Abstract
Introduction
Non-arteritic anterior ischemic optic neuropathy (NAION) is a major cause of vision loss in middle-aged and elderly populations, with no effective treatment currently available. Electrical stimulation (ES) has shown potential in preserving and restoring neural function in optic neuropathies. This study investigates the efficacy of transpalpebral ES in improving visual function in a patient with long-term visual impairment due to NAION.
Methods
A 62-year-old male patient with sequential NAION in both eyes underwent a 60-day transpalpebral ES for 20 min daily. Visual function was assessed at baseline and after 30 and 60 days of treatment, including best-corrected visual acuity (BCVA), contrast sensitivity, visual field (VF) analysis, and microperimetry.
Results
Significant improvements in BCVA were detected, with an overall increase of 11 ETDRS letters in the right eye and 18 in the left eye after 60 days of treatment. Contrast sensitivity also improved in both eyes. Visual field parameters, including mean deviation, pattern standard deviation and visual field index, showed notable enhancements. Microperimetry revealed increased macular sensitivity and improved fixation stability. The patient also reported subjective improvements. No adverse events were noted.
Discussion
ES has been shown to enhance retinal function by modulating neuronal excitability, activating neuroprotective signaling pathways, and potentially supporting neural regeneration. In our case, transpalpebral ES was associated with meaningful improvements in visual function in a patient with long-term NAION-related visual impairment. These results suggest ES as a promising approach for NAION, though further research is needed to confirm these results and optimize treatment protocols.
Introduction
Non-arteritic anterior ischemic optic neuropathy (NAION) represents one of the most important causes of blindness or severely impaired vision in middle-aged and elderly people. This medical condition leads to an acute and painless visual acuity (VA) loss and visual field (VF) defects, associated with sectorial or diffuse optic nerve head (ONH) edema. 1 NAION is typically unilateral at onset, while simultaneous bilateral occurrence is exceptionally rare. However, sequential involvement of the fellow eye has been reported to occur in 15–30% of cases within five years of the first eye damage. 2 Its pathogenesis is complex and not completely known, however, it is commonly accepted that NAION is caused by an acute hypo-perfusion or hypo-ossigenation of the ONH, resulting in an acute ischemic infarction of the optic nerve axons, and secondary apoptosis of the retinal ganglion cells (RGCs). 3 In most cases, NAION results in permanent vision loss, with 40–50% of patients experiencing significant visual impairment or legal blindness in the affected eye 4 leading to a significantly reduction in quality of life and work. 5
Corticosteroids have been proposed for the treatment of acute NAION to accelerate the optic disc edema resolution, thus reducing the compression of capillaries and axons, improving ONH blood flow and increasing the survival of ischemic axons. 6 However, recent clinical trials and meta-analyses found that treatment with systemic corticosteroids significantly accelerates the resolution of the optic disc edema and improves the electrophysiological parameters of the optic nerve in acute NAION but does not significantly improve VA and VF outcomes.7,8 Hyperbaric oxygen therapy 9 and neuroprotective substances 10 have been also used in patients with NAION showing promising results, but studies investigating their efficacy are limited. Currently there is no effective treatment available for NAION. Thus, there is a need to evaluate new therapeutic strategies for the persistent vision loss caused by this optic neuropathy. Several studies indicate that electrical stimulation may help preserve and restore function in damaged neural tissues. It was reported that electrical stimulation (ES) applied to the optic nerve immediately after axotomy in adult rats enhances the survival of axotomized RGCs, suggesting that ES may regulate early neuroprotective mechanisms against axotomy-induced neuronal cell death. 11 A clinical study by Fujikado et al. showed that transcorneal ES can improve the visual functions in patients with optic neuropathy. 12 In addition, it has been shown that repetitive transorbital alternating current stimulation (rtACS) can improve visual performance and vision-related quality of life in patients with long-term optic nerve lesions. 13 In light of these studies, we investigated the efficacy of ES administered via a transpalpebral approach as a method to improve visual function in a patient with long-term visual impairment due to previous sequential NAION in both eyes.
Methods
Case history
A 62-year-old male patient with a history of long-term visual impairment due to consecutive NAION in both eyes was evaluated in an exploratory study at our Low Vision and Visual Rehabilitation Clinic at ASST Santi Paolo e Carlo - San Paolo Hospital in Milan. The patient gave informed written consent to participate in the study, which was carried out in accordance with the Declaration of Helsinki and ethical committee approval.
The patient experienced the first episode of NAION in the right eye (RE) at the age of 55, followed by involvement of the left eye (LE) a year later. At the time of the acute events, comprehensive laboratory investigations were performed, including coagulation studies and inflammatory markers ESR and CRP, all of which yielded results within normal limits. The patient had no history of systemic diseases such as hypertension, diabetes, or cardiovascular conditions and was not on any medication or neuroprotective supplements. However, sleep apnea syndrome had not been investigated during his clinical workup.
For both episodes, the patient presented with sudden, unilateral, painless loss of vision upon awakening, primarily in the lower half of the visual field
After the resolution of the acute phase of the second episode, the patient underwent annual outpatient ophthalmic examinations, which did not reveal any new acute events, and the patient reported no changes in his visual function.
Baseline evaluation
On arrival at our clinic, six years after the second event, the patient reported persistent low VA and inferior VF deficits, causing daily life challenges, including episodes of falls and difficulties at work. He was in good general health and was not taking any medications or neuroprotective supplements.
At the baseline ophthalmological examination BCVA was 71 ETDRS letter score in RE and 60 ETDRS letter score in LE. Pelli-Robson contrast sensitivity was 1.20 in both eyes. Anterior slit lamp examination was unremarkable in both eyes. Intraocular pressure was within normal range in both eyes. Dilated fundus examination revealed an optic disc with a small diameter and a reduced cup-to-disc ratio, associated with pallor in both eyes. (Figure 1) Optical coherence tomography (OCT) (Heidelberg Engineering, Dossenheim, Germany) revealed thinning of the retinal nerve fiber layer (RNFL) in the temporal and nasal superior regions in both eyes, and microcystic macular edema in the inner nuclear layer, (Figure 2) a finding commonly associated with RNFL / RGCs degeneration in optic neuropathies. 14
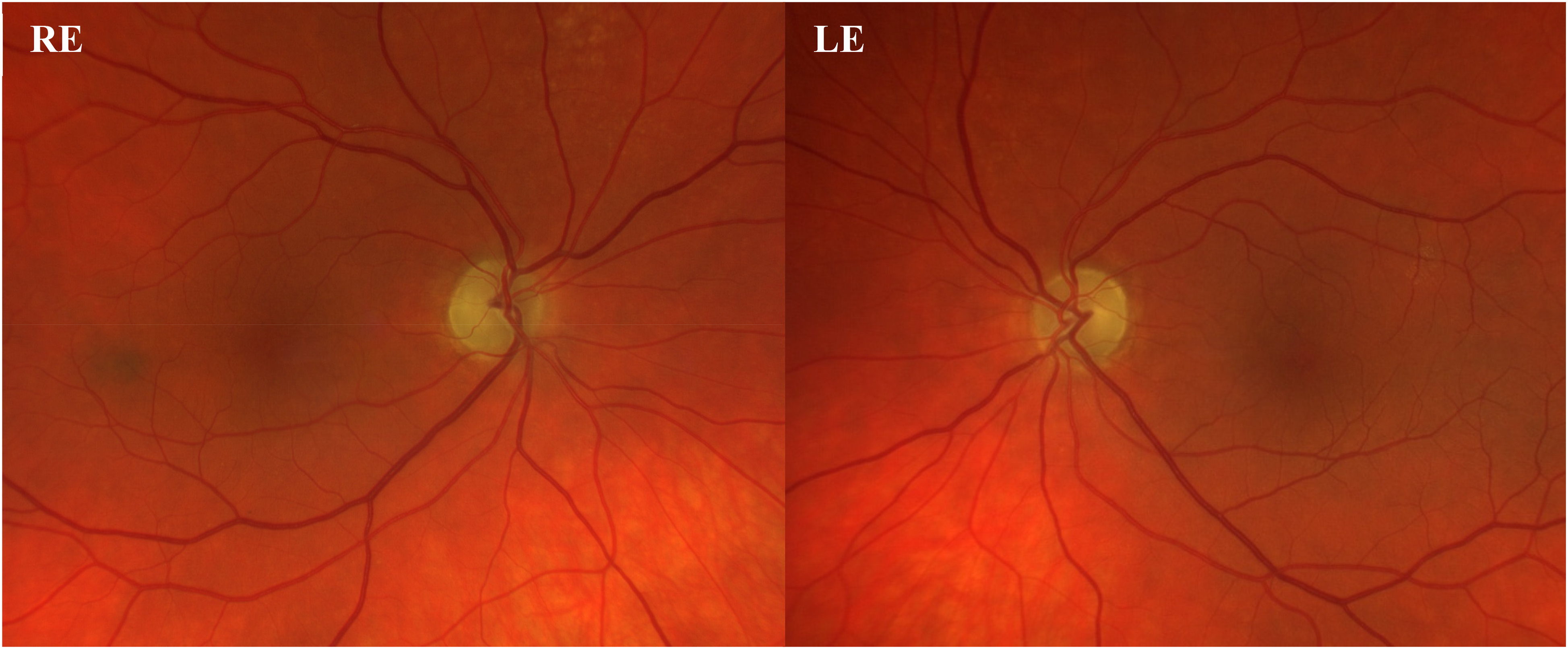
Fundus photography of both eyes revealed small, pale optic discs with a reduced cup-to-disc ratio.
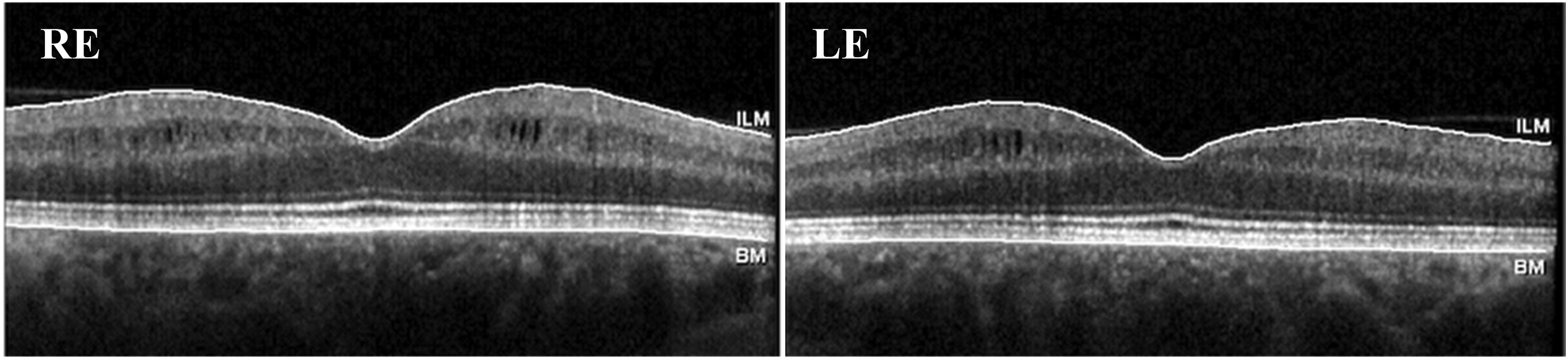
OCT scans of both eyes revealed thinning of the retinal nerve fiber layer (RNFL) and microcystic macular edema in the inner nuclear layer.
VF examination (Humphrey Field Analyzer with SITA-Fast 30-2) again showed a nearly absolute inferior nasal defect in both eyes, and a relative inferior altitudinal defect in RE. MD value was −11.98 dB in the RE (P < 0.5%) and −10.11 dB in the LE (P < 0.5%), PSD value was 12.89 dB in the RE (P < 0.5%) and 10.69 dB in the LE (P < 0.5%), VFI was 69% in RE and 76% in LE.
Macular retinal sensitivity was accurately analyzed with MAIA microperimetry (Centervue, Padova, Italy) using the standard 10−2 test grid with the full threshold 4-2 strategy. The average sensitivity was 15.8 dB in RE and 19.7 dB in LE. In both eyes, the sensitivity map displayed a reduced sensitivity in the superior half of the grid with two distinct areas of absolute scotomas. Comparative analysis of the fixation points cloud and the final PRL on the retinal image obtained via Scanning Laser Ophthalmoscope with the foveal position on OCT scans demonstrates that the patient's PRL does not coincide with the fovea but is situated at an inferior retinal location relative to the fovea. Fixation was relatively unstable in both eyes, with P1 = 55% and P2 = 93% in the RE, and P1 = 48% and P2 = 90% in LE.
Transpalpebral electrical stimulation
Transpalpebral ES was administered in both eyes using the Subeye, a research-use-only device (SubVision, Milan, Italy), as described in a previous work by our group. 15 We chose the transpalpebral approach, with electrodes placed on the eyelids, for practical and safety reasons, as the patient performed the treatment at home independently. The initial treatment period lasted 30 days, during which the patient administered ES at home on his own, five days a week for 20 min each day, excluding Saturdays and Sundays. Follow-up visits were conducted on days 7 and 14 to assess for any potential side effects. At the end of this initial 30-day phase, a follow-up visit was performed to evaluate the effects of the treatment. Subsequently, the treatment was extended for an additional 30 days, and a final follow-up visit was conducted at the end of this extended period.
Results
The patient completed the 60-day treatment regimen at home on his own without reporting any issues with the transpalpebral ES procedure. No adverse events were reported. During the first and second follow-up visits, conducted 30 and 60 days after the initiation of treatment, respectively, baseline assessments were repeated using the same instruments to assess the effects of the treatment.
Optical coherence tomography
Follow-up OCT scans revealed no changes in the macular profile and the RNFL thickness remaining stable compared to previous measurements.
Visual acuity and contrast sensitivity
At the first follow-up visit, after 30 days of treatment, an increase in BCVA was observed in both eyes, with RE achieving a 76 ETDRS letter score and the LE achieving a 71 ETDRS letter score. A further significant improvement was noted at the second follow-up visit after two months of treatment, with BCVA in RE increasing to 82 ETDRS letter score and in LE to 78 ETDRS letter score.
Additionally, the patient demonstrated an increase in contrast sensitivity. In RE, contrast sensitivity improved to 1.50 log CS after 30 days and further to 1.65 log CS after 60 days of treatment. Conversely, in LE, there was an improvement to 1.50 log CS after 30 days of treatment, with no additional increase observed after 60 days of treatment.
The patient also reported subjective improvements, including greater confidence in mobility, even in low-light environments, and increased reading fluency.
Visual field
After treatment, VF examination in RE showed an improvement in the inferior altitudinal defect and an increase in sensitivity, particularly in the central 10-degree area of the inferonasal sector. MD improved from −11.98 dB at baseline to −10.76 dB after 30 days of treatment, and further to −8.95 dB after 60 days. PSD in RE also decreased from 12.89 dB at baseline to 12.38 dB after 30 days and to 9.24 dB after 60 days. Similarly, the VFI in RE showed a progressive increase from 69% at baseline to 72% after 30 days and to 81% after 60 days. (Figure 3)
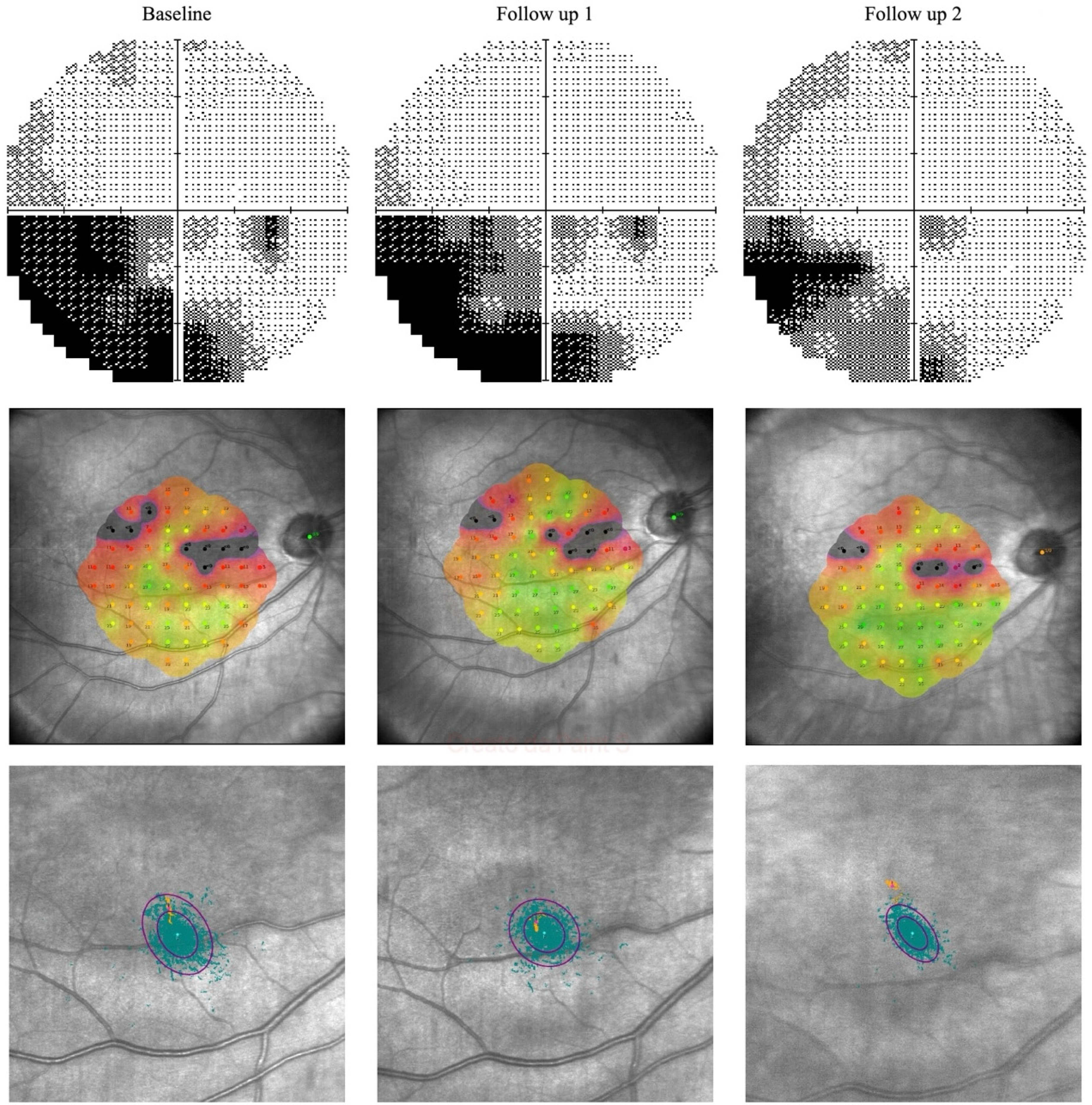
Composite image illustrating the functional changes observed in the right eye at baseline, after 30 days, and after 60 days of transpalpebral ES. The first row shows the visual field progression, with a gradual reduction in scotoma density and increased sensitivity, particularly in the central and inferonasal regions. The second row displays microperimetry maps, which reveal a progressive enhancement in macular sensitivity. The third show increased fixation stability and a progressive shift of the PRL toward the fovea.
In LE, VF examination showed an improvement in sensitivity in the inferonasal sector, particularly within the central 10-degree area. MD improved from −10.11 dB at baseline to −7.80 dB after 30 days of treatment. At the follow up visit after 60 days of treatment, however, it decreased to −8.38 dB, still showing an improvement compared to baseline. The PSD in LE decreased from 10.69 dB at baseline to 9.16 dB after 30 days and to 9.10 dB after 60 days. Additionally, the VFI in LE showed a progressive increase from 76% at baseline to 77% after 30 days and to 81% after 60 days. (Figure 4)
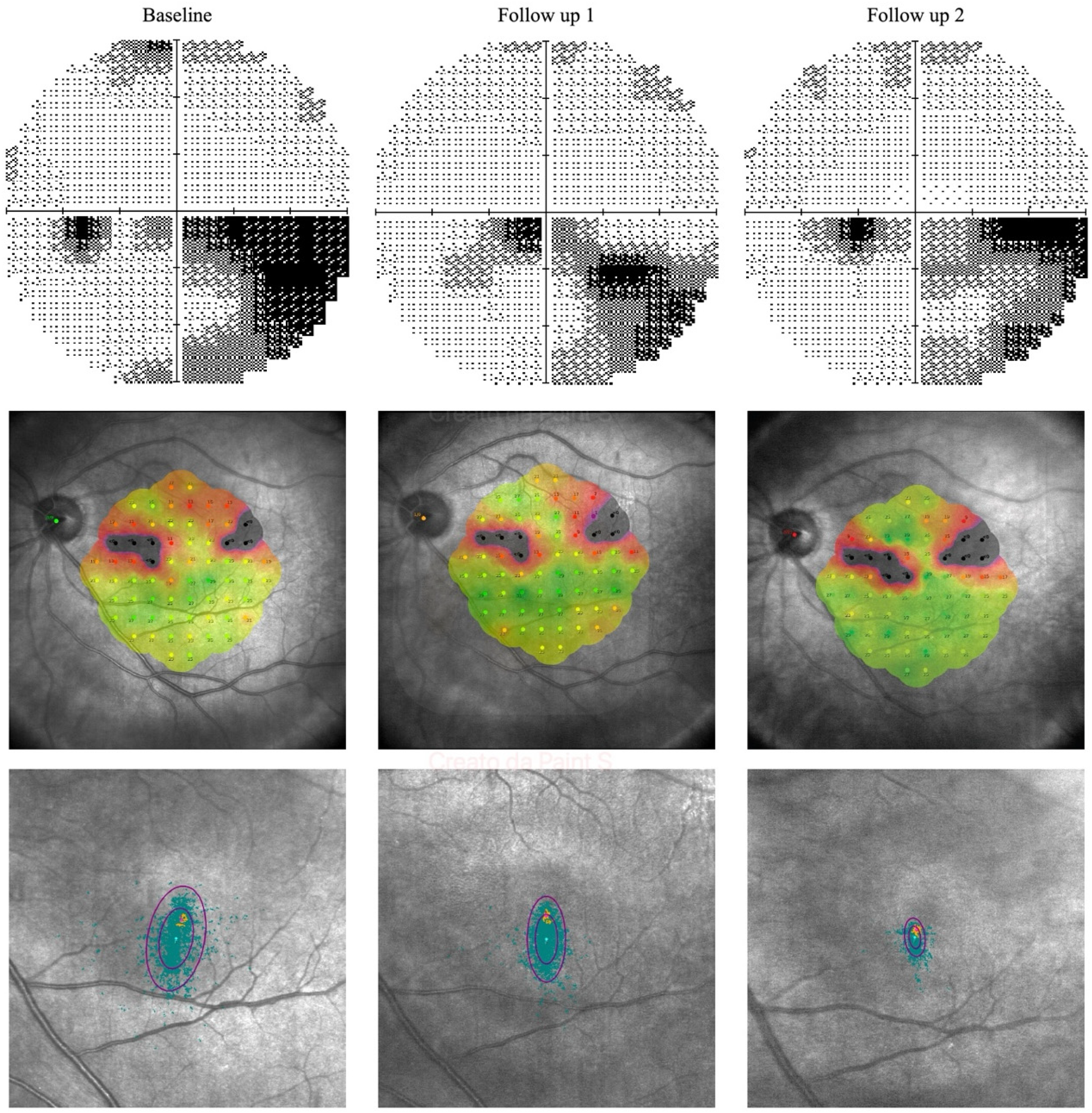
Composite image illustrating the functional changes observed in the left eye at baseline, after 30 days, and after 60 days of transpalpebral ES. The first row shows visual field progression, with a reduction in scotoma density and increased sensitivity compared to baseline. The second row presents microperimetry maps, highlighting a progressive improvement in macular sensitivity. The third row shows increased fixation stability and a gradual shift of the PRL toward the fovea.
Microperimetry
After 30 days of treatment, microperimetry indicated an enhancement in average retinal sensitivity. In RE, average sensitivity increased to 18.40 dB, and in LE, it increased to 20.10 dB.
Following 60 days of treatment, further improvements were noted, with the average sensitivity in RE rising to 19.70 dB and in LE to 20.40 dB.
The sensitivity maps revealed an increase in sensitivity across most tested points, demonstrating a more uniform sensitivity distribution across the grid (Figures 3 and 4).
Fixation stability also improved: in RE, P1 increased to 61% and P2 to 96% after 30 days of treatment and further improved to P1 78% and P2 99% after 60 days; in LE, P1 increased to 65% and P2 to 97% at the first follow-up, with further increases to P1 96% and P2 100% after 60 days of treatment. In both eyes, fixation was classified as stable after 60 days of treatment.
The analysis of the fixation point cloud and the final PRL position showed a progressive shift of the PRL closer to the foveal region in both eyes (Figures 3 and 4).
Discussion
Historically, the central nervous system's limited regenerative capacity has led to the belief that retinal, optic nerve, or cerebral visual injuries result in irreversible visual impairment with no potential for recovery. However, a period of spontaneous visual improvement, typically lasting a few weeks to months and characterized by significant neuroplastic changes, has been documented.16,17 In cases of NAION, the Ischemic Optic Neuropathy Decompression Trial reported that approximately 30% of patients experience spontaneous improvement in visual acuity within three months of onset, with no further significant gains observed during the following 24 months post-onset. 18 Consistent with these findings, our patient exhibited a modest improvement in visual acuity three months after the acute phase in each eye.
In recent years, several studies have demonstrated that patients can achieve improvements in visual function even well beyond the early spontaneous recovery phase.19,20 Lesions to visual structures often do not result in complete damage; instead, certain structures are typically spared or only partially impaired. According to Sabel et al., residual structures can be reactivated or restored through repetitive activation and stimulation, a concept central to their residual vision activation theory. This repetitive activation can be achieved through various methods including visual training or non-invasive ES. 21
ES of the eye represents a therapeutic approach in various clinical applications ranging from retinal dystrophies, age-related macular degeneration, retinal artery occlusion and optic neuropathy. To date, it is understood that the mechanism of action of ES is multifactorial. Several studies have demonstrated its capacity to modulate neuronal excitability, activate neuroprotective pathways, enhance axonal regeneration, increase blood flow, and reduce apoptosis in damaged retinal ganglion cells.22,23
Various noninvasive methods have been employed to deliver ES to patients with optic neuropathy, aiming to improve vision. In a randomized clinical trial involving 42 patients with long-term optic nerve damage, including 8 with NAION, Gall et al. reported improved detection ability and improvements in the vision-related quality of life in patients treated with rtACS. 13 In an observational study by Fedorov et al., a total of 446 patients with optic nerve lesions, including 63 with vascular lesions, underwent a ten days treatment with rtACS, which resulted in long-lasting improvements in VA and VF. Furthermore, a second 10-day course conducted in a subset of patients led to additional improvements. 24 Fujikado et al., in a small clinical study, reported increased visual acuity in patients with NAION and traumatic optic neuropathy following treatment with transcorneal ES. 12
To the best of our knowledge, our case is the first reported case of transpalpebral ES being used for the treatment of NAION and, notably, it is also the first case where the patient administered the treatment independently at home.
After 30 days of transpalpebral ES, the patient showed significant improvements compared to baseline. In line with Fedorov et al., further enhancements were observed following an additional treatment period. Overall, BCVA increased by 11 ETDRS letters in RE and 18 letters in LE. Baseline VF testing showed an inferonasal defect in both eyes, one of the most common patterns in NAION. 25 Bilateral peripheral visual field loss, particularly in the inferior region, can severely impair mobility and increase the risk of stumbling, as also reported by our patient. As evidence of improvements in VF parameters, the patient also experienced increased confidence and independence in mobility, with no further incidents of falling. Microperimetry revealed enhanced macular sensitivity and improved fixation stability, with the PRL progressively shifting closer to the fovea throughout the course of treatment. This appears to be consistent with the patient's reported increase in near reading fluency. Microperimetry in NAION patients reveals reduced macular sensitivity at a very early stage, which is significantly correlated with VA and VF measurements. Therefore, microperimetry can be effectively utilized for therapeutic evaluation and follow-up observation in NAION patients. 26 Currently, there are no established correlations between changes in microperimetry and ES treatment in patients with optic neuropathy. However, in patients with dry age-related macular degeneration, transpalpebral ES has been shown to increase macular sensitivity compared to controls group. 27
This case report supports the potential for meaningful visual improvements even in the chronic phase of optic neuropathy, suggesting that transpalpebral ES may represent a promising non-invasive therapeutic approach. However, this observation is limited by the single-patient design, the relatively short follow-up period, and the absence of electrophysiological assessments (e.g., pattern electroretinography), which would have provided further objective functional assessment.
In conclusion, while these preliminary findings are encouraging, definitive conclusions cannot be drawn from a single case. Randomized controlled studies in larger cohorts are warranted to evaluate the efficacy and long-term benefits of transpalpebral ES in NAION. Future research should also aim to define optimal stimulation protocols and incorporate detailed functional and electrophysiological testing to validate and better understand the mechanisms underlying transpalpebral ES-mediated visual recovery.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study protocol was approved by the local Ethical Committee of ASST Santi Paolo e Carlo and adhered to the tenets of the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient consent
The patient gave their written informed consent to use his anonymized clinical data for publication.
