Abstract
Background
Corneal ulcers are typically caused by external (“exogenous”) factors such as trauma, contact lens use, or preexisting ocular surface diseases. Internal systemic (“endogenous”) sources have not been reported as direct causes of corneal ulcers. Here, we report, to our knowledge, the first documented case of a corneoscleral abscess that resulted from an underlying bacteremia.
Method
This is a single case report.
Result
A previously healthy 35-year-old male presented to the Emergency Department with left eye redness, pain, and blurred vision. Initial examination showed reduced visual acuity (20/150), elevated intraocular pressure (44mmHg), diffuse microcystic corneal edema with mutton-fat keratic precipitates, and anterior chamber cells. No corneal infiltrate, epithelial defect, hypopyon, or vitritis was noted. Within days, his condition rapidly progressed to a corneoscleral abscess with auto-evisceration. Blood and corneal cultures identified hypermucoviscous Klebsiella pneumoniae. Further workup revealed an underlying prostatic abscess with secondary iliac vein thrombus, leading to seeding of other sites, including the lungs and the eye. Despite intensive antibiotic therapy, the ocular condition worsened, requiring urgent left eye evisceration.
Conclusion
This case highlights the rapid progression of hypermucoviscous Klebsiella pneumoniae infection, which first presented with endophthalmitis, followed by corneoscleral perforation.
Introduction
Endophthalmitis is a severe ocular condition that can lead to significant vision loss or even permanent loss of eye if not treated promptly. It can result from both exogenous (traumatic or postoperative) and endogenous (hematogenous spread) cause, the latter occurring particularly in immunocompromised patients. 1
Corneal ulcers are also serious and can lead to devastating ocular complications, including exogenous endophthalmitis.2,3 These ulcers are typically caused by external (“exogenous”) factors such as contact lens use, trauma, preexisting ocular surface diseases, or prior ocular surgeries4,5 that disrupt the protective corneal epithelial barrier, allowing microorganisms to infiltrate. Although immunocompromised state pose as a risk factor for corneal ulcer, internal systemic (“endogenous”) sources have not been identified as direct causes of corneal ulcers. Though corneal ulceration due to bacteremia has not been described, the reverse (severe corneal ulcer leading to bacteremia) has been documented. 6 Here, we report, to our knowledge, the first documented case of a corneoscleral abscess that resulted from an underlying bacteremia.
Case description
A 35-year-old Caucasian male with no significant medical history, except for alcohol use (approximately 15 drinks per week) and previous admission to the Intensive Care Unit due to pneumococcal pneumonia while travelling to Australia eight months ago, presented to the Emergency Department (ED) with a 4-day history of fever and shortness of breath. The patient was tachycardic at 115–120 beats per minute but was afebrile and otherwise hemodynamically stable. Initial workup in ED revealed opacification of the pulmonary artery tree with associated concentric filling defect within the basal lateral left lower lobe segmental artery, in keeping with acute pulmonary thrombus on Computed Tomography (CT) chest. The pulmonary embolism (PE) was thought to have occurred in the context of an upper respiratory tract infection (URTI). The patient was evaluated by the medical team, who consulted hematology, and was started on rivaroxaban. At this time, the patient also reported a 2-day history of left eye redness, pain, and blurred vision, and was referred to ophthalmology.
Ophthalmological examination revealed a reduced unaided visual acuity (VA) of 20/150, elevated intraocular pressure (IOP) of 44 mmHg, diffuse microcystic corneal edema with inferior mutton-fat keratic precipitates (KPs), and 1 + anterior chamber cells (Figure 1a). There was no corneal infiltrate, epithelial defect, or hypopyon. The fundus view was hazy, and a B-scan ultrasound was performed, which did not show any vitritis or gross abnormalities. The patient denied any history of ocular trauma, surgery, or contact lens use. He was diagnosed with possible hypertensive uveitis, and an anterior chamber tap was performed for viral Polymerase Chain Reaction (PCR) testing and to help lower the IOP. He was started on oral valacyclovir (1 g three times daily), topical steroid (prednisolone acetate ophthalmic suspension 1% four times daily), topical antibiotic (moxifloxacin hydrochloride ophthalmic solution 0.5% four times daily), and topical anti-glaucoma medication (4 classes consisting of travoprost 0.004%, timolol 0.5%, brinzolamide 1% and brimonidine tartrate 0.2%), and was re-assessed the following day.
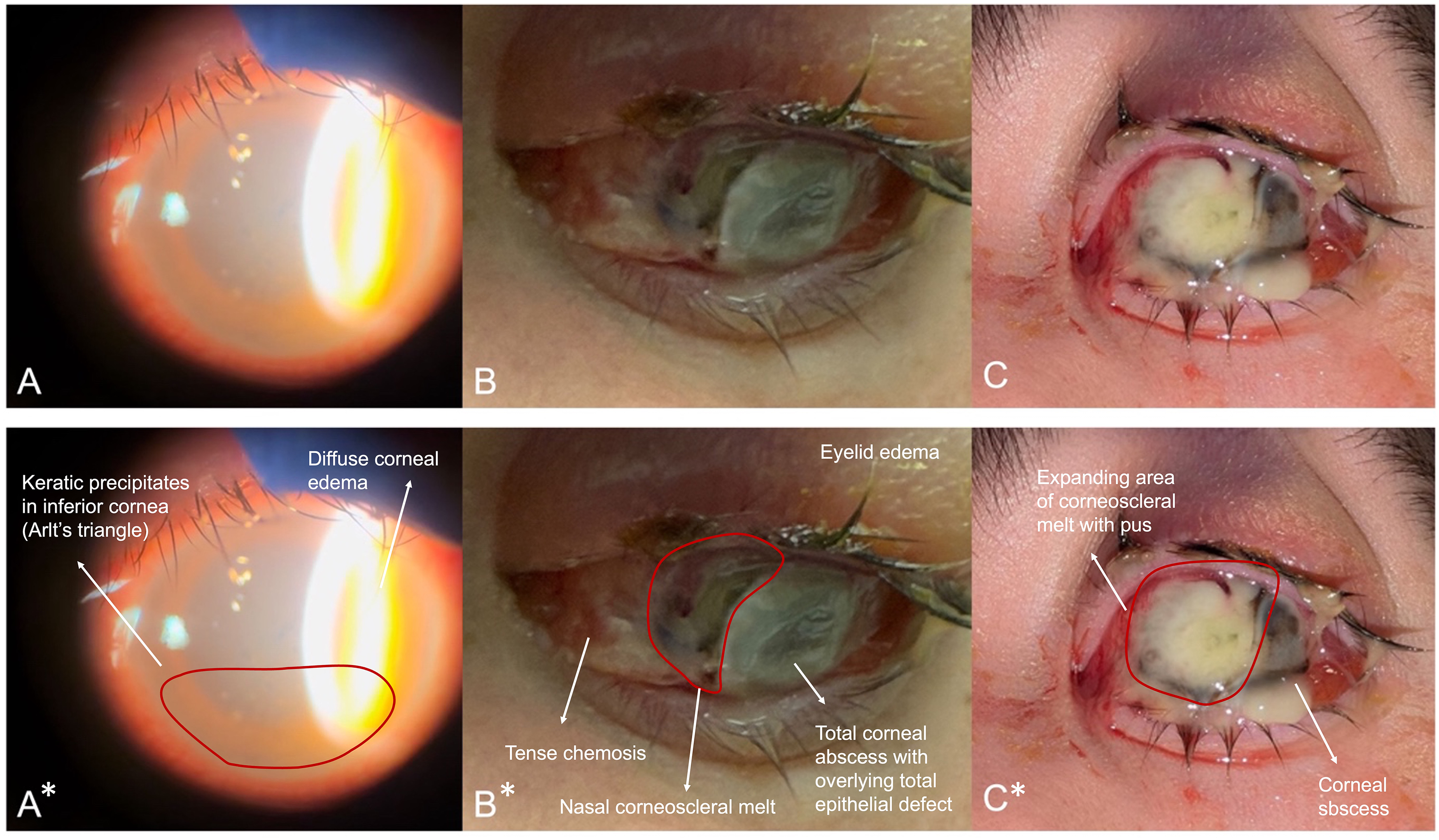
A – First day of presentation showing presence of large keratic precipitates in the inferior cornea with diffuse microcystic corneal edema. No hypopyon, corneal epithelial defect nor infiltrate is seen. B – Third day of presentation showing rapid progression to corneoscleral abscess with large area of perforation in the nasal limbus. There is also significant lid edema, chemosis, and proptosis. C – Fifth day of presentation showing expanding area of perforation with large amount of pus at the site. Bottom row of images with asterisk (*) show annotations.
On day 2, the IOP improved to 17mmHg, but the VA worsened to hand movement vision. The patient had worsening eyelid edema with new significant inferior conjunctival chemosis. The cornea remained diffusely edematous with presence of KPs, and no epithelial defect was seen. There was a hazy view of the anterior chamber, but no hypopyon was seen. The B-scan did not show any vitritis. By then, the tap results were negative for herpes simplex, herpes zoster, and cytomegalovirus. The patient was continued on the above treatment and was reviewed the next day.
On day 3, the patient was referred to and reviewed by the cornea team. There was worsening redness, pain, vision and swelling. His VA deteriorated to light perception (LP), and he developed a total corneal abscess with an epithelial defect. There was adjacent scleral melt with nasal limbal perforation, and underlying vitreous and uvea were seen (Figure 1b). There was no view of the anterior or posterior chamber. There was significant lid edema, chemosis, and proptosis with limitation of extraocular movement in all directions. Given the rapid progression of his ocular findings and the recent URTI and PE history, the patient was admitted immediately for further evaluation and care.
A gentle corneal swab was obtained, and the patient was started on intensive hourly topical fortified vancomycin 33.3 mg/ml and tobramycin 13.6 mg/ml. Topical steroid was discontinued, and intravenous (IV) moxifloxacin was initiated. A CT scan of the orbits showed a rim-enhancing collection in the left anterior globe extending into vitreous chamber, with non-visualisation of lens, and surrounding preseptal cellulitis (Figure 2).
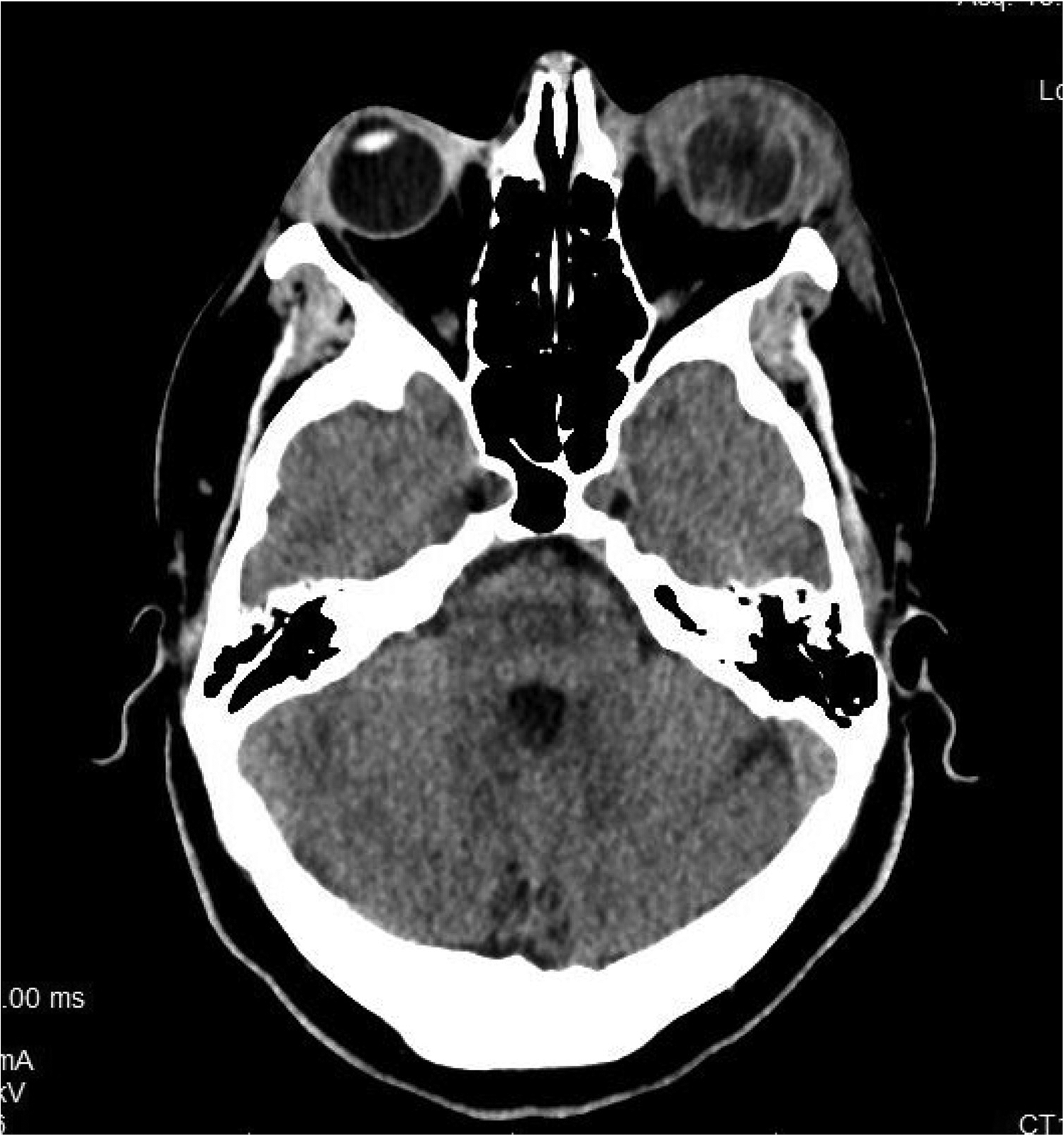
Computed tomography (CT) scan of the orbits showing enhancing collection in the left anterior globe extending into vitreous chamber, with non-visualisation of lens, and surrounding preseptal cellulitis.
Throughout his hospitalisation, the patient was co-managed by internal medicine and infectious disease teams, and an extensive systemic workup was carried out. Blood and eye swab cultures identified Klebsiella pneumoniae, with a hypermucoviscous (HMV) strain confirmed by the string test. Further tests were performed to look for underlying infective foci. Repeat chest CT showed worsening lung parenchymal opacity, while an echocardiogram did not reveal any vegetations. Abdominal ultrasound was negative for hepatic abscesses. A CT scan of the abdomen and pelvis, as well as transrectal ultrasound, revealed a prostatic abscess, which was aspirated and grew Klebsiella on cultures. Additionally, left iliac vein thrombosis was noted. Urine culture and infective screen for other organisms were negative. Human immunodeficiency virus and diabetic mellitus screening were negative. The patient was switched to IV meropenem (2 g every 8 h) during this period and oral valacyclovir was stopped.
Despite intensive antibiotic therapy, the patient's ocular condition worsened. His VA remained LP and a reverse relative afferent pupillary defect was present, along with an expanding area of corneoscleral melt (Figure 1c). By day 5, given the severity of corneoscleral abscess with auto-evisceration, a discussion was held with the patient and his family, as well as the interdisciplinary team, and it was decided to proceed with an urgent left eye evisceration by the oculoplastics team. The pathology report confirmed the presence of gram-negative rods. Following surgery, the patient was switched to topical tobramycin ointment. After all the systemic workups were completed and repeat blood cultures were negative, the patient was placed on a peripherally inserted central catheter line and discharged with a plan for 6 weeks of IV ceftriaxone therapy. The patient made a good recovery with no infection at the surgical site at postoperative month 1.
We present a case of a young, previously healthy male who developed a severe systemic multifocal HMV Klebsiella pneumoniae infection with prostatic abscess, iliac vein thrombosis, pulmonary embolism, and ocular involvement. The likely sequence of events began with a prostatic abscess with secondary iliac vein thrombus resulting in seeding to other sites. Klebsiella bacteremia is well-known to cause endophthalmitis. However, to our knowledge, this is the first documented case of multifocal HMV Klebsiella bacteremia leading to an unusual presentation of predominantly anterior-involving endophthalmitis, prior to seeding to the cornea and leading to perforated corneoscleral abscess.
Discussion
Endogenous Klebsiella pneumoniae endophthalmitis (EKE) is a well-documented, serious complication of gram-negative sepsis, often leading to poor visual outcomes, with a study reporting 41% of cases requiring evisceration or enucleation. 7 While Klebsiella is the leading cause (45.6–60.8%) of endogenous endophthalmitis in East Asia, it is rare in the Western world, where its prevalence ranges from 3.1 to 5.0%. 8 In the Western region, gram-positive bacteria are more commonly responsible for endogenous endophthalmitis. 8 EKE typically stems from liver abscesses or urinary tract infections, 9 though a case involving prostatic abscess has been reported, 10 such as in our case. Diabetes mellitus has been identified as the most significant risk factor for EKE. 9 Poor prognostic factors include rapid onset of symptoms, unilateral involvement, presence of hypopyon, and panophthalmitis. 11
Though main manifestation of Klebsiella in the eye is endogenous endophthalmitis, it can also cause isolated infectious corneal ulcer. Risk factors of Klebsiella keratitis include previous keratoplasty, ocular trauma, pre-existing ocular surface disease, and presence of diabetes mellitus. 12 It can result in rapid progression, with one case report of nosocomial Klebsiella pneumoniae conjunctivitis resulting in infectious keratitis and bilateral corneal perforation, 13 and another study showing that 56.8% of patients with Klebsiella keratitis needed one or more surgical procedures to address the infection. 12
The above clinical presentations are attributable to the fact that Klebsiella pneumoniae is a highly virulent intraocular pathogen. The virulence of Klebsiella, especially strains with certain genetic features like the K1/K2 serotype, hypermucoviscosity (HMV), or the magA gene, contributes to the aggressive nature of the infection and its potential for dissemination. 14 Hypervirulent Klebsiella pneumoniae (hvKP) represents a distinct pathotype characterised by increased virulence and a propensity to cause severe community-acquired infections in otherwise healthy individuals.14–16 Initially emerging in the Asia-Pacific region, this condition has since gained growing attention across Western nations. Key virulence factors that set hvKP apart include multiple iron-acquiring siderophore systems, enhanced capsule production, K1 and K2 capsule types, and the colibactin toxin. 17 One hallmark trait of hvKP is its hypermucoviscous nature, a laboratory-observed characteristic where colonies appear notably sticky. This is assessed using the “string test”, which is considered positive if a colony lifted from an agar plate with a loop or needle forms a viscous string exceeding 5 mm in length. Clinically, hvKP can spread and disseminate rapidly to distant sites, such as the lung, meninges and eye, and result in poor outcomes.14,15 In severe cases, EKE can advance to panophthalmitis, with high risk of need for evisceration or enucleation due to its grave prognosis.
This case is unique in that the patient initially presented with clinical features of endophthalmitis predominantly involving the anterior chamber, before developing a corneoscleral abscess. While corneal abscess can lead to endophthalmitis and potentially bacteremia when severe, 6 the reverse causation has not been previously reported. Though bacteremia can result in endogenous endophthalmitis, there are no prior reports of bacteremia leading to a secondary corneoscleral abscess.
Typically, bacteria preferentially disseminate to the vascular choroid, prior to spreading to the retina and vitreous humour. It is thought that the inflammatory cells and cellular debris then permeate through the zonules into the anterior chamber, leading to a hypopyon. 18 In this case, the patient's initial ocular involvement was predominantly anterior, with large keratic precipitates, severe anterior chamber inflammation, high intraocular pressure, and diffuse microcystic corneal edema, but no hypopyon or significant vitritis. It is unusual for endogenous endophthalmitis to present with such intense AC inflammation alone without vitritis. In this case, given predominantly anterior involvement, and the patient's otherwise healthy, young background, a diagnosis of hypertensive uveitis seemed more plausible at first, before the condition deteriorated rapidly. Moreover, there have been no prior reports of secondary corneoscleral involvement from endogenous source, likely due to the avascular nature of the cornea and sclera. However, since the patient developed a frank corneoscleral abscess and epithelial defect only after the initial presentation of corneal edema and opacification, this suggests the infection originated “inside-out”.
In this patient, it appears that there was a much higher bacterial load in the anterior segment, and it is possible that the highly virulent Klebsiella circulating in the anterior chamber attached to and invaded the cornea from the endothelium. In fact, the keratic precipitates that were initially seen on the first day of ocular presentation could have been early corneal microabscesses. Additionally, it is likely that the bacteria in the bloodstream reached the cornea through the highly vascularised limbal vessels, especially in an inflamed eye, which may explain why the greatest area of corneal melt occurred at the nasal limbus in our patient, leading to perforation in that area. Unfortunately, the aggressive nature of hypervirulent Klebsiella led to the rapid progression of ocular infection, resulting in corneoscleral melt and perforation in our patient, with no improvement on intensive topical and systemic antibiotics.
Conclusion
In conclusion, we present a rare case of severe hypermucoviscous Klebsiella bactermia from prostatic abscess resulting in metastatic septic complications in a young, otherwise healthy patient, resulting in multiple vascular thrombi and panophthalmitis. This case is unique in that the patient initially presented with endophthalmitis with predominantly anterior involvement, followed by secondary development of corneoscleral abscess from an endogenous source. Early diagnosis, thorough evaluation, and prompt initiation of treatment are critical; however, a high risk of disease progression persists, particularly in infections caused by hypervirulent Klebsiella strains, despite intensive topical and systemic antibiotic therapy. Even so, it is crucial to start empirical followed by directed antimicrobial therapy promptly, not only to reduce progression in the affected eye, but to prevent involvement of the fellow eye.
Footnotes
Consent for participation / publication
A written informed consent for participation and publication was provided by the patient.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Ethics approval was not required for this case report. This study followed the tenets of the Declaration of Helsinki and written informed consent was obtained from the patient.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
