Abstract
Background
Recent advances have revealed the important role of the immune system in the progression of age-related macular degeneration (AMD). Monocyte-to-lymphocyte ratio (MLR) is a combined marker reflecting inflammation status. However, research on the correlation between MLR and AMD is limited.
Methods
In this cross-sectional analysis, weighted multivariable logistic regression, multinomial logistic regression, subgroup analysis, smoothed curve fitting and threshold effect analysis were used to investigate the relationship between MLR and AMD based on data from the 2005–2008 National Health and Nutrition Examination Survey (NHANES).
Results
This study included data of 4,655 participants. The proportions of no AMD, early AMD and late AMD were 92.27%, 6.79% and 0.95%, respectively. After adjusting for covariates, weighted multivariable logistic regression analysis revealed that MLR was positively associated with AMD (OR = 3.22, 95%CI 1.32–7.82). Threshold effect analysis revealed that higher MLR associated with prevalence of AMD after MLR was greater than 0.37 (OR = 8.93, 95%CI 2.53–31.60). The diagnostic performance of MLR > 0.37 for AMD was assessed using ROC curve analysis. In addition, MLR was proven to have a significant positive correlation with early AMD (OR = 1.20, 95%CI 1.19–1.21) and late AMD (OR = 9.21, 95%CI 9.10–9.31) through weighted multinomial logistic regression.
Conclusions
Our study demonstrates that an elevated MLR is independently associated with the prevalence of AMD.
Keywords
Introduction
Age-related macular degeneration (AMD) is an acquired progressive degenerative disease of the retina in the elderly, responsible for 8.7% of all blindness cases worldwide. 1 Age, smoking, and genetics are proven risk factors for AMD. 2 Early AMD is featured by drusen and pigmentary abnormalities, while geographic atrophy (dry AMD) or choroidal neovascularization (wet AMD) threaten vision in the late stage of AMD. 3 late AMD imposes a substantial economic burden on patients and caregivers. 4 Currently, multiple studies have demonstrated that immune components including natural killer cells, T lymphocytes, macrophages and complement factor H play a role in the progression of AMD.5–8
Monocyte-to-lymphocyte ratio (MLR) is defined as the ratio of the number of monocytes and the number of lymphocytes and has emerged as a useful prognostic marker in various diseases including lymphoma, prostate cancer and cardiovascular diseases.9–11 Monocytes are a type of leukocyte, serving functions such as phagocytosis and antigen presentation within the immune system. 12 lymphocytes are another type of leukocyte including B lymphocytes, T lymphocytes and natural killer cells, which determine the response to foreign substances like infectious microorganisms. 13 Both monocyte and lymphocyte numbers in the blood vary depending on age, sex and immune status.14,15 Monocytes are mainly associated with innate immunity, while lymphocytes mainly mediate adaptive immunity. Therefore, increased MLR often reflects transitions in immune status, from acute inflammation to chronic disease or immunosuppression. 16
There are rare studies that investigated the association between MLR and AMD. Therefore, we performed an observational cross-sectional study to analyze the relationship between MLR and AMD using data from the 2005–2008 National Health and Nutrition Examination Survey (NHANES). We hypothesized that higher MLR level would be associated with the prevalence and severity of AMD.
Materials and methods
Data source and participants
NHANES is a research program conducted by the National Center for Health Statistics (NCHS), which focuses on health and nutrition of the American population. 17 The data from NHANES (2005–2006 and 2007–2008) on the NHANES website (www. cdc. gov/ nchs/ nhanes/) were chosen to analyze the association of MLR and AMD in this study. Initially, a total of 20,497 participants were enrolled. We excluded 13,416 participants younger than 40 years, 651 participants without blood cell count data, 1,018 participants without gradable retinal imaging, 624 participants with C-reactive protein (CRP) > 1 mg/dL and 133 participants used systemic antibiotics within the past 30 days. 4,655 participants were identified in this survey (shown in Figure 1). To limit potential confounding from acute infections or systemic inflammatory responses, participants exhibiting C-reactive protein (CRP) levels above 1 mg/dL or who had recently undergone systemic antibiotic therapy were excluded from the analysis. The National Centre for Health Statistics (NCHS) Ethics Review Board (ERB) ensured that NHANES research involving human participants protected the rights and welfare of study participants. The appropriate participants’ informed consent was performed in compliance with the Declaration of Helsinki protocols.
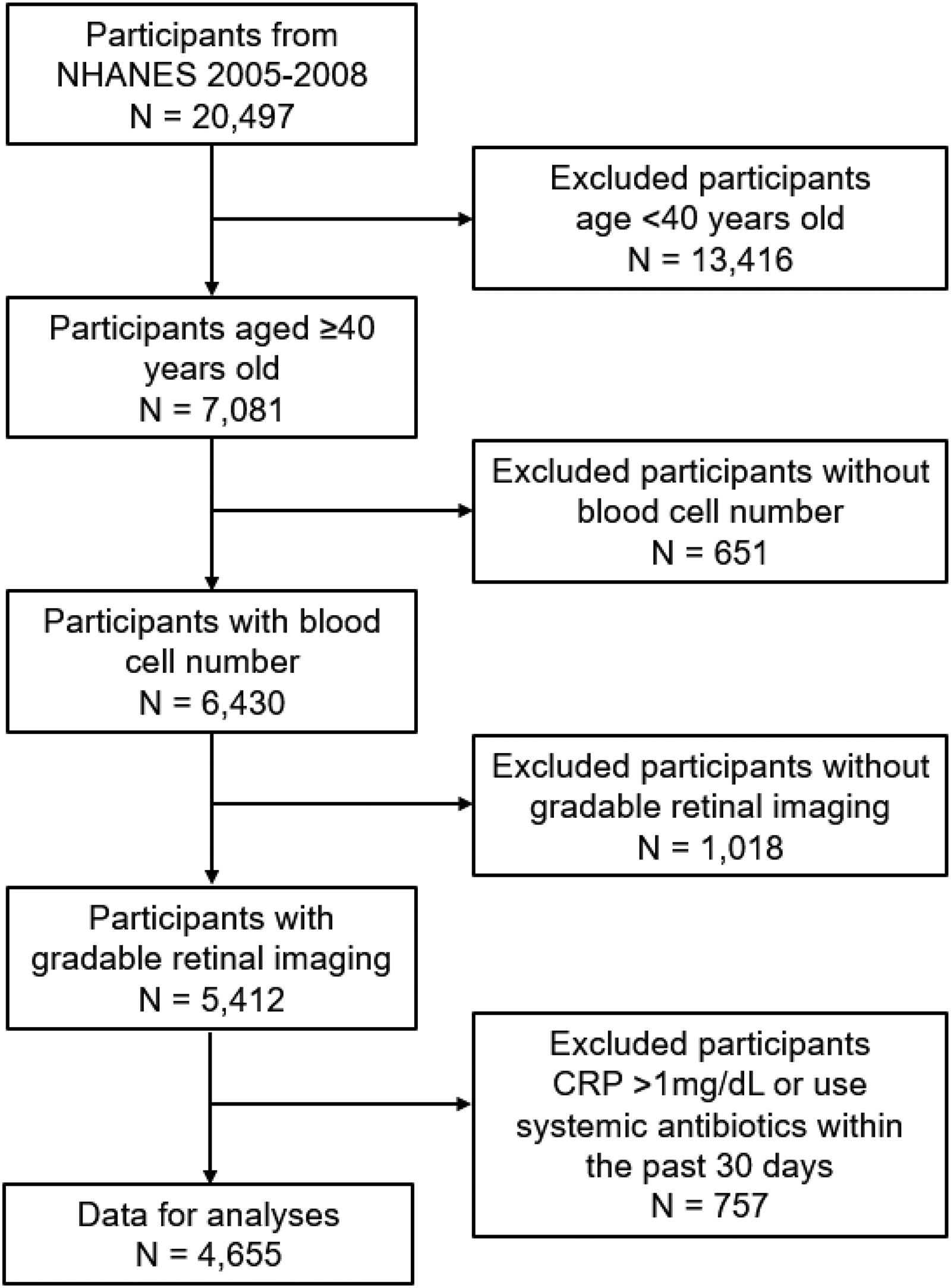
Schematic representation of the selection procedure for the participants included in this study.
Exposure and outcome
The evaluation of retinal photographs was downloaded from the NHANES website. AMD was classified into early AMD and late AMD. Digital fundus images (45-degree field) were taken with a Canon CR6–45NM non-mydriatic camera (Canon Inc., Tokyo, Japan) from subjects aged ≥40 years in a darkened room to allow physiological pupil dilation. Image assessments were performed by at least two trained graders at the University of Wisconsin. AMD was classified according to Age-Related Eye Disease Study (AREDS)-based definition: early AMD was defined by drusen or pigmentary abnormalities and late AMD was identified based on specific criteria including exudative AMD signs or geographic atrophy. In cases where retinal imaging of both eyes was gradable, the eye exhibiting greater AMD severity was selected for analysis. AMD status and its severity were included as outcome variables.
The monocyte-to-lymphocyte ratio (MLR) was defined as the absolute monocyte count divided by the absolute lymphocyte count. 18 Results of complete blood count tests were obtained from publicly released laboratory data on the NHANES website. MLR was treated as an exposure variable in our study.
Covariates
In this study, demographic data, laboratory data and questionnaire data on the NHANES website (https://wwwn.cdc.gov/nchs/nhanes/Default.aspx) were used. In line with prior studies, variables previously shown to be associated with AMD were included as covariates. The following covariates were included: age, gender, race, education level, hypertension, diabetes, body mass index (BMI), CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption.19–25 Among the covariates, age, gender, race and educational level were included in the demographic information. At the Mobile Examination Center (MEC), trained technicians performed standardized assessments including blood pressure and BMI, collected biological specimens including blood and urine and administered questionnaires on psychological and physical health. During the household interview, participants were asked to show all prescription medications taken during the past 30 days. Medication use (statins, antihypertensive, hypoglycemic, glucocorticoid, systemic antibiotics and immunosuppressant drugs) was defined from the NHANES prescription medication file (RXQ_RX). Patients with diabetes were determined based on taking hypoglycemic medications or having a diagnosis of diabetes from the NHANES Diabetes data file (DIQ), having a hemoglobin A1c level ≥ 6.5 from the NHANES Glycohemoglobin data file (GHB). Hypertension was defined as taking antihypertensive medications, a diagnosis of hypertension from the NHANES Blood Pressure and Cholesterol data file (BPQ) or having three consecutive systolic blood pressure measurements ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg from the NHANES Blood Pressure data file (BPX). Alcohol consumption was defined as the intake of twelve or more alcoholic drinks per year from the NHANES Alcohol Use data file (ALQ). Smoking status was determined based on a history of smoking one hundred or more cigarettes during the participant´s lifetime from the NHANES Smoking-Cigarette Use data file (SMQ). CRP data was obtained from the NHANES C-Reactive Protein data file (CRP). BMI was defined as dividing weight (kg) by height squared (m2) from the NHANES Body Measures Examination data file (BMX).
Statistical analysis
Following the NHANES analytic guidelines, sampling weights, strata, and primary sampling units were incorporated in the analyses using WTMEC4YR, SDMVSTRA, and SDMVPSU, respectively. 26 For analyzing weighted cross-sectional characteristics of participants, differences among groups (no AMD, early AMD and late AMD) were evaluated by using survey-weighted linear regression for continuous variables or survey-weighted Chi-square tests for categorical variables. To examine the association between MLR and AMD, weighted multivariable logistic regression analyses were performed, treating MLR as a continuous variable and AMD as a binary variable. Model 1 was adjusted for no covariates. Model 2 was adjusted for race, age and sex. Model 3 was adjusted for age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption. We further conducted multinomial logistic regression to examine the correlation between MLR and severity of AMD (no AMD, early AMD and late AMD), adjusting for age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption. The linearity between MLR and AMD was assessed by smooth curve fitting and the inflection point was further analyzed by threshold effect. ROC curve analyses were performed to assess the diagnostic performance of MLR for AMD. Both unadjusted and multivariable-adjusted models (accounting for age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption) were evaluated. AUC values with 95% confidence intervals were calculated.
In addition, subgroup analyses of the associations between MLR and AMD were performed using stratified multivariable logistic regression models with age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption. Stratified variables were considered as potential effect modifiers. To assess heterogeneity, an interaction term was incorporated into the model, and the likelihood ratio test was employed for evaluation. Because the proportion of missing data was minimal (ranging from 0% to 2.06%) as shown in Table 1, no imputation was performed. Given the negligible level of missingness, complete-case analysis was considered unlikely to introduce bias. 27 All statistical analyses and graphical visualization were performed using R software (version 5.2) and EmpowerStats (version 4.4.3). A two-sided P-value < 0.05 was considered statistically significant.
Weighted cross-sectional characteristics of participants stratified by AMD status from NHANES 2005–2008.
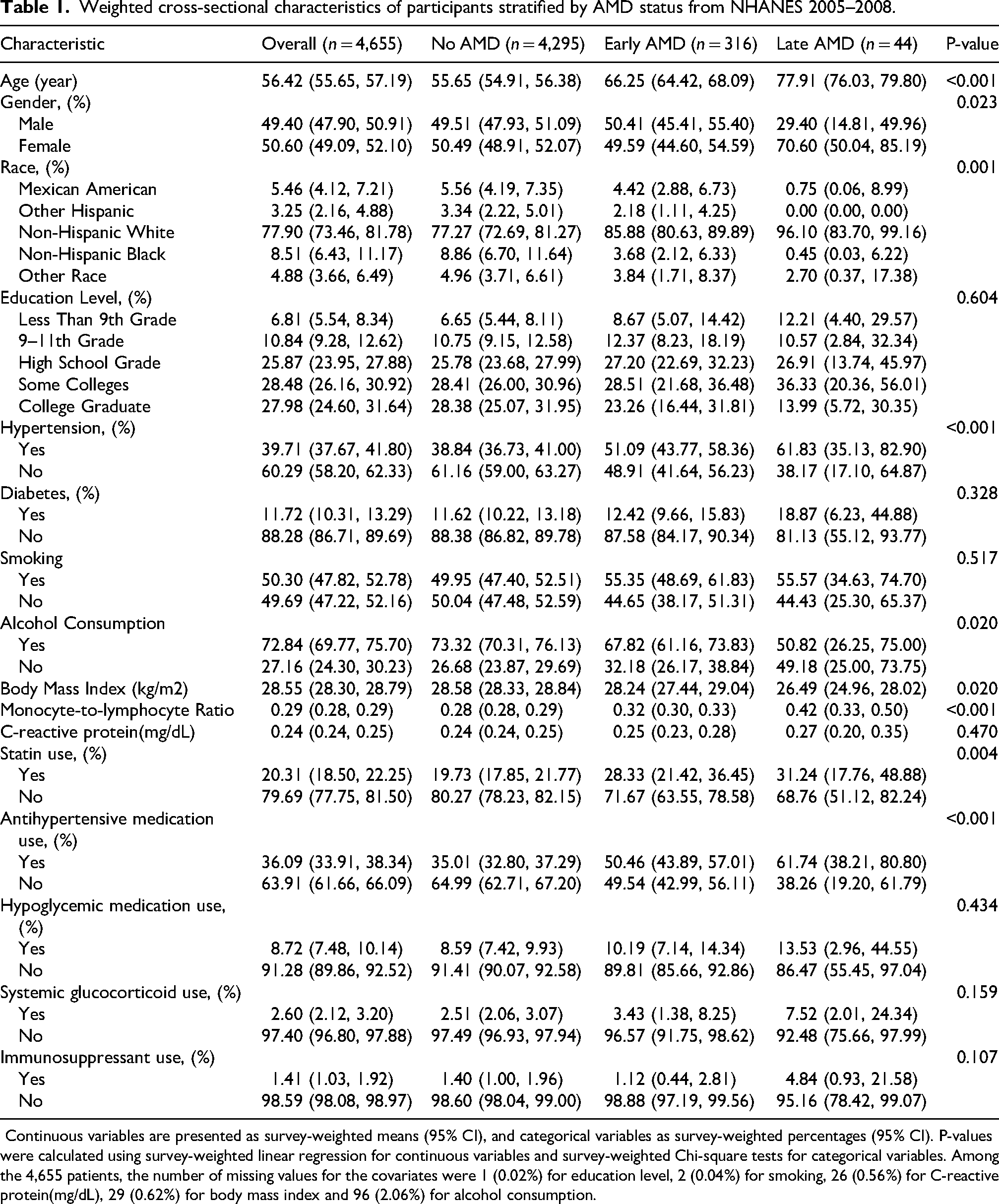
Continuous variables are presented as survey-weighted means (95% CI), and categorical variables as survey-weighted percentages (95% CI). P-values were calculated using survey-weighted linear regression for continuous variables and survey-weighted Chi-square tests for categorical variables. Among the 4,655 patients, the number of missing values for the covariates were 1 (0.02%) for education level, 2 (0.04%) for smoking, 26 (0.56%) for C-reactive protein(mg/dL), 29 (0.62%) for body mass index and 96 (2.06%) for alcohol consumption.
Results
Participants’ characteristics
In this study, 4,655 participants are included, among whom 316 participants (6.79%) had early AMD and 44 participants (0.95%) had late AMD. Table 1 shows the clinical features of the study population based on the severity of AMD. We found statistically significant differences by age, gender, race, hypertension, alcohol consumption, BMI, MLR, statin use, and antihypertensive medication use among groups (all P-values < 0.05). The mean (95%CI) age was 55.65 (54.91, 56.38) years in the no AMD group, 66.25 (64.42, 68.09) years in the early AMD group, and 77.91 (76.03, 79.80) years in the late AMD group. The mean (95%CI) MLR was 0.28 (0.28, 0.29) in the no AMD group, 0.32 (0.30, 0.33) in the early AMD group, and 0.42 (0.33, 0.50) in the late AMD group.
Higher MLR associated with prevalence and severity of AMD
Three regression models were utilized to explore the association between MLR and AMD. The odds ratio (OR) and 95% confidence interval (95% CI) were shown in Table 2. In the non-adjusted model, a significant positive association was discovered between MLR and AMD (OR = 12.01, 95% CI: 5.76–25.04). In the weighted multivariable logistic regression models, after adjusting for age, gender and race, the OR was 2.75 (1.18–6.40); after adjusting for age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption, the OR was 3.22 (1.32–7.82). Smoothed curve fitting also revealed that MLR was positively associated with AMD (Figure 2). In addition, as shown in Table 3, threshold effect analysis revealed that the inflection point was 0.37, and higher MLR associated with prevalence of AMD after MLR was greater than the inflection point value (OR = 8.93, 95%CI: 2.53–31.60). ROC curve analysis was performed to evaluate the diagnostic performance of MLR > 0.37 for AMD. The analysis of MLR alone yielded an AUC of 0.621 (95% CI: 0.57–0.67), indicating modest discriminatory ability (Figure 3(A)). To evaluate its potential clinical utility while accounting for patient characteristics, a multivariable logistic regression model including MLR was constructed and adjusted for age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking status, and alcohol consumption. The model produced an AUC of 0.73 (95% CI: 0.69–0.77), demonstrating good discriminatory ability (Figure 3(B)). These results suggest that while MLR > 0.37 alone has limited predictive power, it could contribute meaningful information in a clinical setting when combined with relevant patient factors.
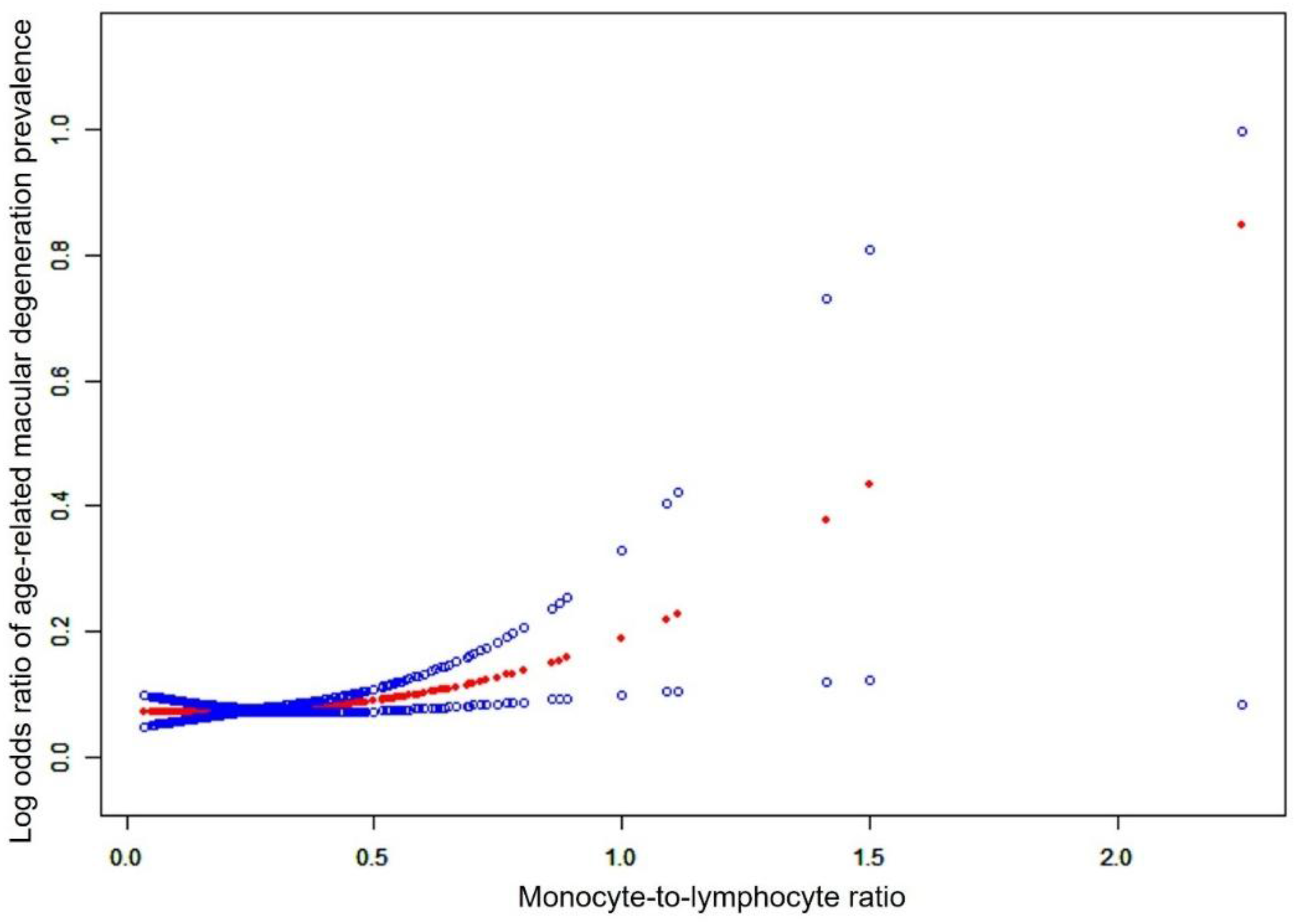
Smoothed curve fitting of the association of MLR with AMD using the generalized additive model. The red line represents the smooth curve fit between variables. Blue bands represent the 95% confidence interval from the fit.
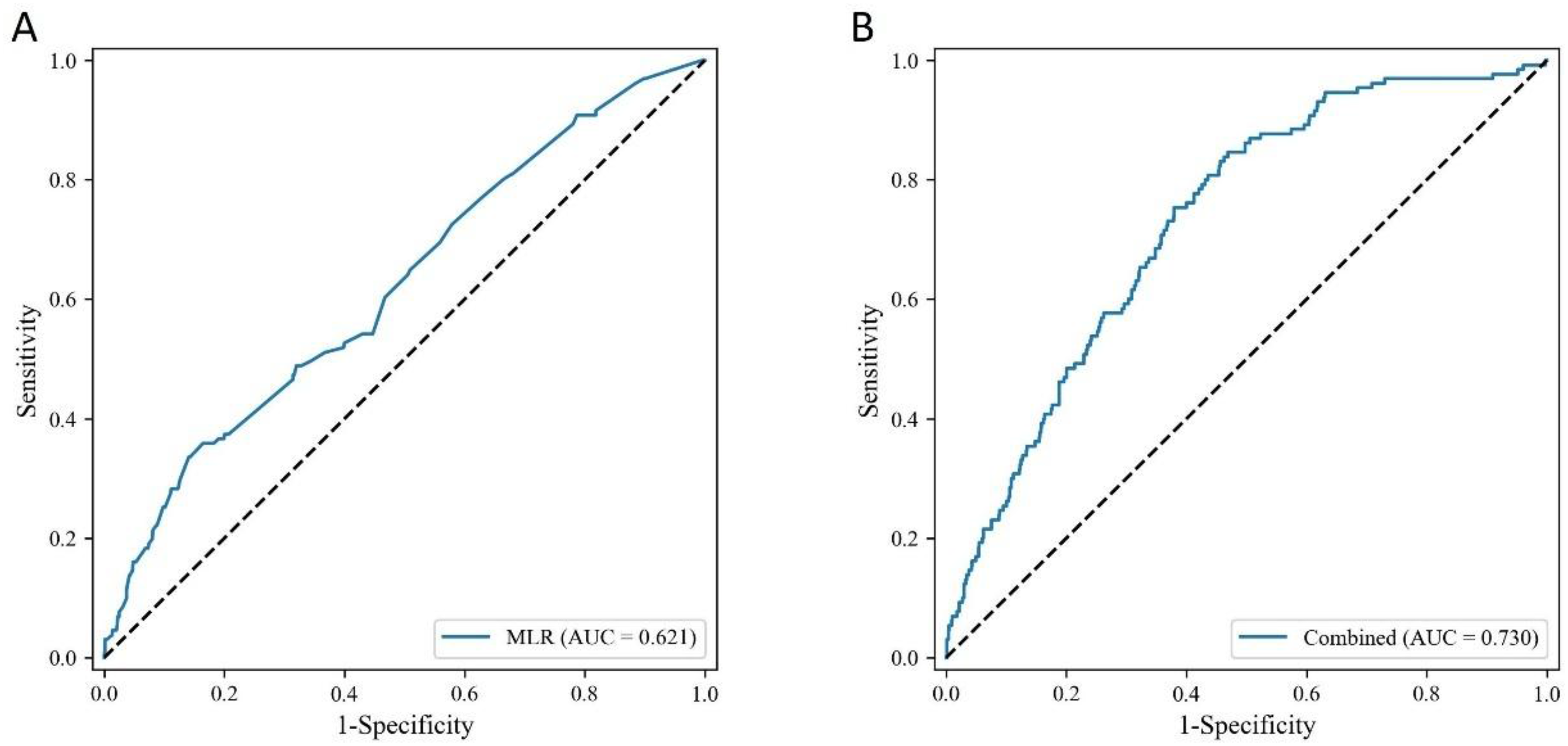
ROC curves illustrating the discriminatory ability of MLR > 0.37 for AMD.
The weighted multivariable logistic regression analysis of association between MLR and AMD.

Model 1: unadjusted. Model 2: adjusted for age, gender, and race. Model 3: adjusted for age, gender, race, education level, hypertension, diabetes, body mass index, C-reactive protein, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption.
Threshold effect analysis of MLR on AMD using two-piecewise linear model.

Model was adjusted for age, gender, race, education level, hypertension, diabetes, body mass index, C-reactive protein, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption.
Further multinomial logistic regression was used to test the statistical significance of the association of MLR with severity of AMD while adjusting for age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption. As shown in Table 4, MLR was significantly associated with early AMD (OR = 1.20, 95% CI: 1.19–1.21) and late AMD (OR = 9.21, 95% CI: 9.10–9.31).
The weighted multinomial logistic regression analysis of the association between MLR and AMD severity.

Models were adjusted for age, gender, race, education level, hypertension, diabetes, body mass index, C-reactive protein, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption.
Subgroup analysis of factors influencing the association between MLR and AMD
Subgroup analysis and interaction test was conducted stratified by age, gender, race, education level, hypertension, diabetes, BMI, CRP, statin use, antihypertensive medication use, hypoglycemic medication use, systemic glucocorticoid use, immunosuppressant use, smoking and alcohol consumption. Our results revealed that the association between MLR and AMD was stable. As shown in Figure 4, the interaction analyses of MLR and age (P for interaction = 0.5452), gender (P for interaction = 0.3362), race (P for interaction = 0.0955), education level (P for interaction = 0.4678), hypertension (P for interaction = 0.2063), diabetes (P for interaction = 0.4252), smoking (P for interaction = 0.1116), alcohol consumption (P for interaction = 0.2546), BMI (P for interaction = 0.6753), CRP (P for interaction = 0.1196), statin use (P for interaction = 0.6678), antihypertensive medication use (P for interaction = 0.7350), hypoglycemic medication use (P for interaction = 0.1072), systemic glucocorticoid use (P for interaction = 0.3503), immunosuppressant use (P for interaction = 0.6017) in regard to AMD were not significant.
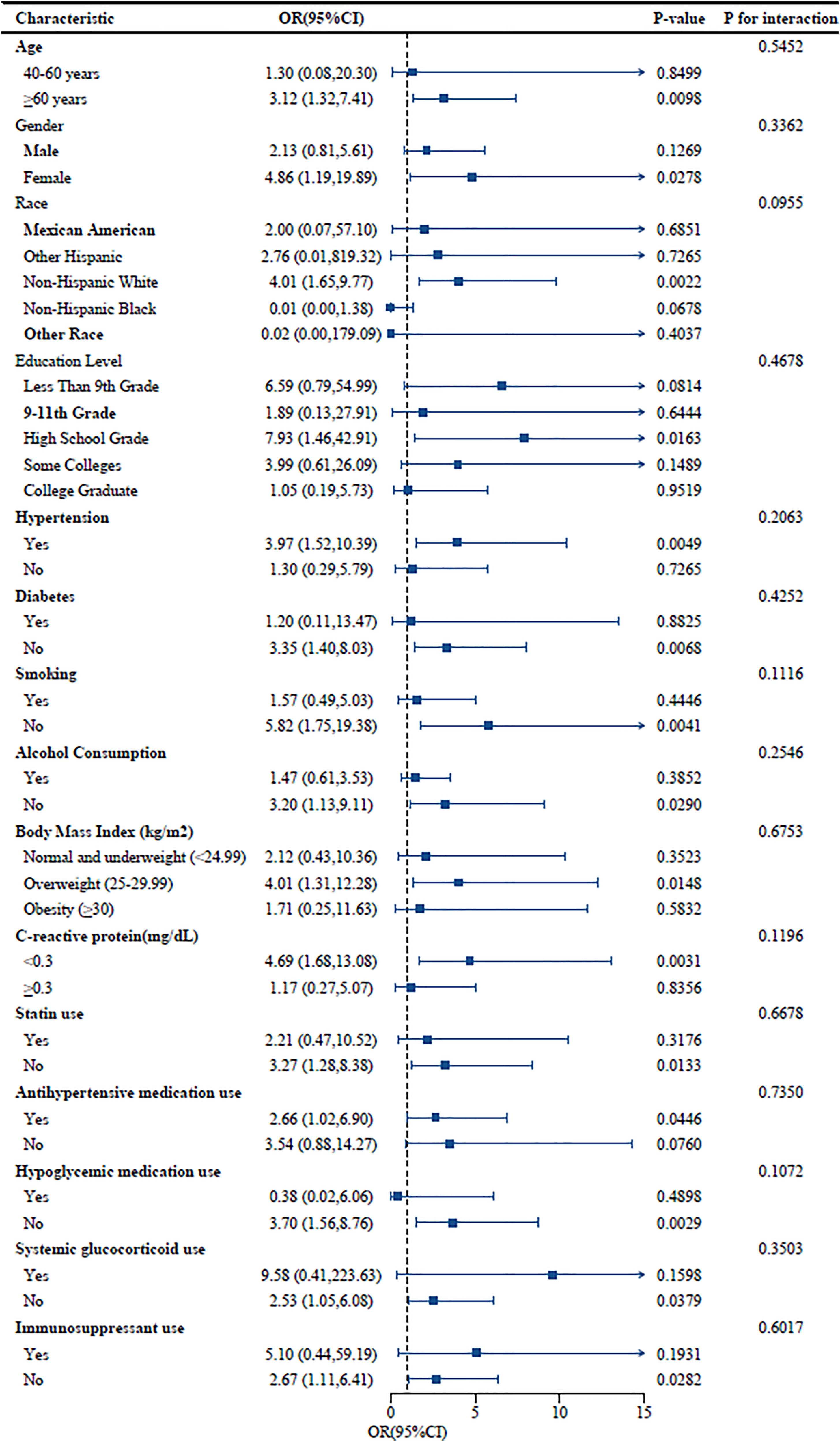
Subgroup analysis for the association between MLR and AMD. Squares represent the adjusted estimates for each subgroup. Horizontal lines indicate the 95% confidence intervals. The dashed vertical line represents the null value (OR = 1). P for interaction was calculated by comparing models with and without the interaction term.
Discussion
This national observational cross-sectional study of 4,655 participants from the 2005–2008 NHANES evaluated the relationship between MLR level and prevalence and severity of AMD. After adjusting for covariates, MLR was positively associated with prevalence of AMD, early AMD and late AMD independently. These findings extend prior evidence from a retrospective study reporting higher MLR levels in patients with neovascular AMD. 28
Emerging evidence highlights the significant role of inflammation in AMD. Levels of the pro-inflammatory cytokines interleukin-8 (IL-8) and interleukin-6 (IL-6) were suggested to be positively correlated to central subfoveal thickness in eyes with neovascular AMD. 29 lexy et al. conducted animal experiments and underlined that IL-6 prolonged subretinal mononuclear phagocyte which was associated with AMD. 30 Human geographic atrophy of the retinal pigment epithelium was observed to contain elevated amounts of inflammasome components. 31 Moreover, Liisborg et al. discovered that patients with myeloproliferative neoplasms characterized by chronic low-grade inflammation have a higher prevalence of AMD. 32
Monocytes produce increased pro-inflammatory cytokines such as IL-6 under certain pathological conditions.33–35 Because independent monocyte and lymphocyte counts can be easily influenced by age, medication use, hormonal changes and other factors, the MLR level provides a more stable marker of immune status.36–38 Previous studies have demonstrated its prognostic value in conditions such as tuberculosis, COVID-19-related sepsis, and cardiovascular diseases.39,40
It is notable that MLR showed a stronger association with late AMD than early AMD in this study. Chronic, low-grade inflammation is evident in early AMD, whereas there is progressive amplification of inflammation including complement activation and macrophages involvement in late AMD. 41 There was a cross-sectional study which revealed that higher monocyte counts (≥ 0.5 × 10⁹/L) are more strongly associated with the risk of intermediate/late AMD (OR 1.58) compared to any AMD (OR 1.45). 42 Patients with neovascular AMD were reported to have significantly increased proportion of non-classical monocytes.43,44
It is the first cross-sectional study to investigate the association between MLR and AMD in a large population. Though there was a retrospective study revealing that greater MLR was significantly associated with neovascular AMD, 28 NHANES database allowed us to use larger sample size and proper adjustment of covariates to enhance the reliability and representativeness of our study. Nevertheless, there are some limitations. First, we cannot establish the causal relationship between MLR and AMD because this is a cross-sectional study. A prospective study is needed in the future. Second, some patients with bilateral AMD or other concomitant ocular diseases might be excluded from this study due to compromised fixation secondary to low vision which made the acquisition of reliable fundus photographs challenging. In addition, NHANES was conducted only in the United States with limited ethnicity, more research is required to verify our results in other populations.
Conclusions
This study indicates that MLR is significantly associated with the prevalence of AMD, including both early AMD and late AMD.
Footnotes
Ethics approval and consent to participate
The research involving human participants underwent a thorough review and received approval from the Research Ethics Review Board of the National Centre for Health Statistics Research Ethics (NCHS).
Consent for publication
Not applicable.
Author contributions
PW and SB designed the study, XC wrote the original manuscript and analyzed data, TL provided statistical advice, and all authors (XC, TL, SJ, PW, SB) critically revised the manuscript for important intellectual content. All authors approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and analyzed in the current study are available from the NHANES website (www. cdc. gov/ nchs/ nhanes/).
