Abstract
Background:
Eighty percent of hemodialysis patients start their dialysis with a tunneled hemodialysis catheter. Catheter related bacteremia is the second most common cause of death in these patients. Side holes near the tips of the tunneled cuffed central venous catheters are associated with accumulation of thrombus, which can lead to catheter dysfunction and, possibly, also to catheter-related infection. To assess the hypothesis that a catheter without side holes would be associated with less bacterial growth, this study compared the susceptibility of a side-hole-free catheter to accumulation of pathogenic bacteria at the catheter tip with that of two catheters which have side holes.
Methods:
Eight tunneled cuffed double-lumen central venous catheters were inserted into both jugular veins of four sheep; one side-hole-free and one control catheter with side holes at the tip in each animal. Staphylococcus aureus bacteria were then infused intravenously to cause bacteremia. Six hours later, the catheters were removed, the clots that accumulated in their tips were collected and cultured, and the bacterial colonies were counted after additional 12 h of incubation.
Results:
Bacteria grew on culture plates seeded with the clot homogenate obtained from the tips of all catheters. The colony counts from the catheters with side holes at the tip exceeded the colony counts of bacteria accumulated in the tips of the side-hole-free hemodialysis catheters by one or more orders of magnitude, with a difference of at least two orders of magnitude observed in three of the four intra-animal comparisons.
Conclusions:
In paired intra-animal post-inoculation comparison made in this limited study, fewer colony forming units of pathogenic bacteria accumulated at the tip of the side-hole-free catheters than at the tips of the catheters which have side holes. This may translate to a decreased rate of catheter-related blood stream infections in the side-hole-free catheters.
Introduction
Blood stream infections are universally recognized as a major determinant of morbidity and mortality associated with hemodialysis, in general, and that administered through long-term tunneled cuffed central venous catheters (CVC), in particular (catheter-related blood stream infections; CRBSI). 1 This complication also accounts for considerable healthcare expenditure. 2 The majority of CRBSI-associated isolates in hemodialysis patients are Gram-positive organisms. Staphylococcus aureus, a pathogen known to cause severe complications and even death, accounts for 21%–43% of all infections, with methicillin-resistant strains being the cause in 12%–38%. 3 Several studies indicated the difficulty of treatment of staphylococcal infections, prompting the National Kidney Foundation’s (NKF) Kidney Disease Outcomes Quality Initiative (KDOQI) to advocate CVC removal with a new CVC replaced at a new site in cases of infections due to Staphylococcus aureus (± “CVC free” duration, whereby the patient has a period when there is no CVC in-situ, with insertion of a temporary CVC as needed for dialysis). 4
Despite the burden of risks, the use of the cuffed tunneled catheters is frequently vital as a standalone treatment modality of acute and chronic renal failure. It is used as a bridge to recovery of renal function, maturation of fistula or until finding a donor organ, as well as a means of permanent access. The United States Renal Data System (USRDS) 2015 and 2017 data show little, if any, change from 2005, with slightly over 80% of patients using a catheter at hemodialysis initiation in the United States.5,6 At 90 days after the initiation, 68.5% of patients are still using catheters. 5 Among prevalent hemodialysis patients, in 2018, catheter use was much higher (52%) for hemodialysis patients ⩽21 years old (vs 19%–21% in other age groups), and was modestly higher for female (22%) than for male (17.7%) hemodialysis patients. 6
Even with the apparently enduring interest in tunneled cuffed catheters and the relentless effort of CVC manufacturers and medical personnel to curb the CRBSI rate through use of antimicrobial and antithrombogenic surface coatings and catheter locking solutions, the incidence of CRBSI remains a major problem (5.2 episodes per 1000 catheter days reported in 2004 7 and up to 5.5 episodes per 1000 CVC days in 2020. 4 It should be noted, however, that although antimicrobial preemptive antibiotic lock installation is not part of the current standard of care in hemodialysis patients with CVCs, some studies showed these might be efficient in reducing CRBSI rates,8,9 leading to a recent KDOQI recommendation to use prophylactic antimicrobial locks in patients in need of long-term CVC who are at high risk of CRSBI and in units with high rates of CRBSI. 4 Overall, the 5 million CVCs inserted annually in the United States are associated with approximately 250,000 instances of CRBSI, 2 and the hospitalization and mortality rates attributed to the increase in bacteremia/sepsis for patients commencing hemodialysis with a CVC are high.10,11
Importantly, the need to attain high blood flow rate, required for adequate hemodialysis posed by hemodialysis machines, to prevent recirculation and to prevent fibrin sheath formation leading to thrombosis and, eventually, bacterial colonization12–14 prompted manufacturers to modify the tip of the catheter. Multiple side holes are frequently introduced in the long-term CVCs, ostensibly, to support inflow in case of obstruction of the end hole. 15 However, side holes in a catheter may have mechanistic and physiological disadvantages. 16 In presence of side holes, blood flow around and through the tip of the catheter almost instantly removes anticoagulant lock solutions, thus practically favoring clot formation at the tip.17–20 In a computational fluid dynamics analysis, distal side holes present clotting risk at the catheter tip due to the creation of a low flow zone. 21 Finally, creation of side holes can be riddled with imperfections of the cut surfaces, to which thrombi have been shown to attach firmly and irretrievably.15,22 Considering all of the above, the purported association of catheters with side holes with higher infection rates, compared to catheters lacking side holes, 23 follows logically.
Hypothetically, a side hole-free catheter would then be associated with less bacterial growth than a one with side holes. Validating this hypothesis requires attention to potential confounders arising from other disparities in the tip design. 24 The number of these confounders may be reduced by limiting the number of design dissimilarities to the extent possible; for instance, by not comparing staggered-tip (also known as step-tip) and symmetrical tip catheters.
Similar to the symmetrical-tip Palindrome (Medtronic plc, Dublin, Ireland) and the symmetrical-tip, albeit to a lesser extent, GlidePath (Becton, Dickinson and Company, Franklin Lakes, NJ, USA), the Pristine hemodialysis catheter (Pristine Access Technologies Ltd., Tel Aviv, Israel; Figure 1) features symmetry in its split tip configuration but, unlike the two former catheters, its tip is devoid of side holes. This makes it a suitable comparator for the two former catheters, in view of the aforementioned experimental set up considerations.

Pristine hemodialysis catheter features a split symmetrical tip design free of side holes. The tip should be placed in the upper right atrium, oriented in the anterior posterior position to reduce the likelihood of positional occlusion.
Methods
Animals and experimental setup
The study was approved by The Council for Experiments on Animal Subjects, Ministry of Health, Israel. Four Assaf ewes (>24-months old; after one lambing) were used in this study. Animals were allowed free access to food until 24 h before the procedure, at which time access to food was denied. Water was provided ad libitum at all times. Before the procedure, animals were sedated with ketamine 9 mg/kg + midazolam 0.45 mg/kg, intubated and connected to a mechanical ventilator. Anesthesia consisted of 1%–2.5% isoflurane. Ketoprofen was administered at 4 mg/kg before first incision. Tunneled cuffed double-lumen CVCs (insertion length of 19 cm) were inserted in both jugular veins of the four animals according to the instructions for use provided by the catheters’ manufacturers. Y-tip Pristine hemodialysis catheters were percutaneously inserted in one jugular vein in each animal. Inserted in the other jugular vein was either a Palindrome Symmetric Tip Dual Lumen Catheters (hereinafter called Palindrome) or GlidePath Long-Term Dialysis Catheters (hereinafter called GlidePath). Upper right atrium tip position was confirmed fluoroscopically, for all catheters.
The procedures were performed on two separate days, 1 week apart. Two animals were used on each day. Eight tunneled cuffed double-lumen central venous catheters were inserted in the jugular veins of the four animals. Four of the CVCs were the Pristine hemodialysis catheters, 2—Palindrome and 2—GlidePath catheters. The Pristine hemodialysis catheters were inserted in each of the four animals (two in the right internal jugular vein (RIJV) and two in the left internal jugular vein (LIJV). Palindrome and GlidePath were inserted in RIJV and LIJV of two animals each, contralaterally to the Pristine hemodialysis catheter.
Bacterial inoculation and accumulation
To replicate conditions of a systemic bacteremia, Staphylococcus aureus subsp. aureus Rosenbach (American Type Culture Collection (ATCC) strain 6538) bacteria were infused to each animal, bilaterally and simultaneously, through a peripheral 16 G catheter (Becton, Dickinson and Company; Franklin Lakes, New Jersey, United States) placed in the jugular veins cranial to each CVC compared in this study, at a concentration of roughly 0.9 × 1010 colony-forming units (CFU) per kilogram of animal weight. Bacterial infusion lasted 15 min. After bacterial infusion was initiated, all CVC catheters were flushed with normal saline to remove any preexisting clot and to mimic flushing performed in clinical practice. As this was an acute study, animals were kept under anesthesia for 6 h after the bacteria were infused. Catheters were then gently rinsed with normal saline and removed, under sterile conditions; and catheter tip was cut at approximately 4 cm from the distal end.
Euthanasia
Animals were euthanized with an injected barbiturate (pentobarbital) overdose, in agreement with the American Veterinary Medical Association (AVMA) acceptable method for euthanasia of small ruminants. Death was confirmed by the animal facility veterinarian after assessing heartbeat, respiration, and pupillary response to light.
Specimen preparation and bacterial culture
Catheter tips were gently rinsed with normal saline and clot was collected using forceps and homogenized in trypsin-EDTA solution (0.25% and 0.02%, respectively; Biological Industries Israel Beit Haemek LTD, Kibbutz Beit-Haemek, Israel) supplemented with 10% fetal bovine serum (Biological Industries Israel Beit Haemek LTD, Kibbutz Beit-Haemek, Israel). The homogenate was diluted 1:10 in tryptic soy broth (TSB; Hy Laboratories Ltd, Rehovot, Israel) and 20 µL of the mixture were dispersed on tryptic soy agar (TSA; Hy Laboratories Ltd, Rehovot, Israel) plates. TSA plates on which 20µL of a 1:10 TSB dilution of the stock bacteria (ATCC strain 6538) was dispersed served as positive controls. Finally, TSA plates to which an equal amount of the TSB diluent was added and untreated TSA plates were used as negative controls. A blood sample was collected from a vascular sheath placed in the jugular vein and also cultured. The cultured specimens were incubated at 37°C for 12 h before counting colonies.
Results
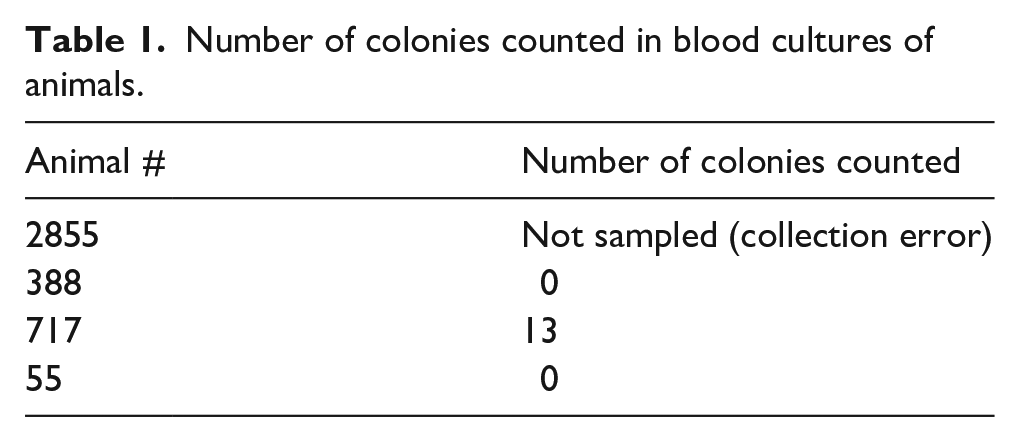
Following the bacterial infusion, one of the animals (717) showed clinical signs of septic shock that resolved with administration of fluids. This was later evident from the results of peripheral blood culture collected 6 h after the inoculation (Table 1), at which time the catheters were removed.
Number of colonies counted in blood cultures of animals.
Clots were present in the tips of all removed catheters (Figure 2). The specimens prepared from the catheters’ tips contents were plated and incubated, and colonies were counted 12 h thereafter.
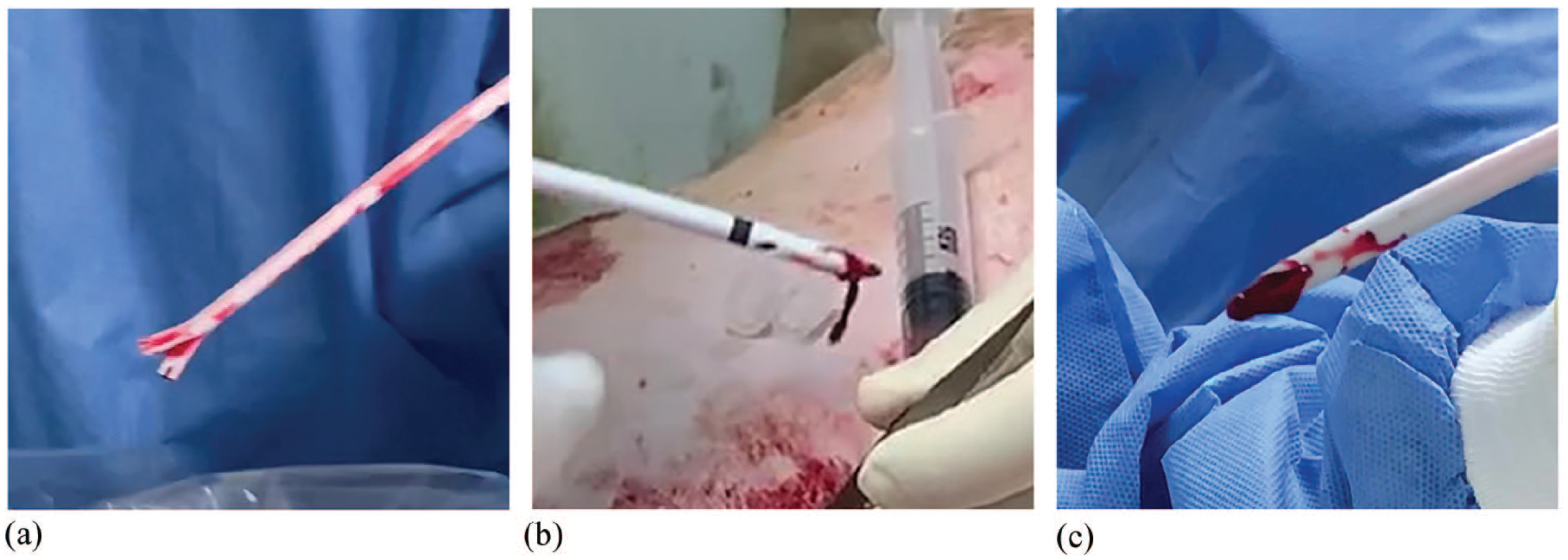
Blood clots accumulated on the catheter tips following removal: (a) pristine, (b) palindrome, and (c) glidepath.
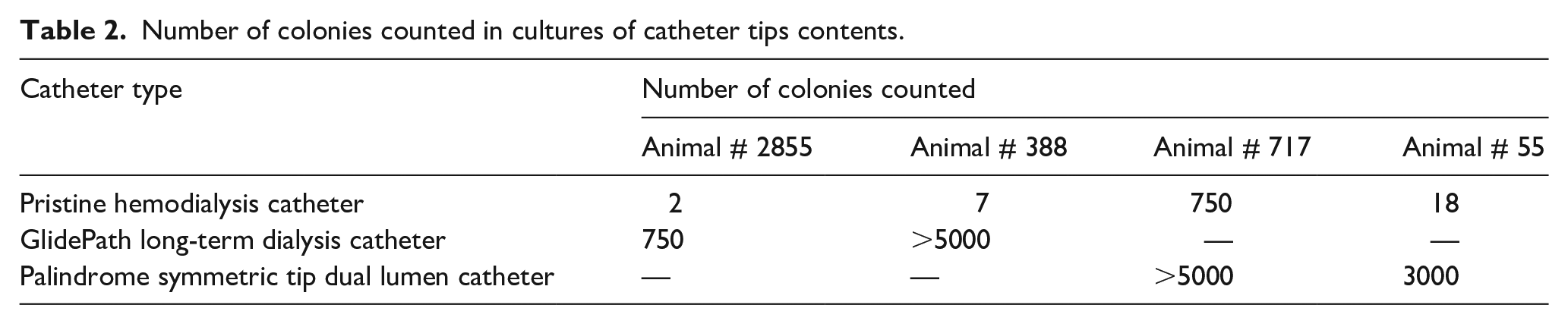
Bacterial colony counts obtained from the tips of both Palindrome and GlidePath amounted to several hundreds to several thousands, exceeding their counterparts collected from the Pristine hemodialysis catheter by at least an order of magnitude (Table 2). In fact, a difference of more than two orders of magnitude was reported for three of the four paired observations, with the only “outlier” being the animal with bacteremia.
Number of colonies counted in cultures of catheter tips contents.
Discussion
Use of cuffed, tunneled catheters is generally indicated for renal replacement therapy longer than 2 weeks.25–27 Cuffed, tunneled dialysis catheters allow for a prompt use without vein puncture during each hemodialysis and, reportedly, associate with a much lower bacteremia rate than that reported for their non-tunneled counterpart.28,29 The 2020 KDOQI guideline further acknowledges that cuffed, tunneled, dual-lumen CVCs have become an acceptable form of hemodialysis vascular access when an arteriovenous access is not suitable or available, despite the associated high complication rates and mortality risk. 4
Still, the CRBSI incidence during CVC use seems to be impervious to change,4,7 despite repeated attempts of all stakeholders. Septic complications of the long-term CVCs may result from contamination of CVC connections, bacterial colonization at the insertion site, and/or along the catheter shaft. Much thought is invested in the material design of modern dialysis catheters to decrease infection rates and to prolong the long-term outcome of catheterization.30–32 On a related note, antimicrobial coating of the catheter surface introduced by some manufacturers—and driving the catheter price up 33 —has been shown to reduce infection in intensive care units, 34 but not to provide significant benefit in the chronic maintenance dialysis population. 35
Thrombosis is a yet another formidable opponent in the battle with CVC-related infections. Microscopically, the fibrin sheath that develops around indwelling catheters promotes the adherence of bacteria, including staphylococci and Candida species.36–38 Clinically, complicated catheter-related septicemia was linked to infected mural (septic) thrombi. 39
The Palindrome catheter introduced in 2005 has a double-D-shaped cross-section of the mid-shaft. Both of its lumens are identical in length, with oppositely angled symmetrical side ports. 40 Aspiration and return of dialyzed blood are carried out through lumens terminating at the same position within the device. The arterial and venous blood admixture is curtailed by the 180° offset of the lumens achieved with a septum with angled crosscuts. This design shows relatively low recirculation. 40 The GlidePath catheter is another, more recent, double D catheter. Admixture of blood in this catheter is reduced through introduction of curved distal apertures on opposing sides of the catheter. The guidewire aperture at the distal tip, as part of the venous lumen, and the offset side holes contribute asymmetry to the otherwise symmetrical GlidePath’s tip. In computational analysis, Palindrome and Glidepath showed comparable percentages of blood moving out of the catheter from the tip, as well as comparable flow rates through side holes. 41 In that computational analysis, the most prominent flow stagnation regions were detected around side holes and terminal apertures, where laminar flow from the catheter tip is interrupted by inflow from the side holes. 41
Since frequent vascular access places hemodialysis patients at increased risk for accidental inoculation and transient bacteremia, the clot in the catheter tip may become infected. 16 As hypothesized, in this limited in vivo study of three catheters featuring tip symmetry, the side-hole-free Pristine hemodialysis (cleared by the United States Food and Drug Administration and CE-marked) catheter outperformed the Palindrome and GlidePath catheters with respect to the bacterial load associated with the aforementioned clot. The difference in the amount of accumulated bacteria reported here may translate to a reduced CRBSI rate and subsequent infectious complications in end-stage renal disease (ESRD) patients. Despite the obvious limitations of this study, such as the low number of animals involved, the relatively short catheter dwell time, and the heavy inoculation performed with only one organism, the observed difference, varying between one and three orders of magnitude, may stem from a genuine determinant of significance rather than be attributed to a statistical chance of an error. The reduced amount of bacteria accumulating at the symmetric side-hole-free tip of a hemodialysis catheter, compared to its counterpart at the symmetric tips of catheters that have side-holes, may decrease the risk of CRBSI and have a positive impact on ESRD patients.
Footnotes
Acknowledgements
We thank Dr. Leonard Mermel for advising on the animal model, Dr. Igor Ruvinsky for his help in drafting the manuscript, and Da-Ta Biotech (Rehovot, Israel) for assistance with the in vitro study model.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have equity in Pristine Access Technologies Ltd., which manufactures and markets the Pristine hemodialysis catheter.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by Pristine Access Technologies Ltd.
