Abstract
Purpose
To demonstrate non-inferiority on efficacy and safety of preservative-free bimatoprost 0.01% versus preserved and higher-concentration bimatoprost formulations for the treatment of glaucoma or ocular hypertension (OHT), using a network meta-analysis (NMA).
Methods
A systematic review and Bayesian NMA were conducted according to a pre-registered protocol (PROSPERO: CRD420250650769) and PRISMA-NMA guidelines. Randomised controlled trials comparing monotherapies of bimatoprost (preserved or preservative-free) in adults with glaucoma or OHT were included. The primary endpoint was mean intraocular pressure (IOP) at Week 12. Conjunctival hyperaemia, were descriptively analysed. Risk of bias was assessed using the Cochrane RoB 2 tool.
Results
Four studies were included. The Bayesian NMA demonstrated that preservative-free bimatoprost 0.01% was non-inferior to all other bimatoprost formulations in lowering IOP, with all 95% credible intervals falling within the 1.5 mmHg non-inferiority margin. Sensitivity analyses using a frequentist model confirmed these findings. Safety analysis indicated that preservative-free bimatoprost formulations were associated with lower rates of hyperaemia compared to preserved versions. At Week 12, more patients had no hyperaemia with preservative-free bimatoprost 0.01% (57.3% vs. 43.6%) and fewer showed worsening scores (18.3% vs. 30.4%). Hyperaemia reported as an adverse event was also less frequent with bimatoprost 0.01% than 0.03% (28.6% vs. 37.4%).
Conclusions
Preservative-free bimatoprost 0.01% provided IOP-lowering efficacy similar to preserved or higher-dose bimatoprost, with less occurrence of conjunctival hyperaemia. Further long-term studies are needed to confirm these benefits.
Keywords
Introduction
Glaucoma is a group of progressive optic neuropathies marked by the degeneration of retinal ganglion cells, ultimately resulting in vision loss and blindness. 1 To date, elevated intraocular pressure (IOP) is the only modifiable risk factor associated with glaucoma. 1 To manage elevated IOP, a range of topical ocular hypotensive agents are used, with prostaglandin analogs being among the most commonly prescribed first-line treatments.1,2
Traditionally, preservatives such as benzalkonium chloride (BAK) have been added to topical antiglaucoma medications to maintain sterility. BAK may also enhance corneal penetration, associated with the hope to increase the efficacy of active pharmaceutical ingredients. 3 However, not all studies found increased penetration 4 and multiple clinical trials have demonstrated that preservative-free formulations offer comparable IOP-lowering efficacy with reduced ocular surface toxicity.5–7 It is now well established that BAK can disrupt the lipid layer of the tear film, cause conjunctival inflammation, and lead to epithelial cell damage with chronic use due to its detergent properties.8,9 In addition, it is well established that the lowest effective dose of an anti-glaucoma drug should be used providing the target therapeutic effect while minimizing adverse events. 10 Overall, the adverse effects related to BAK, other preservatives, and/or high concentration of active ingredients can reduce patient comfort, induce poor adherence, and decrease quality of life, critical concerns in the chronic management of glaucoma.11,12
Since poor treatment adherence leads to poor IOP control,13–15 efforts have been made to address these tolerability issues. This includes removing the preservative and lowering the concentration of active ingredients in topical therapies. Bimatoprost, a synthetic prostamide analogue related to prostaglandin F2α, is widely used for lowering IOP.16–18 The original formulation, bimatoprost 0.03% preserved with BAK, demonstrated robust IOP-lowering efficacy (25–30% from baseline), but was frequently associated with adverse events such as conjunctival hyperaemia.19–21 To improve tolerability without sacrificing efficacy, a series of reformulations were developed: first, a preservative-free formulation of bimatoprost 0.03%, and later, a lower concentration formulation (0.01%), but containing higher preservative concentration than preservative-free bimatoprost 0.03%. Clinical studies confirmed that the preserved 0.01% formulation maintained comparable efficacy while significantly reducing side effects.5–7 In this context, a new preservative-free formulation of low-concentration bimatoprost 0.01% has been introduced.
Despite these developments, there remains a lack of comprehensive data directly comparing the efficacy and safety of the preservatove-free 0.01% formulation with other available bimatoprost formulations. This evidence gaps creates uncertainty for clinicians when selecting the most appropriate therapy, particularly for patients at risk of ocular surface disease.
To address this need, we performed a network meta-analysis (NMA) to evaluate whether preservative-free bimatoprost 0.01% demonstrates non-inferiority of the efficacy, and safety compared with other monotherapies of bimatoprost at different concentrations and formulations available for the treatment of glaucoma or ocular hypertension (OHT). The results of this analysis aim to provide evidence-based guidance for clinicians when considering preservative-free or low-concentration options for patients at risk of ocular surface complications.
Methods
Eligibility criteria were based on PICO method.

Results
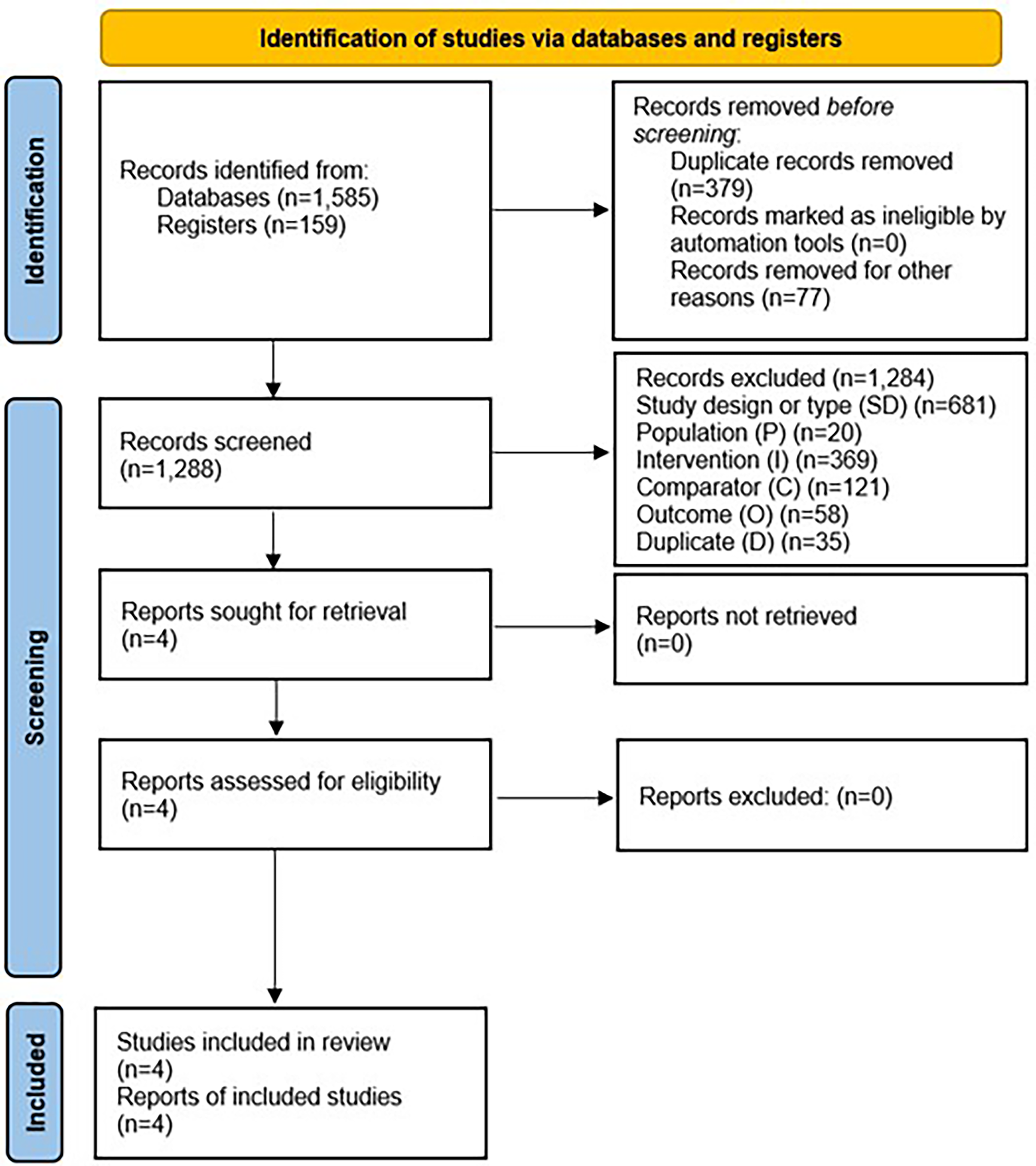
PRISMA flow .
Description of included studies.
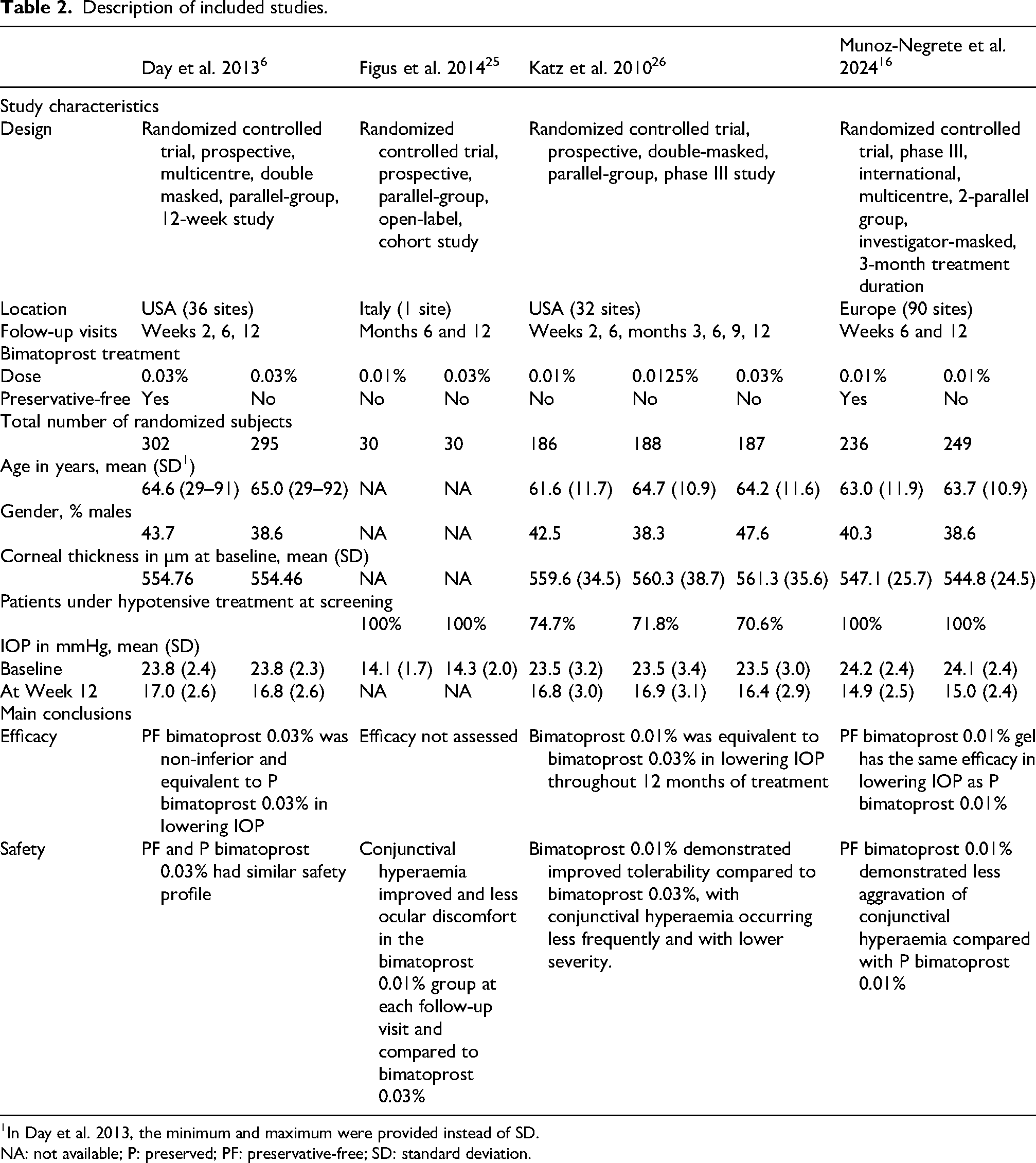
In Day et al. 2013, the minimum and maximum were provided instead of SD.
NA: not available; P: preserved; PF: preservative-free; SD: standard deviation.
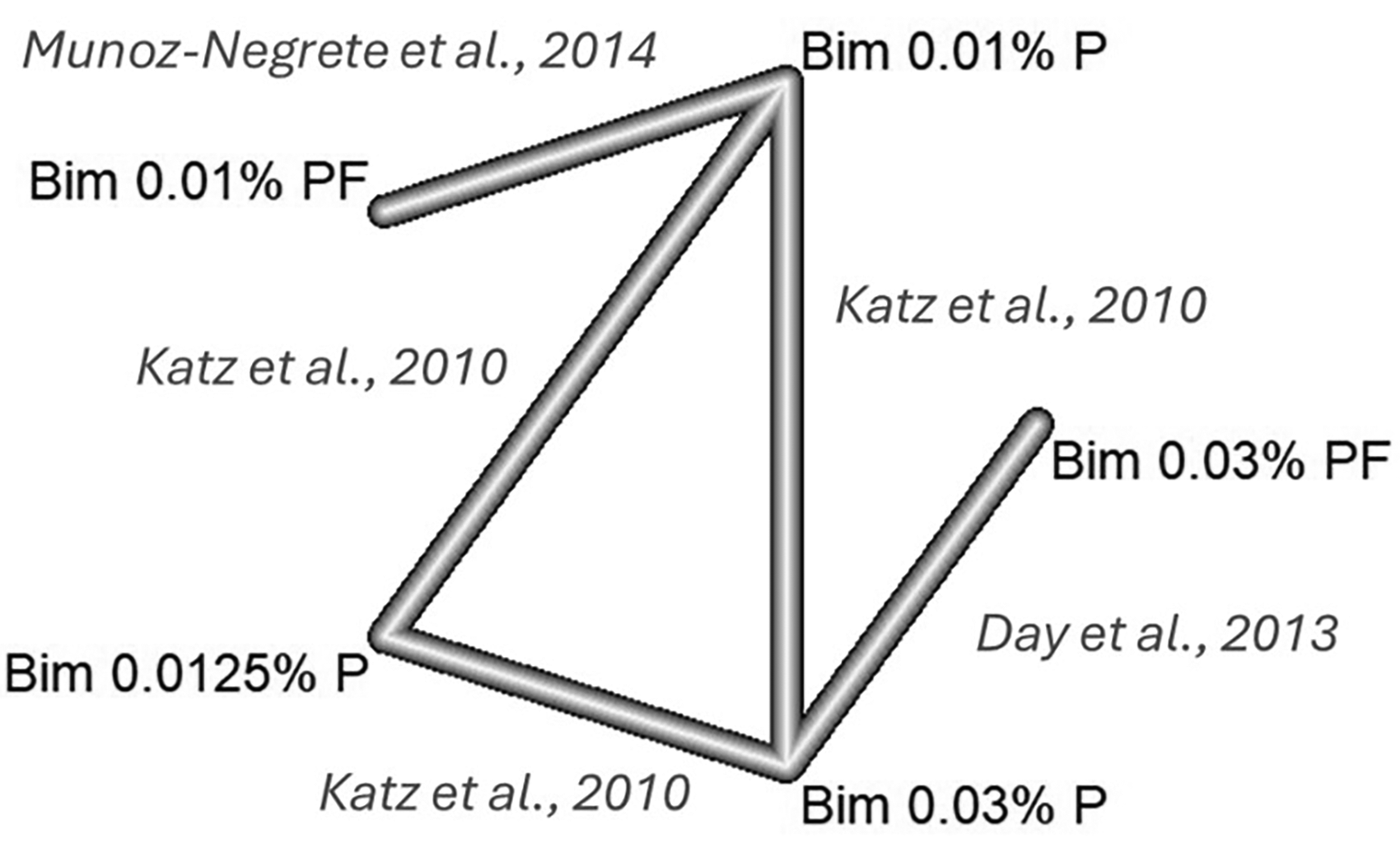
Network for mean intraocular pressure at week 12 (P: preserved; pf: preservative-free)
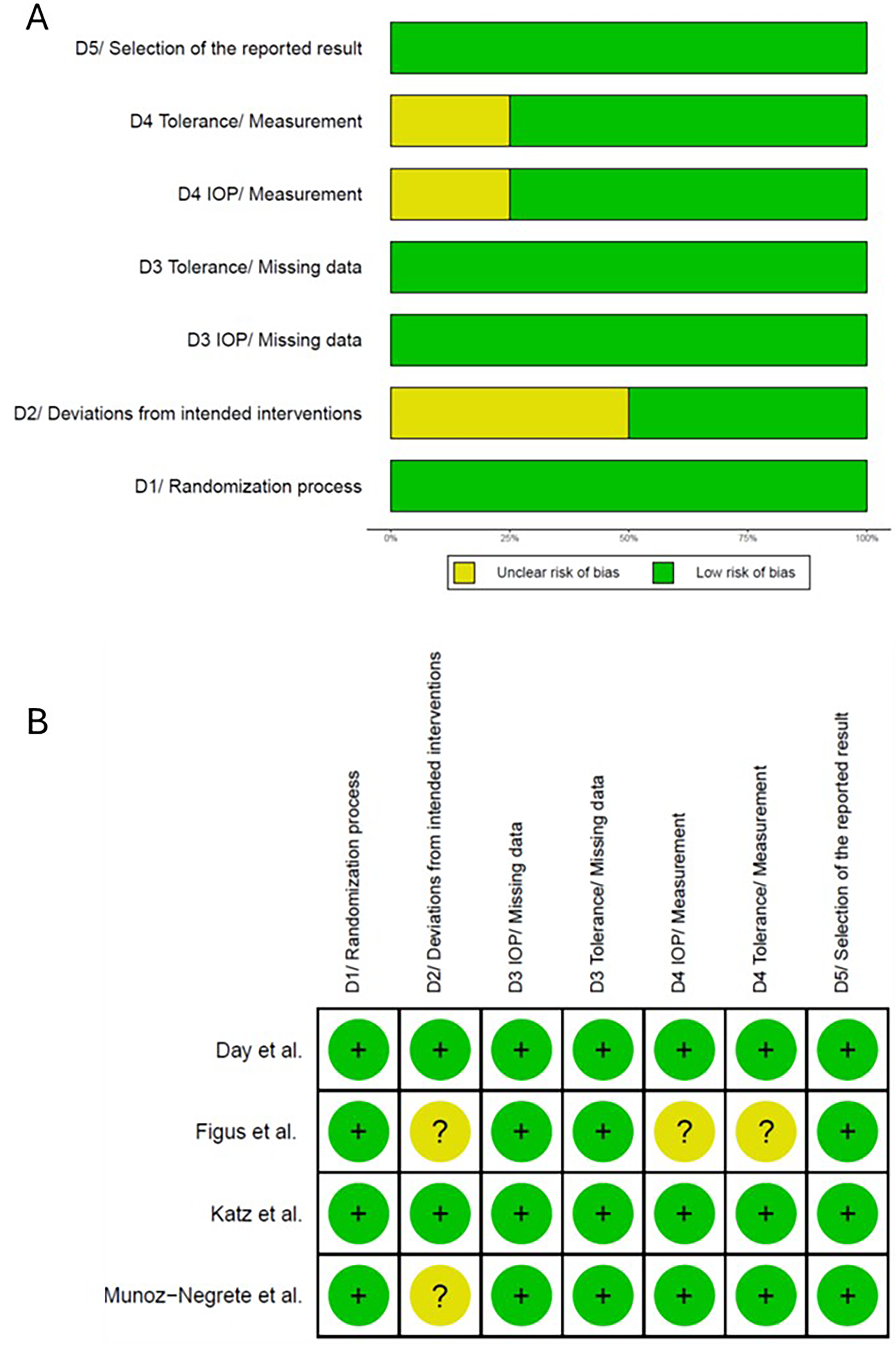
Risk of bias assessment.
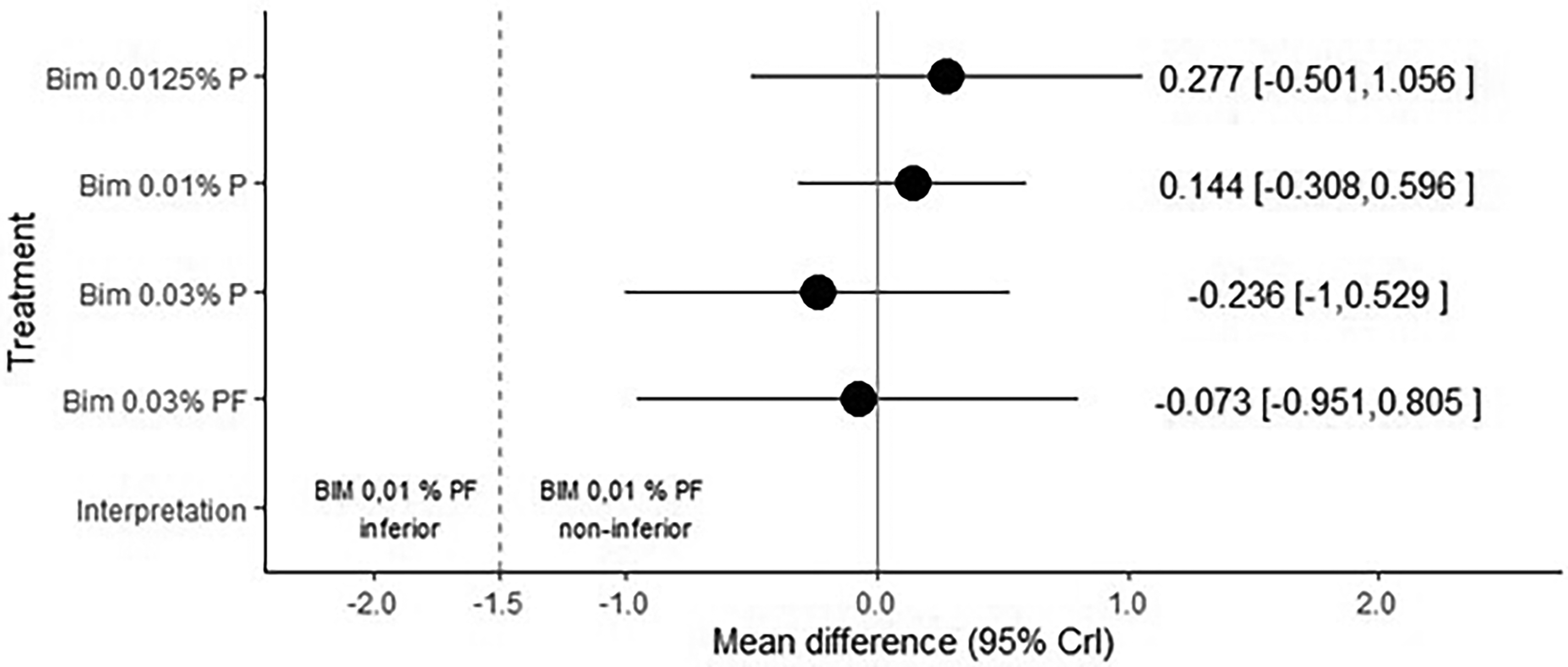
Treatment estimates – Bayesian fixed effects NMA at week 12 compared to preservative-free bimatoprost 0.01% (CrI: credible intervals; P:preserved; pf:preservative-free)
When reported as an adverse event, conjunctival hyperaemia occurred significantly less frequently in patients treated with bimatoprost 0.01% than in patients receiving bimatoprost 0.03% (28.6% versus 37.4%, p = 0.044). 25 Preservative-free formulations also showed a trend towards reduced conjunctival or ocular hyperaemia compared to preserved formulations (bimatoprost 0.01%: 6.4% versus 10.4% 16 ; bimatoprost 0.03%: 23.9% versus 26.1% 6 ).
Discussion
This NMA included four randomized controlled trials evaluating formulations of bimatoprost (preserved and preservative-free bimatoprost from 0.01% to 0.03%) in patients with glaucoma or OHT treated with a bimatoprost monotherapy for at least 3 months. This time frame was chosen to ensure adequate assessment of IOP efficacy and conjunctival hyperaemia, as 12 weeks is a standard duration in clinical trials for evaluating the sustained effects of glaucoma therapies.27,28
Efficacy was assessed in three out of the four studies, with one study per comparison. The NMA of mean IOP at Week 12 suggests that preservative-free bimatoprost 0.01% gel has similar IOP-lowering effects to other formulations, including preserved and preservative-free 0.03% bimatoprost, although these results are based on limited availability of evidence. The fact that this was confirmed for the mean IOP measured at three different timepoints during the day suggests that bimatoprost dosed once daily is enough to control IOP over 24 h. A non-inferiority margin of 1.5 mmHg was selected based on prior regulatory guidance and clinical trials, where differences below this threshold are not considered clinically meaningful for IOP efficacy in patients with glaucoma or OHT.29–31 Overall, the IOP results reinforce previous evidence that reducing the concentration of the active ingredient from 0.03% to 0.01% may maintain efficacy,25,32,33 aligning with the clinical need to balance IOP reduction and tolerability.
Our analysis supports that preservative-free formulations offer comparable efficacy with an improved safety profile. This included a lower incidence of conjunctival hyperaemia, a common adverse effect associated with prostaglandin analogues, which may be exacerbated by preservatives such as BAK. Conjunctival hyperaemia and other ocular surface symptoms are known contributors to treatment discontinuation and poor adherence.11,12 Preservatives such as BAK can damage the ocular surface by disrupting the tear film, causing inflammation and epithelial harm, especially with long-term use.34,35 Preservative-free formulations were developed to reduce these effects while maintaining IOP-lowering efficacy. Our analysis confirmed that across included studies, a higher proportion of patients receiving preservative-free bimatoprost had no conjunctival hyperaemia, and fewer experienced worsening of conjunctival hyperaemia compared to those treated with preserved formulations.6,16 This trend was shown at Week 12 and was consistent regardless of bimatoprost concentration and of the assessment methods, including photographic grading scales and through adverse event reporting. Additionally, low-concentration bimatoprost formulations (0.01%) were associated with less conjunctival hyperaemia compared to the 0.03% formulations. The preservative-free bimatoprost 0.01% gel is formulated with a near-neutral pH, which helps minimize ocular irritation commonly associated with more acidic or alkaline solutions.36,37 Additionally, its increased viscosity contributes to prolonged corneal contact time, allowing for sustained drug release while reducing the frequency and intensity of adverse sensations such as burning, stinging, and blurred vision.38,39 The reduced occurrence of conjunctival hyperaemia is clinically significant because it has the potential to enhance patient comfort and adherence to treatment, thereby improving long-term glaucoma management outcomes. These findings are corroborated by real-world data,7,32,33,40–42 which consistently reported less conjunctival hyperaemia with preservative-free and low-concentration bimatoprost treatments. In these real-world studies, other safety assessments were performed, (e.g., such as ocular surface disease index, tear break-up time or fluorescein staining 7 ), and preservative-free and lower-concentration bimatoprost consistently demonstrated a similar or more favourable safety profile, reinforcing the robustness of this observation across varied clinical settings.7,32,33,40–42
Several limitations temper the interpretation of these findings. The analysis included a limited number of studies, with only one study informing each comparison between formulations, precluding the assessment of heterogeneity or inconsistency within the network. Nevertheless, the included studies had adequate sample sizes for efficacy outcomes. Due to missing or inconsistently reported data, it was not possible to construct a connected network for change-from-baseline IOP or safety endpoints, limiting the scope of comparative safety conclusions. As a result, the impact of tolerability on discontinuation and adherence could not be assessed, and the absence of these data further constrained our ability to build an appropriate and informative safety network. Furthermore, safety outcomes primarily relied on subjective grading scales and were evaluated over a relatively short follow-up period of three months. This duration may be insufficient to fully capture the impact of long-term therapy on ocular surface health. The open-label design of some studies and unclear methodological aspects, such as the washout period in the Figus et al. trial 26 and missing outcome data in others, introduce potential biases that may affect the reliability of the results.
This NMA suggests that preservative-free bimatoprost 0.01% may be associated with comparable IOP-lowering effects and a trend toward lower conjunctival hyperaemia compared with higher-dose or preserved formulations. These findings could support its use as an effective and well-tolerated treatment option for patients with glaucoma and OHT in clinical practice, particularly for patients requiring long-term topical therapy. Enhanced tolerability could be relevant for patient comfort and may have implications for treatment adherence, although this was not directly assessed in the included studies. While preservative-free, lower-concentration bimatoprost formulations may be considered as an option for patients, particularly those with ocular surface concerns, clinical decisions should be individualized. Further adequately powered, long-term studies are needed to confirm these observations and to better characterize their impact on safety, adherence, and long-term clinical outcomes.
Footnotes
Acknowledgements
The authors thank Marina SCHVERER, PhD, who provided medical writing service.
Ethical approval and informed consent statements
Not applicable
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Laboratoires Théa.
Declaration of conflicts of interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Alina Popa Cherecheanu is a speaker for Laboratoires Théa. Ronald Marinus Petrus Cornelis de Crom is a consultant for Abbvie, paid instructor for Santen, consultant and paid instructor Laboratoires Théa, and paid instructor for Tramedico. Louise Baschet has no conflicts of interest. Cindy Mary-Lynn Hutnik is a speaker for Allergan, Aequus, Bausch & Lomb, Labtician, Laboratoires Théa, PharmaDrug, and Valeo, a consultant for Bausch & Lomb, Citrus Therapeutics, Heidelberg Engineering, Ivantis, Labtician, Laboratoires Théa, PharmaDrug, Valeo, and Viatris, and received in-kind support from Heidelberg Engineering. Leopold Schmetterer is consultant for Laboratoires Théa.
Data availability statement
Not applicable.
