Abstract
Background:
The aim of this pilot clinical study is to evaluate the safety and efficacy of the Selution Sustained Limus Release (SLR)™ sirolimus-coated balloon (M.A. MedAlliance SA, Nyon, Switzerland) for improving the patency of failing arterio-venous fistulas (AVF) in hemodialysis patients. We also present herein a pre-clinical pharmacokinetic and safety evaluation of Selution™ to justify its first use in hemodialysis patients for endovascular access salvage.
Methods and results:
This is an investigator-initiated prospective single-center, non-blinded single-arm trial. Forty patients with clinically significant de novo or recurrent stenoses in a mature AVF circuit will be recruited. All stenotic lesions will be prepared with high pressure non-compliant conventional balloon angioplasty (CBA) prior to deployment of the Sustained-Release Selution™ sirolimus drug-eluting balloon. The primary efficacy endpoint is 6-month target lesion primary patency and the primary safety endpoint is freedom from localized or systemic serious adverse events through 30 days. Secondary endpoints of interest include technical and clinical success rates and circuit access patency at 3 and 6 months. Follow-up will occur for 2 years for those patients whose AVFs remain patent. Pharmacokinetic and histological animal safety studies performed with the Selution™ coating formulation showed prolonged arterial tissue retention of sirolimus with therapeutic levels up to 60 days and non-toxic and rapidly declining blood levels. Histological results in animal models demonstrated safety, freedom from intraluminal thrombus, reduction in restenosis by sirolimus elution compared to CBA, and no evidence of embolic phenomena indicative of adverse particulate effects.
Discussion:
Long release sirolimus coated balloons may serve as a promising novel alternative therapy to paclitaxel-based technology for treating conduit stenosis secondary to neointimal hyperplasia. Pre-clinical pharmacokinetic and histological animal data are encouraging and provide suggestion of safety and efficacy in this setting. This single-center trial will provide a first step toward demonstration of efficacy and safety of this device for treatment of stenotic fistulas.
Keywords
Introduction
The recently updated National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI) Clinical Practice Guidelines for Vascular Access has moved toward an individualized patient-driven life plan addressing chronic kidney disease progression in patients. 1 For end-stage kidney disease, preference for those undertaking hemodialysis is the use of an autogenous arterio-venous fistula (AVF) or graft rather than a central venous dialysis catheter, which carries a higher risk of infection. However, the Achilles heel of an AVF is the development of conduit stenosis, which is the most common cause of circuit dysfunction and remains a significant cause of morbidity and hospital readmissions for hemodialysis patients. 2 The KDOQI guidelines recommend treating hemodialysis access stenoses of more than 50% when associated with clinical dysfunction such as reduced flow rate and elevated venous pressures. 1
An endovascular technique is considered as effective as surgery in dealing with venous stenosis, and conventional high-pressure balloon angioplasty (CBA) is the current standard of care for the treatment of these stenotic lesions with this approach. Despite its widespread availability, minimally invasive nature and safety, primary access patency rates in the medium and long-term following CBA are low—at best achieving a 40%–50% 12-month primary patency,3–5 with high incidence of recurrent stenosis requiring repeat interventions and eventually loss of access and circuit patency. 6 Hence, there is a clinical need to improve the duration of patency in dialysis vascular access.
The advent of successful drug-eluting technology for the treatment of the coronary vascular bed and accumulating evidence of favorable results with this technology in the peripheral arterial circulation has prompted the introduction of paclitaxel coated balloons (PCB) for treatment of venous stenosis and in-stent restenosis in the access fistula circuit. Recent studies suggest that DCBs may significantly reduce re-intervention rates on native and recurrent lesions.4,7,8 The restenosis process is in part or in whole the result of neo-intimal hyperplasia (NIH) formation and is considered the main culprit phenomenon in access circuit target lesion stenosis and access circuit failure. 9 NIH is the blood vessel’s response not only to hemodynamic changes when the circuit becomes arterialized but also the vascular response to repeated needlestick injuries during the subsequent hemodialysis sessions and to the trauma associated with the balloon angioplasty process. Furthermore, factors that influence endothelial dysfunction such as uraemia, oxidative stress, and inflammation in the setting of end-stage renal disease are thought to also play a role. 10
Paclitaxel is a cytotoxic drug, which inhibits NIH formation and slows the restenosis process, leading to improved patency rates.8,11 Results of meta-analyses and randomized controlled trials for the use of PCBs in AV access are mixed to date. A meta-analysis performed by Khawaja et al. 12 seemed to suggest that PCBs conferred some benefit in improving target lesion primary patency (TLPP) in AVFs. The recent IN.PACT AV RCT showed that PCB was superior to CBA for the treatment of stenotic lesions in dysfunctional AVF at 6 months. 8 However, the largest RCT to date of PCB versus CBA in AVF, reported no improvement in TLPP at 6 and 24 months follow-up. 13 Interestingly, there was a significant difference in TLPP in favor of PCB at the 9 and 12-month timepoints and a reduction in reintervention rate for the PCB group at the 9-month interval only. Another recent reported RCT using the Biotronik Passeo-18 Lux® PCB did not show a significant superior patency after 6 and 12 months compared to high pressure CBA, although there was a trend of less relapse and greater survival in the PCB treated group. 14 Another recent meta-analysis found that PCB showed no statistically significant improvement over conventional balloons in decreasing fistula stenosis. 15 Furthermore, PCB use has been linked to longer term safety concerns. 16 The results of pooled data from 28 RCTs of paclitaxel-eluting technologies consisting of 4663 patients showed that all-cause death at 2 years was significantly higher following the application of paclitaxel-coated devices (risk ratio 1.68, 95% CI, 1.15–2.47). Similarly, all-cause death up to 5 years was significantly higher in the DCB/DES arm (pooled risk ratio 1.93, 95% CI, 1.27–2.93). Meta-regression analysis indicated a highly significant association between paclitaxel dose-time product and absolute risk of death. However, it must be noted that there does not seem to be any difference in all-cause short- and midterm mortality in patients who underwent dialysis access treatment using PCBs compared with CBA. 17 Furthermore, the 24-month data from the Lutonix IDE study did not demonstrate any significant difference in mortality between the DCB and CBA groups. 13
In light of these issues, attention has turned recently from paclitaxel-based technologies to sirolimus coated platforms. 18 Sirolimus, like paclitaxel, is a potent antiproliferative agent, which has been found to prevent restenosis in the coronary bed 19 and more recently in the peripheral vasculature 20 but to date there have been few studies in AVF circuits.The aim of ISABELLA is to determine the safety and efficacy of the MedAlliance SELUTION Sustained LimusRelease (SLR)™ 018 sirolimus coated balloon (SCB) in the treatment of failing AVFcircuits due to conduit stenosis in patients undergoing hemodialysis. We also present pre-clinical pharmacokinetic and histological data, to justify its use in the endovascular access salvage arena.
Materials and methods
Hypothesis
We hypothesize that the application of SCB to cover the target venous stenotic lesion after successful preparatory effacement using a high pressure balloon is safe, minimizes NIH, and improves AVF target lesion patency compared to CBA.
Study design
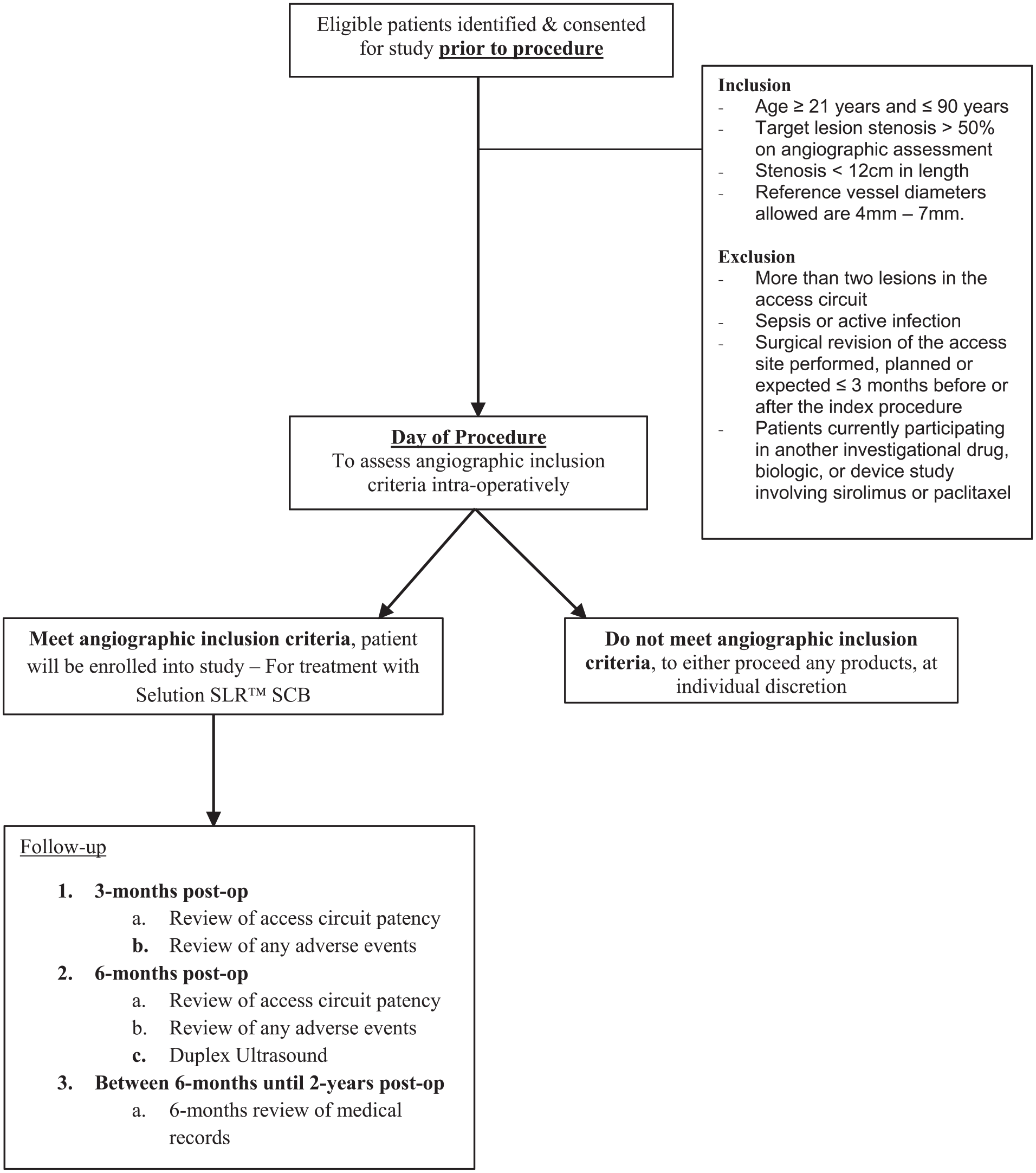
This is a Singapore General Hospital (SGH) investigator-initiated multi-investigator, single arm, non-randomized, prospective pilot study. Forty end-stage renal disease patients with failing AVFs on follow-up with the Departments of Vascular Surgery and Renal Medicine will be enrolled. Follow-up will be at 6, 12, and 24 months. The trial will be carried out under an investigational device exemption (GN27) from the local Health Services Authority (HSA) of Singapore. Approval was obtained from the local Human Research Ethics Committee (CIRB ref: 2020/
Inclusion and exclusion criteria
The primary inclusion criterion is a native mature AVF in the arm and forearm, which had at least one clinical indicator of dysfunction as defined by the KDOQI. 1 Thrombosed and immature AVFs, those AVFs that have been previously stented, presence of central venous stenosis, and arterio-venous grafts will be excluded. There will be no restrictions based on gender and race.
Inclusion criteria
- Patient aged ⩾21 years and ⩽90 years.
- Native AVF was created more than 2 months prior to the index procedure and had undergone 10 or more hemodialysis sessions utilizing two needles.
- Target lesion location located between the anastomosis and the axillary-subclavian vein junction, as defined by insertion of the cephalic vein.
- On initial fistulogram, target lesion stenosis >50% on angiographic assessment and in keeping with the clinical indications for intervention.
- Stenosis <12 cm in length (to allow for potential treatment with one SCB available in lengths up to 15 cm).
- Stenosis has to be initially treated successfully with a high-pressure POBA prior to SCB treatment as defined by:
(a) No clinically significant dissection (flow limiting).
(b) No extravazation requiring treatment/stenting.
(c) Residual stenosis ⩽30% by angiographic measurement.
(d) Ability to completely efface the lesion waist using the pre-dilation CBA.
- No more than one additional (“nontarget”) lesion in the access circuit that has to be also successfully pre-treated (⩽30% residual stenosis) before SCB deployment. Separate lesion is defined as being at least 3 cm in distance from the target lesion.
- Reference vessel diameters allowed are 4–7mm.
Exclusion criteria
- Women who are pregnant, lactating, or planning on becoming pregnant during the study.
- Subject with more than two lesions in the access circuit.
- Subject has a secondary non-target lesion that could not be successfully pre-treated.
- Sepsis or active infection.
- Surgical revision of the access site performed, planned or expected ⩽3 months before or after the index procedure.
- Patients who were taking immunosuppressive therapy or are routinely taking ⩾15 mg of prednisone per day.
- Patients currently participating in another investigational drug, biologic, or device study involving sirolimus or paclitaxel.
- Contraindication to aspirin or clopidogrel usage.
- Where final angioplasty treatment requires a stent or drug-eluting balloon >8 mm in diameter.
- Metastatic cancer or terminal medical condition.
- Blood coagulation disorders.
- Limited life expectancy (<12 months).
- Allergy or other known contraindication to iodinated media contrast, heparin, or sirolimus.
Endpoints
The primary efficacy end point is target lesion primary patency (TLPP) at 6 months defined as freedom from clinically driven reintervention of the target lesion or access thrombosis and no significant restenosis (lumen diameter <2.7 mm) on Duplex ultrasound. 21 A patient can undergo reintervention on the AVF without TLPP ending provided that the reintervention did not include retreatment of the target lesion. The primary safety end point is freedom from localized or systemic serious adverse events through 30 days that reasonably suggest the involvement of the arteriovenous access circuit. These would include life-threatening events or those resulting in death, requiring hospitalization, resulting in permanent disability, or requiring intervention to prevent permanent impairment; the latter definition includes thrombosis.
Secondary endpoints include access circuit primary patency at 3 and 6 months. Access circuit patency loss is defined by a development of a stenosis in any region of the AVF circuit requiring reintervention. Other secondary endpoints include target lesion primary patency and access circuit primary patency at 3 months, procedural success, access circuit thrombosis, need for open bypass revision surgery, access site abandonment, number of interventions required to maintain access circuit primary patency at 3 and 6 months, and mortality.
Investigational device
The SELUTION SLR™ 018 SCB is a combination product consisting of a standard PTA balloon catheter coated with a drug (sirolimus) at 1 μg/mm2. The drug coating is formulated with sirolimus-loaded bioresorbable poly(lactic-co-glycolic acid) (PLGA) microspheres. The use of a solid solution of Sirolimus in the PLGA microspheres allows for sustained release of sirolimus at the treatment site as the smaller drug molecule diffuses through the polymer network. PLGA has a long history of safe clinical use in biodegradable medical devices. The drug coating formulation is sprayed onto the inflated balloon surface with a proprietary Cell Adherent Technology (CAT™) amphipathic transfer membrane that contains and protects the microspheres during balloon insertion, lesion crossing, and inflation. Upon contact with the vessel wall, the coating is further designed to increase absorption of the drug into the vessel wall by utilizing the amphiphatic properties of the coating. The sizes of balloons available for this study are 4–7 mm in diameter, available on an 0.018″ wire platform in 80 and 150 mm balloon lengths and 150 cm shaft length.
Treatment
All procedures will be performed in a hybrid operating theatre using standard percutaneous techniques under local anaesthesia. Unfractionated heparin (2000 iu) will be used for peri-procedural anticoagulation. An initial fistulogram will be obtained. The stenosis will be crossed with a 0.018″ guidewire (Boston Scientific, Marlborough, MA, USA) and support catheter. Mandatory pre-dilatation of the target lesion with a standard high-pressure non-compliant balloon (Mustang®, Boston Scientific, Marlborough, MA, USA) for 2 min, will be used to match the calibre of the adjacent normal vessel. The Selution SLR™ SCB will then be inserted and inflated for 2 min to rated burst pressure of 10 ATM, which according to the design specifications of the SCB will allow maximal drug transfer to the vessel wall. Balloon length shall be chosen to be at least 2 cm longer than the area treated during pre-dilatation (1 cm overlap proximal and distal) in order to avoid geographical miss. No post-dilatation will be allowed.
Adjuvant medical therapy
Post-procedure, all patients will receive daily aspirin (100 mg) and clopidogrel (75 mg) plus a proton pump inhibitor for 1 month followed thereafter by anticoagulation therapy employing a single antiplatelet agent.
Follow-up
All subjects will be followed-up after successful intervention by a telephone call from one of the study co-ordinators, to assess patency at 1 month, any complications such as localized infection and hematoma of the AVF, compliance to antiplatelet therapy, and to identify any adverse effects of treatment. Three and six-month clinic assessments will follow with a dedicated Duplex ultrasound at 6 months. Problems with hemodialysis including difficulties with cannulation, prolonged bleeding after needle removal, hematoma due to mis-cannulation, high venous pressure, and inadequate dialysis clearance will be recorded. From 6 months onwards, there will be extended follow-up for patients whose fistulas remain patent. Adverse events will be recorded until either primary patency is lost, or 2 years post-intervention is reached. Follow-up during the 6 months to 2-year period will be flexible and will be tailored to patient requirements, but data will be collected at least once every 6 months at a minimum. No imaging is mandated during the extended follow-up period (Figure 1).
ISABELLA trial flowchart.
Sample size calculation and statistical analysis
Based on a historical 6-month primary patency of approximately 50% with CBA, 5 we hypothesize that 6-month primary patency will improve to 70% following CBA/SCB treatment and that the estimated sample size needed is 28 subjects using an alpha risk of 0.05 and a power of 0.8. Hence a follow-up of n = 40 at 6 months should suffice to allow for dropouts along the way. The probability of re-intervention over time will be calculated using competing risks analysis using the cmprsk package in R, as death is a competing risk. Cause-specific sub-distributions will be compared across groups using Gray’s test. All analyses will be performed in R version 3.5.1. 22
Pharmacokinetic/histological animal safety studies using Selution™ SLR
The first animal studies using the SELUTION SCB were pharmaco-kinetics (PK) studies in 22 rabbit iliac arteries (first cohort) and 32 rabbit iliac arteries (second cohort). Additionally, light microscopy and SEM of treated vessels were conducted at 1 and 24 h post-treatment. Protocols were submitted to the Institutional Animal Care and Use Committee (IACUC) prior to study initiation.
Combining the two cohorts yielded timepoints of 1, 28, 60, 90, 112, and 180-days for analysis of arterial drug content. Sirolimus assay found therapeutic levels were likely still present out to 60 days but were not quantifiable at 90 days. Based on these successful pharmacokinetics results, the rabbit artery evaluation refocused on histological evaluation of the remaining 112- and 180-day cohorts. Downstream skeletal muscle of SCB treated segments were also collected for assessment. Tissues were dissected, carefully excised, and immediately frozen in liquid nitrogen. Measurements of sirolimus blood level and iliac artery sirolimus concentrations were performed by liquid chromatography-tandem mass spectrometry analysis.
A third animal study was conducted on Yucatan mini swine. This study compared SCB and uncoated control balloons. Each swine provided four arteries for treatment; the right and left external femoral, right and left internal femoral arteries. Blood samples were drawn at three time points to assess sirolimus levels in the bloodstream, which were found to be in the range of 1–8 ng/mL post SCB deployment. Excised arteries were used to assess, via histological evaluation, the safety and vascular biocompatibility of the SCB in healthy porcine peripheral arteries at 28 and 90 days as compared to uncoated control balloons. Downstream muscle samples were collected from all eight animals treated, consisting of gluteal muscle, gastrocnemius muscle, and coronary corium (coronary bands).
Pre-clinical results
Sirolimus blood content PostDCB
Sirolimus in whole blood was quantified for the rabbit PK studies after iliac intervention at 30 min, 1, and 4 h for all animals. Sirolimus blood levels peaked at 30 min postprocedure, were low by 24 h, and were below quantification limits by 7 days. Twenty-four hours became the last time-point for blood measurements (Figure 2) due to limit of quantification (LOQ). Sirolimus concentration in rabbit arteries was found to be therapeutic at 28 days, with a measured content of 21.5 µg of sirolimus per gram of tissue (Figure 3). In Yucatan mini swine, the maximum early porcine systemic sirolimus blood concentration of 7.77 ng/mL was below the peak blood level of 211 ng/mL found to be safe in human studies. The mean blood concentration beyond 24 h was below the therapeutic range of sirolimus (9–17 ng/mL) recommended for organ transplant applications.
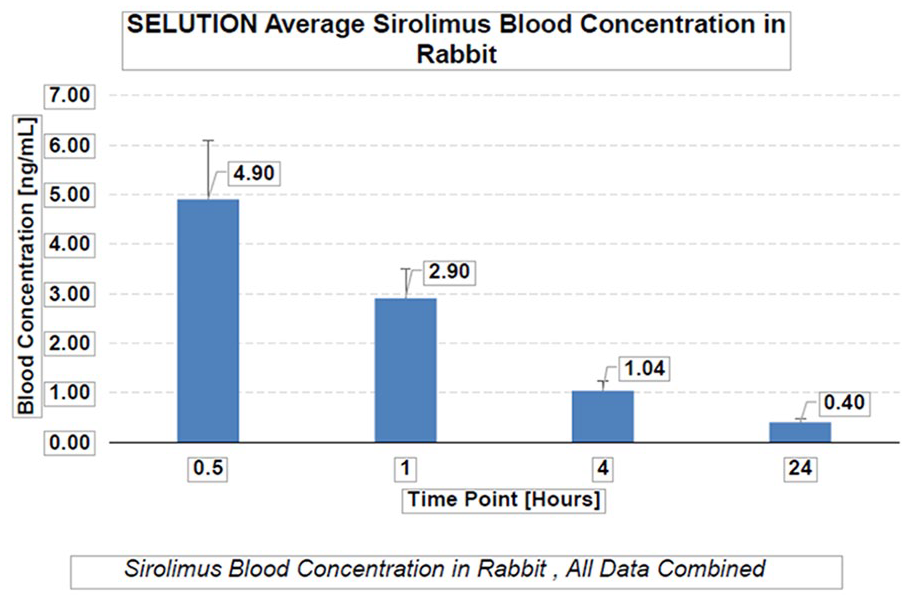
Average sirolimus blood concentration in rabbits.
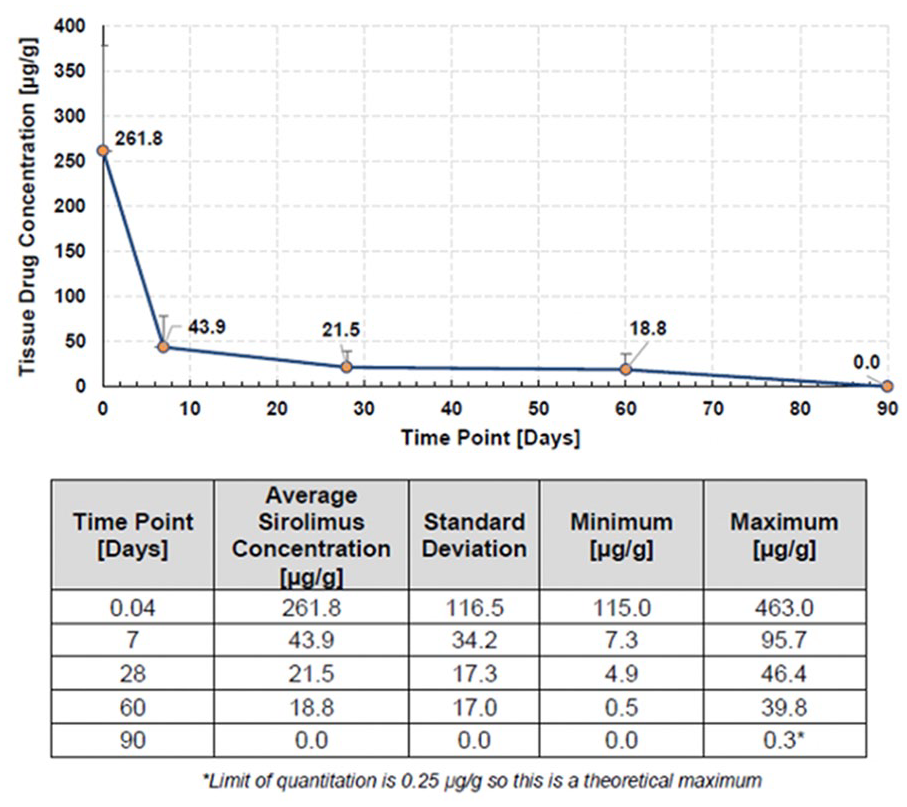
Sirolimus tissue concentration out to 90 days in rabbit iliacs.
Tissue and organ evaluation
For the rabbit study, a minimum of two pieces of each organ from an animal sacrificed 1 h after treatment and from an animal sacrificed 24 h after treatment underwent gross evaluation. Evaluated organs included left and right kidneys, liver, lung, spleen, and skeletal muscle from the left and right gastrocnemius. These are all locations where blood is filtered through selectively smaller orifices and therefore where particulate introduced by the catheter could be expected to accumulate. There was no evidence of drug embolization.
Histological evaluation in rabbits
Histologic examination by confocal, scanning electron microscopy (SEM), and light microscopy at 1 and 24 h was performed in conjunction with the rabbit studies. Microspheres were observed on the interior surface of excised arteries at 1 and 24 h post-treatment demonstrating that the coating had been successfully transferred to the arterial wall (Figure 4). Medial smooth muscle cell (SMC) loss (combination of drug effect and injury) was observed at the 112-day time point. No incidence of occlusive thrombosis was seen in any of vessels at either 112- or 180-day time points; focal regions of neointimal proteoglycan accumulation associated with vacuolization and foamy macrophages was observed, presumably from microsphere resorption; but no incidence of fibrin, calcification, or red blood cells was noted in any vessel; endothelialization was nearly complete with no evidence of surface fibrin/platelet thrombus. Morphometry of cross-sectional vessel areas was used to calculate percent stenosis for the 112- and 180-day rabbit time points. Average stenosis at 112 days was 43.20% ± 5.78%. Average stenosis at 180 days was 17.03% ± 14.39%. Thus, a significant decline in percent stenosis was observed between 112 and 180 days. All rabbit histology samples were noted to be unremarkable except for lung tissue, which was noted to be “mottled tan-brown, dark red and brown-red,” which according to the pathologist was unrelated to DCB treatment and typically seen as damage caused by mechanical ventilation of the animal. An overall conclusion was there was no indication of infarction/scarring or distal emboli involving PLGA microspheres in any of the organ and muscle tissues assessed.

Microparticles on surface of rabbit artery 24 h after treatment.
Histological evaluation of porcine study
Both 28- and 90-day cohorts used 9 DCB’s and 7 control (POBA, uncoated) balloons amongst the 16 available arteries in the four animals. All eight animals completed the in-life phase successfully and there was no incidence of occlusive thrombosis in any vessel examined. At both time points, majorities of both DCB and POBA treated vessels appeared normal with minimal neointimal growth, absence of medial injury, and no medial smooth muscle cell loss. Endothelization was nearly complete, and improvement is seen from 28 to 90 days with a reduction in neointimal proteoglycans and neointimal inflammation. At 90 days, there was no significant difference in histology scoring between SCB and control and the majority of vessels, appeared normal whether treated with SCB or control. There was a higher loss of SMC in the SCB group along with greater amount persistent fibrin in the medial layer in this group (Figure 5). Angiography findings at time of sacrifice further confirmed that the vessels treated by the SCB remained widely patent for the duration of the in-life phase of the studies, with no incidence of occlusive thrombosis.
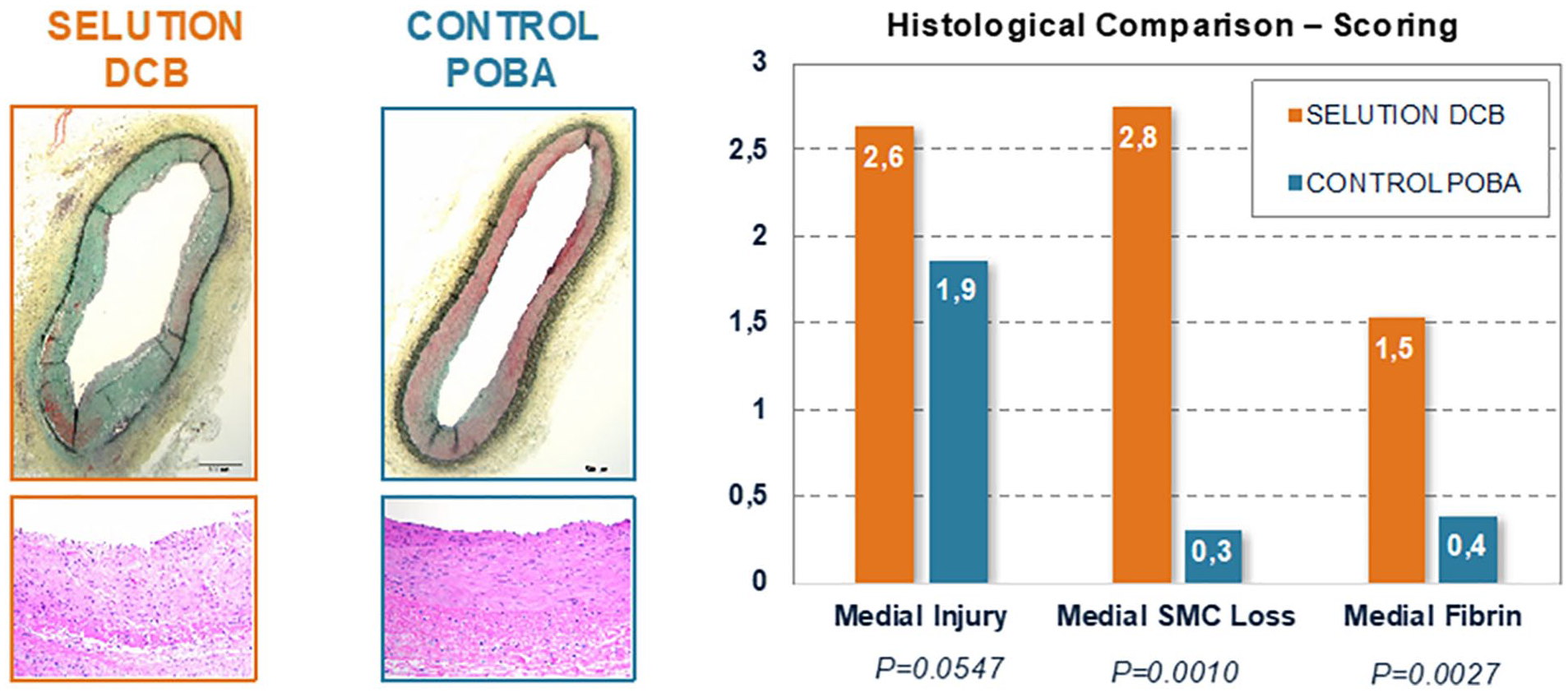
28-day histological evaluation of Selution™ SLR.
Discussion
The conservation of a patent, well-functioning AVF is one of the most difficult clinical problems in the long-term treatment of patients undergoing hemodialysis. Conduit stenosis is the most frequent cause of AVF dysfunction. PCBs have been used with some success to prolong luminal patency but as discussed, the data are far from conclusive. Potential reasons why outcome data were variable in previous studies may be related to how the stenotic lesion is prepared prior to drug elution and the high-speed blood flow in dialysis access circuits, which may wash a large amount of the paclitaxel away from the target lesion soon after application. A measurement in swine showed that only 20%–30% of paclitaxel was taken up into the coronary artery wall in vivo, 15–25 min after PCB application. 23 Another explanation for the heterogeneity of PCB efficacy in the AVF circuit, is probably related to the inconsistent use of study definitions and endpoints.
Historically, paclitaxel has been the drug of choice for use in drug-coated balloon catheters for treating the peripheral circulation, yet large-scale trials have demonstrated the superior efficacy of sirolimus and it’s close analogs as coatings for drug-eluting stents, especially in the coronary circulation. 24 Among cardiologists, sirolimus is perceived as superior to paclitaxel, mostly because of lower restenosis rates in the coronary bed following limus-eluting versus paclitaxel-eluting DES. 25 The absence of an effective SCB in the peripheral vascular market until recently has been due to the difficulty of creating an effective sirolimus coating on a balloon platform, which can deliver the anti-restenotic agent to the vessel wall in sufficient quantity and duration to inhibit the NIH process. A further difficulty was the high washout rate of first-generation sirolimus coatings under arterial blood flow. Both sirolimus and paclitaxel can impact endothelial function, but while paclitaxel causes cell death by interfering with microtubule formation during cell division (cytotoxic), sirolimus analogs have a cytostatic mode of action that prevents SMC activation after vascular injury by halting cell progression through the G1 phase of the cell cycle, with a greater margin of safety, and additional anti-inflammatory properties. 24 An additional theoretical advantage of sirolimus is that it is equally distributed in the vessel layers in contrast to paclitaxel, which accumulates in the adventitia and is thought to play an inferior role in retarding the restenotic cascade in in-stent-stenosis. 24
The Selution SLR™ SCB utilizes micro-reservoirs and a novelCAT™ transfer membrane. This technology increases uptake of sirolimus into the artery wall, prolongs exposure to the drug, reduces dose lost to the circulation and minimizes distal embolization of large, random sized particles of the balloon coating. The coating is further engineered to enhance sirolimus uptake by cells inhabiting the interior lining the vessel lumen due to the amphiphilic properties of the coating. Combined with a wider safety margin of sirolimus compared to paclitaxel, these features allow a lower dose of drug on the balloon per mm², thus facilitating the use of larger and multiple balloons during intervention. The Selution SLR™ balloon was found to be safe and effective at 6 months with a late lumen loss of 0.19 mm and TLR rate of 2.3% in the peripheral vasculature
20
—outcomes at least as good as paclitaxel DCBs in the same vessel beds. Furthermore, a sirolimus coating employing a fatty acid amphilimus formulation in the NiTiDES stent system (CID, Saluggia, Italy), released through an abluminal reservoir technology, showed safety and efficacy in the ILLUMINA trial for femoropopliteal interventions.
26
Recent data has emerged from our center using the MagicTouch™ sirolimus-coated balloon (SCB) catheter (Concept Medical Inc., Tampa, FL, US) in the dialysis access circuit, which utilizes proprietary nanolute technology designed to improve the lipophilicity and bioavailability of sirolimus.27,28 Sirolimus is converted into sub-micron sized particles and encapsulated into phospholipid-drug nanocarriers, which allows more effective transfer to the vessel wall. The
The pharmacokinetic and histological animal safety studies performed with the SCB coating formulation in peripheral applications revealed that the test devices performed as intended, there was survival of all study animals with no observed complications and a robust and prolonged arterial tissue retention of sirolimus with therapeutic levels up to 60 days. Blood levels declined rapidly after DCB treatment and were not detected after 7 days. Despite the use of biodegradable PDLA microspheres to deliver sirolimus to the artery over extended periods, there was no evidence of downstream emboli or tissue reaction indicative of adverse particulate effects, which have previously been linked to the use of paclitaxel-coated devices. 16 Based on previously reported clinical studies, the potent antiproliferative properties of sirolimus make it a useful agent to reduce the risk of restenosis for at least several months post-angioplasty, justifying its use on DCBs.20,27,28 Sirolimus release interferes with fibroblast and smooth muscle cell proliferation as well as the proliferation of inflammatory cells. These three cell types are largely responsible for the restenotic cascade resulting from tissue responses to vascular injury secondary to angioplasty.
An interesting finding from the rabbit model was a restenosis rate of 43% at 112 days, which may draw concern regarding the effectiveness of the SCB. The rabbits were subjected to what we refer to as a “high injury model,” which is the worst case scenario and not seen in most animal safety studies, which are conducted in low injury porcine models. There was a much higher drug and excipient application than what would typically be used in a porcine safety study or actual human use of the SCB product. The excipient is mostly PLA microspheres that house the sirolimus drug. Degradation of the PLA normally causes some temporary pH shift, which leads to transient inflammation/restenosis. The larger amount of excipient (larger pH shift) coupled with high mechanical injury to the rabbit vessels (the vessels were fully denuded in vessel preparation) likely caused a temporary increase in restenosis in this particular animal experiment. Viewing the extreme test conditions, we view the 43% restenosis at 112 days as acceptable. It was significantly resolved at 180 days.
There may be also variations in biological sensitivity to the Selution SLR™ coating between animal species but this is just a theory. No animal model perfectly duplicates the response of a diseased human vessel because the partitioning of the drug in the vessel wall is different and humans seem to have a more effective anti-restenotic response than animal models when studying drug-coated balloons. Animal models are primarily used to evaluate safety, not effectiveness of DCBs.
The durability of the results from a single SCB application,which we hypothesize will be observed in the ISABELLA study will need to be confirmed with our more prolonged observations extending out to 2 years, especially in view of the fact that endothelial regeneration occurs typically 1 month or longer after injury, highlighting the need for prolonged anti-inflammatory therapy.29,30 Controlled release of drug from the SELUTION SLR™ SCB, although unproven in the clinical arena, may provide therapeutic concentrations of drug within the vessel wall for at least 60 days post-angioplasty, which is a major advantage over other drug-coated balloons in dealing with the extended nature of the NIH process. In comparison, the paclitaxel dose that penetrated and remained within the vessel wall in animal models showed its disappearance in time (26.7% at 28 days) 31 and may be an explanation for the decline in patency at 6 months using these paclitaxel devices and hypothesizes the need to perform repeated procedures with more drug loaded balloons to maintain longer target lesion patency.
Conclusions
Animal model data using the device demonstrate reasonable pre-clinical safety following peripheral treatments in rabbit and swine models, with arterial tissue retention of sirolimus. Furthermore, animal histological data showed comparable safety response to the SCB compared to CBA with trends toward increased SMC loss, a potential indicator of reduction in NIH. Although no specific testing has been conducted using animal models of AVF, we believe the existing safety and feasibility data in other peripheral vessel beds can be translated to the AV indication. ISABELLA will be the first study to report clinical safety and efficacy data of SCB angioplasty using the SELUTION SLR™ balloon catheter combined with high pressure CBA vessel preparation, for dysfunctional AVF circuits in Asian hemodialysis patients.
Footnotes
Author contributions
TYT (study PI) was primarily involved in study design and protocol development. He will lead the protocol implementation and analysis of the data, as well as patient recruitment. CST, RYT, HYY, LHTT, CTT will be involved in patient recruitment and will edit the final draft of the final study manuscript. CJQY, SXYS will coordinate the project and patient communication and telephone follow-up at 1 month and will be involved in manuscript preparation with TYT. SLC and SXYS will aid with the data analysis and statistical prowess. AVF, SB, DH, and RTD contributed the pre-clinical pharmacokinetic and histology data. All authors have read and approved this final edition of the protocol manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TYT and TTC have received physician-initiated study grants from M.A. MedAlliance SA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is kindly funded by an investigator-initiated unrestricted grant from the Manufacturer (M.A. MedAlliance SA) to defray costs of running the study and analysing the data. Funding source had no role in the design of this study and will not have any roleduring its execution, analyses, interpretation of data or decision to submit results.
Trial registration
Selution SLR™ Agent Balloon for Endovascular Latent Limus therapy for failing AV Fistulas (ISABELLA) Trial (NCT04629118).
Guarantors
TYT/AVF.
