Abstract
Intestinal failure (IF) patients are dependent on central venous access to receive parenteral nutrition. Longstanding central venous catheters are associated with life-threatening complications including infections and thromboses resulting in multiple line exchanges and the development ofprogressive central venous stenosis or occlusion. The Haemodialysis Reliable Outflow (HeRO) graft is an arterio-venous device that has been successfully used in haemodialysis patients with ‘end-stage vascular access’. We describe a case series of HeRO graft use in patients with IF and end-stage vascular access. Four HeRO grafts were inserted into IF patients with end-stage vascular access to facilitate or support intestinal transplantation. In all patients the HeRO facilitated immediate vascular access, supporting different combinations of parenteral nutrition, intravenous medications, fluids or renal replacement therapy with no bloodstream infections. In a highly complex group of IF patients with central venous stenosis/occlusion limiting conventional venous access or at risk of life-threatening catheter-related complications, a HeRO® graft can be a feasible alternative.
Keywords
Introduction
Intestinal failure (IF) is a life-threatening condition that is diagnosed when patients lack the ability to adequately absorb sufficient nutrients via the gut and consequently become reliant on intravenous nutritional supplementation to maintain their health. 1 This is dependent on adequate central venous access, which can become problematic to maintain and preserve, typically requiring management from specialist intestinal and vascular access services. Patients established on intravenous nutrition, have in-dwelling central venous catheters (CVCs) for prolonged periods and are at high risk of a serious infection, catheter dysfunction and central vein pathology. Catheter-related bloodstream infections (CRBSIs) are the main indication for repeated catheter replacements and in themselves carry high morbidity and an estimated 12.5% to 25% mortality.2,3 Infections are frequently preceded by some form of catheter dysfunction, resulting from intraluminal blockage, a fibrin sheath, catheter tip thrombus or mural thrombus. Once established, CRBSIs are not only medically challenging to manage but are also associated with a large economic burden. 4
The combination of repeated intimal trauma exerted by the catheter against the vessel wall along with episodes of line CRBSIs and thrombosis leads to permanent and irreversible scarring and fibrosis. Over time, patients develop central venous stenosis and occlusions which make subsequent access placement increasingly challenging. Less favourable sites such as the groin veins or lumbar veins are often attempted, but carry an even greater infection and thrombosis. A vicious cycle of consuming venous real estate ensues, resulting in ‘End-Stage Vascular Access’. 5 This, combined with recurrent CRBSIs, poses major risks to IF patients and consequently, can be an indication for intestinal transplantation.
Several alternative approaches have been adopted to deliver PN in patients with catheter-related complications. One of these alternatives, is the formation of peripheral limb arteriovenous fistulas (AVF) such as those typically used in end-stage renal failure patients for haemodialysis access, therefore offering a peripheral access opportunity for IF patients. However, despite demonstrating lower infection rates and higher delivery flow rates (shortening TPN delivery times), success has been limited by poor access patency rates. 6 This poor patency is likely attributable to the inability to overcome the pre-existing central stenosis caused by multiple previous CVCs, as the AVF’s have often been created late in the patients IF journey rather than at an earlier point before multiple lines have been inserted. In addition, an AVF created in the limbs in the presence of central vein pathology can often result in marked morbidity from the resultant venous hypertension manifested by limb and sometimes facial and neck swelling.
The Hemodialysis Reliable Outflow (HeRO®) graft
The Hemodialysis Reliable Outflow (HeRO®) graft (Merit Medical Systems, Inc, South Jordan, Utah) is a novel device licenced in 2008 by the Food and Drug Administration (FDA) for use in haemodialysis (HD) patients with central venous stenosis or obstruction described as ‘end-stage vascular access’.5,7 It is a two-component device with an outflow stent graft and an inflow ePTFE (Polytetrafluorethylene) graft (Figure 1). Crucially, the HeRO® graft has no external component which is thought to be critical in avoiding the higher infection rates that are observed with central catheters, which have these components. 8 The only alternative intervention for central venous stenosis is repeated radiologically guided venoplasty, but this often futile, resource intensive and unsustainable in the long term. 9
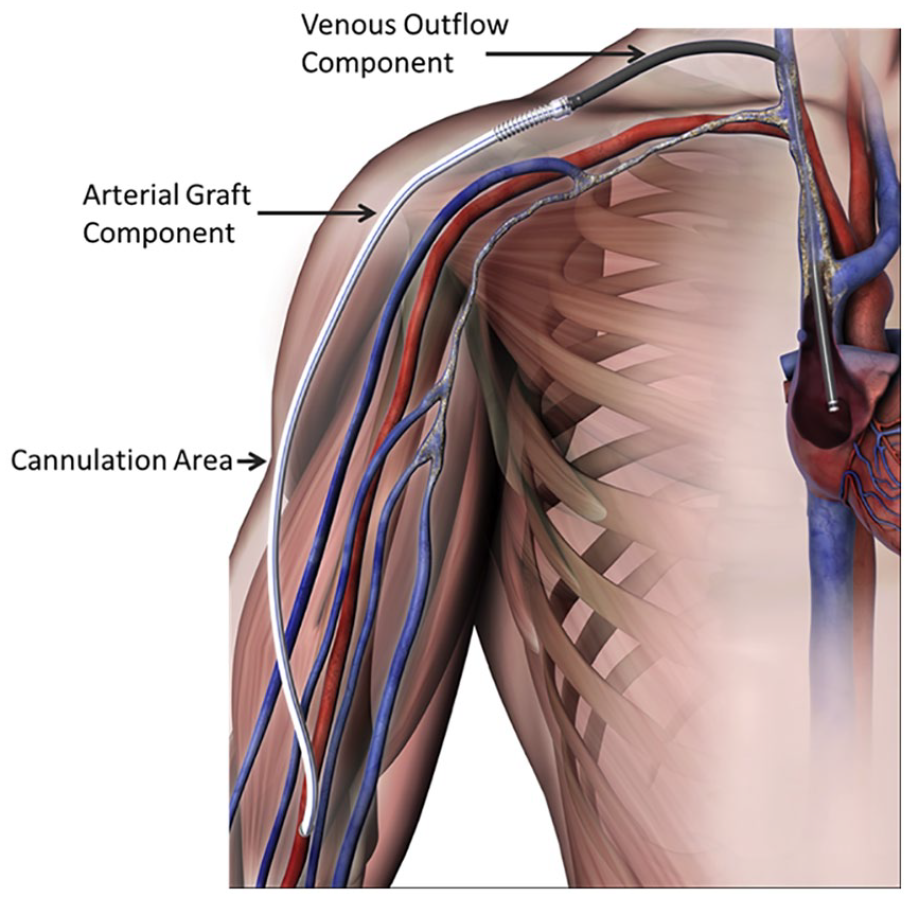
HeRO graft: demonstrating a right brachial arterial inflow, arterial cannulation area (graft), connector and venous outflow component.
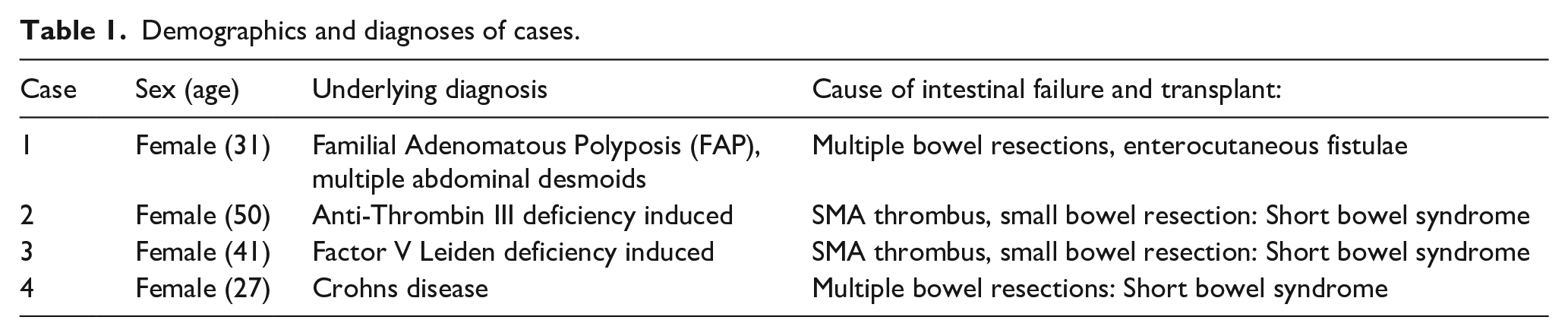
We describe the first reported experience of the use of HeRO® graft in four intestinal failure and intestinal transplant recipients (Table 1) with complex vascular access.
Demographics and diagnoses of cases.
Description
Case 1
A 31-year-old female with Familial Adenomatous Polyposis Coli, desmoid tumours and intestinal failure due to multiple small bowel resections. She underwent intestinal transplantation and lost central venous access in the immediate post-operative period while she remained dependent on supplemental PN, intravenous fluids and medications. A venogram demonstrated bilateral subclavian stenoses and superior vena cava (SVC) stenosis, thrombus in both common femoral veins (CFVs), an occluded left common iliac vein (CIV) and a stenosed but patent infra renal inferior vena cava (IVC). A decision was made to insert a HeRO® graft. The outflow component was placed into the right atrium via the left internal jugular vein (IJV) following a venoplasty of the SVC and the graft obtained inflow from the left brachial artery. The HeRO® graft was able to support her ongoing nutritional and fluid requirements and facilitate a recovery sufficient to allow her transplant graft to function. The graft was successfully used for phlebotomy, additional fluid therapy and for PN at home and as an inpatient. At the time of writing primary and secondary patency were 1235 and 1748 days respectively with eight salvage procedures and no other complications.
Case 2
A 50-year-old female with Anti-thrombin III, multiple pulmonary emboli and short gut syndrome due to small bowel resection following a superior mesenteric artery (SMA) thrombus in 2006. She had a functioning intestinal allograft for 5 years when she presented with severe cytomegalovirus disease with pulmonary manifestations and further pulmonary emboli. Critically unwell, she required haemofiltration, TPN and IV vasoactive medications. After failure to obtain central venous access, a venogram was performed which showed longstanding occlusion of the left axillary vein with drainage via collaterals. Superior cavograms showed occluded subclavian veins (SCVs) and innominate veins bilaterally and an occluded SVC. Inferiorly, she had bilateral external iliac vein occlusions but a patent IVC. Due to a lack of upper limb access, a lower limb HeRO® graft was sited. This was done in a J-configuration, for additional graft length, from the left superficial femoral artery via the left CFV with the outflow stent located in the patent IVC. The HeRO® graft facilitated renal replacement therapy (RRT), PN, intravenous fluids and medications for 52 days without complications and despite prolonged intensive treatment she died of multi-organ failure with a functioning HeRO® graft.
Case 3
A 41-year-old female with short bowel syndrome due to small bowel resection following an SMA thrombus precipitated by underlying Factor V Leiden deficiency. She underwent intestinal transplantation and 3 years later developed ischaemic enteritis of the intestinal allograft requiring explant and return to PN. Definitive central venous access was required to permit this procedure. A venogram showed the SVC and left brachiocephalic vein were occluded, with the drainage of her right arm going into a very large azygous vein. She underwent a HeRO® graft insertion with access to the SVC via her right IJV and after an SVC venoplasty, the outflow component was positioned in the right atrium through the SVC stenosis. Her arterial inflow was via the right brachial artery. She was discharged self-cannulating the graft and other than an MRSA needle site infection, which was treated with oral antibiotics, she had 177 days of graft use without complication. She decided upon withdrawal of treatment and died with a functioning HeRO® graft.
Case 4
A 27-year-old female with fistulating Crohn’s disease who underwent multiple small bowel resections and abdominal operations for pelvic sepsis. She underwent intestinal transplantation but developed chronic rejection and renal failure following repeated episodes of dehydration and kidney injury. She required RRT and TPN simultaneously and a HeRO® graft was inserted to address these requirements while avoiding an in-dwelling catheter. Her venogram showed absent IJVs bilaterally and stenosed but patent subclavian veins. A decision was taken to insert a HeRO® graft to address her ongoing RRT, intravenous fluids and TPN requirements while avoiding an in-dwelling catheter that might cause a life-threatening bacteraemia or further episodes of endocarditis that would disqualify her eligibility for intestinal transplantation. A HeRO® graft was inserted via her left subclavian vein into her right atrium, with inflow coming from her left brachial artery. She had primary patency of 23 days and secondary patency of 359 days, successfully using the HeRO® graft for haemodialysis, TPN and IV fluids. She required 15 interventions to maintain the patency of the graft, after experiencing recurrent episodes of graft thrombosis, but had no other graft-related complications (Table 2). Due to the number of procedures, the patient decided to have a tunnelled haemodialysis catheter Tesio®line (MedCOMP, Harleysville, Pennsylvania) inserted and the graft removed.
Outcomes of HeRO graft in intestinal failure patients.
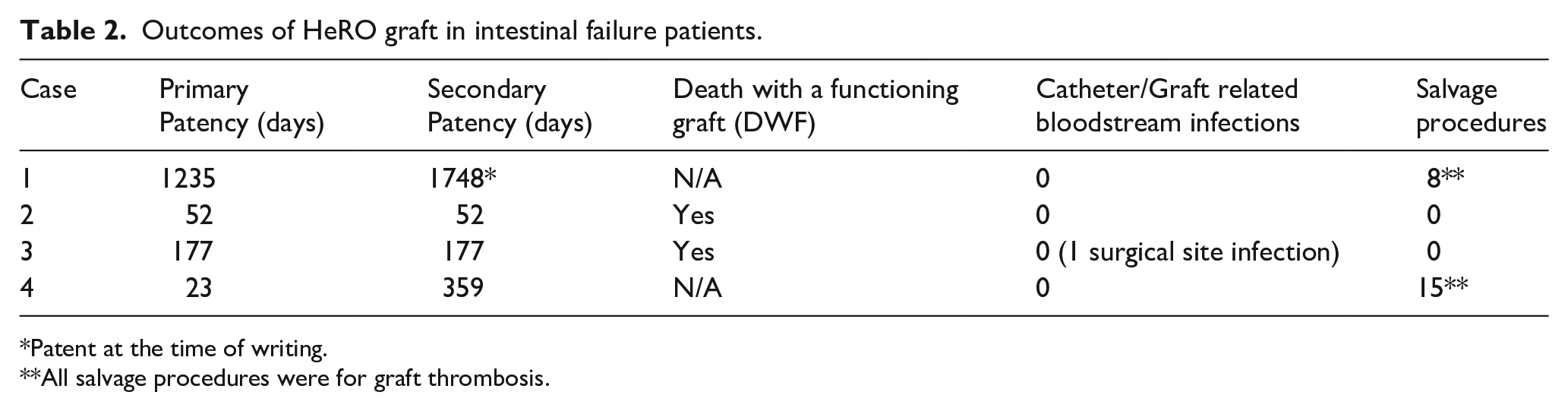
Patent at the time of writing.
All salvage procedures were for graft thrombosis.
Discussion
This case series represents the first reported experience of HeRO® grafts for central venous access in intestinal failure patients. Our results indicate this is a safe and effective option in end-stage vascular access patients with intestinal failure or intestinal transplants with an ongoing requirement for PN, intravenous medications, fluids or RRT. The HeRO® graft in this context has demonstrated acceptable primary and secondary patency rates and minimal infective complications. Two of the four patients in our series died with functioning access and of the two surviving patients (at time of writing) one patient has functioning access and the other had the HeRO® graft removed due to patient preference. There was one infective complication, which was a superficial skin infection in Case 3 which grew MRSA on a skin swab, but this was managed with oral antibiotics and did not result in a HeRO® graft related bloodstream infection. Our findings are in keeping with the data from previous HeRO® experience in the haemodialysis population where the most recent systematic review of HeRO® graft use shows significantly reduced bacteraemia episodes and comparable primary and secondary patency rates to other secondary access series in the complex vascular access literature. 10 This combination of low infective complication rate while managing to overcome the central venous pathology in IF patients who have been burdened with multiple catheters makes the HeRO® a compelling option in IF. However, there were some drawbacks, as was seen in Case 4. The HeRO® graft can be associated with multiple salvage operations related to thrombosis, which significantly impacts the patient experience and economic burden. Due to the high number of salvage procedures, the patient requested the graft to be removed, specifically highlighting the issue of multiple interventions. Furthermore, the lack of widespread exposure of clinical staff to HeRO® grafts necessitates specialist nurse input and individualised patient training with regard to needling, graft surveillance and overall access management. 7 The adoption of this treatment option is therefore contingent on access to these skills, which are typically only available in Nephrology/Haemodialysis departments that are not necessarily co-located with IF and intestinal transplant centres. There were some auxiliary advantages of using the HeRO® graft in IF patients identified in this series. An arteriovenous circuit with flow rates between 700 and 1500mls/min returning to the right atrium provided the opportunity to deliver the daily PN volume over faster rates and substantially shorter periods. This was of course done being mindful of re-feeding syndrome and we found no discernible biochemical or haemodynamic consequences. Furthermore, the cannulation of the grafts was successfully taught to patients who were able to easily administer home PN via the graft using an innovative closed needling system that reduced the number of graft cannulations from daily to every 72 h without any bloodstream infections.
Presently, the HeRO® graft is only indicated in late-stage complex vascular access patients with established central venous stenosis. However, intervening earlier in the patient journey, before the development of central venous pathology with the formation of arteriovenous peripheral access may reduce CRBSIs, avoid the central pathology, and preserve venous real-estate while facilitating more efficient PN delivery in a more efficient way than traditional tunnelled central catheters. It should be noted that with only four cases, the experience is still very limited and the encouraging outcomes are from a single institution with a large experience of HeRO® graft use, intestinal failure and transplant patient management.
Conclusion
This series has demonstrated that use of the HeRO® graft in a carefully selected group of patients may offer an innovative solution to end-stage vascular access patients with intestinal failure (IF) or intestinal transplant with multiple ongoing requirements for PN, intravenous medications or RRT where a high risk of catheter-related infections or complications is unacceptable. HeRO® grafts are associated with minimal infective complications, acceptable secondary graft patency, and have proven safe and effective in this patient population. Further experience, where appropriate, should be sought of HeRO® grafts in intestinal failure with complex vascular access to identify the patients that benefit most from this treatment option.
Footnotes
Acknowledgements
The authors would like to thank the specialist nurses Jo Carter and Juniya John for their exceptional work in the clinical management of the vascular access for the cases included in this manuscript.
Author contributions
F.D. contributed to the acquisition and analysis of the data, the interpretation and drafting of the manuscript. J.H., J.G. and G.V. contributed to the conception and design of the manuscript, all authors were involved in data interpretation and ensured the accuracy and integrity of clinical information presented.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: J.G. reports receipt of speaker and advisory fees from The Merit Medical, the distributor of the HeRO® device. No other authors have any financial disclosures.
Ethical approval
Informed consent for use of anonymised data for audit was sought and obtained at the time of treatment. Ethics approval is not required for case reports or case series and they are not deemed to constitute research at our institution.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
