Abstract
Acetabular augmentation is a challenge for complex primary and revision hip surgery. Several methods exist to recreate a supportive, hemispherical acetabulum where a defect exists, however, these can be complex, expensive, time consuming, and limited by available size and shape. We present a simple, inexpensive, quick and customisable method of acetabular augmentation using cementless polyethylene with titanium coating. The first case is presented with follow-up up to 8 years.
Introduction
A supportive acetabulum is a prerequisite for good acetabular fixation, necessary to support a hemispherical implant and provide primary stable fixation. Where the acetabulum is defective, the reconstruction remains a challenge. Severe acetabular defects can be found in some primary and revision cases where the shape of the acetabulum is incompatible with hemispherical preparation, thus compromising implant stability and fixation. Several methods exist to address this problem, including tantalum augments, 1 bulk allograft, 2 mesh and impaction grafting, 3 jumbo cup, 4 oblong cup, 5 cages and custom implants.6,7 However, customisable solutions (trabecular metal and custom implants) are expensive, and require advanced ordering or storage of multiple sizes, while other methods give concern with regards to poor long-term survival and osteointegration of graft and excessive removal of bone and high hip centre.8,9
We present a simple technique to custom fit a cementless polyethene augment to reconstruct acetabular defects for implantation of a standard cup. This method has been successfully used in our institution since 2011 in patients with defects that would result in an unsupported cup, and we present the first case with long-term follow-up.
Surgical technique
Surgery is performed through the standard posterior approach in the lateral position. Standard acetabular preparation is performed according to the hip centre and using the transverse acetabular ligament (TAL) as guide. 10 The acetabular defect is evaluated (in this case Paproski Type 2b), with an augment used if the cup is unsupported (<50% coverage) (Figure 1(a)). Standard preparation, as for trabecular metal augments, is achieved with acetabular reamers to create a hemispherical shape for the desired augment (Figure 1(b)). 1 A line-to-line sized augment is chosen (Figure 1(c)). The augment is custom-made from a segment of the all-polyethylene RM Pressfit cup (Matthys Ltd, Bettlach, Switzerland). As it is made of polyethylene with a single layer of titanium, it can be reamed in situ to the required acetabular size, in a similar manner to bulk bone graft, however, it is cut with a reciprocating or oscillating saw prior to implantation while on the cup holder to reduce the reaming required (Figure 2). It is our observation that the reamer cuts through the titanium without any excess torque, with any residual titanium shell being thin enough to cut flush to the polythene with a knife. The augment is impacted into the reamed defect and secured with screws through pre-drilled holes to augment the obtained press fit (Figure 3(a)). The polyethylene is soft enough that additional screw holes can be drilled if required using a standard 2.5-mm drill and countersink. Once the unfinished augment has been fixed to the defect it is reamed using sequential reamers, to match the size and position of the pre-reamed acetabulum positioning it parallel to the transverse acetabular ligament (TAL) for version and hip centre level (Figure 3(b) and (c)). Reamings of polyethylene are removed manually or by washing in similar manner reaming out a cemented cup, or removing a well-fixed RM cup. 11 The cementless acetabular component is then implanted in the usual way (Figure 4). In all our cases primary stability was satisfactory but additional prophylactic screws are used to enhance stability due to the complexity of the cases allowing fixation of the complete cup to the augment to create a monoblock, as well as to bone. No additional fixation such as cementation between components has been used or required.
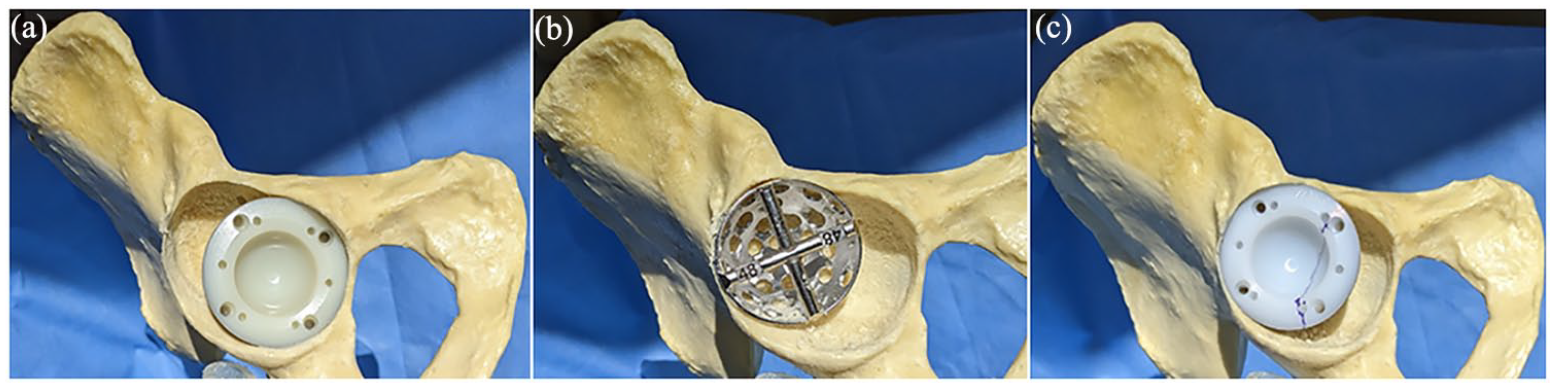
(a) Cup placed in reamed acetabulum to assess defect, (b) defect reamed to accept augment, and (c) RM augment 161 placed in defect and marked.

(a) Acetabular component cut with recipricating saw and (b) cut acetabular component.
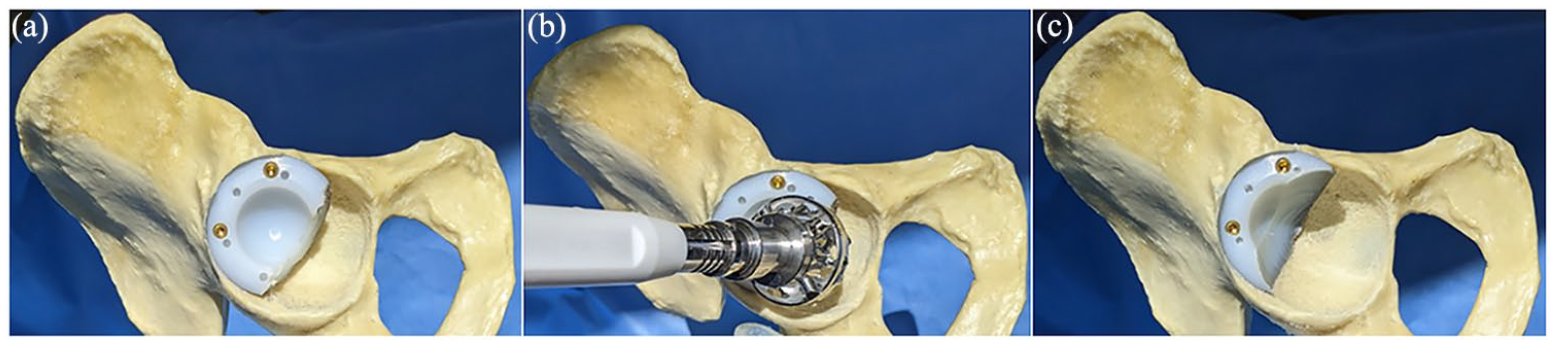
(a) Cut RM augment screwed into defect, (b) RM augment reaming, and (c) Reamed RM augment in situ.
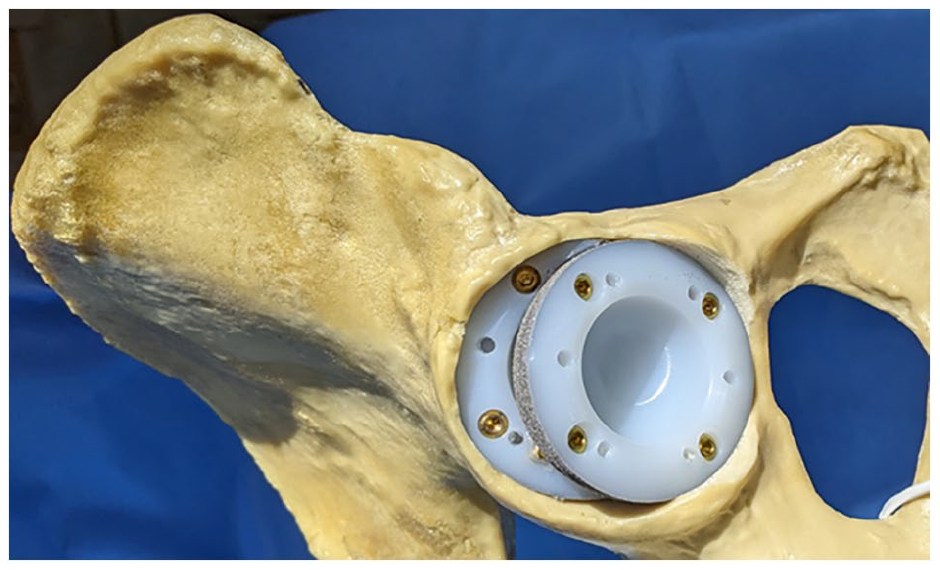
Final augment and cup position.
Case presentation
A 58-year-old woman with a 3-year history of left hip pain presented for total hip arthroplasty. On examination she was found to have 4½ cm of shortening, and x-ray revealed gross destruction of femoral head and acetabulum (Figure 5(a)). The acetabular defect was prepared and reconstructed as described above, with an uncemented cup placed in the reconstituted acetabular hemisphere (Figure 5(b)). Her Oxford Hip Score was 33 preoperatively, improving to 47 at 1 year. She was followed-up to 8 years and reported very good satisfaction with her pain and function. Computed tomography (CT) and x-ray showed satisfactory cup position (Figure 5(c) and (d)).
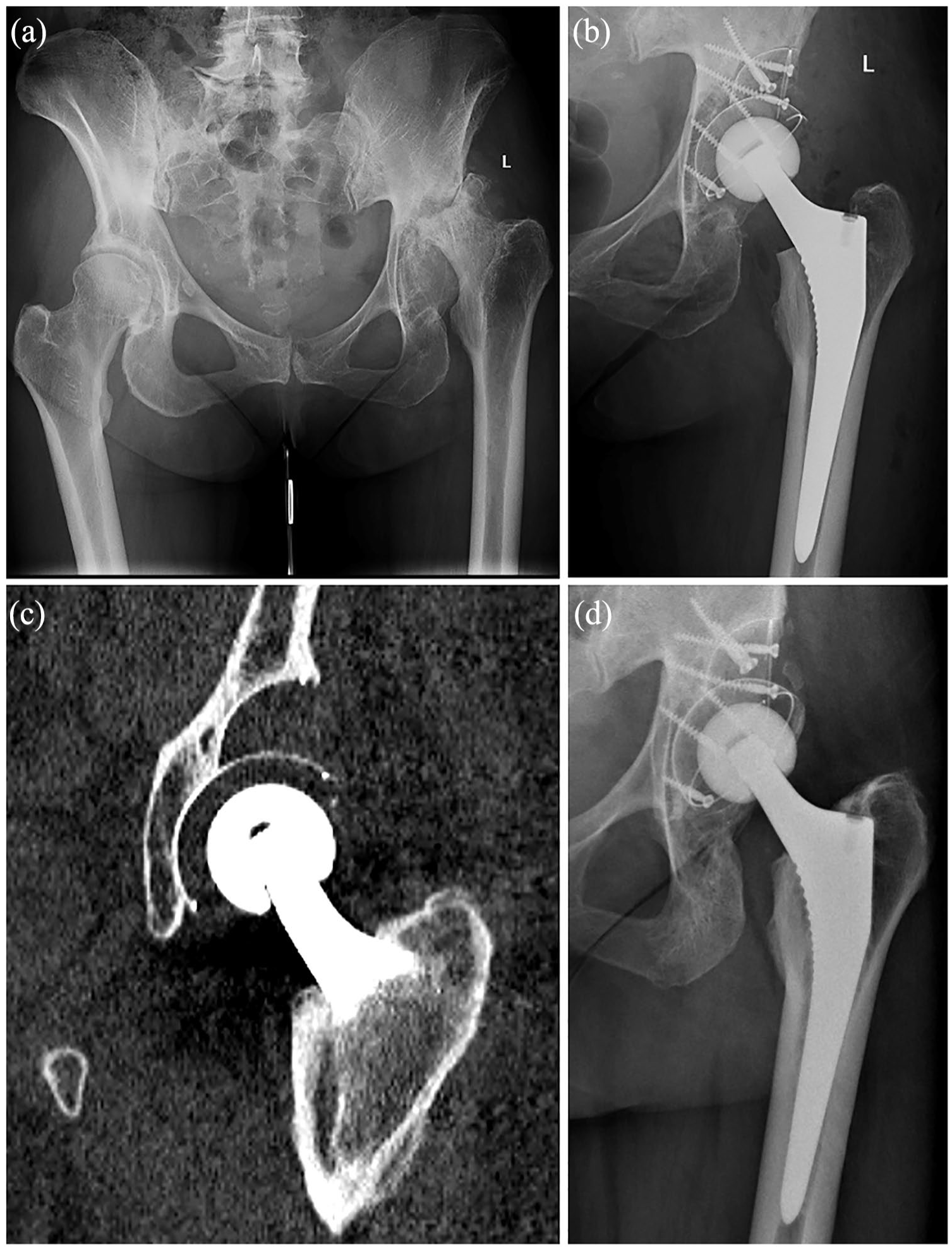
58-year-old lady with left hip pain and gross femoral head and acetabular destruction: (a) preoperative 174 radiograph, (b) acetabular reconstruction with all polyethylene augment and uncemented cup, (c) 8-year follow-up CT 175 scan, and (d) 8-year follow-up CT scan showing osteointegration.
Discussion
Acetabular reconstruction in the presence of large defects continues to be a challenge in both primary and revision hip surgery. While many methods exist to deal with this challenge, each has associated limitations. Customisable solutions (trabecular metal and custom implants) are expensive and require advanced ordering or storage of multiple sizes. Bulk bone graft has high reported rate of late failure due to poor osteointegration while jumbo cups have good survival, but raise the joint centre and can result in removal bone such that re-revision is difficult.8,9
The use of cementless polyethylene acetabular augments has not been described before. The all-polyethylene augment described in this paper is based on the RM Pressfit cup (Mathys Ltd, Bettlach, Switzerland). This is an all polyethylene monoblock cementless cup coated with titanium particles. This allows the cup to remain isoelastic with the surrounding bone. The coating comprises a thin layer of non-correlating, individually anchored titanium particles that have no structural stiffness between them. This fact coupled with the elasticity of the ultra-high molecular weight polyethelene, which is similar to that of human cancellous bone, 12 ensure that the load transfer and the biomechanical behaviour of the cup remain physiological after implantation, with the aim of preventing stress shielding. 13 The RM Pressfit cup achieves its primary stability through an equatorial press-fit. Aided by the flattening of the polar region, compressive forces are guided towards the periphery of the acetabular cup, 14 As the augment is made of a section of this polyethylene cup, it can be reamed in situ after filling the defect to the required acetabular size to provide pressfit fixation for the acetabular component in a similar way to bulk bone graft. This is an advantage over conventional metal augments, which are very difficult to customise, and does not interfere with restoration of the hip centre.
Conclusion
This paper describes a simple, customisable and inexpensive method for reconstructing acetabular deficiencies in primary and revision hip arthroplasty. It is quick, requiring no additional specialist equipment or technique, and does not require additional kit ordering or storage, while preserving bone and restoring hip centre.
Footnotes
Acknowledgements
Mathys Medical (Bettlach, Switzerland) – manufacturer of the RM Pressfit cup used to create the augment - provided the sawbones, instruments and implants used in the photographs in the article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KS and JB have received fees for lecturing for Mathys Medical. KS and JB have contributed to research studies funded by Mathys Medical.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
