Abstract
Purpose:
To report the patency rates after implantation of an interwoven nitinol stent to salvage failing arteriovenous grafts (AVGs) caused by intragraft stenoses.
Methods:
Between May 2018 and May 2020, 21 Supera stents were placed in 20 patients (18 women; mean age: 79.9 years) who had a failing AVG due to repeat intragraft stenoses. Recurrent AVG dysfunction with same intragraft stenosis within 3 months after first time angioplasty was a criterion for stenting. Those with concurrent treatment for other lesions were excluded.
Results:
The technical success rate was 100%. Intragraft stenoses were treated at a median of 19.7 (interquartile range: 6–36) months after graft creation. Access circuit primary patency rates after stent placement were 84% and 35% at 6 and 12 months, respectively. Access circuit secondary patency rates were 100% at 6 and 12 months and 89% at 18 months. Only one patient presented with graft failure due to proximal drainage vein occlusion. The target lesion patency rates were 100% at 6 months and 75% at 12 months. The rate of reintervention for intragraft lesion was 0.15 procedures per year. Stent distortion did not occur under regular cannulation.
Conclusion:
The interwoven nitinol stent is a promising treatment for failing AVGs with recurrent intragraft stenoses. The 1-year access circuit primary, secondary, and target lesion patency rates were acceptable, with a low reintervention rate. Stent fracture does not occur in areas of needle puncture.
Introduction
The National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI) recommends arteriovenous fistula (AVF) as the first access for hemodialysis (HD) but arteriovenous grafts (AVGs) are still an appropriate option for vascular access in certain HD patients. 1 The use of AVGs has historically been hampered by low patency rates, with reported secondary patency of 76% at 6 months and 55% at 18 months. 2 In one study, 77% of AVGs developed stenosis or thrombosis within the first year. 3 Hemodynamically significant stenosis at the venous anastomosis is the most common cause of prosthetic AVG dysfunction in patients with HD, followed by arterial anastomosis and venous outflow stenosis. 4 The underlying pathogenesis of these stenoses is neointimal hyperplasia resulting in progressive luminal narrowing.5,6 The rate of intragraft stenoses varies from 6% to 20%.4,7 Unlike stenotic lesions occurring at other sites, stenoses within the AV graft is correlated with repeat needle puncture, causing fibroplastic ingrowth via needle puncture tracts and graft structure distortion.8,9 The primary patency rates of intragraft stenoses after conventional balloon angioplasty range from 56% to 75% at 6 months and 18% to 33% at 1 year.7,10 For recurrent lesions, there are only few studies analyzing the outcomes of deploying a stent on intragraft stenoses. Although the results were acceptable, the use of stents within AVGs has been associated with stent fracture due to repeat puncture. 9 Supera stent is made of nitinol wires woven around a central lumen and is not laser cut from a nitinol tube, like most other nitinol stents. This interwoven design is resistant to kink formation, especially during the compression. 11 Therefore, it can maintain the integrity of graft lumen and allow for repeat needle puncture. The current study aimed to report our experience of using an interwoven nitinol stent for salvaging failing synthetic AVGs in patients with recurrent intragraft stenoses.
Materials and methods
Procedural database review was performed to identify all patients with intragraft stenoses treated with interwoven nitinol stents from May 2018 to May 2020. In total, 20 patients (18 women and 2 men; mean age: 79.9 (range: 38–89) years) who had failing AVGs due to recurrent intragraft lesion within 3 months were treated with the Supera stent (Abbott Vascular, Santa Clara, CA, USA). This retrospective study was approved by the institutional review board. The requirement for consent was waived. Data including patient demographics, hemodialysis parameters, and procedural details were collected. The indication for intragraft stenosis treatment was significant stenosis (>50%) on ultrasonography or angiography, which meets the treatment criteria established by the KDOQI. 1 Meanwhile, those with concurrent treatment for other lesions, particularly venous anastomosis, were excluded. Intragraft stent placement was the only nonsurgical approach that can preserve graft function.
All procedures were performed by a single endovascular surgeon in a hybrid operating room while the patient was under local or general anesthesia. All intragraft stenoses were initially treated with balloon angioplasty using a 6-, 7-, or 8-mm diameter high-pressure noncompliant balloon. If intragraft stenosis was the main culprit lesion for reintervention, stenting was performed. If the waist of the stenosis did not resolve with a high-pressure angioplasty balloon catheter, stenting was not performed. Considering the mechanism underlying intragraft lesion correlated with injury caused by needle cannulation, different stenosis statuses were found over both puncture sites for all patients. For this reason, all patients required a long stent (12 or 15 cm in length) to cover the whole AVG area, including both arterial and venous puncture sites. The AVGs of all patients were created using 6-mm ringed polytetrafluoroethylene (PTFE) grafts (WL Gore & Associates, Inc., Flagstaff, AZ, USA). The stent diameter was selected to match the graft diameter dilated by the angioplasty balloon. The 6.5-mm Supera stent was preferred if a 6- or 7-mm balloon was used for dilatation. The 7.5-mm Supera stent was only selected if a balloon with 8-mm diameter was used. All stent deployments were conducted via the retrograde access, starting 2 or 3 cm away from the arterial anastomosis and extending toward the venous anastomosis (Figure 1). If a covered stent was over the venous anastomosis, the Supera stent was extended into it. If there was no covered stent over the venous anastomosis, the stent was deployed 1–2 cm close to venous anastomosis without extending over it. For forearm AVG, the proximal drainage vein was commonly selected for the retrograde access. If a covered stent was found over the venous anastomosis or proximal drainage vein, the preferred route was direct puncture over the covered stent and stent placement was performed via a 6Fr sheath introduced in retrograde method. For upper-arm AVG, due to a shortage of relatively superficial proximal drainage veins (commonly axillary or subclavian veins), it is difficult to perform retrograde access for stent deployment since the proximal drainage vein was relatively deep to approach. The femoral vein was considered the preferred retrograde access for stent placement. Either right or left femoral vein puncture was obtained after decision for stenting was made following angioplasty of upper arm AVG. A long 6Fr sheath was introduced and a 0.018-inch wire was used to cross over the subclavian vein, venous anastomosis, and finally the arterial anastomosis. After stenting, post-dilatation was then performed and final angiography was obtained.

Angioplasty of intragraft stenosis and deployment of the Supera stent within the graft. (a) Digital subtraction angiography (DSA) after the use of two 6-Fr sheaths for catheter-directed thrombectomy showing severe stenosis over the arterial (white arrow) and venous (open arrow) cannulation sites. The asterisk denotes the covered stent placed over the venous anastomosis site. (b) Fluoroscopic image of the 6.5-mm × 12-cm Supera stent deployed within the AVG, covering both the cannulation sites and extending into the covered stent. (c) Final DSA showing complete expansion and patency of the stent within the whole graft.
Patients were prescribed with clopidogrel daily for 3 months and were followed-up every 3 months. The routine follow-up included clinical examination, measurement of graft flow rates and pressure, and ultrasonography of the AVG. Patients with abnormal findings were referred for further angiographic assessment. Clinical and operative reports were reviewed to identify the incidence of graft surgeries, renal transplantation, and mortality. Adverse events and complications within 30 days after the index procedure were recorded.
Statistical analysis
The data of all patients were collected by a surgical service personnel during workup for the initial procedure and during follow-up for recurrent graft dysfunction. Technical success was defined as <30% residual angiographic stenosis. Access circuit primary patency was defined as the interval following the initial percutaneous intervention on the index intragraft stenosis until the next access thrombosis or percutaneous intervention for a lesion anywhere within the access circuit. Access circuit secondary patency was defined as the interval following the index percutaneous intervention until surgical revision or abandonment of the access circuit. Target lesion patency was defined as the interval following the initial percutaneous intervention until the development of ⩾50% stenosis at the site of the initially treated lesion. A Kaplan–Meier life table analysis was performed to assess access patency rates. Censoring of endpoints was applied to death, renal transplantation, graft resection caused by infection, functioning AVG at the end of the study period, and loss to follow-up.
Results
In total, 21 Supera stents were deployed in 20 patients (Tables 1 and 2). Seventeen (85%) patients were women. The median length of time between the creation of an AVG and the placement of the Supera stent was 19.7 (interquartile range (IQR): 6–36) months. Approximately 95% of these AVGs had recurrent venous anastomosis dysfunction treated with covered stent before.
*Baseline characteristics of the 20 study patients and procedure details.
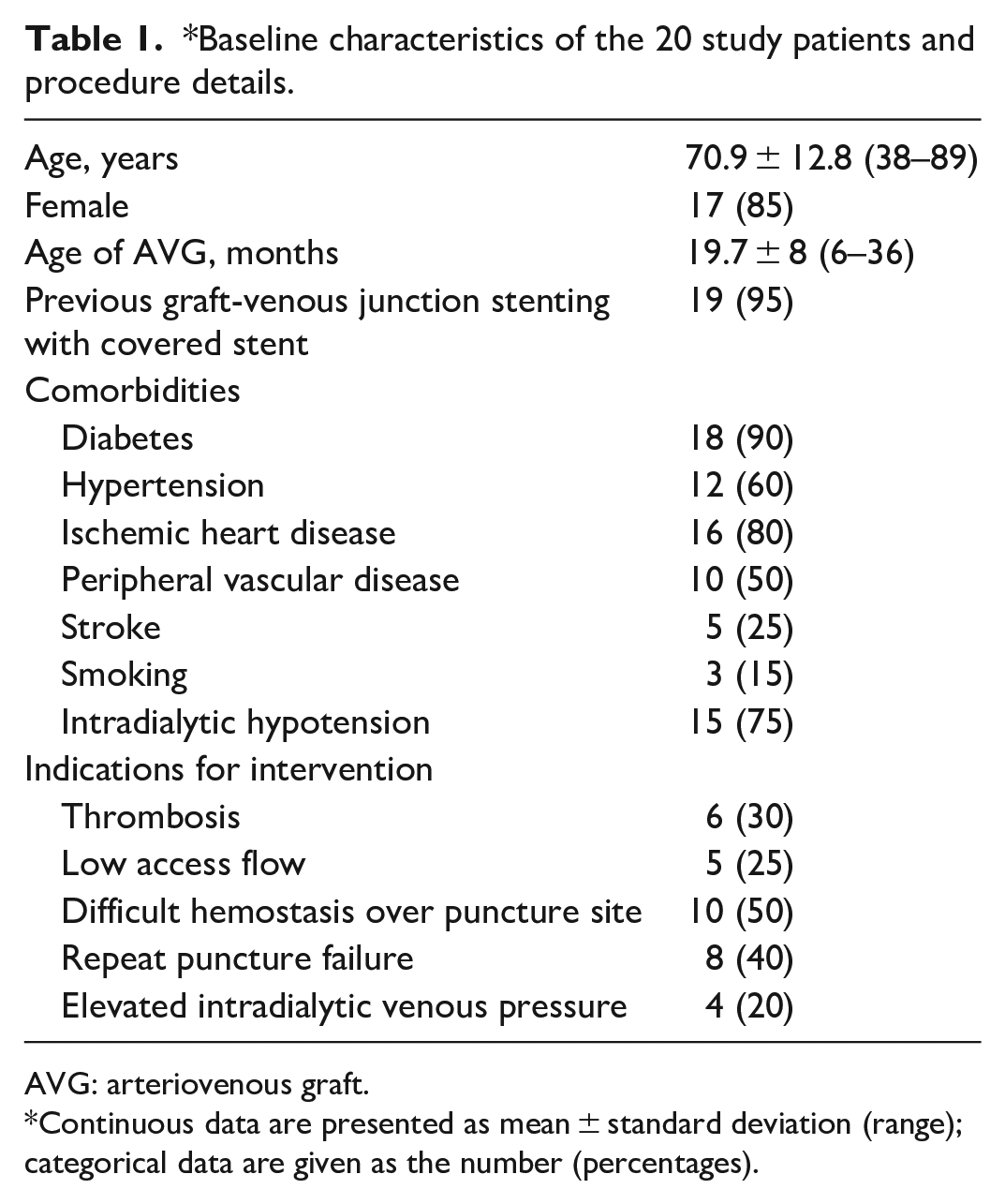
AVG: arteriovenous graft.
Continuous data are presented as mean ± standard deviation (range); categorical data are given as the number (percentages).
*Characteristics of the 20 AV grafts comprising the study population.
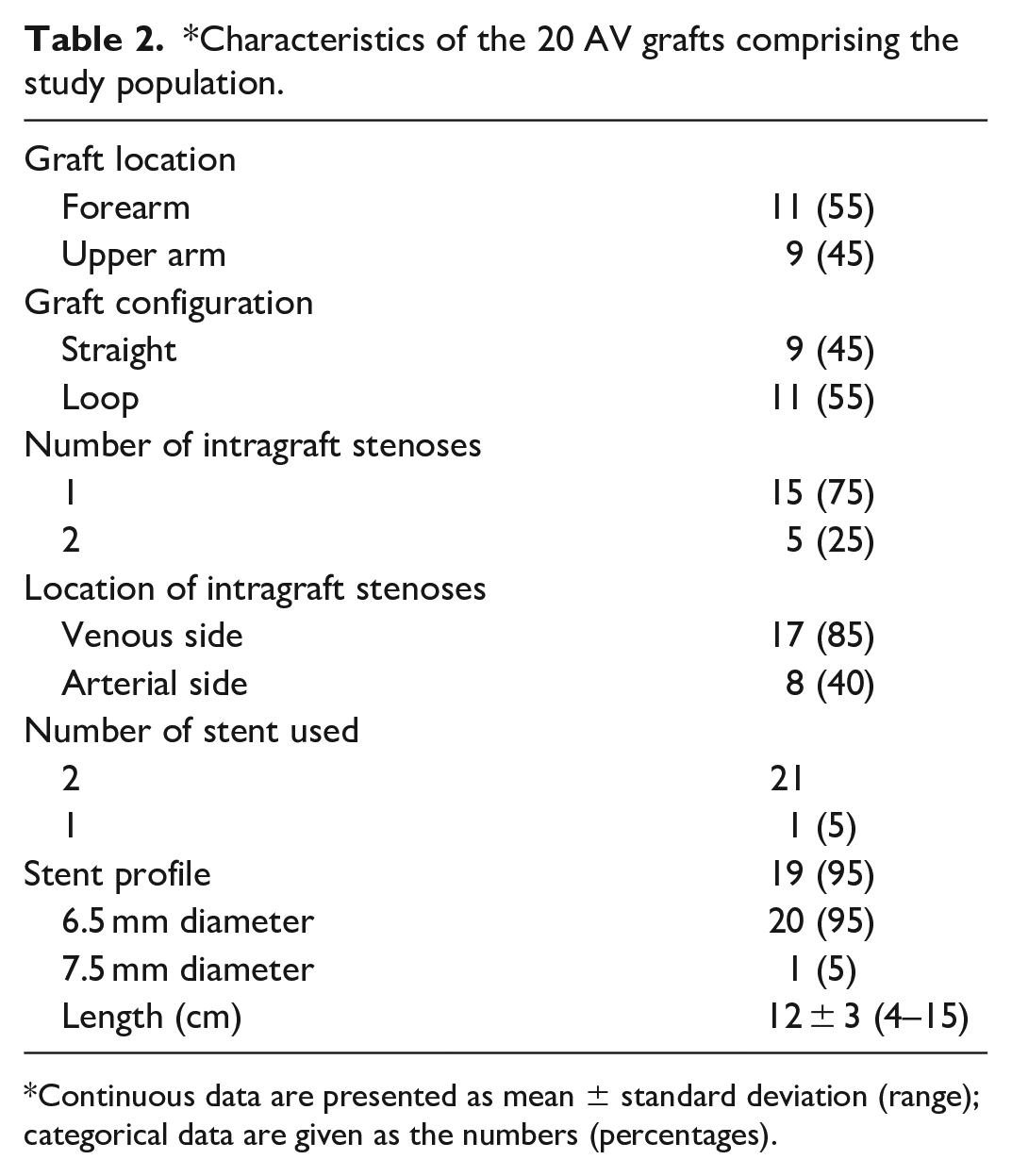
Continuous data are presented as mean ± standard deviation (range); categorical data are given as the numbers (percentages).
All intragraft stenoses were initially treated with a balloon. Recurrent stenoses within 3 months were observed during follow-up. Six (30%) patients presented with acute thrombosis. Seventeen (75%) patients exhibited stenosis over the venous puncture site. Five patients had stenoses over both puncture sites. There was only a patient who had a larger length of AV loop graft over forearm received stenting first over the venous site but developed significant stenosis over the arterial side after 5 months. Another 4 cm Supera stent was placed over the arterial side (Figure 2). In 95% of patients, 6.5-m supera was used for stenting. Only one 7.5-mm Supera stent was used in one patient due to severe recoil and success dilatation with an 8-mm angioplasty balloon. The median stent length was 12 (range: 4–15) cm. The technical success rate was 100%, without any procedural complications. In the retrograde access for stent deployment, direct manual compression was performed on patients with forearm AVGs. For patients with upper-arm AVGs and those who successfully achieved the retrograde access via the femoral vein, direct manual compression was performed while the patients were in supine position after the femoral sheath was removed. Then, patients were monitored for bleeding in the recovery room for 1 h, and none presented with post-procedural bleeding or hematoma.

A 67-year-old woman with left-forearm arteriovenous loop graft who presented with recurrent difficult cannulation and graft thrombosis within 3 months despite the placement of the Supera stent at the venous cannulation site 5 months previously. (a) Digital subtraction angiography (DSA) via a retrograde 6-Fr sheath revealing new stenosis over the arterial cannulation site (white arrow). (b) A 7-mm × 8-cm noncompliant balloon used to dilate the arterial cannulation site. (c) Final DSA showing the whole coverage of the arteriovenous graft with two Supera stents after deployment of another 6.5-mm × 8-cm Supera, beginning from the arterial anastomosis and extending into the previously implanted Supera stent.
Access circuit primary patency rates were 95% at 3 months, 84% at 6 months, and 35% at 12 months after stent placement (Figure 3). The median time to loss of primary access patency was 11 (95% CI: 5.5–16) months. The secondary patency was maintained at 100% at 3, 6, and 12 months. However, it reduced to 89% at 18 months (Figure 3). The target lesion patency rates after Supera stenting were 100% at 3 months, 100% at 6 months, and 75% at 12 months (Figure 3). During follow-up, nine patients required one or more reinterventions for AVG (total: 13 procedures). Only three procedures required intragraft reintervention within the 12-month period. The reintervention rate was 0.15 procedures per year. During follow-up, only one AVG was abandoned due to recurrent stenosis over the proximal drainage vein. The pathogenesis of intragraft restenosis was correlated with progressive neointima thickening along the inner wall of the Supera stent after repeat needle puncture. Two patients presented with difficult cannulation during dialysis. Another patient presented with acute graft thrombosis that was successfully rescued after thrombectomy, followed by balloon angioplasty.
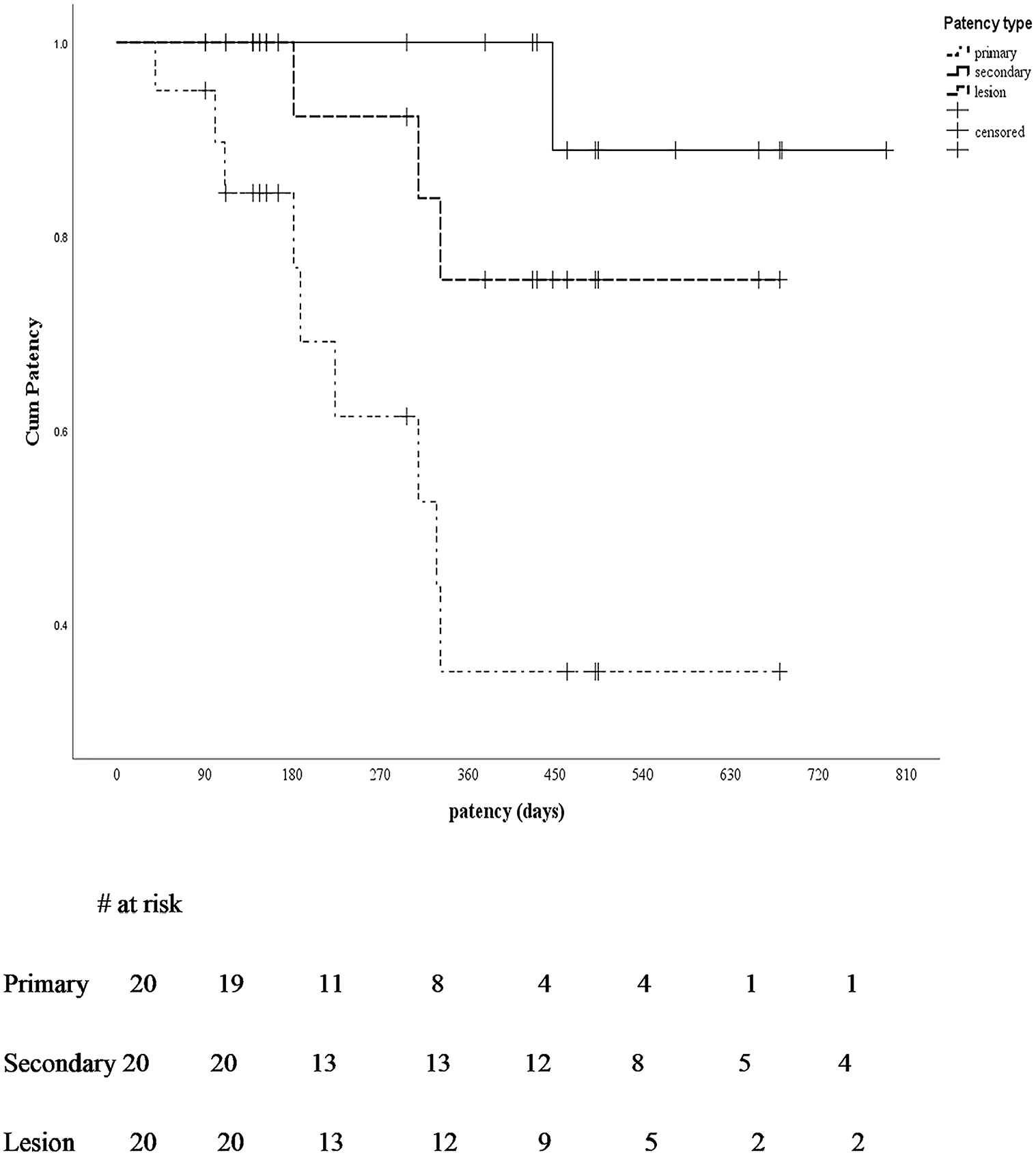
Kaplan–Meier curve of the primary, secondary, and target lesion patency after endovascular intervention for intragraft stenosis.
There were no major complications after Supera placement over the intragraft stenosis. No patient was lost to follow-up during the study period. Further, no graft infection or stent fracture was reported. AVG puncture was successful in all patients who underwent Supera stenting over an AVG intragraft lesion.
Discussion
The prevalence of AVG in Taiwan is approximately 20%–30% of all dialysis vascular access type. 12 Grafts have a relatively short life span and are at risk of recurrent stenosis. 13 Intragraft stenoses account for 2%–28% of all AVG lesions.4,7 Most of the intragraft stenosis occurred at a median age of 19.7 months and contributed for late complication of AVGs. Inferior result after balloon angioplasty for intragraft stenosis will result in access failure or abandonment. 14
Beathard 7 showed that the 3- and 6-month primary patency rates for intragraft stenoses in 285 patients were 76% and 56%, respectively. Angioplasty of intragraft stenoses compared with venous anastomotic lesions had a lower primary patency. In another large retrospective review by Bautista et al., 14 229 intragraft stenoses were identified in 183 grafts. The primary patency rates at 6 and 12 months were 40% and 23%, respectively. Both studies showed that plain balloon angioplasty was inferior to treat intragraft stenoses but only stent deployment could prolong lesion patency. The mechanisms underlying intragraft stenoses are correlated with repeat needle puncture, resulting graft structure distortion and graft intima dissection.9,15 The graft permanently loses its strength to sustain needle trauma. Plain balloon angioplasty has an inferior outcome since repeat needle trauma on the same lesion is inevitable due to the lack of new cannulation area in these patients.
Reports about the use of stents for the treatment of intragraft stenosis are rare. Bautista et al. showed that 15% of intragraft stenoses required stents or covered stents. 14 In their study, stent deployment became an independent predictor of a high lesion patency rate. Zaleski et al. 9 showed the use of wall stents (Boston Scientific, Natick, MA, USA) for treating intragraft stenoses in 19 patients who had recurrent graft thrombosis within 2 weeks after the initial thrombolysis, with resulting 6-month secondary patency rates of 91%. Stent deployment could improve treatment outcomes in intragraft stenosis. The application of stents is effective for preventing surgical revision, which is complicated by temporary loss of graft function and higher procedural risks. However, none of these stents are designed for needle puncture. Stent distortion, particularly in wall stents, eventually developed after repeat needle puncture. 9 Cannulation of a covered stent will also damage the underlying metal framework that supports the PTFE fabric layer, 16 as observed in one of our patients (Figure 4). Cannulation of the covered stent for HD are not approved by the US Food and Drug Administration, owing to the absence of high-quality evidence on its effectiveness and safety.

Deployment of the Supera stent within the arteriovenous graft (AVG). (a) A 78-year-old female patient with left-forearm loop AVG who presented with repeat stenosis over the venous puncture site due to repeat needle puncture over the covered stent (asterisk). (b) Fluoroscopic image of the inserted 6.5-mm × 12-cm Supera stent before deployment. The white arrows indicate the actual length of the Supera stent before deployment. The double arrow shows the distance between the Supera stent before deployment and the expected final location of the stent (approximately 4–5 cm). A 0.035-in wire (white circles) was used as a marker to identify both puncture sites for a precise stent deployment guidance. (c) Final digital subtraction angiography showing acceptable stent elongation in the estimated length that can completely cover both puncture sites.
The Supera stent has six pairs of nitinol wires interwoven to form a helical structure. This interwoven design is resistant to kink formation even when there are torsion, compression, and flexion/extension. It has been postulated that this stent can provide extra supportive force over the intragraft lesion, even when the lesion is going to be punctured after stenting. The interwoven cell design within the stent can accommodate a dialysis needle, and relative low stent distortion is encountered after repeat puncture at the same lesion, unlike others nitinol bare mental stent or covered stent.
The Supera stent has excellent outcomes in the treatment of femoropopliteal occlusive disease. 17 Thomas et al. performed the first study involving 42 patients who had a failing AVF due to juxta-anastomotic stenosis and successfully treated with the interwoven Supera stent. 18 Our study first showed the use of the Supera stent in patients with failing AVGs caused by intragraft stenoses. After stenting over intragraft stenoses, Access circuit primary patency rates were 84% at 6 months and 35% at 12 months. With the Supera stent over the intragraft lesion, the secondary patency rate was up to 89% at 18 months. Most reinterventions for the target lesions occurred after 6 months, with a final target lesion patency of 75% at 18 months. Three patients developed in-stent restenosis 6 months after deployment. Repeat puncture inevitably became an aggressive stimulus for restenosis in specific patients. Despite repeat needle trauma, none of our patients presented with stent distortion or fracture in the follow-up period and the risk of injury by stent fracture to patient and health care stuff is negligible
Deployment of the Supera stent in femoropopliteal lesions is challenging. The vessel should be prepared to an optimal size, and the stent must be deployed in a nominal shape to preserve its radial strength. The deployment is different in AVG. The 6.5-mm Supera stent was commonly used because all patients received a 6-mm PTFE graft for AVG creation. A 7-mm noncompliant balloon was used for predilatation if no severe recoil was noted. Subsequently, we deployed a slightly elongated Supera stent to widen the interstices of the stent to facilitate a smooth needle puncture. In our experience, the 12-cm Supera stent can be elongated to 15–17 cm, and the 15-cm Supera stent to 18–20 cm (Figure 4). Instead of packing within a graft to achieve the nominal diameter, the stent is likely to be pulled during deployment to facilitate elongation. The retrograde access is the preferred route for precise deployment.
This study had several limitations. First, it has a single-center retrospective design, and the sample size was limited. Second, there was no comparison group. Third, surveillance angiograms at standardized intervals were not performed for the assessment of lesion patency. Since there was no direct comparison between different stents available for intragraft lesions, the superiority of the Supera stent was not validated. Another concern is its off-label use for cannulation. Only few reports about this technique are available. Therefore, further studies should be conducted to evaluate stent structure damage after cannulation.
In conclusion, interwoven nitinol stent placement is a promising treatment for failing AVGs caused by intragraft stenoses. Further, access circuit primary, secondary, and target lesion patency rates are acceptable. Since the Supera stent is resistant to compression and fracture, it can be placed over an intragraft lesion to sustain direct cannulation for dialysis and to decrease the risk of stent distortion or fracture. With consideration of the significant improvement in patency with intragraft deployment of the Supera stent, the life span of AVGs can be prolonged in patients on HD with a limited vascular access.
Footnotes
Acknowledgements
We would like to thank Wallace Academic Editing for English language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s note
Authors are affiliated with the Taiwan Association of Thoracic & Cardiovascular Surgery.
