Abstract
Background:
The two ends of arteriovenous graft (AVG) are anastomosed to the upper limb vessels by surgery for hemodialysis therapy. However, the size of upper limb vessels varies to a large extent among different individuals.
Methods:
According to the shape and size of neck vessels quantified from the preoperative computed tomography angiographic scan, the ethylene-vinyl acetate (EVA)-based AVG was produced in H-shape by the three-dimensional (3D) printer and then sterilized. This study investigated the function of this novel 3D-printed AVG in vitro and in vivo.
Results:
This 3D-printed AVG can be implanted in the rabbit’s common carotid artery and common jugular vein with ease and functions in vivo. The surgical procedure was quick, and no suture was required. The blood loss was minimal, and no hematoma was noted at least 1 week after the surgery. The blood flow velocity within the implanted AVG was 14.9 ± 3.7 cm/s. Additionally, the in vitro characterization experiments demonstrated that this EVA-based biomaterial is biocompatible and possesses a superior recovery property than ePTFE after hemodialysis needle cannulation.
Conclusions:
Through the 3D printing technology, the EVA-based AVG can be tailor-made to fit the specific vessel size. This kind of 3D-printed AVG is functioning in vivo, and our results realize personalized vascular implants. Further large-animal studies are warranted to examine the long-term patency.
Introduction
Patients whose renal function progress to end-stage kidney disease necessitates regular hemodialysis therapy, which requires a functional vascular access. While arteriovenous fistula (AVF) surgically anastomoses an artery to a vein, an arteriovenous graft (AVG) is a synthetic looped tube that connects an artery to a vein. In the case of limb vessels being inadequate for AVF creation or the created AVF failing to mature, AVG is the choice of vascular access.
The most commonly used material for the construction of AVGs is the expanded polytetrafluoroethylene (ePTFE). However, the relative bio incompatibility of the synthetic tubes with living vessels tends to cause hemodialysis vascular access vasculopathy and failure. Compared to native fistulae, ePTFE grafts possess a three-fold increased risk of adverse events such as thrombosis, resulting in morbidity, hospitalization, and even mortality.1 –4 Three-dimensional (3D) printing is a promising approach for designing and engineering human organs and tissues by providing a flexible, automated, and on-demand platform for fabricating complex living architectures.5 –7 Moreover, 3D printing enables customization of the shape and size of AVG, an unachievable feat using previous techniques.
The graft manufactured solely from natural proteins failed to achieve appropriate mechanical properties.8,9 Synthetic polymers (e.g. poly(ε-caprolactone) (PCL) and polylactide (PLA)) have been added to collagen for fabricating vascular grafts due to their good biocompatibility.10,11 However, the PCL or PLA-based 3D-printed vascular grafts are limited by their rigid properties, resulting in a compliance mismatch between the graft and the native vessel. Furthermore, their biodegradability poses a risk of blood leak and thus prevents them from long-term implantation.12,13 In contrast, ethylene-vinyl acetate (EVA) is a novel, printable, non-biodegradable, and Food and Drug Administration (FDA)-approved biocompatible material and has been recently developed to used as a drug-loaded implant intra-uterinely or subcutaneously.14 –16 The soft and biostable EVA elastomer is featured by its high flexibility and strength, which accentuates its advantages of being favorable for manufacturing the 3D-printed vascular graft. However, EVA-based vascular graft has not been investigated in the past.
The present study took advantage of preoperative 3D-reconstructed computed tomography (CT) angiography to tailor-make the AVGs. We incorporated the biocompatible EVA copolymer with 3D printing technology to fabricate vascular grafts with personalized geometry (i.e. shape and size). Furthermore, we aimed to examine the in vivo functionality of the innovative tailor-made AVGs in the animal model. With the advances in material science and 3D printing technology, we believe that combining these two tiers, ideal biomaterial and tailor-made manufacturing, will help perfect the vascular graft.
Methods
Biomaterials
We used poly(ethylene-co-vinyl acetate) (EVA) copolymer (Sigma-Aldrich, St. Louis, MO, USA) as a biomaterial for the 3D printing process because of its biocompatible nature, optical clarity, thermoplasticity, and tunable stiffness, with Young’s moduli between 3 and 42 MPa, as previously reported.17,18 EVA is an FDA-approved biocompatible material that can support cell growth and be applied in vivo as a long-term implant. 14 The printability significantly depends on the thermoplastic polymer’s glass transition temperature (Tg) and melting temperature (Tm). For EVA copolymers with the proportion of 12%–40% (w/w) vinyl acetate with a Tm range of 60°C–70°C, the Tg ranges from −19°C to −10°C, which were measured by dynamic mechanical analysis using TA Instruments Q800 at a heating rate of 10°C min−1 between −80°C and 80°C (Supplemental Figures S1–S5), and decreases while increasing the ratio of vinyl acetate. 18 Supplemental Figure S1 shows the thermogravimetric analysis of EVA copolymers with different proportions of vinyl acetate. Also, Supplemental Table S1 summarizes the characterization of EVA copolymers examined in this study. Polymers suitable for the 3D printing process are characterized by low Tg, low Tm, low fluidity, and high thermal-extrusion temperature. As a result, the EVA copolymer composed of 40% vinyl acetate possessed the best printability and was thus selected for 3D printing.
In vitro endothelial cell culture
Human umbilical cord vein endothelial cells were purchased from the Food Science Research Institute (BCRC) in Taiwan. Before culturing, human endothelial cells (ECs) were mixed with 10% fetal bovine serum (FBS, Hyclone), 1% penicillin/streptomycin (P/S, Hyclone), and 1% endothelial cell growth supplement (endothelial cell growth supplement), the cells were cultured in 5% CO2 at 37°C. ECs were cultured on the flat EVA surface with the proportion of 40% (w/w) vinyl acetate, which was pre-attached on the well surface of the non-tissue culture plate (Falcon™ 48-Well, Non-Treated, Flat-Bottom Microplate, Corning, New York, NY, USA). To examine whether the EVA surface is suitable for EC adhesion and proliferation, ECs were also cultured directly on the non-tissue culture plate to serve as the negative control group, while ECs cultured on multi-well plates with the Nunclon™ Delta surface treatment to promote maximum adhesion (Nunc™ Cell-Culture Treated Multidishes, ThermoFisher Scientific, Waltham, MO, USA) were the positive control group. Before culturing, a cell attachment factor solution (Cat. 123-100; Cell Applications, San Diego, CA, USA) was added in all the wells at 37°C for 1 h to facilitate cell attachment. Next, ECs were seeded at the density of 5 × 104 cells/mL.
The crystal violet assay was used to determine cell viability. The crystal violet dye binds to proteins and DNA. Cells that undergo cell death lose their adherence and are subsequently lost from the population of cells, reducing the amount of crystal violet staining in a culture. Cells were then fixed for 10 min in a solution of buffered formalin (3.7%), washed with PBS (pH 7.3), and then subsequently stained with a 1.0% crystal violet solution for 10 min. After removing the excess stain by ddH2O, the crystal violet stained cells were counted under optical microscopy (CKX53, Olympus, Tokyo, Japan). Cell adhesion and viability were analyzed using one-way ANOVA with the GraphPad Prism 6.0 (GraphPad Software, CA, USA). Fisher’s LSD multiple comparison tests were used for post hoc analysis. A p-value of less than 0.05 was considered statistically significant. ImageJ software (National Institutes of Health, Bethesda, MD, USA) was used to compare pixel number per cell in the microscopic field. The results are presented in Figure 1.
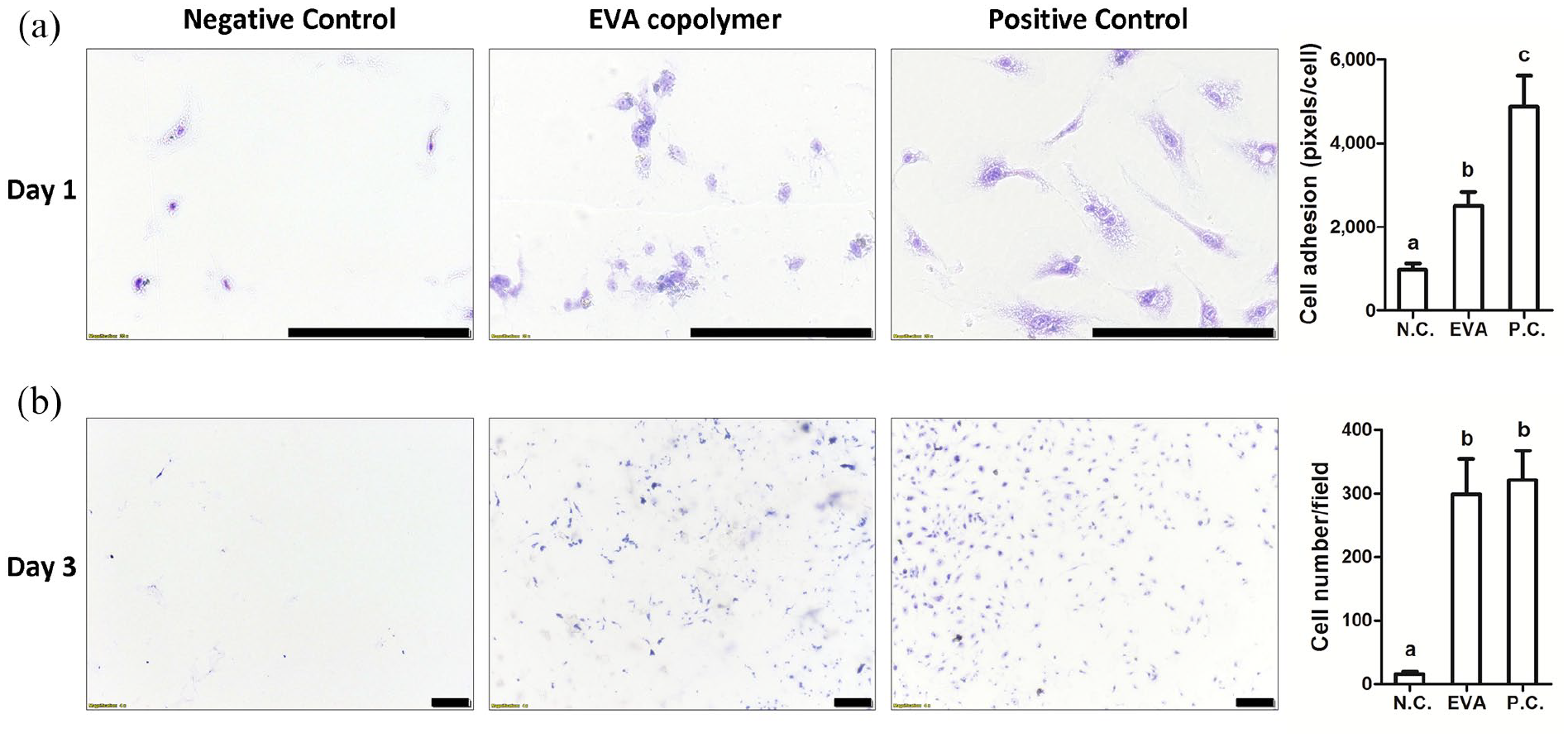
In vitro culture of human endothelial cells on the EVA surface: (a) Cell adhesion on day 1 with relevant quantification analysis and (b) cell viability on day 3 with relevant quantification analysis. N.C. and P.C. denote negative and positive control groups, respectively. ANOVA, p < 0.05, different characters represent different levels of significance. Scale bar = 200 μm.
Preoperative CT angiography
Rabbit neck CT angiographic scans were performed on a 128-slice multi-detector CT scanner (Ingenuity, Philips Healthcare, Best, The Netherlands). Under general anesthesia, the rabbits were restrained at the center of the CT scanner in a prone position. Before scanning, 3 mL/kg of non-ionic contrast medium (Omnipaque 300, GE Healthcare Ireland Limited, Ireland) was injected into the ear vein at the rate of 1 mL/s using an automated contrast injector. Following the contrast medium injection, 10 mL of normal saline was flushed at the same rate. The scanning parameters consisted of slice thickness of 1 mm, tube voltage of 120 kV, and effective mAs of 120 mA.
3D Printing
Based on the CT angiographic image of the rabbit, a specific H-shape AVG, with internal diameters of 1.30–1.54 mm and 1.80–2.10 mm for rabbit common carotid artery and common jugular vein, respectively, was printed. The wall thickness was 0.2 mm. Such 3D-printed AVG was designed using BioCADTM software (RegenHU, Villaz-St-Pierre, Switzerland) and then additively manufactured on a flat glass slide by a polymer extrusion printing head of a regenHU 3D Discovery printer (RegenHU 3D Discovery, Villaz-St-Pierre, Switzerland; DD135H printing head equipped with a 0.10 or 0.2 mm inner diameter needle, sample extrusion rate is 1–5 mm/s under air pressure at 0.18 MPa) via thermal extrusion at 140°C (Supplemental Video S1; time-lapse photography). As shown in Supplemental Video S1, it took approximately 3 h to complete the printing process of a specific H-shape AVG. Therefore, the printed AVG’s external diameters were 1.70–1.94 mm and 2.2–2.5 mm to fit rabbit common carotid artery and common jugular vein, respectively (Figure 2).
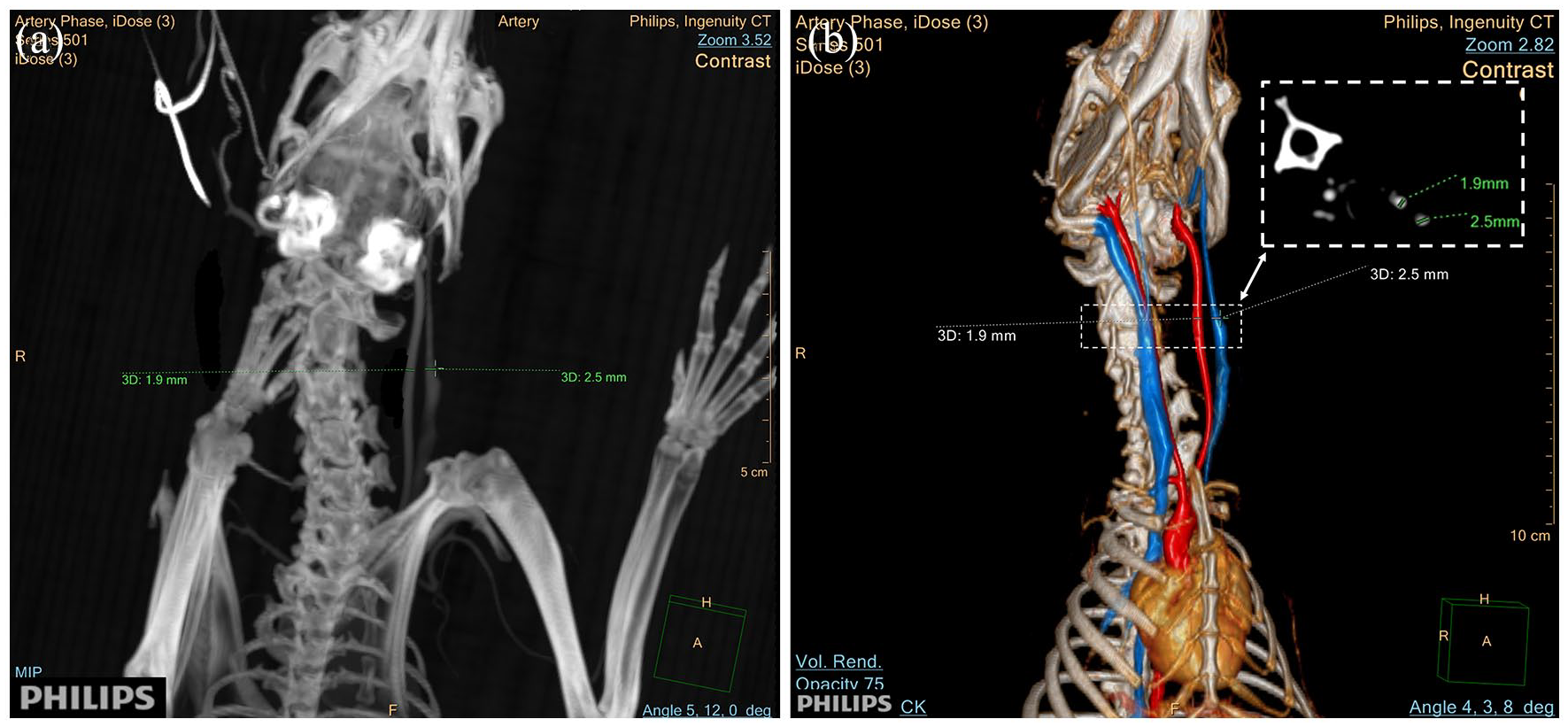
The representative computed tomography (CT) angiography of the rabbit neck vessels for pre-3D printing evaluation: (a) the internal diameters of neck vessels were measured on the coronal section of CT scan and (b) 3D-reconstructed CT angiographic image of the left common carotid artery (red) and the left common jugular vein (blue).
Animals
New Zealand White rabbits (male, n = 6; weight, 2.5–3.0 kg) were purchased from Hui-Jun Rabbit Farm, Changhua, Taiwan. All rabbits were housed individually in a stainless steel cage system under standard environmental conditions (20.2 ± 2°C, 30%–70% relative humidity, 12:12-h light: dark cycle, and 12–15 air changes hourly). Rabbits were allowed ad libitum access to a commercial rabbit diet and filtered water. The operative practices were developed under the supervision of a veterinary surgeon. Euthanasia was done by injection of a potassium chloride solution. All animal experiments were performed following the guidelines of the Institutional Committee for Animal Experimentation of the institute.
Surgical procedure
The surgical procedure of 3D-printed vascular graft implantation was summarized in Figure 3. Rabbits were pre-medicated with Zoletil® 50 (0.1 mL/kg, IM) (25 mg/mL tiletamine and zolazepam 25 mg/mL, Virbac, France) and Rompun® (0.5 mL/kg, IM) (20 mg/mL; Bayer, Germany), followed by injections of Zoletil® 50 (0.2 mL/kg, IV) every 30 min. As the EVA is used above its Tg, which is less than room temperature, the planar H-shape AVG (Figure 4(a)) is flexible thus easily bendable. When the AVG was implanted in vivo, a reverse-loop configuration was formed (Figure 3). A para-median cervical incision was made in each animal; this incision was then dissected layer-by-layer to expose the trachea. The left common carotid artery and ipsilateral common jugular vein were then identified. First, the left common carotid artery and left common jugular vein were freed by 2 cm each, and both were clipped at the distal and proximal ends using damage-free vascular clamps (Figure 4(b)). A longitudinal incision, approximately 1.5 mm in length, was made in the middle of the common carotid artery and common jugular vein parallel to its vertical axis. The heparinized saline was perfused into both the vessel lumen and the graft. The graft’s larger-diameter limbs were consequently inserted into the common jugular vein using a forceps that slightly lifted the upper venous wall to expose the lumen (Figure 4(c) and (d)). Next, the other smaller-diameter limbs were inserted into the arterial lumen (Figure 4(e) and (f)). Next, a few drops of 3 M™ Vetbond™ Tissue Adhesive was applied to the graft-vessel insertion connective part and then waited for 5 min (Figure 5(a) and (b)). No single suture was required between the prosthesis and vessels. Finally, the vascular clamps were released (Figure 5(c)), and then the blood was recirculated at once (Figure 5(d)–(g)). In this manner, a common carotid artery-common jugular vein bridge was built to bypass arterial blood flow into the venous system.
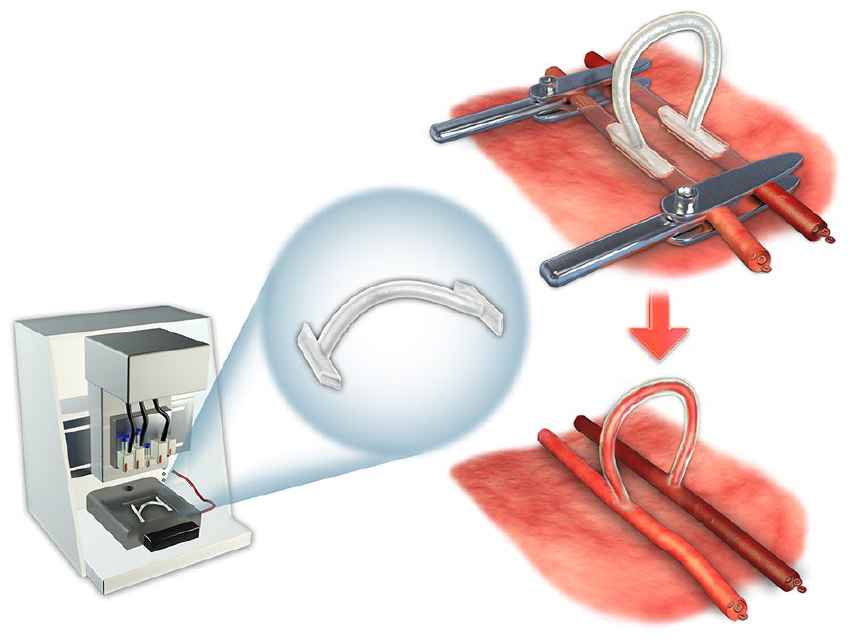
The schematic diagram of the three-dimensional (3D)-printed vascular graft implantation. After surgically free of the rabbit’s common carotid artery and common jugular vein, the 3D-printed vascular graft was trimmed and inserted into the blood vessels. The blood recirculates at once after releasing the vascular clamps.
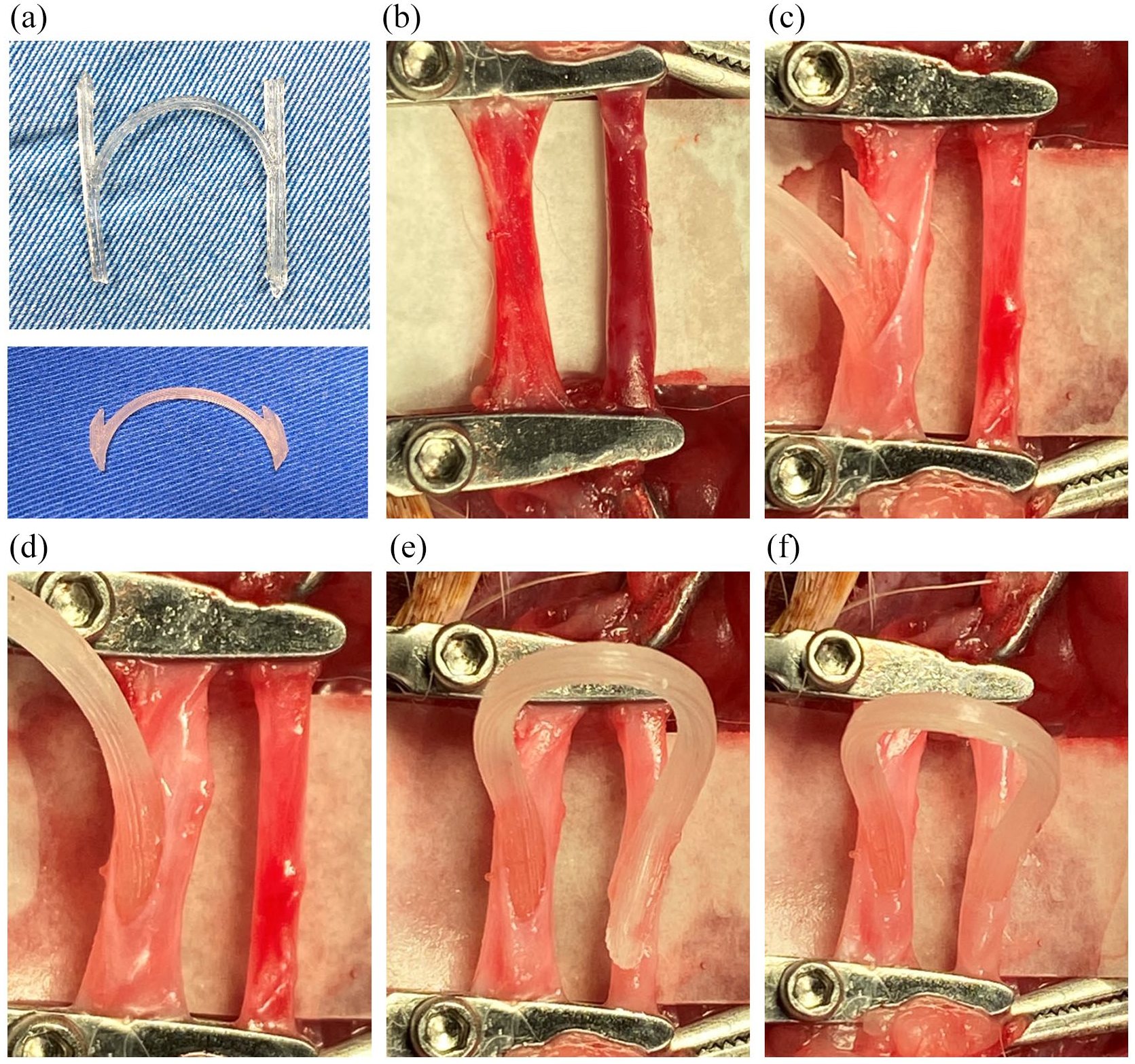
The surgical procedure of the 3D-printed arteriovenous graft (AVG) implantation: (a) four limbs of the manufactured 3D-printed AVG (upper panel) were trimmed to proper length (lower panel) for vascular insertion, (b) freed rabbit common carotid artery and common jugular vein were clamped, (c and d) after heparinized saline perfusion, two limbs of AVG were consequently inserted into the vein, and (e and f) the remaining two limbs of AVG were inserted into the artery.

The surgical procedure of the 3D-printed AVG implantation (continued): (a and b) the tissue adhesive was applied to the junction sites, (c) the distal ends of blood vessels were released, and (d–g) once the proximal ends of blood vessels were released, the blood recirculated at once, as revealed by serial high-speed photographs. The AVG was functioning in situ.
In vitro biomaterial recovery property analysis of the ePTFE AVG versus the EVA-based 3D-printed AVG
Firstly, we quantified the water flow rate within the ePTFE AVG and the EVA-based 3D-printed AVG, whose internal diameters are both 6 mm. Next, the biomaterial recovery property was analyzed by quantifying the water amount leaking from the needle puncture hole within a fixed period of time using the 16-gage needle, which is the size that clinicians use on patients during hemodialysis cannulation.
Results
In vitro endothelial cell adhesion and viability on the EVA surface
Compared to the negative and positive control groups, ECs adhered well to the EVA film on day 1 and remained viable on day 3 (Figure 1). The EVA copolymer with the proportion of 40% vinyl acetate has both desirable printability and good biocompatibility with the vascular cell.
Blood loss during the surgery
Since the vessel wall incision was short in length with no suture, the blood loss was minimal during the surgery. Five minutes after applying tissue adhesive at the vessel wall incision site, the vascular clamps were released (Figure 5(c)). Incisional site bleeding was minimal to trivial, which was easily managed through additional tissue adhesive application.
Measurement of blood flow velocity within the AVG
Blood passed through the AVG from the common carotid artery to the common jugular vein with velocity so fast that the measurements had to be taken with a high-speed camera (Figure 5(d)–(g)). The videos were presented at regular speed and slow-motion in Supplemental Videos S2 and S3, respectively (Supporting Information). The blood only took 0.203 ± 0.068 s to pass through the AVG, and the corresponding blood flow velocity was 14.9 ± 3.7 cm/s.
AVG patency and surgical complications
AVG relies on the cooperation of the subject to avoid compression, where restriction of movements is essential. We are not able to control the motion of the rabbit’s neck after general anesthesia wears off. Meanwhile, small-diameter vascular implants (e.g. <6 mm) are characterized by low long-term patency due to thrombogenicity. 19 Therefore, we focused on the peri-operative period. After the AVG was placed, we observed for another 30 min before we closed the wound. We found that all six AVGs were patent in all six rabbits (100%). There was no hematoma formation after the surgery, at least for 1 week.
In vitro biomaterial recovery property analysis between the ePTFE AVG versus the EVA-based 3D-printed AVG
Firstly, quantification of access flow rate shows no difference between ePTFE and EVA-based 3D-printed AVG. Next, quantification of water leakage from the needle puncture hole showed that the 3D-printed AVG significantly leaked less water than the ePTFE AVG did. This finding indicates that the recovery of the puncture hole is better in the EVA-based AVG than in the ePTFE AVG, probably because the EVA copolymer possesses a glass transition temperature lower than room temperature (Figure 6).
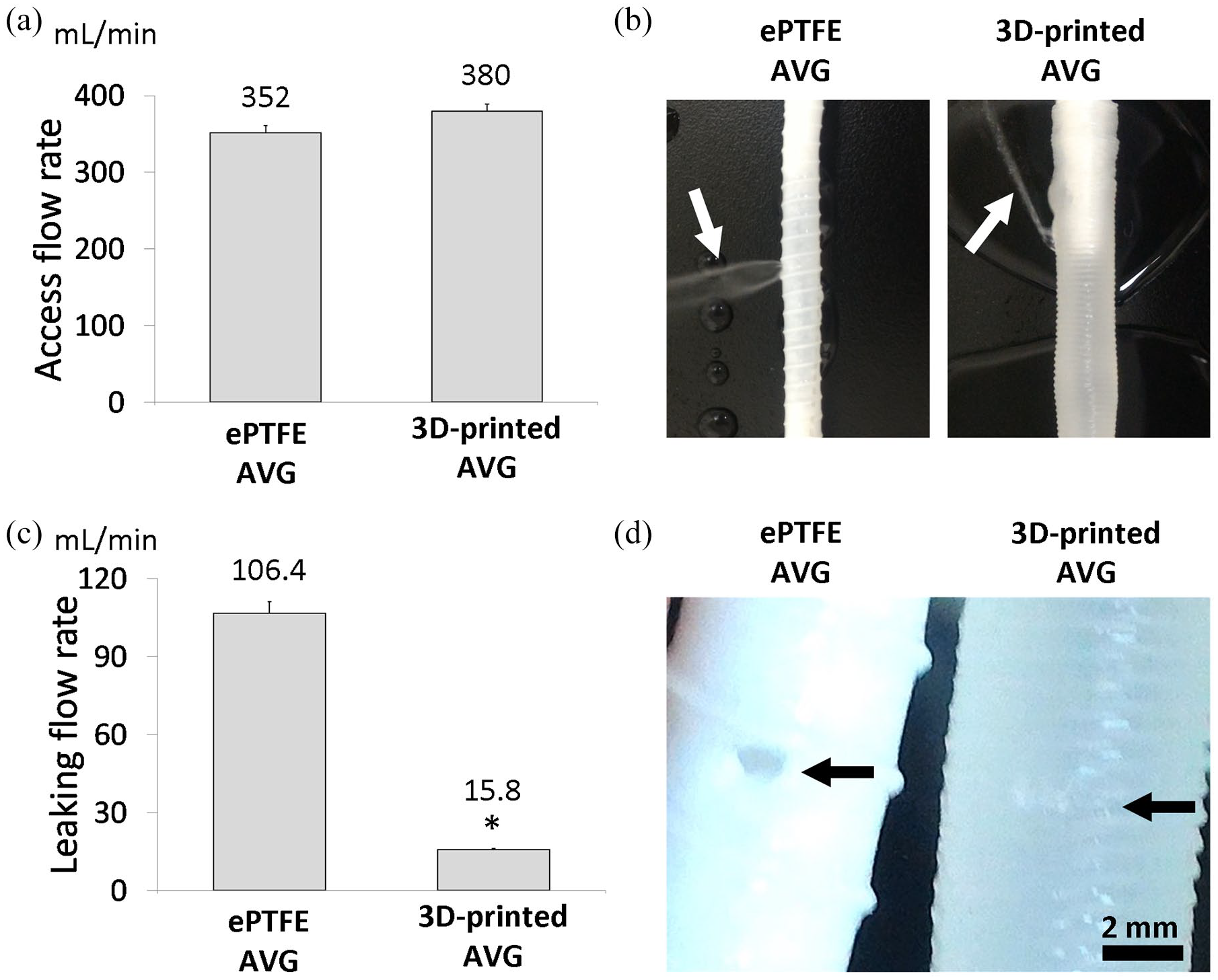
The needle puncture and water leakage tests on the arteriovenous grafts (AVGs) were performed using the 16-gage needle, which is the size that clinicians use on patients during hemodialysis cannulation. The tests were compared between expanded polytetrafluoroethylene (ePTFE) AVG and ethylene-vinyl acetate (EVA)-based 3D-printed AVG, whose internal diameters are both 6 mm. (a) Quantification of access flow rate shows no difference between ePTFE and 3D-printed AVG. (b) The 3D-printed AVG leaks less water than ePTFE AVG (white arrows). (c) Water that leaked via the puncture hole was collected and recorded. Quantification of water leakage from the puncture hole showed that the 3D-printed AVG significantly leaked less water than the ePTFE AVG did. (d) the recovery of the puncture hole is better in 3D-printed AVG than in the ePTFE AVG (black arrow).
Discussion
Our results indicated that 3D printing allows the manufacture of individual-specific EVA-based AVGs with the sutureless techniques to connect the graft and blood vessels. The procedure is quick and straightforward, with a precise fit based on vessel size. Furthermore, the blood loss is minimal. To the best of our knowledge, we are the first to successfully tailor-made 3D-printed AVGs proven to be functional in vivo.
3D printing, also known as additive manufacturing, provides significant innovations in many areas, such as engineering, manufacturing, art, education, and medicine. Recent advances have enabled the 3D printing of biocompatible materials, cells, and supporting components into complex and functional living tissues. Wang et al. 20 have demonstrated that 3D printing in conjunction with clinical image reconstruction is a promising method for coronary artery disease research. This finding echoes the present study, in which we utilized the preoperative CT scan to evaluate the vessels to be implanted and then fabricated the vessel-specific AVG accordingly.
An optimal 3D biological structure should possess multiple functional, structural, and mechanical components and properties.21 –24 The printed construct can then be transplanted in vivo, in some cases after a period of in vitro maturation, or is reserved for in vitro analysis. However, the multifunctional vascular grafts from the synthetic polymers, anti-thrombogenic additives, and peptides by 3D bioprinting technology, as well as study the controlled release of anti-thrombogenic additives from the grafts, have been explored only sporadically. Once introducing these additives to our tailor-made AVG, we can expect promising results in future studies.
We tested the EVA-based 3D-printed AVG in animals and confirmed that the surgical procedure was quick and straightforward. We have also verified the immediate graft patency. There is no synthetic AVG model in small animals since the long-term patency of AVG depends on the internal diameter of AVG.25 –31 Only an autologous venous graft model in rabbits has been reported, in which an excised external jugular vein graft was anastomosed to replace a removed segment of the ipsilateral common carotid artery in an end-to-end manner.32,33 Therefore, our 3D-printed AVG should be examined for a long-term patency rate using the large animal model.
Additionally, the ideal tissue-engineered vascular graft should fulfill several requirements. The graft needs to be covered by thrombogenic ECs that are resistant to thrombosis. It should also have comparable biomechanical properties to avoid the intimal thickening, which could have serious consequences such as loss of graft patency, proteolytic degradation of extracellular matrix, and development of degradation-associated vascular dilation or aneurysm.34 –36 Thus, the prevention of thrombosis, vascular intimal thickening, and aneurysm formation is essential for a tissue-engineered vascular graft’s successful post-implantation function. Further large animal studies are warranted to examine long-term EC behaviors on the 3D-printed AVG.
The small-sized (⩽4 mm) prosthetic vascular graft is easily thrombogenic because of the lack of an endothelial layer inside the graft even after long-term implantation. However, such a shortcoming does not affect the clinical performance of large-sized vascular implants. Therefore, scientists urged a proper selection of animal models for a specific study purpose.37,38 Given the fact that the failure of small-sized vascular grafts does not mainly depend on the biomaterial or properties of the prosthesis, long-term graft patency should be examined in a large animal model.28,39 The present study is proof-of-concept research that mainly focused on the 3D-printing technique elaborating its advantage by customizing the shape and size of vascular implants. Further large-animal studies are warranted to confirm the long-term patency of this tailor-made 3D-printed vascular graft in large sizes.
Several questions may be answered in future studies. First, long-term graft patency should be examined in the large animal whose vessel size is closer to human forearm vessels—for example, the porcine model of carotid-jugular AVG. Second, cannulation of AVG should be tested in the large-size 3D-printed AVG. Nevertheless, the present study demonstrated the realization of in vivo functioning 3D-printed AVG.
By applying the 3D printing technology, our study demonstrates that EVA-based AVG for hemodialysis can be tailor-made. The surgical procedure can be done with ease, blood loss is minimal, and most exceptionally, no suture is required. Although this study was conducted in small animals, it proves that the 3D-printed AVG functions in vivo. Further studies in large animal models are warranted to validate our promising results.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298221086173 – Supplemental material for Functioning tailor-made 3D-printed vascular graft for hemodialysis
Supplemental material, sj-pdf-1-jva-10.1177_11297298221086173 for Functioning tailor-made 3D-printed vascular graft for hemodialysis by Ming-Chia Li, Pu-Yuan Chang, Huai-Rou Luo, Ling-Yuan Chang, Chuan-Yi Lin, Chih-Yu Yang, Oscar Kuang-Sheng Lee, Yan-Hwa Wu Lee and Der-Cherng Tarng in The Journal of Vascular Access
Footnotes
Acknowledgements
We thank Bo-Sheng Wu (Institute of Clinical Medicine, School of Medicine, National Yang Ming Chiao Tung University, Taipei, Taiwan) for his English editing and valuable input.
Author contributions
Ming-Chia Li: Conception and design, acquisition of data, data analysis and interpretation, manuscript writing, final approval of the manuscript. Pu-Yuan Chang: Acquisition of data, data analysis and interpretation, manuscript writing, final approval of the manuscript. Huai-Rou Luo: Acquisition of data, data analysis and interpretation, manuscript writing, final approval of the manuscript. Ling-Yuan Chang: Acquisition of data, data analysis and interpretation, manuscript writing, final approval of the manuscript. Chuan-Yi Lin: Acquisition of data, data analysis and interpretation, manuscript writing, final approval of the manuscript. Chih-Yu Yang: Conception and design, financial support, administrative support, provision of study material, acquisition of data, data analysis and interpretation, manuscript writing and revising, final approval of the manuscript. Oscar Kuang-Sheng Lee: Conception and design, financial support, manuscript revising, final approval of the manuscript. Yan-Hwa Wu Lee: Conception and design, financial support, manuscript revising, final approval of the manuscript. Der-Cherng Tarng: Conception and design, financial support, manuscript revising, final approval of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Institutional Animal Care and Use Committee (IACUC) of the Taipei Veterans General Hospital (#2020-034) on April 18, 2020.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support for research purposes by the “Yin Yen-Liang Foundation Development and Construction Plan” of the School of Medicine, National Yang-Ming University, Taipei, Taiwan (107F-M01-0504 and 107F-M01-0510), the Ministry of Science and Technology (MOST), Taiwan (MOST 105-2628-B-010-017-MY3, MOST 110-2221-E-A49-002-MY3, MOST 108-2923-B-010-002-MY3, MOST 108-2813-C-010-034-B, MOST 109-2314-B-010-053-MY3, MOST 109-2926-I-010-502, MOST 109-2823-8-010-003-CV, MOST 109-2811-B-010-532, MOST 110-2811-B-010-510, MOST 110-2813-C-A49A-551-B, MOST 110-2923-B-A49A-501-MY3, and MOST 110-2321-B-A49-003), grants from Taipei Veterans General Hospital, Taipei, Taiwan (V111C-155, V111D63-003-MY2-1, VGHUST111-G6-7-2), and the “Center for Intelligent Drug Systems and Smart Bio-devices (IDS 2 B)” from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. The funders have no role in study design, data collection, analysis, interpretation, or manuscript writing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
