the patient’s history clearly described and presented as a timeline?
Q3. Was the current clinical condition of the patient on presentation clearly described?
Q4. Were diagnostic tests or assessment methods and the results clearly described?
Q5. Was the intervention(s) or treatment procedure(s) clearly described?
Q6. Was the post-intervention clinical condition clearly described?
Q7. Were adverse events (harms) or unanticipated events identified and described?
Q8. Does the case report provide takeaway lessons?
Discussion
With an increasing number of ESRD patients having a longer duration of dialysis dependence and life expectancy, more patients are expected to develop complications relating to hemodialysis access such as CVOD.45,46 In this systematic review, we assessed the indications, technical aspects, and outcomes of RA bypass grafting for treating CVOD for patients on hemodialysis from an assemblage of real-world evidence.
From our systematic review, we identified 21 relevant articles with a total of 55 patients who underwent RA bypass grafting to treat hemodialysis-related CVOD. Unsurprisingly, the evidence was limited to mainly individual case reports and case series as RA bypass grafting is an invasive procedure usually performed when endovascular treatment options are not available or if patients have recalcitrant or resistant CVOD and have exhausted all other treatment options for CVOD. CVOD is a debilitating disease that is difficult to treat. According to the most recent 2019 KDOQI clinical practice guideline for vascular access, an endovascular approach with balloon angioplasty should be the first-line treatment in hemodialysis patients with symptomatic CVOD. 1 In the era of endovascular therapy, the majority of hemodialysis-related CVOD is managed with endovascular techniques such as balloon angioplasty or stenting. In a retrospective study by Saleh et al., 47 plain balloon angioplasty of patients with symptomatic CVOD demonstrated a 1-year primary and secondary patency of 51.7% ± 7.4% and 67.2% ± 6.9%, respectively. Although plain balloon angioplasty is associated with a high technical success rate, long term patency outcomes are poor, and patients commonly recur symptomatically. As such, other endovascular strategies have been evaluated for the treatment of CVOD, such as drug-coated balloons and stenting. A randomized controlled trial performed by Kitrou et al. 48 demonstrated a more extended intervention-free period in the paclitaxel-coated balloon group compared to the plain balloon group. Similarly, a more recent retrospective cohort study by Chong et al. 49 also demonstrated favorable outcomes for drug-coated balloon angioplasty for treatment of CVOD with mean primary patency of 164 days compared to 140 days when plain balloons were used. Whilst drug-coated balloons have shown improved results compared to plain balloon angioplasty, most studies have a short follow-up period with a paucity in longer-term data.
Endovascular stenting is also commonly performed and has also been evaluated in the treatment for CVOD, either as primary stenting or as secondary stenting after recurrence or failure of balloon angioplasty. Although early studies have reported unfavorable patency outcomes associated with endovascular stenting in the treatment of CVOD, such as in a study by Gray et al. 50 that reported primary patency of only 20% at 12 months after bare metal stenting, more recent studies with improved stent designs and constructions have actually achieved better outcomes. A recent systematic review by Andrawos et al. 51 reported pooled 12-month primary patency, assisted primary patency, and secondary patency of 53.8%, 70.9%, and 94%, respectively, after endovascular stenting for the treatment of hemodialysis related CVOD. Similarly, Akkakrisee and Hongsakul 52 reported a primary patency of 61.8% at 12 months when a venous specific bare metal stent was used to treat CVOD.
Another option that can be used to manage patients with CVOD would be hybrid grafts such as the Hemodialysis Reliable Outflow (HeRO) graft, provided that wire access can be obtained into the superior vena cava and RA for placement of the central venous component. The HeRO graft consists of a synthetic graft that is anastomosed to an inflow artery (commonly the brachial artery) and connected to a central venous component in the superior vena cava or right atrium. A systematic review by Shakarchi et al. found that the pooled primary and secondary patency rates in patients who underwent implantation of the HeRO graft to be 21.9% and 59.4% respectively. Although the pooled patency rates associated with HeRO graft implantation reported in the systematic review appeared to be low and may have been biased by smaller studies, it is important to note that the largest series that reported outcomes after HeRO graft implantation had favorable results. Gage et al. performed a retrospective review of 164 patients who underwent HeRO graft implantation, and reported primary and secondary patency at 12 months of 48.8% and 90.8%, respectively, and primary and secondary patency at 24 months of 42.9% and 86.7%, respectively. In the same study, the authors also reported a low incidence of access-related infections, affecting only 4.3% of patients. 53 More recently, newer studies have also corroborated the technical success and safety profile of HeRO graft implantation. Illig et al. 54 reported that 89% of patients could undergo hemodialysis via graft cannulation and discharged without a tunneled catheter after HeRO graft implantation. The authors also reported a low rate of postoperative mortality and surgical site infections.
In more extreme cases of CVOD, especially when wire access to the superior vena cava and RA is no longer possible, other options such as arterioarterial grafts or the use of transhepatic or translumbar catheters may be considered. 14 The use of arterioarterial grafts have been described widely in patients with unreconstructable CVOD, or in patients with severe heart failure. Arterioarterial grafts are generally constructed using synthetic grafts that are tunneled and anastomosed in a loop configuration to the mid arm brachial artery or infraclavicular axillary artery. 55 A systematic review by Grima et al. 56 reported encouraging results with the arterioarterial grafts with a 12-month primary patency rate between 61.7% and 87.9% and a 12-month secondary patency rate between 83% and 90.7%. However, despite the favorable patency outcomes associated with arterioarterial grafts, we must be aware of complications such as upper limb ischemia and the development of pseudoaneurysms. Alternatively, other authors have described the use of translumbar or transhepatic catheters for hemodialysis access. Inston et al. 14 systematically reviewed and summarized the data pertaining to translumbar or transhepatic catheters and found that the reported outcomes in terms of patency or complications are extremely variable and wide-ranging. For example, the 6-month catheter patency rate ranged from 25% to 83%, while the 12-month patency rate ranged from 7% to 73.2%. However, what seemed to be consistent in the use of translumbar or transhepatic catheters was the need for frequent catheter changes due to catheter dysfunction.
In our systematic review, we found that 30-day mortality after RA bypass grafting was 4%, while the incidence of postoperative surgical site infection, postoperative bleeding, and early access thrombosis was 0%, 4%, and 4%, respectively. The risk of postoperative mortality and morbidity in ESRD patients is increased due to several factors. In ESRD patients receiving hemodialysis, the incidence of cardiovascular disease is estimated to be 77% according to the United States Renal Data System, which increases the risk of perioperative major adverse cardiac events and mortality. 45 Patients with ESRD are at increased risk of developing wound complications because they are immunocompromised due to uremia and chronic inflammation. 57 The risk of wound complications is further increased if the patient has concomitant diabetes mellitus. 58 Patients with ESRD also have an increased risk of perioperative bleeding and requiring blood transfusions, as highlighted in a meta-analysis by Acedillo et al. 59 The increased risk of perioperative bleeding can be attributed to ESRD-related platelet dysfunction and coagulopathy, as well as the presence of abnormal venous collaterals in the chest wall due to CVOD. The incidence of postoperative complications reported in our study, such as surgical site infections, appears to be consistent with other studies that evaluated postoperative complications in cardiothoracic surgery. Lemaignen et al. 60 reported an incidence of wound infections in ESRD patients who had undergone cardiac surgery of 4.1%, while Figuerola-Tejerina et al. 61 reported a similar incidence of 4%. On the other hand, our reported 30-day mortality of 4% appeared to be lower compared to studies reporting mortality after cardiothoracic surgery. Several large cohort and registry studies have reported 30-day or in-hospital mortality of ESRD patients above 10% after cardiac surgery.62,63 This discrepancy in 30-day mortality could be attributed to procedural differences such as need for cardiopulmonary bypass or the presence of a highly selected population chosen for RA bypass grafting, but also to underreporting or publication bias. Nevertheless, RA bypass grafting for the treatment of CVOD in ESRD patients requiring hemodialysis remains an option for selected patients with recalcitrant CVOD or for patients in whom endovascular techniques have failed or are not available. Strict and careful patient selection is mandatory for RA bypass grafting, as ESRD patients are at higher risk of developing postoperative mortality and morbidity compared to patients without renal failure. 64
The management of hemodialysis-related CVOD is constantly evolving. We now have more options to treat CVOD with endovascular techniques that offer good outcomes and low complication rates. For example, in patients with chronically occluded central veins where conventional wire access and wire passage are not possible, the Surfacer Inside-Out Access Catheter System can be used to restore wire access and facilitate placement of a tunneled catheter or HeRO graft. As such, RA bypass grafting should be reserved as one of the last-resort options in the management of CVOD.
This is the first study that comprehensively summarizes the indications, technical aspects, and relevant clinical outcomes of RA bypass grafting for the treatment of CVOD in hemodialysis patients. The findings from our systematic review contribute to the current literature, as long as they are interpreted judiciously with the following limitations in mind. First, most of the included articles did not adhere to standard dialysis access reporting guidelines when reporting patency outcomes and postoperative complications. 16 Although we attempted to extract data from each article in a structured manner using predetermined definitions, there could still be inaccuracies in the data. Secondly, it is regrettable that the larger case series included in this systematic review, such as the articles by Glass et al. 29 and Anaya-Ayala et al. 33 did not report results on patency, which could lead to publication bias and small study effects. Third, readers need to be aware that the studies included in our systematic review are susceptible to substantial reporting, where only positive outcomes are reported and published. In this systematic review, none of the included studies reported a catastrophic outcome such as intraoperative or early postoperative mortality. Such a bias could lead to an underestimation of the incidence and risk of postoperative morbidity and mortality associated with RA bypass grafting. Finally, given the evolving treatment strategies of CVOD, it is difficult to determine exactly when RA bypass grafting should ideally be performed. More studies are needed to prove the safety and efficacy of this invasive treatment strategy.
Conclusion
This systematic review summarizes the patient characteristics, technical features, and outcomes of RA bypass grafting in the treatment of hemodialysis-related CVOD. However, in current practice, less invasive endovascular strategies such as balloon angioplasty or stenting are still preferred. There might be a role for RA bypass grafting in patients with recalcitrant CVOD, where endovascular strategies have failed or are not available.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
