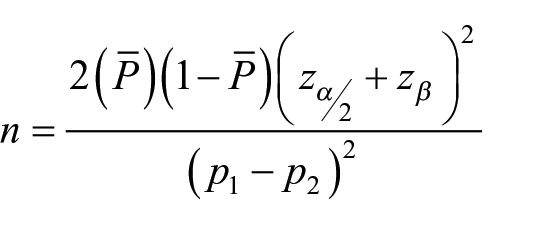Abstract
Background:
Peripheral intravenous catheters (PIVCs) are frequently used in clinical settings for intravenous access. Multiple attempts of PIVC insertions leads to patient discomfort, delay in treatment, associated complications, and extensive expenditure cost. Reduced number of attempts causes patient/nursing personnel satisfaction and expenditure costs. The present study evaluated performance efficacy of BD Venflon™ I with Instaflash needle technology (investigational device) as compared to the BD Venflon™ without Instaflash needle technology (control device).
Methodology:
The PIVC insertions were randomized in the ratio 1:1 using either investigational or control device and were monitored for first stick success rate, ease of insertion, and patient satisfaction. Data was analyzed using R 4.0.3 and Microsoft Excel. Chi square test was used to establish association between two categorical variables.
Results:
In total, 1402 patients were analyzed for first attempt insertion success which showed 98.72% success rate in investigational device as compared to 88.87% success rate in case of the control device (p = 0.0004). Marginal differences were observed in ease of insertion in investigational (98.71%) and control devices (99%) signifying high satisfaction levels of nursing personnels. Positive responses were observed in investigational (98.01%) and control devices (99%) underlining satisfactory performances of overall patient experiences.
Conclusion:
The present study showed that BD Venflon™ I with Instaflash needle technology enhanced first attempt insertion success rate along with marginal differences in its efficacy in comparison with the BD Venflon™ without Instaflash needle technology thus enhancing patient and nursing personnel satisfaction in turn making it a better alternative to be used in hospitals.
Introduction
Peripheral intravenous catheters (PIVCs) have been used for intravenous access of about 70% patients present in acute care hospitals. 1 Currently, PIVCs are the most common choice among the vascular access devices in clinical settings. PIVC insertion is one of the most common as well as amongst highly invasive clinical procedures.2,3 Insertion of PIVCs can be extremely challenging even for skilled personnel which in turn results in increased attempts of cannulation and thereby, causing delay in patient treatment, increase in complications, as well as reduction in patient satisfaction. 4
PIVC insertion procedures practiced currently are of sub-optimal standards. The rates of first-attempt peripheral insertion failure have been identified to be in the range 12%–26% for adults whereas 24%–54% for children. 5 There are several studies stating number of factors influencing the rate of success involved in first-time PIVC insertion procedures. A recent study elaborated different patient related factors like age, gender, body mass index, swelling, obesity, dialysis status, vasculature, diseases like diabetes and sickle-cell anemia, history of chemotherapy, as well as history of recent hospitalization which influence PIVC insertion failure. 6 Multiple number of insertion attempts has been observed to cause pain, stress, increased infection rates, and decreased satisfaction in PIVC patients.7,8 Hence, significant reduction in number of attempts required for PIVC insertion should be a major concern. The reduced number of attempts for PIVC insertion in turn results in saving expenditure considering the expenses affecting treatment options, hospital stay, and use of disposable PIVC packs per attempt. 9 Thus, it is important to make use of various assessment tools as well as clinical prediction rules to bring about improvement in PIVC insertion procedures.10,11
There are several studies which evaluate ease of PIVC insertion along with its short term and long-term efficacy.12,13 However, such studies lacking from the perspective of adult Indian population. Also, there is need of comparative analysis of currently used PIVCs with that using Instaflash needle technology to understand its efficiency in first stick success. Therefore, present study was designed to do a comparative analysis of BD Venflon™ I with Instaflash needle technology with the BD Venflon™ without Instaflash technology in hospitalized patients requiring PIVC insertion.
Materials and methods
Study design and devices
This was a prospective, non-blinded, randomized, controlled, post-market study which was performed in three different units of a tertiary care hospital in Bangalore, Karnataka, India. The present study was carried out for a time duration of 120 days between January 2021 and April 2021 from screening of patients up to data collection. PIVC insertions were randomized in the ratio 1:1 using either investigational or control device by participating nursing personnels. The study was monitored and approved by the Institutional Ethics Committee and registration of the study in Clinical Trial Registry, India (REF/2021/06/044189). All the PIVC insertions were performed depending upon clinical status of the patients by following standard precautions.
In the present study, BD Venflon™ I with Instaflash needle technology IV Catheter (BD Medical, Franklin Lakes, NJ, USA) was used as the device under study (investigational device) whereas BD Venflon™ IV Catheter (BD Medical, Franklin Lakes, NJ, USA) was used as the control device present under current use. Gauge of PIVC used is 18G and 20G. Study IV catheter that is, BD Venflon™ I is present with a notch stylet, while BD Venflon™ doesn’t, thereby facilitating early visualization of blood flashback during PIVC access. Primary outcome of this study was assessment of first stick success of PIVC insertion procedures, secondary outcomes evaluated ease of insertion and confidence while using BD Venflon™ I with Instaflash needle technology. In addition, the tertiary outcomes evaluated degree of patient satisfaction.
Study population
Nursing personnels authorized to perform PIVC cannulation with varied years of experience inserting at least two PIVCs per week for a minimum period of 3 months were included in this study. The baseline demographic information and PIVC insertion experience of all the eligible nursing personnels participating in the study was recorded. These nursing personnels were trained for the study protocol, BD Venflon™ I PIVC Catheters along with its insertion technique for a period of 12 h within a week. Training included video demonstration as well as observation of procedure performed by experts and a hands-on with BD Venflon™ I. A total of 28 nursing personnels participated in the study where each nursing personnel performed 50 insertions in total.
A total of 1402 patient volunteers with age between >18 and <60 years were included in the study. Prevalence of patients experiencing pain during PIVC insertion with and without Instaflash needle technology has been assumed to be 50% and 60% respectively with 95% confidence level and 90% power. A minimum of 520 subjects per group has been calculated by using the following sample size calculation:
where n is the required sample size per group, p1 and p2 is the percentage occurrence of a state or condition (proportion or prevalence), z
All the patients who were required to receive a PIVC and were willing to participate in the study were enrolled as a study subject after signing of informed consent. However, pediatric and geriatric patients as well as pregnant and lactating women were excluded from the study.
Data collection
All the data was collected and recorded after obtaining informed consent as well as patient demographic details. Simple 1:1 randomization was performed using investigational device or control device. Similarities are observed between primary diagnosis as well as type of admitting patients between groups. Depending upon the primary diagnosis, reason for cannulation as well as site of insertion was determined in all of subject patients.
For each PIVC insertion, one nursing personnel was allowed a total of two venepuncture attempts to gain vascular access. In case of insertion failure, PIVC insertion was repeated by a different nursing personnel or clinician. The needle size was also changed in the case of failure beyond the second attempt which would in turn result in higher degree of pain in those patients for both cases. An observer observed the insertion process and recorded the data regarding number of attempts, first stick success, nurse satisfaction score, as well as patient satisfaction score for each successful attempt.
Study outcomes
Primary outcome of this study was assessment of first stick success of PIVC insertion procedures, secondary outcomes evaluated ease of insertion and confidence while using BD Venflon™ I with Instaflash needle technology whereas the tertiary outcomes evaluated degree of patient satisfaction. For every successful first attempt insertion, observer made a note regarding number of attempts required as well as indicated taken for successful insertion and also indicate their agreement using a 5-point Likert’s scale of satisfaction with remarks like strongly disagree, disagree, neutral, agree, and strongly agree using the scoring system of 1 for strongly disagree as well as 5 for strongly agree.
Statistical analysis
Data is analyzed using statistical software R 4.0.3 and Excel. Categorical and continuous variables were presented in the form of frequency and mean ± standard deviation. Chi square test was used to check the association between two variables. p ⩽ 0.05 was considered as statistically significant.
Results
A total of 1402 patients participated in the present study with mean age 45.42 ± 13.04 years. Out of these 1402 patients, there were 818 males (58%) and 584 females (42%).
All the patients were allocated to one of two treatment groups, either investigational device or control device, with 701 patients using simple randomization in 1:1 ratio. There were no reports regarding randomization errors. There were no significant differences observed between the control and treatment groups in various subject characteristics which were measured during the study.
Tables 1 and 2 provide demographic data regarding nursing personnels as well as participating subject patients respectively:
Demographic data of nursing personnels.

Demographic data of subject patients.

All the patients were treated using BD Venflon™ I with Instaflash needle technology as well as BD Venflon™ without Instaflash needle technology. The design of the two catheters were similar and the main difference was visualization of primary blood return. The visualization was within the catheter lumen with BD Venflon™ I and in the flash chamber with BD Venflon™. Patients were cannulated for medication via intravenous routes (90.8%), infusion therapy (5.49%), blood transfusion (2.14%), radiological investigation (0.78%), as well as other causes (0.78%). Different sites used for cannula insertion included metacarpal (58.63%), cephalic (21.68%), basilic (12.2%), antecubital (7.06%) along with other locations (0.43%) as well.
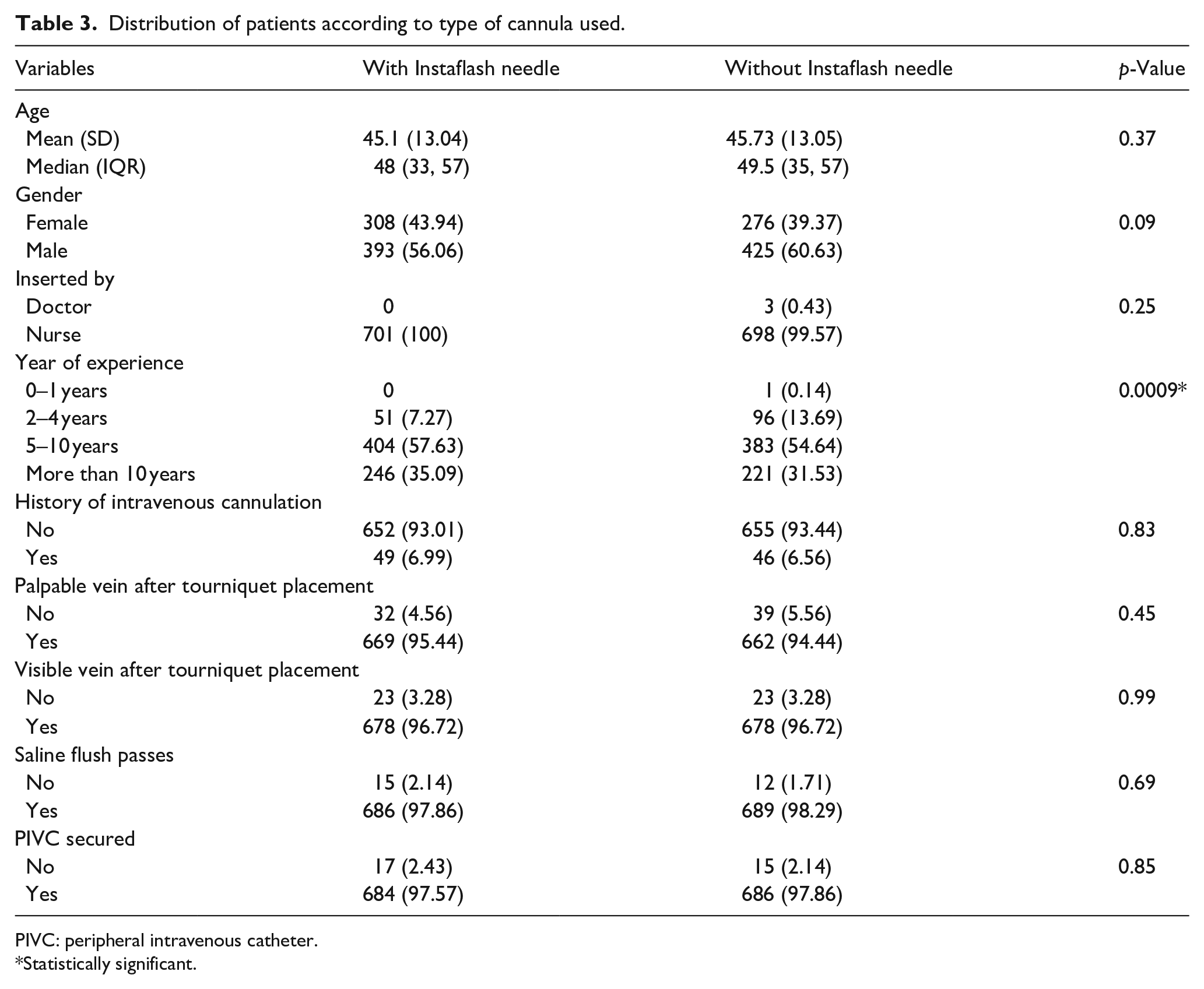
Comparative analysis was performed using BD Venflon™ I with Instaflash needle technology as well as BD Venflon™ without Instaflash needle technology for various parameters so as to estimate the superiority of the investigational device over the currently used control device. Even though there was no associated bias between the experience levels of nursing personnels and use of investigational or control device, it was observed that experience of nursing personnels and first attempt success of PIVC insertion procedures show high levels of association with p value = 0.0009 using Chi square test which is highly significant.
Table 3 represents different parameters monitored according to the type of cannulation:
Distribution of patients according to type of cannula used.
PIVC: peripheral intravenous catheter.
Statistically significant.
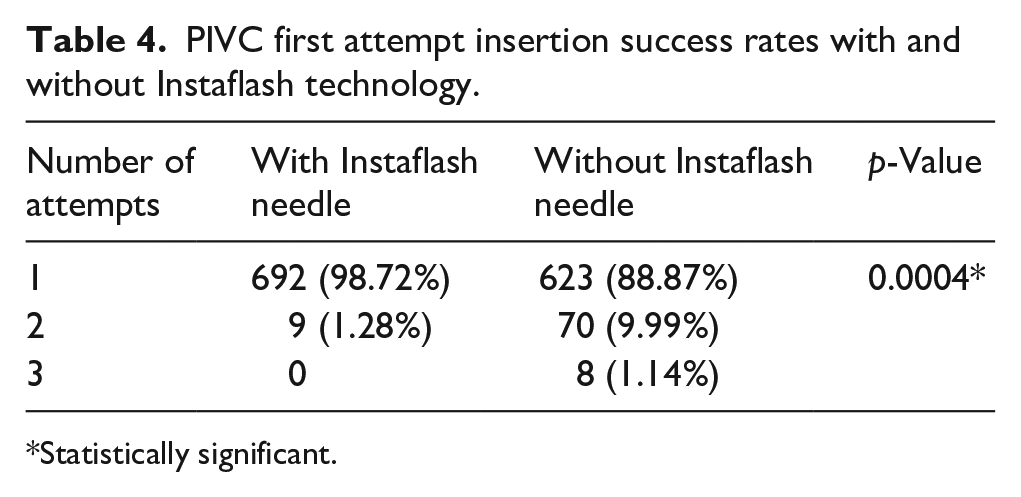
First attempt insertion success was defined as the ability of the nursing personnel to successfully perform PIVC insertion procedures in the subject patients with a maximum of two venepuncture attempts. Table 4 represents first attempt insertion success rates of PIVC with and without Instaflash technology. For the investigational device with Instaflash needle technology, 692 out of the 701 insertions (98.72%) were reported to be successful in the first attempt in comparison with 623 out of the 701 insertions (88.87%) for the control device without Instaflash needle technology. Moreover, no third attempts were required in case of investigational device as compared to eight insertions (1.14%) in case of control device. Using Chi square test, first attempt insertion success rates of the investigational device are significantly higher as compared to control device with positive association between first stick success and use of investigational device with p value = 0.0004.
PIVC first attempt insertion success rates with and without Instaflash technology.
Statistically significant.
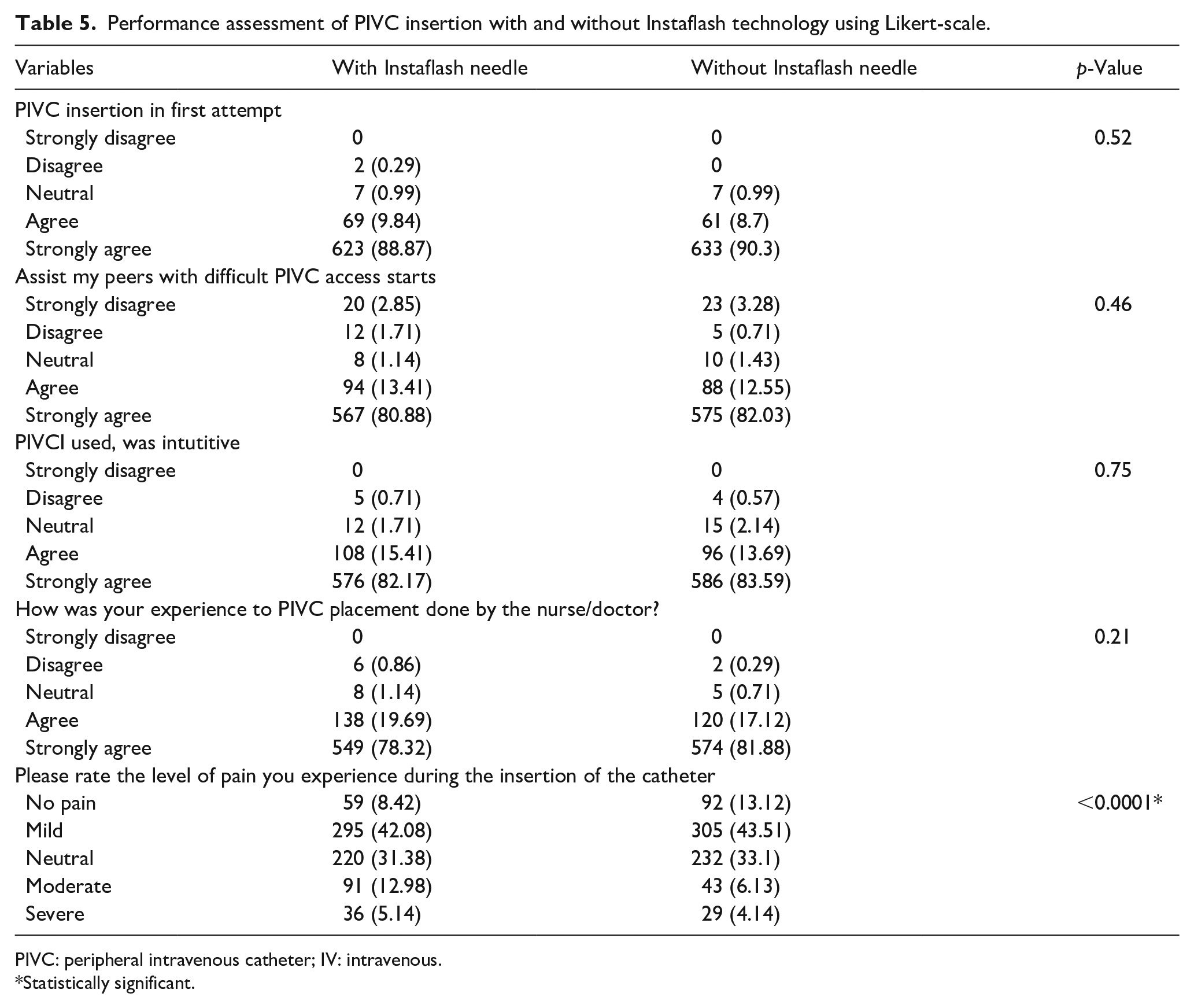
Clinical usability was used to assess the performance of PIVC insertion using various parameters like first attempt insertion success, assistance during multiple attempts, intuitive nature of PIVC, level of pain experience during PIVC insertion, and overall experience after each insertion. The nursing personnels as well as subject patients submitted Likert-scale rated responses about their agreement levels on the scale of 5. Comparative analysis has been done regarding efficiency of PIVC with and without Instaflash technology by assessing responses of subject patients as represented in Table 5.
Performance assessment of PIVC insertion with and without Instaflash technology using Likert-scale.
PIVC: peripheral intravenous catheter; IV: intravenous.
Statistically significant.
After overall assessment of both the devices, positive feedback was given regarding the clinical usability of both devices. Clinicians and nursing personnels stated that the investigational device with Instaflash needle technology showed higher insertion rates in first attempt as compared to control devices without Instaflash needle technology even in case of nursing personnels with low experience which could help in reducing multiple PIVC insertions.
Discussion
PIVC insertion procedures are amongst the most common invasive procedures frequently used in clinical settings. Safety and durability are two crucial parameters to be considered by choosing the type of catheters.14,15 Failures during PIVC insertion procedures are quite frequent, because of which multiple attempts are needed for its successful insertion. Multiple number of insertion attempts results in complications, delay in treatment, lack of patient satisfaction, as well as higher economic expenditure.16–18 In order to overcome these shortcomings, newer technologies are being developed to be used in PIVC insertion procedures so as to decrease the number of attempts. Thus, first attempt insertion success would be better as a result of seeing blood return quicker and thus avoiding through puncture of the vein/transfixation.
The present study was focussed on minimizing the number of attempts at PIVC insertion using Instaflash needle technology. Satisfaction levels of nursing personnels and subject patients during the use of PIVC with and without Instaflash technology were also monitored through their responses. The results observed while using the investigational and control device are quite similar. Remarkable improvement in PIVC insertions with first stick success of 98.72% in investigational device as compared to 88.87% of the control device was observed while maintaining the current standards of performance as well as clinical accessibility. Minimizing the number of attempts involved in PIVC insertion procedures has been associated with reduction in the patient treatment cost. 19 Reduced insertion attempts are also associated with higher satisfaction levels in clinicians and nursing personnels due to ease of operation.20,21 Affirmative responses regarding PIVC insertion in single attempt were observed for both investigational (98.71%) as well as control devices (99%) which emphasized on similar ease and satisfaction while using BD Venflon™ I with Instaflash technology as well as BD Venflon™ without Instaflash needle technology. The positive responses also underlined similar satisfaction levels of subject patients during PIVC insertion with (98.01%) and without Instaflash technology (99%). Thus, similar satisfaction levels of subject patients and nursing personnels with higher stick success rate makes BD Venflon™ I with Instaflash technology a more promising alternative to be used for PIVC insertion.
However, this study shows some associated limitations as well. As this study majorly covers adult population, its use cannot be generalized for all age groups. Evaluation of pediatric population can further help in understanding the usability of the device in that respective age group. Moreover, more detailed analysis of catheter characteristics as well as associated complications and ways to counter them needs to be studied as these factors might affect the overall efficiency of the investigational device in first attempt PIVC insertions.
Conclusion
The present study evaluated performance efficacy of BD Venflon™ I with Instaflash needle technology as compared to the BD Venflon™ without Instaflash needle technology on the basis of several factors. Higher rates of first attempt success makes the investigational device a better alternative over the control device. However, detailed evaluation regarding catheter dwelling time and associated complications is necessary for better understanding regarding the choice of catheter to be used.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partially funded by BD.