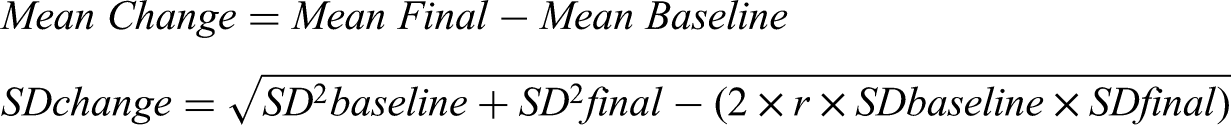Abstract
Purpose
This systematic review and meta-analysis aims to compare BT and DT in terms of intraocular pressure (IOP) reduction, safety, and patient preferences.
Methods
Following PRISMA and Cochrane guidelines, we searched PubMed, Scopus, Web of Science, CENTRAL, and Embase up to December 30, 2024. Twelve studies (11 randomized controlled trials [RCTs]) involving 1,885 patients were included. Primary outcomes were IOP reduction and adverse events. Statistical analyses were performed using Review Manager 5.4.1, with mean differences (MD) and risk ratios (RR) calculated for continuous and binary outcomes.
Results
BT demonstrated a statistically greater reduction in the morning IOP compared to DT at 12 weeks or more (BT: MD = 0.56 mmHg, P < 0.001; BTFC subgroup: MD = 0.66, P < 0.001), with consistent benefits observed at 8 and 4 weeks. No significant difference was observed in the evening IOP reduction. BT combination was associated with a 49% lower risk of eye irritation (RR = 0.51, P < 0.001) but a threefold higher risk of blurred vision (RR = 3.14, P < 0.001). Patient preference studies favored brinzolamide/timolol fixed combination (BTFC) due to reduced ocular discomfort.
Conclusion
BT showed a statistically greater reduction in morning IOP than DT; however, the absolute difference was modest, and its clinical relevance is uncertain. BT was associated with less ocular irritation but higher blurred vision risk. High heterogeneity for evening IOP and overall adverse events limits interpretation. Considering these findings, the choice of treatment usually depends on the patient's tolerance to higher initial ocular irritation in DT or blurring of vision in BT. Longer-term trials with 24-h IOP and preference are needed to assess these outcomes meaningfully.
Introduction
Glaucoma, a leading cause of irreversible blindness worldwide, poses a significant public health challenge, with an estimated prevalence projected to reach approximately 111.8 million individuals by 2040.1,2 The primary goal in glaucoma management is to lower intraocular pressure (IOP), as elevated IOP is a major risk factor for disease progression. Among the topical medications used, beta-blockers and carbonic anhydrase inhibitors (CAIs) are commonly prescribed due to their efficacy in reducing IOP. 3 The current guidelines suggest initiating the treatment protocol with monotherapy using an IOP-lowering drug, and escalating to a combination of two IOP-lowering drugs, if tolerability is acceptable and further IOP lowering is required. 4
Brinzolamide/timolol (BT) and dorzolamide/timolol (DT) combinations are commonly used in clinical glaucoma management. These combinations may be administered either as fixed-combination eye drops, brinzolamide/timolol fixed combination (BTFC) or dorzolamide/timolol (DTFC), or as non-fixed regimens involving separately administered drops. This review includes both types of formulations to reflect real-world prescribing patterns and the design of included studies. BTFC and DTFC are two widely used fixed-combination (FC) therapies in the clinical management of glaucoma. Dorzolamide, the first topical CAI, was introduced in 1994 and later combined with timolol, a beta-blocker, to form DTFC in 1998. This combination has since been extensively utilized and demonstrated significant efficacy in lowering IOP for glaucoma patients. 5 Brinzolamide, another CAI, became available in 2000 and was subsequently combined with timolol to create BTFC in 2005, which has also gained widespread use in clinical practice. 6 Both FCs exert their IOP-lowering effects through complementary mechanisms: brinzolamide and dorzolamide reduce aqueous humor secretion by inhibiting carbonic anhydrase, while timolol decreases aqueous humor production by blocking beta-adrenergic receptors. 7
Existing literature suggests that BTFC may be preferred by patients for better ocular comfort, potentially due to its near-physiologic pH of 7.2 compared to the more acidic pH of dorzolamide/timolol (5.6). 8 However, the clinical significance of these preferences and whether dorzolamide/timolol is equally effective remain uncertain. Some studies indicate a superior IOP-lowering effect with brinzolamide, but this has not been consistently demonstrated across all trials. 9 This systematic review and meta-analysis aims to address these gaps in knowledge by comparing the safety, efficacy, and patient preferences associated with BT (fixed and non-fixed combination) and DT (fixed and non-fixed combination) in glaucoma management. By synthesizing data from randomized controlled trials (RCTs) and relevant studies, this review seeks to provide evidence to guide clinical decision-making, particularly in regions with limited healthcare resources, where treatment adherence and patient comfort are critical. The findings could have significant implications for optimizing glaucoma management strategies.
Methods
The guidelines of the Cochrane Handbook and PRISMA were adhered to throughout our systematic review and meta-analysis.10,11 The review protocol was pre-specified and registered on PROSPERO (CRD42025635413).
Eligibility criteria
We included studies comparing BT and DT combinations in open-angle glaucoma and/or ocular hypertension patients, focusing on efficacy and safety. Both fixed and non-fixed combinations were eligible for inclusion, as they are thought to have similar efficacy. 4 Fixed combinations were written as BTFC and DTFC, while non-fixed combinations were written as “non-fixed BT” and “non-fixed DT” to differentiate them. All included DTFC interventions were preserved formulations; no preservative-free DTFC formulations were evaluated. Some studies involving other intraocular hypertension cases concentrating on patients’ preferences and switching from DTFC to BTFC were included for qualitative synthesis only. Only RCTs were included in the quantitative synthesis to ensure high-quality evidence. There were no restrictions on language, time period, or journal. We excluded case reports, reviews, and conference abstracts. Studies that were duplicate publications, lacked extractable data, or had no available full text were also excluded.
Information sources and search strategy
PubMed, Scopus, Web of Science, Cochrane Central Register of Controlled Trials (CENTRAL), and Embase were searched from inception until the 30th of December 2024 for possible studies using a combination of keywords for glaucoma and combinations included in our review. The PubMed search strategy was: “((Glaucoma OR POAG OR “intraocular hypertension” OR “ocular hypertension”) AND (Brinzolamide OR Azarga OR brinzolamide 1%) AND (Dorzolamide OR dorzolamide 2% OR Cosopt) AND (Timolol OR timolol 0.5% OR beta blocker OR Beta blocker))”. Furthermore, a manual search was done through Google Scholar, CENTRAL, and references of included studies to search for potential studies to include.
Screening and data extraction
Two authors (Alnabihi A and Albishry A) screened the results of the initial search. First, the titles and abstracts were screened using Rayyan, a web-based tool designed to facilitate and streamline the screening process in systematic reviews. 12 Second, full texts were retrieved and screened using a Google Sheet. Disagreements were resolved through discussion with a third reviewer (Alamoudi A). For data collection, a predefined extraction form was employed to gather essential details, including the study ID (first author's last name and publication year), study design, country, number of patients, interventions, average patient age and sex, baseline IOP, and a summary of the study's findings. Adverse event terms were extracted verbatim as reported and summarized in a clarification table.
Statistical analysis
Statistical analyses were carried out using Review Manager (RevMan) version 5.4.1 on Windows 11. Our primary outcomes were the difference between the two combinations in IOP reduction and adverse events. For continuous outcomes, the mean difference (MD) in change scores was calculated along with 95% confidence intervals (CI), while binary outcomes were expressed as risk ratios (RR) with their corresponding 95% CI. The chi-square test for heterogeneity and the I2 test were used to assess heterogeneity where a P value <0.1 and I2 > 50% were considered significant heterogeneity and a random-effects model based on the DerSimonian and Laird method was used. Otherwise, a fixed effect model (inverse-variance) was used. Studies using both fixed and non-fixed combinations were included in the main meta-analysis, and a subgroup analyses were conducted to examine any effect of formulation type. A p-value of less than 0.05 was considered statistically significant. As recommended by Cochrane guidelines, publication bias was not assessed due to the limited number of included studies (< 10) in our primary outcome analysis. No sensitivity analyses were conducted due to the limited number of studies per outcome and consistent directionality of effect sizes. Mean changes were derived by subtracting pre-treatment from post-treatment values in studies that provided these data. Standard deviations for these changes were estimated using an appropriate formula and a correlation coefficient of 0.5, as recommended by Cochrane guidelines.
Risk of bias and quality assessment
The quality of the included studies was assessed using the Risk of Bias 2 (RoB-2) tool by Cochrane. 13 This tool evaluates study quality based on five domains: the randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. Studies are then categorized as having low risk, some concerns, or high risk of bias.
The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) framework was used for appraising the certainty of evidence of the outcomes that were meta-analyzed. 14
Results
Studies selection and characteristics of included studies
Out of the initially screened 794 reports, 25 were eligible for full-text screening, of which only 12 studies were included in our review (Figure 1). The included studies comprised 11 RCTs and one prospective single-arm trial, pooling data from 1,885 patients.8,9,15–23 The studies were conducted across multiple countries, including India, Japan, Germany, United Kingdom, Italy, Egypt, South Korea, Spain, United States of America, Argentina, Brazil, and Chile. The mean age of participants ranged between 42.3 to 66.7 years and males constituted on average between 29.3% and 69% of the samples. Three studies compared non-fixed combinations (BT vs DT), one study assessed the effects of switching from DTFC to BTFC while the rest compared BTFC and DTFC.
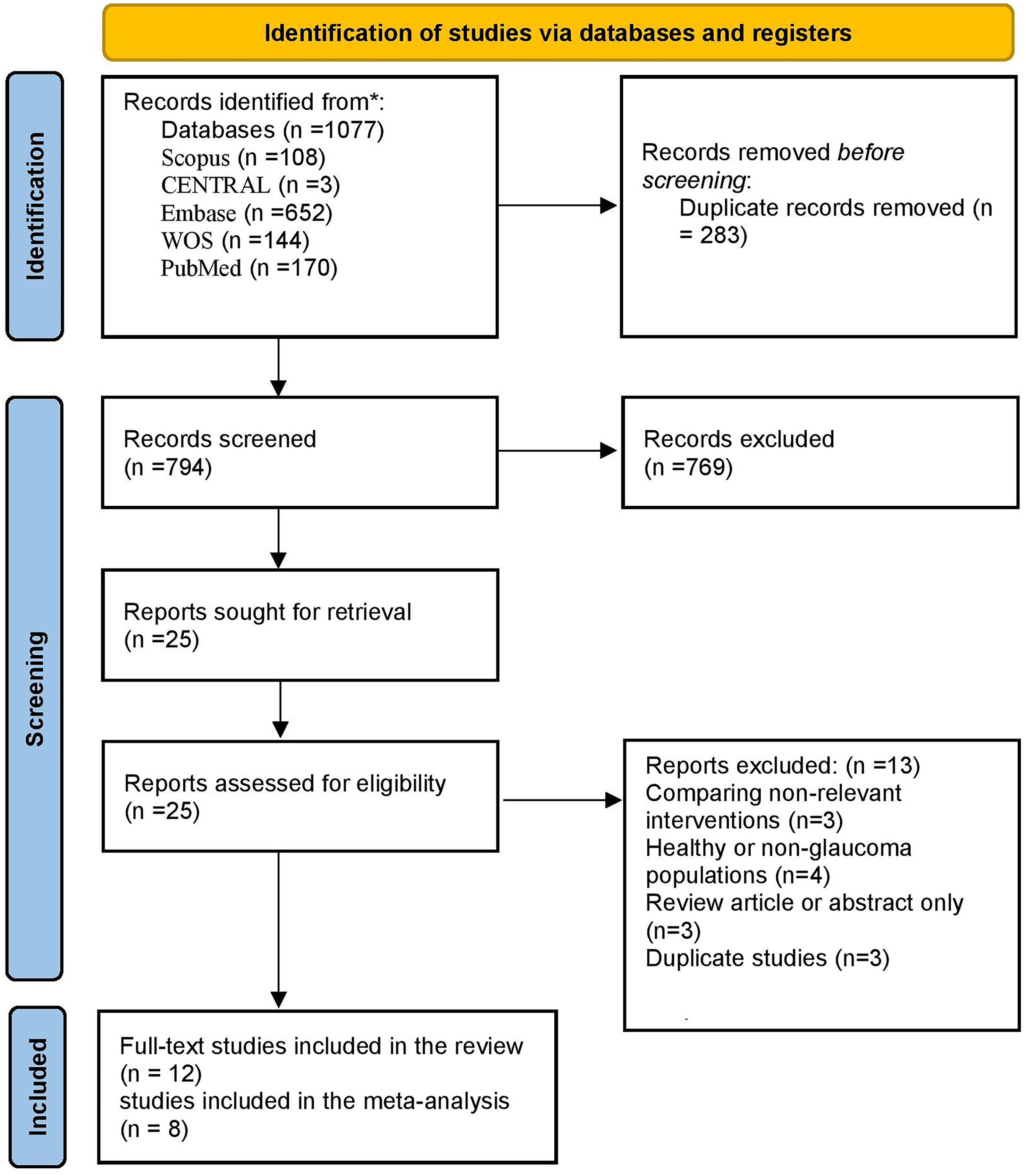
PRISMA flow diagram of the selection process.
All included DTFC interventions used preserved formulations; no studies evaluated preservative-free DTFC. The fixed-combination concentrations were consistent across studies: brinzolamide 1% + timolol 0.5% BID for BTFC, and dorzolamide 2% + timolol 0.5% BID for DTFC—except in Japan, where the approved DTFC formulation contains dorzolamide 1% + timolol 0.5% BID (Table 1). The reductions in mean IOP at different follow-up intervals demonstrated that both BT and DT maintained consistent efficacy. The included studies reported similar amounts of mean IOP reduction during the time intervals evaluated (Table 2). Because the terms eye discomfort, eye irritation, and eye pain are clinically similar but variably defined across studies, we summarized their exact usage and definitions, where reported, in Table 3 to clarify this aspect of the safety analysis.
Characteristics of included studies.
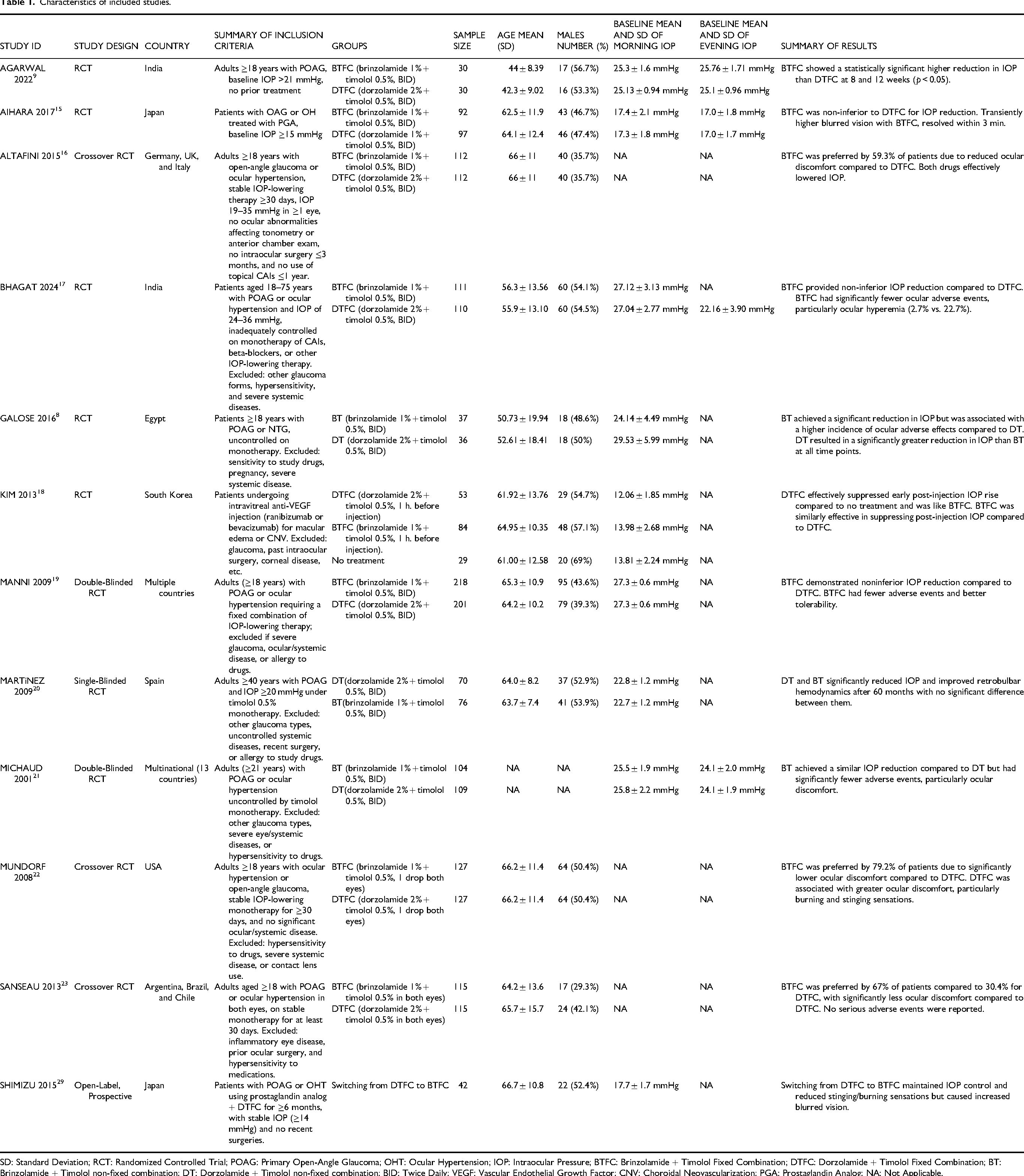
SD: Standard Deviation; RCT: Randomized Controlled Trial; POAG: Primary Open-Angle Glaucoma; OHT: Ocular Hypertension; IOP: Intraocular Pressure; BTFC: Brinzolamide + Timolol Fixed Combination; DTFC: Dorzolamide + Timolol Fixed Combination; BT: Brinzolamide + Timolol non-fixed combination; DT: Dorzolamide + Timolol non-fixed combination; BID: Twice Daily; VEGF: Vascular Endothelial Growth Factor; CNV: Choroidal Neovascularization; PGA: Prostaglandin Analog; NA: Not Applicable.
Summary of intraocular pressure (IOP) outcomes at multiple time points in patients treated with brinzolamide/timolol (BT) versus dorzolamide/timolol (DT).
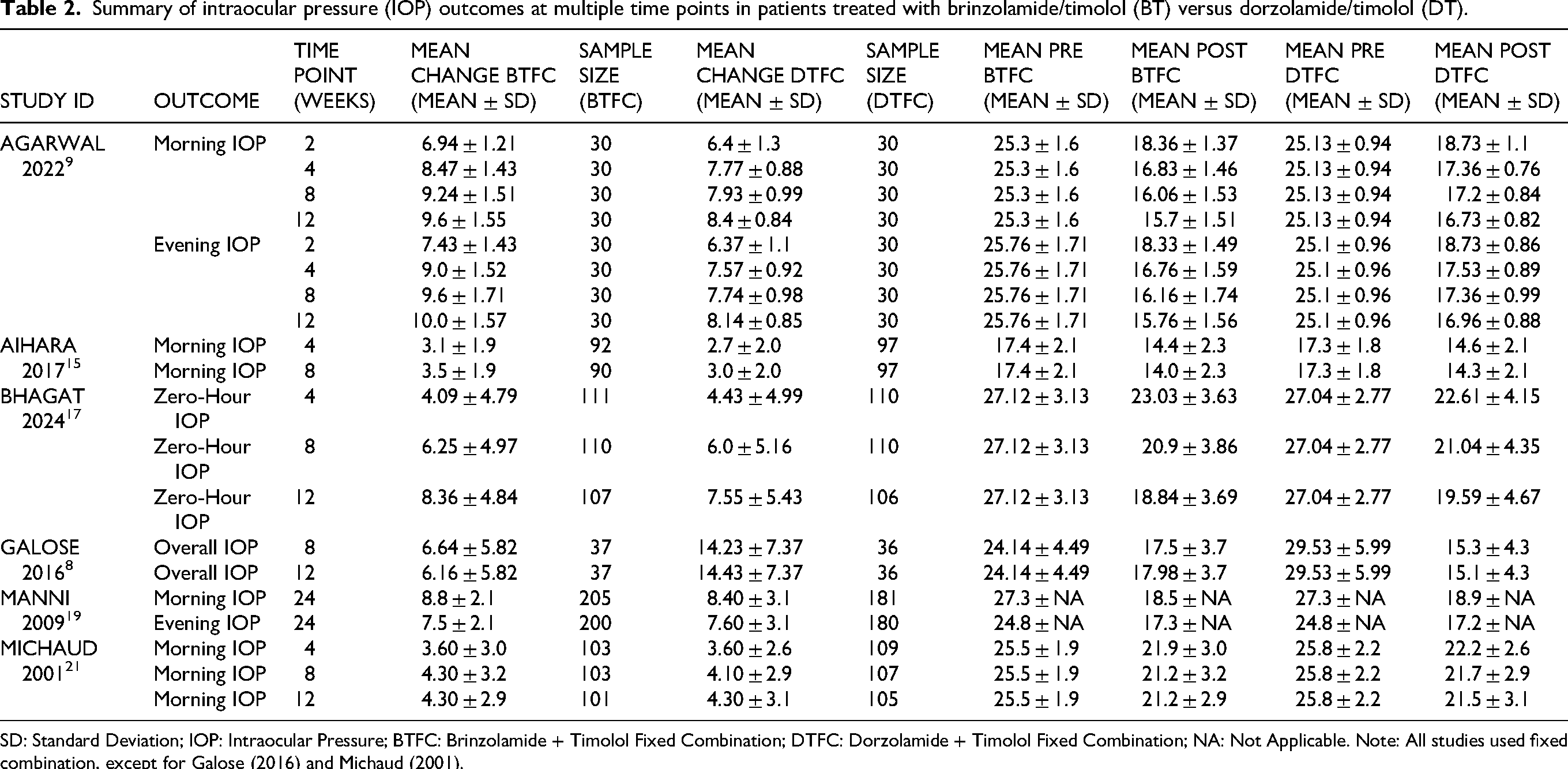
SD: Standard Deviation; IOP: Intraocular Pressure; BTFC: Brinzolamide + Timolol Fixed Combination; DTFC: Dorzolamide + Timolol Fixed Combination; NA: Not Applicable. Note: All studies used fixed combination, except for Galose (2016) and Michaud (2001).
Reported definitions for eye irritation, discomfort, and pain across the included studies.
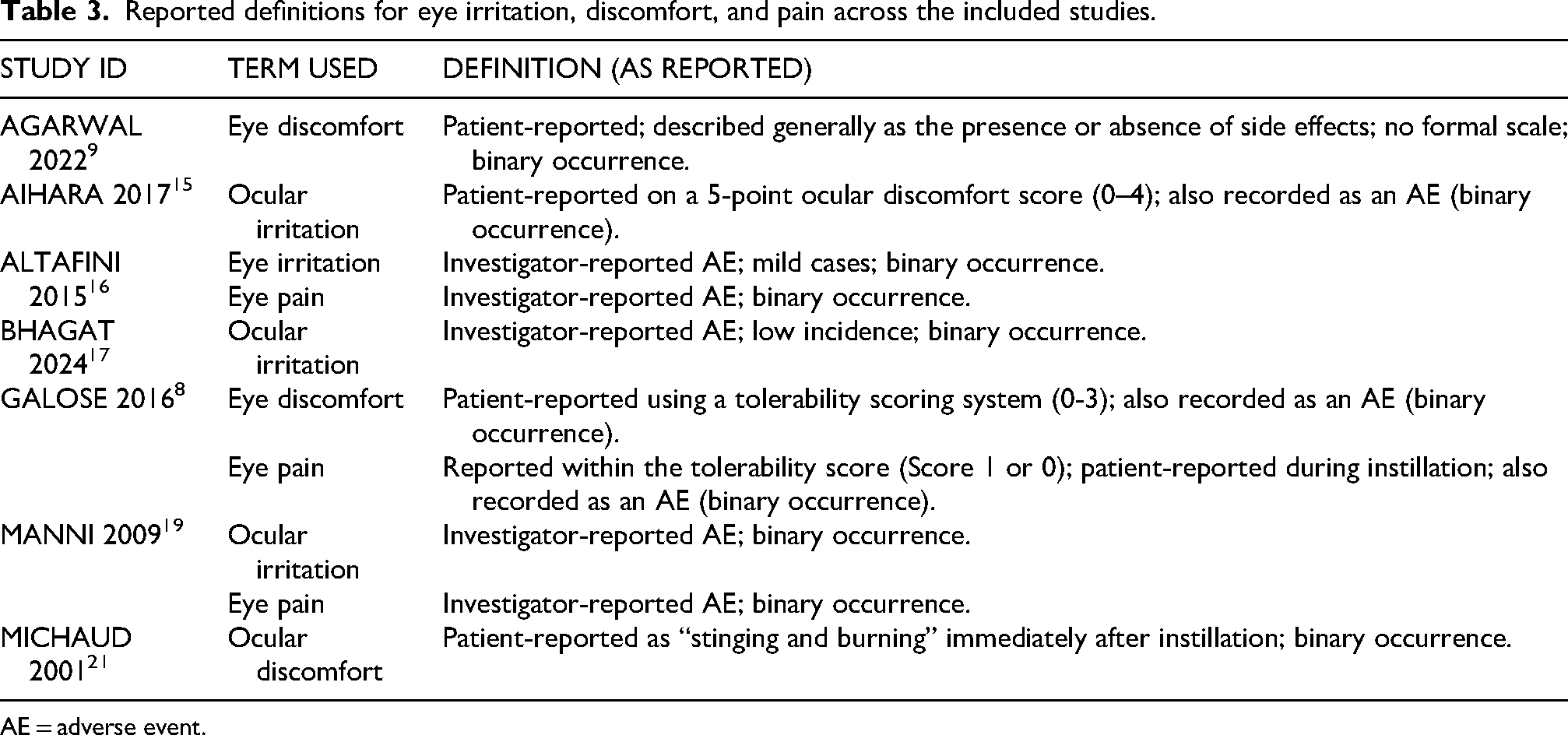
AE = adverse event.
The risk of bias assessment of the 11 RCTs included in our review revealed that 7 studies had an overall low risk of bias, adhering to high methodological standards; 2 studies had some concerns, and 2 were rated as having a high risk of bias, warranting cautious interpretation of their results (Figure 2).
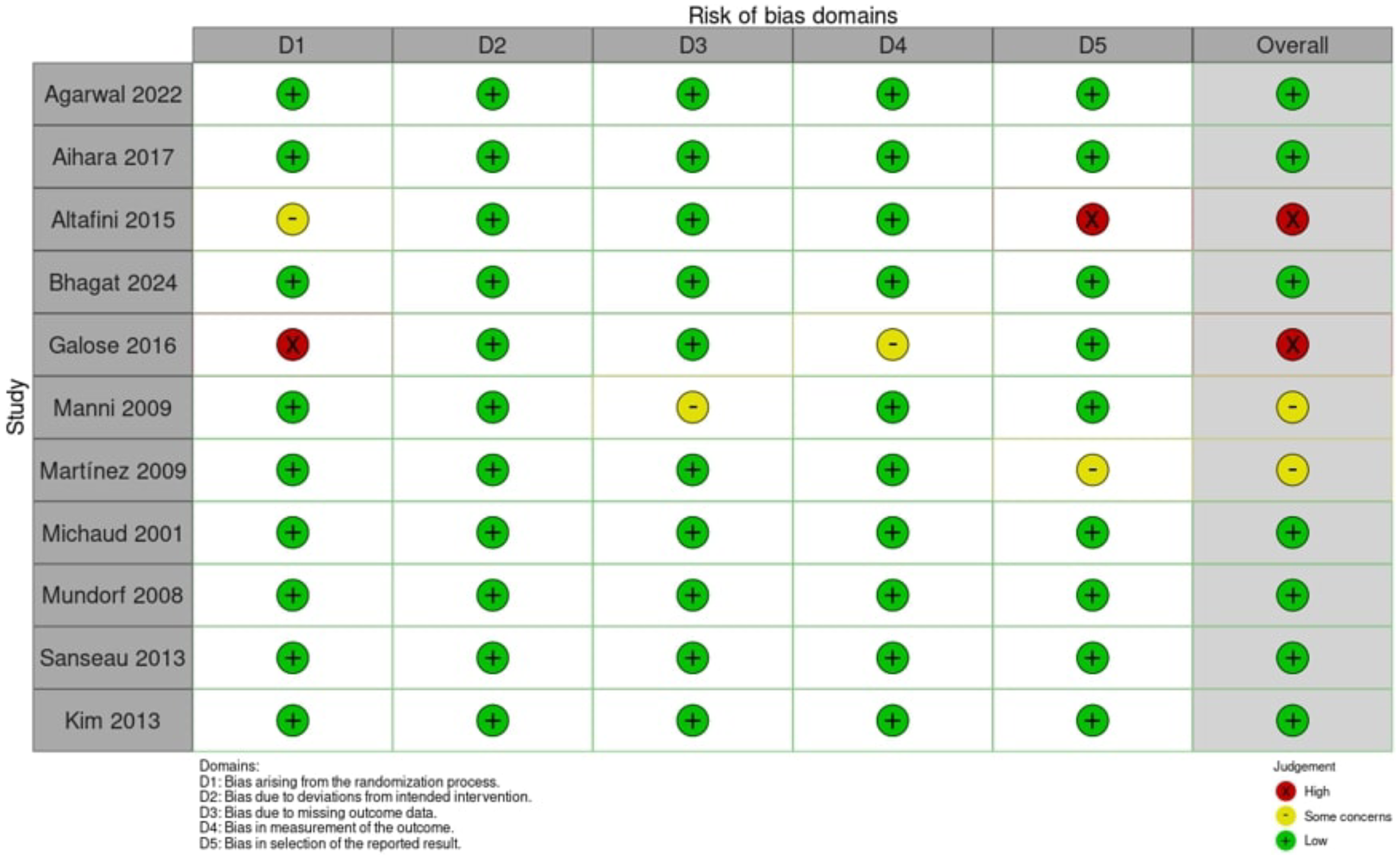
RoB-2 results of randomized controlled studies.
Meta-analysis of IOP reduction
For morning IOP reduction at 12 weeks or more, five studies (n = 535 for BT, n = 523 for DT) showed that overall BT (fixed and non-fixed combination) provided a significantly greater reduction in IOP compared to DT (fixed and non-fixed combination) (MD = 0.56 mmHg; 95% CI [0.27, 0.86]; P < 0.001, I2 = 39%). In the fixed-combination subgroup, BTFC demonstrated significantly better IOP reduction than DTFC (MD = 0.66 mmHg; 95% CI [0.34, 0.98]; P < 0.001; I2 = 26%), whereas the non-fixed combination subgroup showed no significant difference (MD = 0.00 mmHg; 95% CI [−0.76, 0.76]; P = 1.00). However, the test of subgroup differences was not significant (P = 0.11) (Figure 3A).
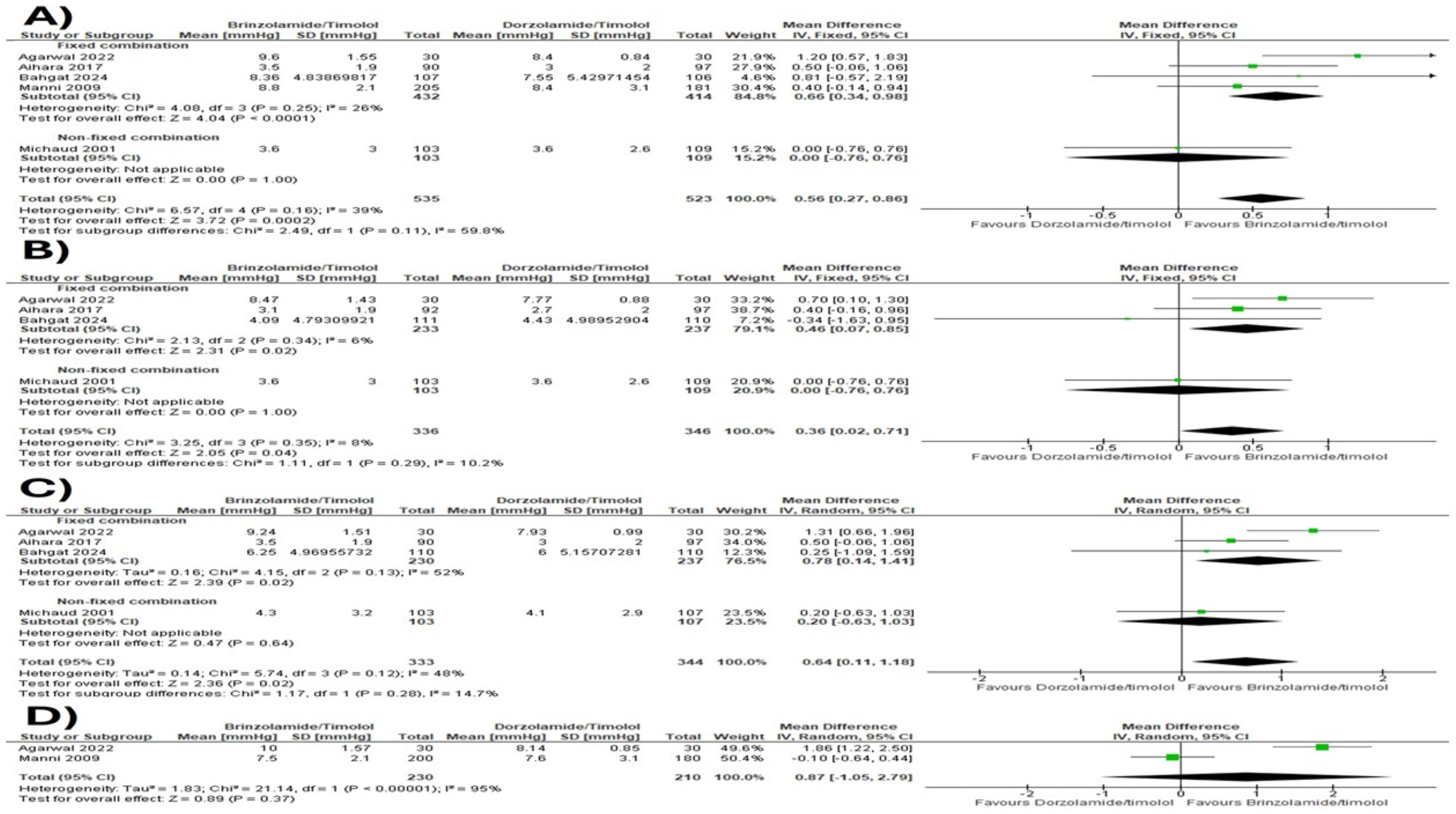
Meta-analysis of A) morning IOP reduction at 12 weeks or more B) morning IOP reduction at 8 weeks C) morning IOP reduction at 4 weeks D) evening IOP reduction at 12 weeks or more.
These results were consistent at 8 and 4 weeks, where overall BT (fixed and non-fixed combination) use achieved a significantly greater reduction in IOP compared to DT (fixed and non-fixed combination) (MD = 0.36 mmHg; 95% CI [0.02, 0.71]; P = 0.04, I2 = 10.2%), (MD = 0.64 mmHg; 95% CI [0.11, 1.18]; P = 0.02, I2 = 48%), respectively. Similarly, BTFC also achieved higher reduction rates than DTFC at 8 and 4 weeks (MD = 0.46 mmHg; 95% CI [0.07, 0.85]; P = 0.02; I2 = 6%), (MD = 0.78 mmHg; 95% CI [0.14, 1.41]; P = 0.02; I2 = 52%), respectively (Figures 3B and 3C). In contrast, the analysis of evening IOP reduction at 12 weeks or more was not significant (MD = 0.87 mmHg; 95% CI [−1.05, 2.79]; P = 0.37, I2 = 95%) (Figure 3D). Evening IOP at 4 and 8 weeks was not included in the meta-analysis, as it was reported by only one study 9 ; the findings are summarized in Table 2.
Meta-analysis of adverse events
Our meta-analysis included six studies (n = 671 for BT, n = 684 for DT) evaluating the risk of any adverse events and found no significant difference in the risk of adverse events between BT and DT (RR = 0.75; 95% CI [0.49, 1.15]; P = 0.19; I2 = 80%) or between BTFC and DTFC (RR = 0.78; 95% CI [0.43, 1.41]; P = 0.41; I2 = 76%). The test of subgroup differences was not significant (P = 0.85), indicating no evidence of difference in the risk of adverse events between fixed and non-fixed formulations (Figure 4).
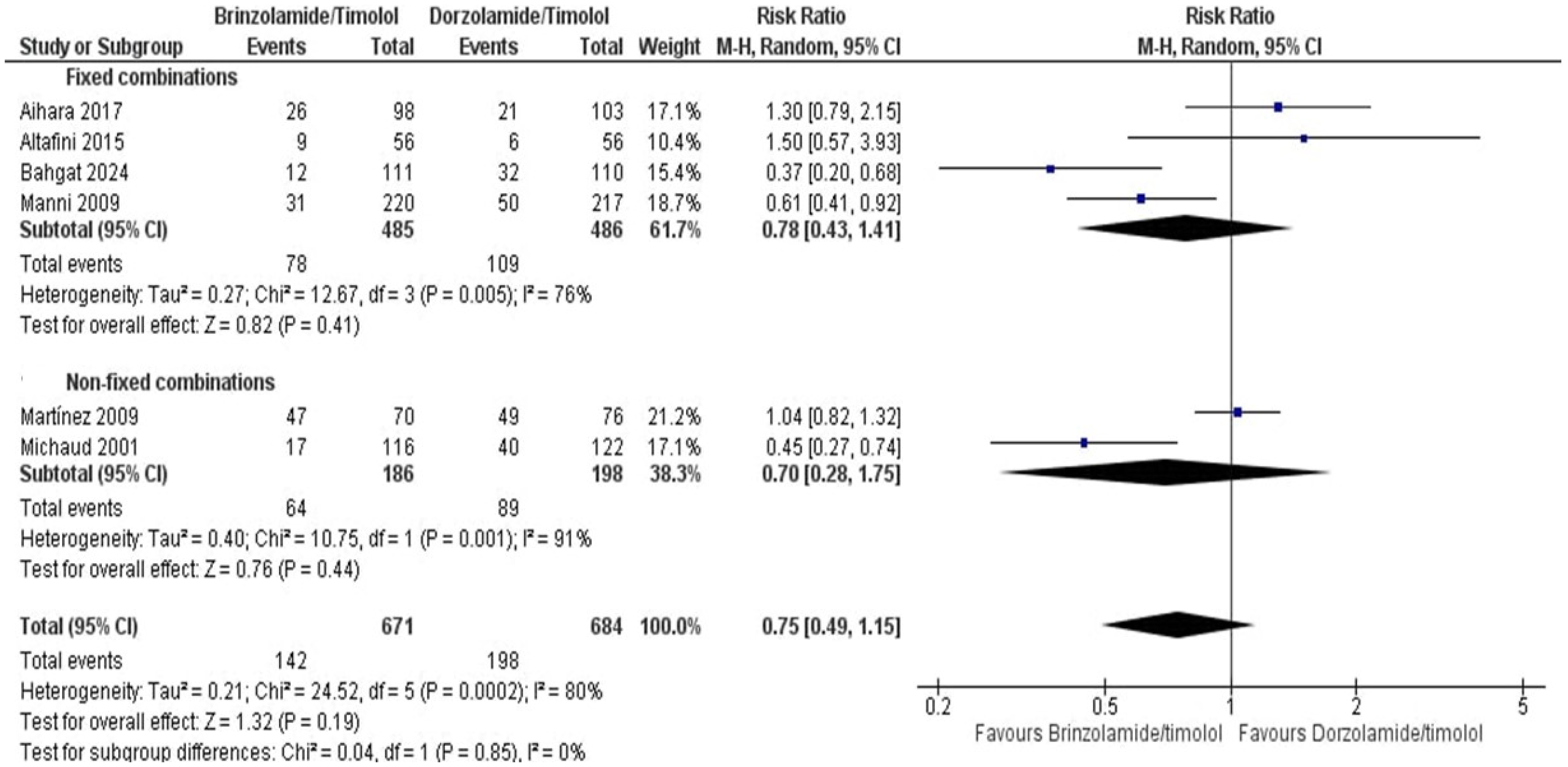
Meta-analysis of any adverse events.
However, an analysis of five studies reporting eye irritation adverse events (n = 561 for BT, n = 556 for DT) demonstrated that BT was associated with a significantly lower risk of eye irritation compared to DT (RR = 0.51; 95% CI [0.35, 0.75]; P = 0.0005; I2 = 47%; Figure 5A). In contrast, analyses of three studies reporting eye discomfort (n = 183 for BT, n = 188 for DT) showed no statistically significant difference (RR = 0.50; 95% CI [0.11, 2.28]; P = 0.37; I2 = 81%; Figure 5B), and three studies reporting eye pain (n= 313 for BT, n = 309 for DT) similarly found no significant difference (RR = 0.96; 95% CI [0.10, 8.79]; P = 0.97; I2 = 76%; Figure 5C).
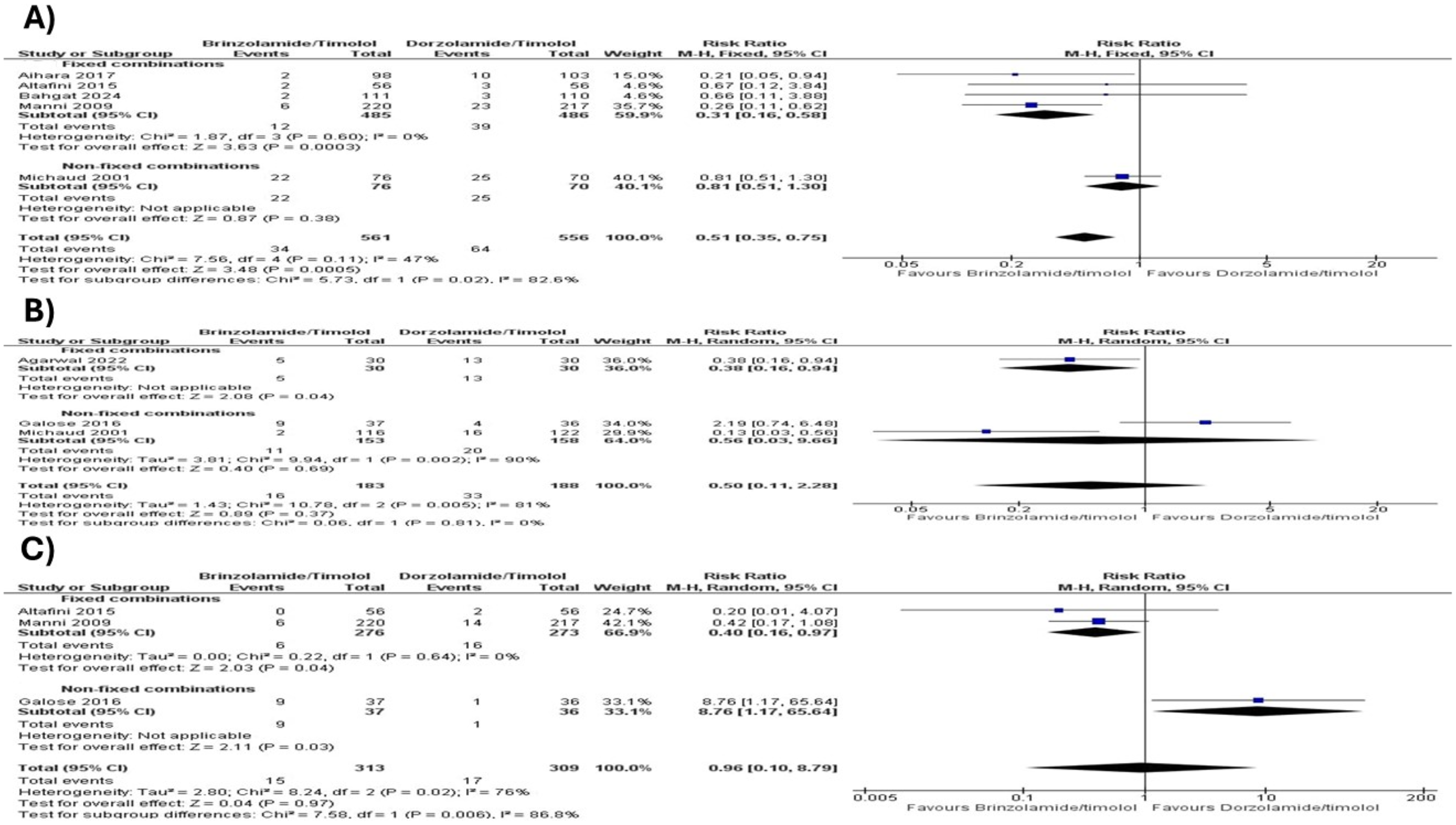
Meta-analysis results of A) eye irritation B) eye discomfort C) eye pain.
Interestingly, a meta-analysis including six studies (n = 714 for BT, n = 714 for DT) evaluating the risk of blurred vision revealed that BT (fixed and non-fixed combination) was associated with a significantly higher risk of blurred vision compared to DT (fixed and non-fixed combination) (RR = 3.14; 95% CI [1.93, 5.09]; P < 0.001; I2 = 0%). This finding was consistent across both fixed combinations (RR = 2.68; 95% CI [1.43, 5.01]; P = 0.002; I2 = 0%) and non-fixed combinations (RR = 3.87; 95% CI [1.79, 8.34]; P < 0.001; I2 = 27%) (Figure 6).
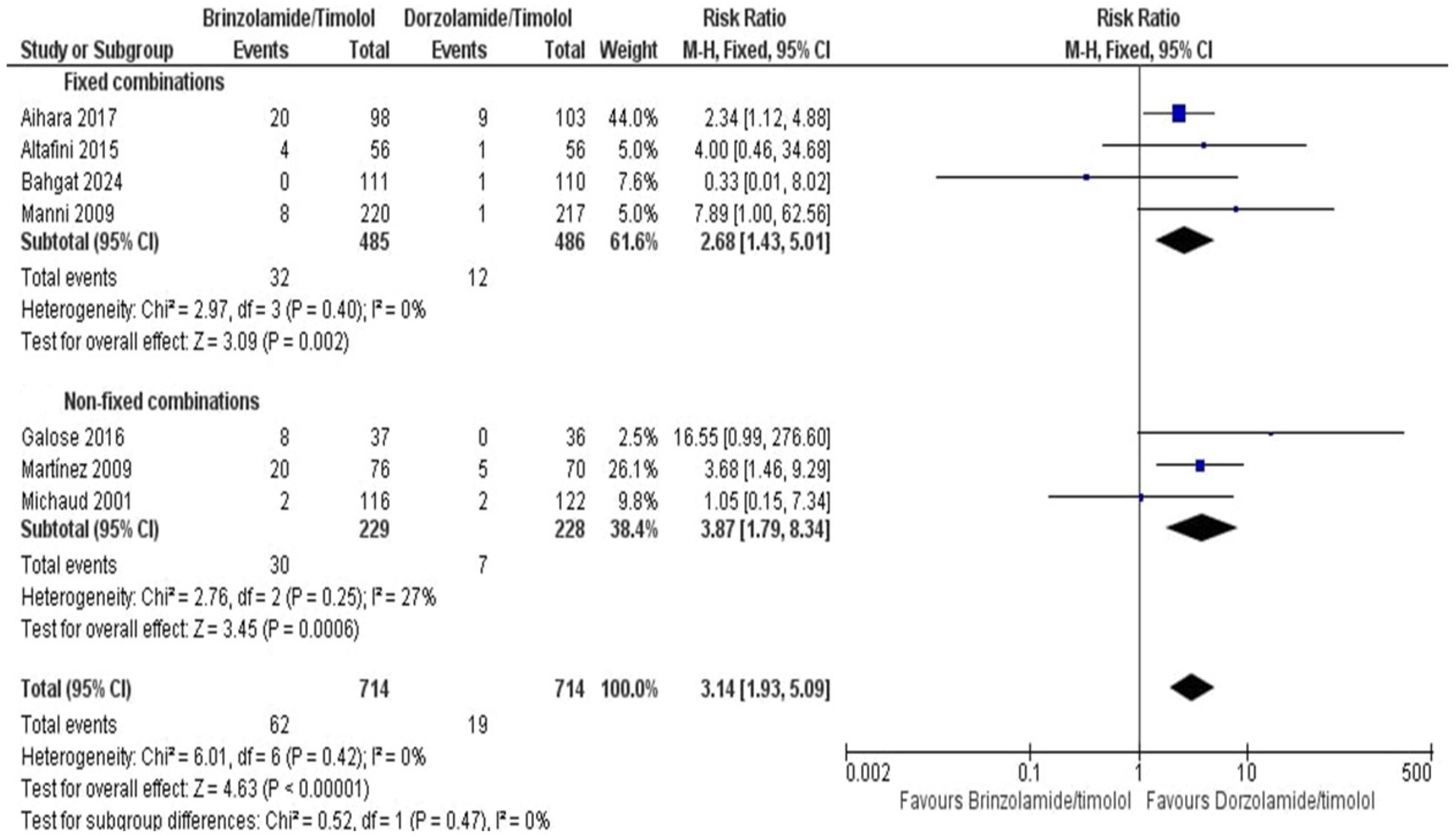
Meta-analysis of blurred vision.
Patients’ preferences
Two crossover RCTs compared patients’ preferences for BTFC and DTFC in the short term. Mundorf et al. conducted a study on 127 glaucoma patients over two days, comparing both BTFC and DTFC. 22 Among the 106 patients who completed the study and provided preference data, 79.2% preferred BTFC, while 20.8% preferred DTFC (P < 0.001). Ocular discomfort was significantly lower with BTFC compared to DTFC (1.4 ± 1.6 vs. 2.9 ± 2.5, respectively; P < 0.001). Ocular irritation and ocular pain were reported more frequently with DTFC (17.3% and 6.9%, respectively) than with BTFC (5.5% and 0.8%). However, blurred vision was more commonly reported with BTFC than with DTFC (14.8% vs. 0.8%). Similarly, Sanseau et al. found that among 112 patients who tried both BTFC and DTFC for two days, a significantly higher percentage preferred BTFC over DTFC (67.0% vs. 30.4%; P = 0.001), with DTFC being associated with higher eye discomfort scores (P < 0.001). 23 A longer crossover RCT (seven days) reported a higher but non-significant preference for BTFC over DTFC (59.3% vs. 40.7%), along with a higher incidence of blurred vision with BTFC (3.6%) and eye irritation with DTFC (5.4%). 16
Grading of recommendations, assessment, development, and evaluation (GRADE) framework
This Summary of Findings (SoF) table presents the key outcomes assessed in the meta-analysis comparing brinzolamide/timolol (BT) versus dorzolamide/timolol in the treatment of glaucoma (Table 4). The GRADE framework was used to rate the certainty of the evidence for each outcome.
Summary of findings table (GRADE approach).
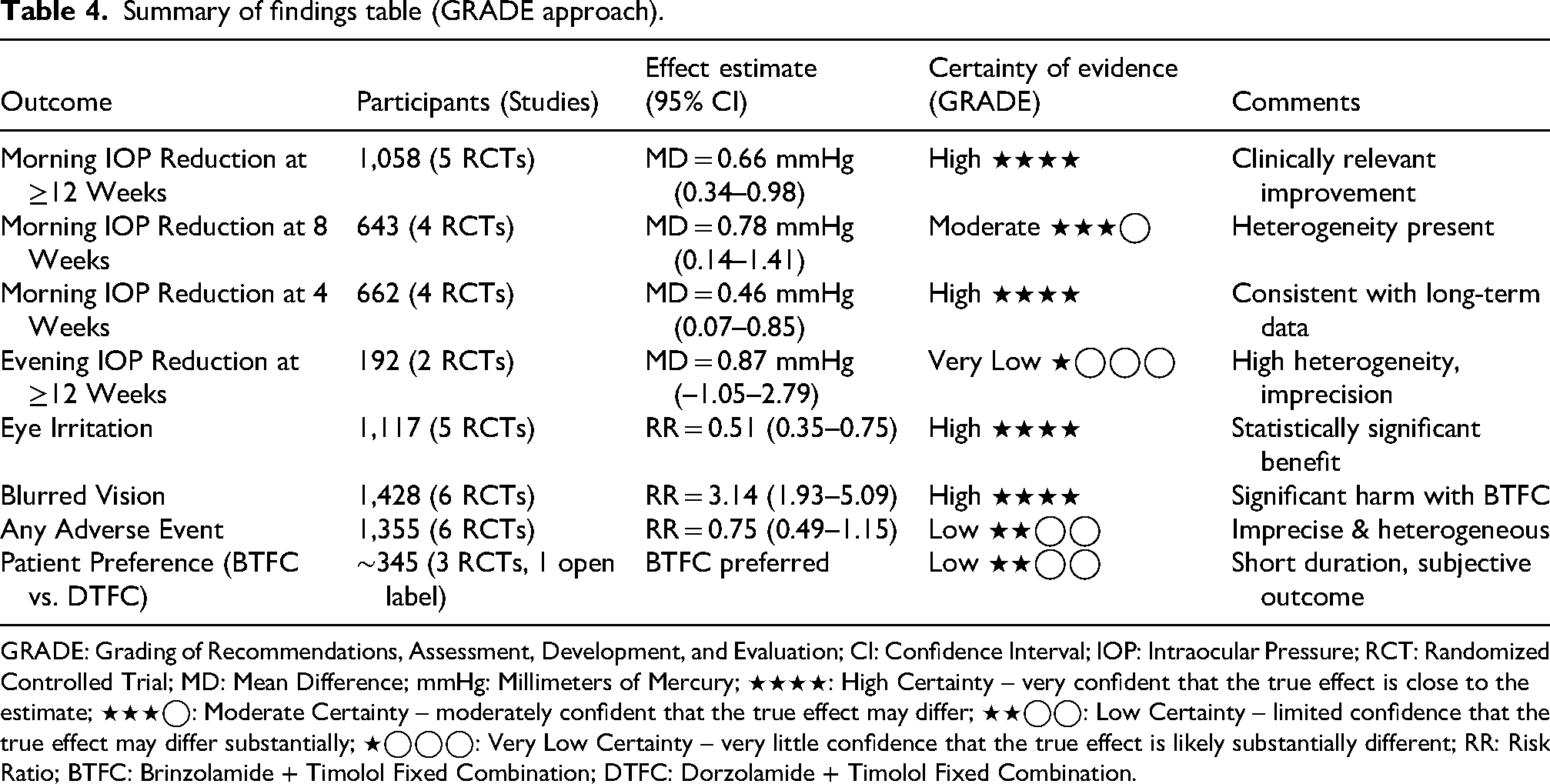
GRADE: Grading of Recommendations, Assessment, Development, and Evaluation; CI: Confidence Interval; IOP: Intraocular Pressure; RCT: Randomized Controlled Trial; MD: Mean Difference; mmHg: Millimeters of Mercury; ⭑⭑⭑⭑: High Certainty – very confident that the true effect is close to the estimate; ⭑⭑⭑◯: Moderate Certainty – moderately confident that the true effect may differ; ⭑⭑◯◯: Low Certainty – limited confidence that the true effect may differ substantially; ⭑◯◯◯: Very Low Certainty – very little confidence that the true effect is likely substantially different; RR: Risk Ratio; BTFC: Brinzolamide + Timolol Fixed Combination; DTFC: Dorzolamide + Timolol Fixed Combination.
Discussion
Although CAI–beta-blocker combinations are not typically first-line therapy for primary open-angle glaucoma or angle-closure disease, they are widely used as step-up or adjunctive treatment when additional IOP lowering is required beyond prostaglandin analogs, or when clinicians aim to reduce the number of drops used with combination therapy. 4 In routine practice, dorzolamide/timolol and brinzolamide/timolol are among the most common options within this class, and the choice between them is often driven by tolerability trade-offs (stinging/irritation vs transient blur), patient preference, and local availability. To the best of our knowledge, this is the first systematic review and meta-analysis to investigate the comparative safety and efficacy of BT (fixed and non-fixed combination) in comparison with DT (fixed and non-fixed combination). It includes 12 studies (11 RCTs) comprising evidence from 1,885 patients. BTFC provided a statistically significant greater reduction in morning IOP compared to DTFC formulations at 12 weeks or more (MD = 0.66 mmHg). This benefit was consistent at 8 and 4 weeks, with BTFC showing superior IOP reduction over DTFC (MD = 0.78 mmHg at 8 weeks; MD = 0.46 mmHg at 4 weeks). The significant difference was not affected by the inclusion of a non-fixed combinations study in the analysis (Michaud et al.), though the pooled effect was attenuated (MD = 0.56 mmHg at 12 weeks, 0.36 mmHg at 8 weeks, 0.20 mmHg at 4 weeks). 21 However, no significant difference was observed in the evening IOP reduction at 12 weeks or more. The observed advantage of BT/BTFC in morning IOP reduction was statistically significant, but the absolute magnitude was modest. However, even a one mmHg reduction in IOP can lead to a 10% reduction in the risk of glaucoma progression. 24 Therefore, the clinical relevance of the slightly lower morning IOP with BTFC remains uncertain and warrants confirmation in longer-term trials.
Morning IOP measurements were more consistently reported across studies, while evening measurements were less frequently assessed. Although evening IOP changes were not statistically significant, morning reductions consistently favored BT. The evening IOP outcome showed substantial heterogeneity, likely reflecting the contribution of only 2 studies and non-uniform timing of “evening” measurements across trials. Given the small number of studies contributing to this outcome and inconsistent reporting windows, additional stratified analyses were not considered reliable, and the evening IOP estimate should be interpreted cautiously. Most studies did not report the average of morning and evening IOP values, limiting the interpretation of diurnal IOP control. The need for 24-h IOP monitoring remains a known challenge in glaucoma research. 25 In our included studies, morning measurements were typically taken around 9 a.m., and evening measurements around 4 p.m., in accordance with standard office hours. This should be taken into consideration, as IOP is known to exhibit diurnal variation, with potential spikes occurring in the early morning or during sleep. 26 While traditional office-hour measurements are commonly used, they often fail to capture the full circadian variation of IOP, particularly during nocturnal fluctuations. Some technologies may help achieve 24-h IOP recording, such as the contact lens sensor (CLS) 27 or home tonometry. 28 CAIs, especially in combination therapies, have shown IOP-lowering efficacy during the day but to a lesser extent at night. 29 Furthermore, Kim et al. found both BTFC and DTFC to be comparable in reducing the early post-VEGF injection rise in IOP. 18
In terms of safety, no significant differences were found in the overall risk of adverse events or in specific outcomes like eye discomfort and pain. However, BT was associated with a 49% lower risk of eye irritation compared to DT but a significantly higher risk of blurred vision, increasing by up to 214%. We could not perform a subgroup analysis for fixed and non-fixed combinations due to the low number of studies and the observed heterogeneity of safety outcomes. Studies with less than a week of drug administration were excluded from the analysis. These findings are further supported by our review of short trials assessing patient preferences, where BTFC was consistently preferred over DTFC in the short term, despite its documented higher risk of blurred vision in both short- and long-term use. Similar findings on preference were reported in longer trials. Agarwal et al. reported a preference for BTFC over DTFC after 12 weeks of use, although no specific numerical data were provided. 9
These results are further supported by the findings of Shimizu et al., who switched 44 patients who had been using DTFC for more than six months to BTFC for six months 30 and found no significant differences in IOP changes. Although stinging/burning side effects were less common (P = 0.042) and blurred vision was more common (P = 0.003), approximately 31% of patients preferred DTFC, while 29% preferred BTFC, and 40% preferred neither. These findings are particularly important for reducing drug discontinuation rates. A previous study reported a 16% dissatisfaction rate and a 12% discontinuation rate for DTFC. 31 These findings indicate that while patients may prefer BTFC in the short term, these differences may diminish in the long term. This should be considered given the high cost of these drugs to healthcare systems. In a study analyzing the cost of glaucoma medications, DTFC was the most expensive drug among 10 glaucoma treatments. 32 However, evidence comparing the costs of BTFC and DTFC remains limited. BTFC is a potential candidate for being more cost-effective, offering similar or better comfort and a lower discontinuation rate while maintaining comparable efficacy. 33
The meta-analysis included both fixed and non-fixed combinations that reflect the many regions where patients receive separate bottles of medications instead of a fixed combination. The previous literature shows that fixed and non-fixed combinations have similar efficacy but adherence, convenience and tolerability may differ. 34 Fixed combinations reduce the number of instillations which may improve adherence, minimize dosing errors, and increase patient satisfaction. In contrast, non-fixed combinations provide more flexibility in dose adjustments and tolerability management. The observed heterogeneity in our safety analyses may have been influenced by this variability. This may reflect differences in adverse-event definitions, reporting practices, follow-up duration, baseline ocular surface status, and formulation characteristics. In addition, brinzolamide formulations are suspensions and may cause transient post-instillation blur, whereas dorzolamide formulations are typically solutions, which may contribute to differences in subjective tolerability.30,35 Clinicians should interpret these findings in the context of the specific formulation (fixed vs. non-fixed) that is available to their patients.
Although our study comprised evidence from a large number of studies, the number of included studies in each analysis was limited. Publication bias could not be reliably evaluated because fewer than ten studies contributed to each primary outcome analysis. Significant heterogeneity was observed in some analyses, which may affect the applicability of the findings. Moreover, the included studies did not assess the safety of preservative-free DTFC formulations. The availability of preservative-free DTFC in some markets allows for better ocular surface tolerance than preserved DTFC.35,36 Our safety findings should be interpreted with this limitation in mind. Future research should compare preserved and preservative-free DTFC directly. Furthermore, the inclusion of both fixed and non-fixed combinations, though clinically relevant, introduced variability in the analysis, which we addressed by using random-effects models and subgrouping when needed. A key limitation is that most included RCTs were short-term (typically weeks to a few months) and did not assess glaucoma progression outcomes. Preference crossover studies were short (days) and likely captured early comfort and immediate post-instillation effects rather than long-term tolerability, adherence, or ocular surface outcomes. DTFC is generally associated with initial irritation that tapers later, while BTFC's blurring of vision risk persists for longer periods, which these short studies do not capture. This distinction is important when applying preference findings to chronic therapy. The reliance on office-hour IOP measurements may not fully capture circadian variations in IOP, particularly nocturnal spikes, which are critical for glaucoma management. Future studies should prioritize 24-h IOP monitoring using advanced technologies like CLS or home tonometry to better assess circadian fluctuations. Long-term studies comparing BTFC and DTFC are needed to evaluate sustained efficacy, safety, and patient adherence. Cost-effectiveness analyses should be conducted to determine the economic impact of these treatments, especially in resource-limited settings. Finally, patient-centered outcomes, including quality of life and long-term preferences, should be incorporated into future trials to guide clinical decision-making.
Conclusion
This meta-analysis suggests higher efficacy of BT in reducing morning IOP compared to DT, along with better tolerability regarding ocular irritation. However, this higher efficacy was modest and potentially not clinically significant. Additionally, BT was associated with a higher risk of blurred vision, and there were no significant differences in evening IOP reduction. Future research should focus on 24-h IOP monitoring, long-term outcomes, and cost-effectiveness to optimize glaucoma management strategies. Clinicians should consider patient preferences, side effect profiles, and overall IOP control when choosing between BT and DT.
Footnotes
Acknowledgment
“Supported in part by an Unrestricted Grant from Research to Prevent Blindness, New York, NY, to the Department of Ophthalmology & Visual Sciences, University of Utah.”
ORCID iDs
Author contributions
Anas Alamoudi: Study conception, literature screening, data extraction and analysis, and drafting the manuscript. Ahmed Alnabihi: literature screening, data extraction, risk of bias assessment, and drafting the manuscript. Sultan Al-Qahtani: drafting and reviewing the manuscript. Asim M Albishry: literature screening, data extraction, and risk of bias assessment. Waleed K. Alsarhani: critical manuscript review. Hady Saheb: in-depth critical manuscript review and feedback. Iqbal Ike K. Ahmed: Project supervision, and final manuscript review.
Statement of human and animal rights,or ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study informed consent is not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
Anas Alamoudi: None. Ahmed Alnabihi: None. Sultan Al-Qahtani: None. Asim M Albishry: None. Waleed K. Alsarhani: None. Hady Saheb: Consultant – Alcon, AbbVie, Glaukos, Valeo, Bausch, Thea; Honoraria – Zeiss, Bausch, Alcon, Abbvie; President – Canadian Glaucoma Society. Iqbal Ike K Ahmed: Acucela Inc (Seattle, Washington, USA): consultation; Aerie Pharmaceuticals Inc (Irvine, California, USA): consultation, research grant/support; Alcon (Geneva, Switzerland): consultation, honoraria, research grant/support; Allergan (Dublin, Ireland): consultation, honoraria, research grant/support; ArcScan Inc (Golden, Colorado, USA): consultation; Bausch and Lomb (Rochester, New York, USA): consultation; Beaver-Visitec International Inc (Waltham, Massachusetts, USA): consultation; Camras Vision Inc (Durham, North Carolina, USA): consultation, research grant/support; Carl Zeiss Meditec AG (Jena, Germany): consultation, honoraria; Centervue (Padova, Italy): consultation; Ellex Medical Lasers (Adelaide, Australia): consultation; ElutiMed (New Orleans, Louisiana, USA): consultation; Equinox (Newport Beach, California, USA): consultation; ForSight Labs (Menlo Park, California, USA): consultation; Genentech Inc (San Francisco, California, USA): consultation; Glaukos Corp (San Clemente, California, USA): consultation, research grant/support; Gore (Newark, Delaware, USA): consultation; IanTECH (Reno, Nevada, USA): consultation; InjectSense Inc (San Francisco, California, USA): consultation; Iridex Corp (Mountain View, California, USA): consultation; iSTAR Medical (Wavre, Belgium): consultation; Ivantis Inc (Irvine, California, USA): consultation, research grant/support; Johnson and Johnson Vision (Jacksonville, Florida, USA): consultation, honoraria, research grant/support; KeLoTec Inc (Orange County, California, USA): consultation; LayerBio Inc (Boston, Massachusetts, USA): consultation; Leica Microsystems (Wetzlar, Germany): consultation; MicroOptx (Maple Grove, Minnesota, USA): consultation; New World Medical Inc (Rancho Cucamonga, California, USA): consultation, research grant/support; Omega Ophthalmics (Lexington, Kentucky, USA): consultation; PolyActiva (Melbourne, Australia): consultation; Sanoculis Ltd (Kiryat Ono, Israel): consultation; ScienceBased Health (Oak Ridge North, Texas, USA): consultation; Sight Sciences Inc (Menlo Park, California, USA): consultation; Stroma Medical (Laguna Beach, Cal- ifornia, USA): consultation; TrueVision (Goleta, California, USA): consultation; Vizzario (Venice, California, USA): consultation.
Data access
The data used in this review were extracted from published sources. Extracted datasets used during the current study are available from the corresponding author upon reasonable request.
Précis
Comparative analysis suggests that morning intraocular pressure control and patient comfort may favor Brinzolamide/timolol over dorzolamide/timolol combination, emphasizing the importance of formulation differences in tailoring glaucoma therapy.