Abstract
Purpose
Age-related macular degeneration (AMD) is a leading cause of irreversible vision loss, particularly neovascular AMD (nAMD). This study aimed to investigate the real-world treatment patterns, effectiveness, and safety of intravitreal anti-vascular endothelial growth factor (anti-VEGF) therapies for anti-VEGF naïve nAMD patients in Italy.
Methods
RADIANCE is a retrospective, observational, multicenter cohort study conducted at 13 clinical sites across Italy. The study enrolled all consecutive patients with nAMD, naïve to any intraocular anti-VEGF treatment and who initiated therapy with aflibercept, ranibizumab, or bevacizumab between January 2017 and November 2018. The primary objective of this study was to evaluate changes in visual acuity (VA) 52 weeks after initiating treatment with anti-VEGF.
Results
A total of 405 patients were enrolled; of these, 263 patients had at least two VA measurements and were included in the completer analysis (CA) set. At 52 weeks, the median VA change from baseline in the CA set was +1 letter, with 41.1% showing ≥ 5-letter improvement. Stratified by anti-VEGF agent, no statistically significant differences were observed. Overall, patients received a median of 5.0 (25th–75th percentile 3–6) injections of the initial anti-VEGF agent during the first year. Patients receiving ≥ 6 injections in the first year showed better VA outcomes. Undertreatment was evident at decreasing injection frequency over time.
Conclusion
The results of the RADIANCE study suggest an overall moderate effectiveness after 1 year of treatment with anti-VEGF in naïve patients with nAMD in Italy. Real-world outcomes demonstrated suboptimal treatment with no significant differences among anti-VEGF agents.
Keywords
Introduction
Age-related macular degeneration (AMD) is a chronic eye disease characterized by the presence of progressive, degenerative abnormalities in the central retina and is one of the leading causes of visual impairment and irreversible vision loss.1,2
Neovascular AMD (nAMD) is an advanced form of AMD characterized by neovascularization, mainly caused by an abnormally high expression of vascular endothelial growth factor (VEGF),3–8 which leads to progressive, severe, and irreversible retinal damage if untreated.9–11 Anti-VEGF agents have proven to be efficacious and safe options for the management of patients with nAMD12–15 and have changed the treatment paradigm of the disease. 16 Although there is no causative cure, timely treatment with anti-VEGF agents can help achieve treatment goals by removing exudative fluid from the retina and suppressing the formation of leaking new blood vessels, thus improving or maintaining visual acuity (VA) over time. 7
Anti-VEGFs administration frequency after the three-monthly loading doses is not yet standardized and depends on physician practice and individual cases. 17 The most common approaches are the reactive Pro-Re-Nata (PRN) method and proactive Treat and Extend (T&E) and fixed bimonthly regimens. In the PRN method the loading phase is followed by flexible dosing based on monthly monitoring of VA and/or macular morphology.18,19 In clinical practice, the reactive nature of this approach does not allow for optimal outcomes. 20 The risk of undertreatment is high because of the difficulty of maintaining strict compliance with regular, uninterrupted, and frequent follow-ups. Proactive protocols attempt to determine and anticipate the time to intraocular fluid recurrence. 20 Fixed bimonthly dosing requires continuous bimonthly injections for at least 1 year.12–14 In clinical practice it has shown improved outcomes compared with PRN regimen 21 ; however, it may still be associated with a considerable treatment burden. 16 In the proactive T&E regimen, anti-VEGF is administered at every scheduled visit. After the loading phase, the treatment interval can be gradually extended or reduced according to the anatomical and VA status to determine the maximum time between injections without disease recurrence.7,16,21,22 This approach allows the extension of treatment intervals while reducing the overall number of visits and appears to be associated with better VA outcomes.23–25
The
Material
RADIANCE is a retrospective, observational, multicenter cohort study conducted at 13 clinical sites across Italy (Supplementary Material 1). The study enrolled all consecutive subjects with a diagnosis of nAMD, naïve to any intraocular anti-VEGF treatment, aged 50 + who initiated anti-VEGF therapy between January 2017 and November 2018 (index period). The study evaluated the three anti-VEGF agents available in Italy during the index period: aflibercept, a recombinant decoy-receptor type of inhibitor of VEGF, and placental growth factor, 26 ranibizumab, a recombinant humanized monoclonal antibody fragment against VEGF-A, 27 and bevacizumab, a humanized monoclonal antibody fragment against VEGF-A. 28 Both ranibizumab and aflibercept have received marketing authorization from the European and Italian regulatory authorities for the treatment of nAMD, whereas bevacizumab in Italy can be administered on an off-label regimen. 29
The study included living patients who signed the informed consent form (ICF) and deceased and lost to follow-up patients, whose data were collected according to local regulations on data privacy. 30
Data were retrieved from hospital charts/patient's clinical records and collected from the index date (date of the first anti-VEGF injection) up to the last available visit before ICF collection during the observational period (January 2017–December 2022).
The study was notified to the Ethics Committee of each participating institution and was conducted in accordance with the national legislation on observational studies, 31 the principles of Good Practice,32,33 and the tenets of the Declaration of Helsinki.
The primary objective of the study was to evaluate the change in VA 52 weeks after starting treatment with aflibercept, ranibizumab, or bevacizumab. VA assessment was not standardized among all participating centres; therefore, all data were converted to the approximate ETDRS letter score (Supplementary material 2). The key secondary objectives reported in this paper were to 1) describe the treatment patterns, 2) evaluate the relationship between clinical effectiveness and the number of injections per year, and 3) describe the safety and tolerability of anti-VEGFs.
Statistical analysis
The study was planned to enrol 400 patients, and the following analysis sets were considered: safety analysis (SA, defined as all enrolled patients who received at least one dose of intravitreal anti-VEGF treatment), effectiveness analysis (EA, including all patients in SA set with at least two VA measurements after the one at baseline), and completer analysis (CA, including all patients in EA set who had a VA measurement at week 52 ± 10).
VA improvements were computed as the absolute difference between a post-baseline VA measurement and that at baseline. VA improvement was defined using the measurement closest to 52 weeks during the time window 42–62 weeks after the index date, if available; otherwise, VA improvement was considered missing for the analysis in the CA set and was imputed following the Last Observation Carried Forward method for the analysis in the EA set. For patients who discontinued the initial treatment because of adverse events (AE) or lack of efficacy, all observed values were used regardless of the occurrence of the intercurrent event. Patients who switched to another anti-VEGF before week 52 were allocated to the treatment they received for the longest period, and all observations were used in the analysis regardless of the occurrence of the intercurrent event.
More details on the statistical methods are reported in Supplementary Material 3.
Results
Patient disposition and baseline characteristics
A total of 405 patients were enrolled: 387 (95.6%) were eligible for the SA set, 344 (84.9%) for the EA set, and 263 (64.9%) for the CA set (Figure 1).
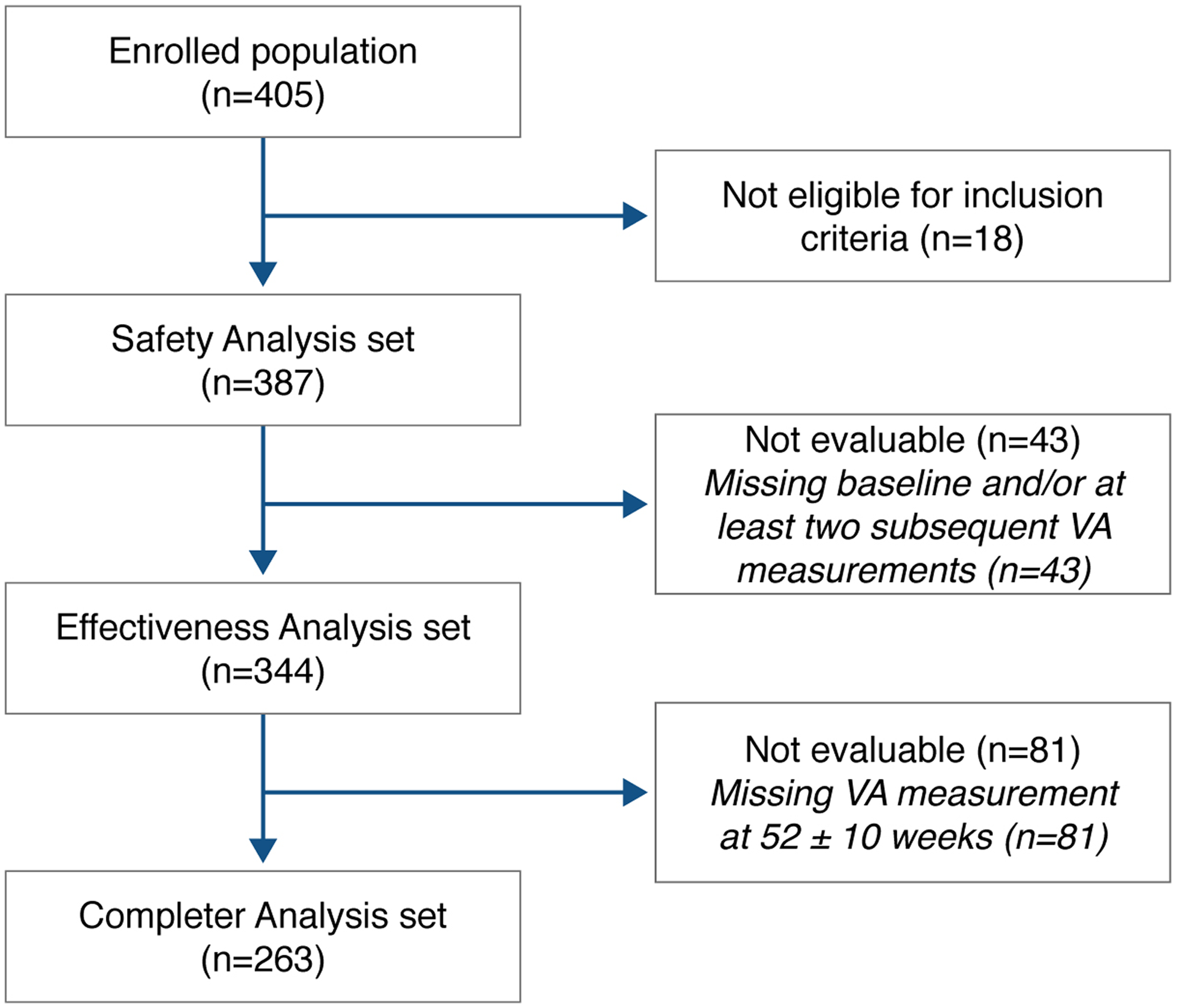
Patient disposition. Note: Safety analysis (SA) set = all enrolled patients who received at least one dose of intravitreal anti-VEGF treatment, Effectiveness analysis (EA) set = all patients in the SA set with at least two VA measurements after the baseline one; Completer analysis (CA) set = all patients in the EA set who had a VA measurement at week 52 ± 10) Abbreviations: VS: visual acuity.
Among the EA set, 150 (43.6%) patients received ranibizumab as the first anti-VEGF treatment, 131 (38.1%) received aflibercept, and 63 (18.3%) received bevacizumab. Sociodemographic and clinical characteristics at the index date of patients in the EA set are reported in Table 1.
Sociodemographic and clinical characteristics overall and stratified by initial anti-VEGF treatment at baseline (EA set).
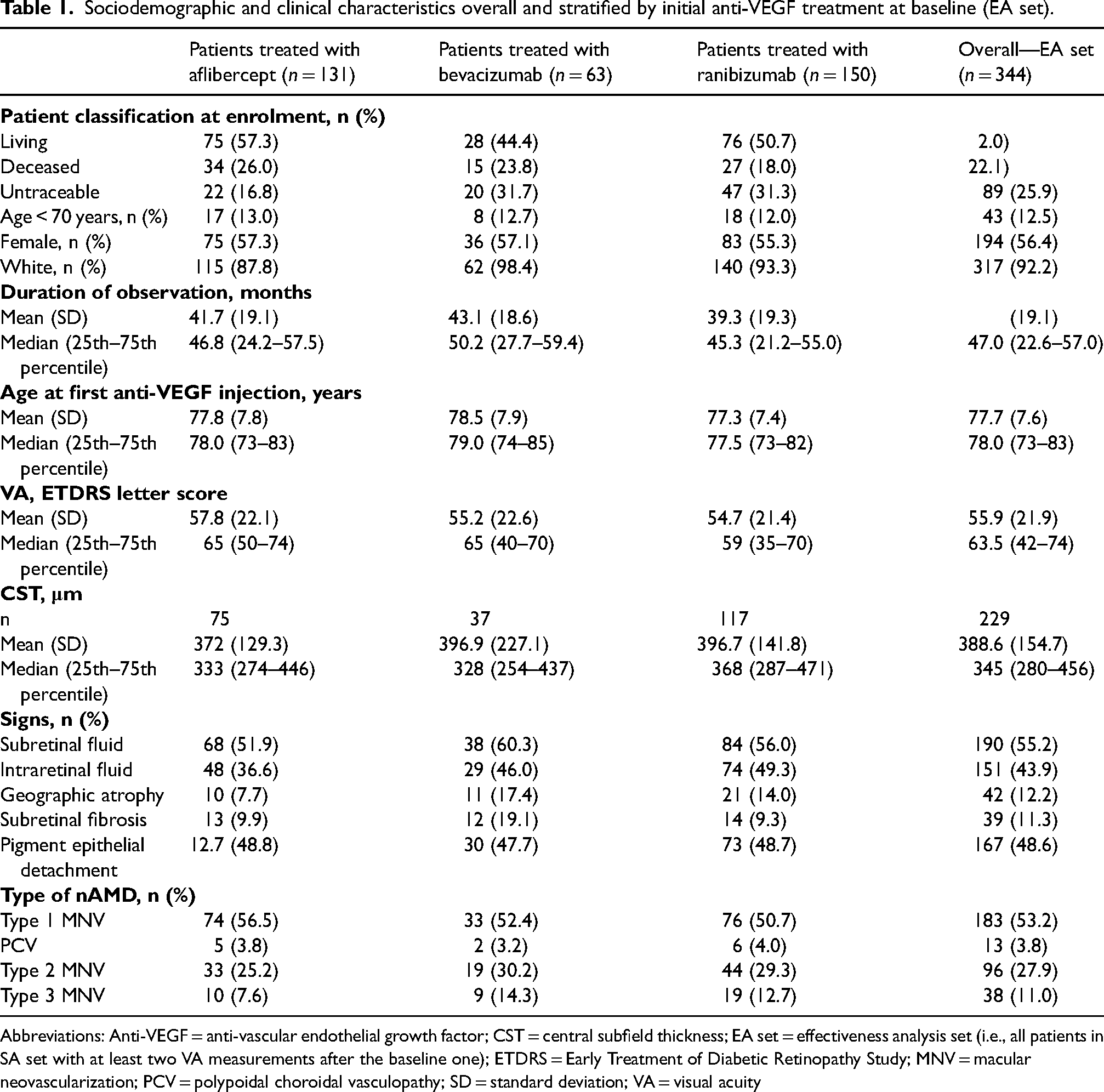
Abbreviations: Anti-VEGF = anti-vascular endothelial growth factor; CST = central subfield thickness; EA set = effectiveness analysis set (i.e., all patients in SA set with at least two VA measurements after the baseline one); ETDRS = Early Treatment of Diabetic Retinopathy Study; MNV = macular neovascularization; PCV = polypoidal choroidal vasculopathy; SD = standard deviation; VA = visual acuity
Ocular comorbidities were reported in 119 patients (34.6%), the most prevalent being cataract (49.6%) and glaucoma (26.1%). Only 5 patients (1.5%) had previously received treatment for macular degeneration other than anti-VEGF in the affected eye and 218 patients (35%) had previously undergone ocular surgery.
The median baseline VA was 63.5 letters (25th–75th percentile: 42–74). Central subfield thickness (CST) was reported in 229 patients, and the median value at baseline was 345 μm (25th–75th percentile: 280–456). Similar median baseline VA and CST were observed between the treatment groups.
Primary objective
At baseline, the median VA in the CA set was 65 letters (25th–75th percentile: 5–74). Overall, after 52 weeks, the patients showed a median change of 1 letter (25th–75th percentile: −5–+10); the difference among treatments was not statistically significant (p = 0.2388) (Table 2).
Primary objective: VA changes at 52 weeks overall and stratified by the number of injections

Abbreviations: CA set = completer analysis set (i.e., all patients in EA set who had a VA measurement at week 52 ± 10); ETDRS = Early Treatment of Diabetic Retinopathy Study; SD = standard deviation; VA = visual acuity
Only 109 (41.4%) patients in the CA set had a VA improvement ≥ 5 letters between baseline and week 52 (Table 2). By stratifying VA changes by the number of injections per year, patients treated with > 6 injections in the first year of treatment showed a median VA improvement of 3 letters (25th–75th percentile: −1–+11) while no evident improvement was observed in patients treated with a lower number of injections. Among patients treated with > 6 injections, those treated with bevacizumab showed no change in median VA change (0 letters; 25th–75th percentile: 0–15) as compared to ranibizumab (2.0 letters; 25th–75th percentile: −5–+11) and aflibercept (5 letters; 25th–75th percentile: 0–11) (Table 2).
A linear mixed model for repeated measurements with VA changes from baseline until week 52 and after was also evaluated according to VA at baseline (< 58 and ≥ 58 letters). Anti-VEGF treatment showed a positive effect on VA at 42 months for patients with a VA at baseline < 58 (overall 0.96 [SE:3.08]). In contrast, patients with VA at baseline ≥ 58 showed a greater VA deterioration at 42 months.
Secondary objectives
Overall, in the EA set, 293 (85.2%) patients underwent unilateral treatment. Most patients (73.5%) began anti-VEGF treatment with three loading doses, after which, as the study investigators self-declared, 161 (46.8%) patients were treated with a PRN approach, 90 (26.2%) with a T&E approach, 75 (21.8%) used a fixed dosage, and 18 (5.2%) patients had other regimens(16 followed a proactive regimen and 2 discontinued the regimen before completing the loading phase) (Figure 2). Proactive and reactive regimens were equally distributed in the EA set (52.7% vs 46.8%). Proactive regimens were most commonly used with aflibercept (60%), whereas reactive regimens were most commonly used with bevacizumab (64%).
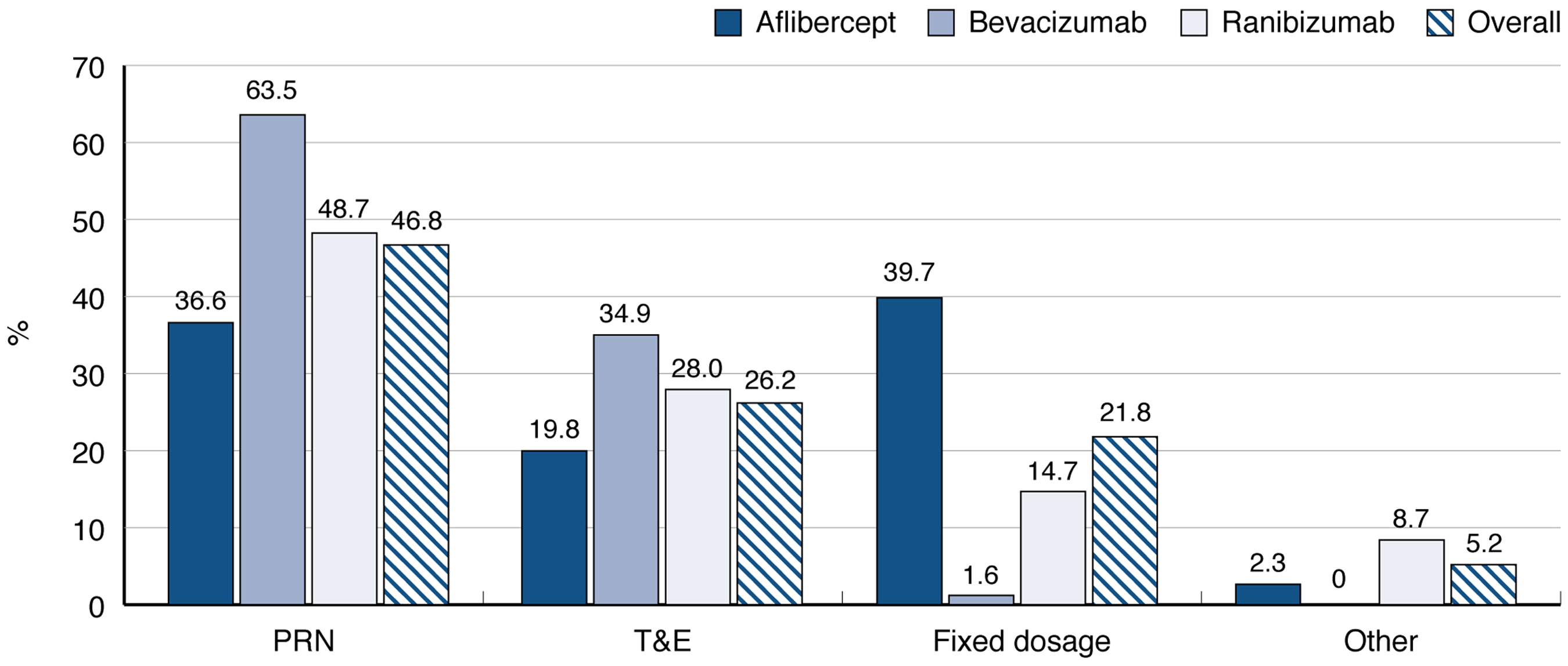
Prescribed regimen after loading. Note: Percentage was computed from the total number of patients treated with a specific initial anti-VEGF. Abbreviations: PRN: Pro-Re-Nata; T&E: Treat and Extend.
Patients received a median of 5.0 (25th–75th percentile: 3–6) injections (mean = 5.1) of the initial anti-VEGF treatment during the first year (Table 3). This rate decreased in the following years, with a median of 3 (25th–75th percentile: 2–4), 2 (25th–75th percentile: 1–3), and3 (25th–75th percentile: 1–4) injections during the second, third, and fourth year, respectively. Moreover, the number of patients treated with the initial treatment decreased over time: patients who received ≥ 1 injection of the initial anti-VEGF treatment were 180 (52%), 95 (28%), and 51 (15%) during the second, third, and fourth year, respectively. Overall, 262 (76.2%) patients permanently discontinued initial anti-VEGF treatment, and the most frequent reasons were investigator decision (65.3%), lack of efficacy (17.2%), and persistent fluid (13.4%). The median duration of the initial anti-VEGF treatment was 14 months (25th–75th percentile: 7.6–27.2).
Treatment patterns in the EA set
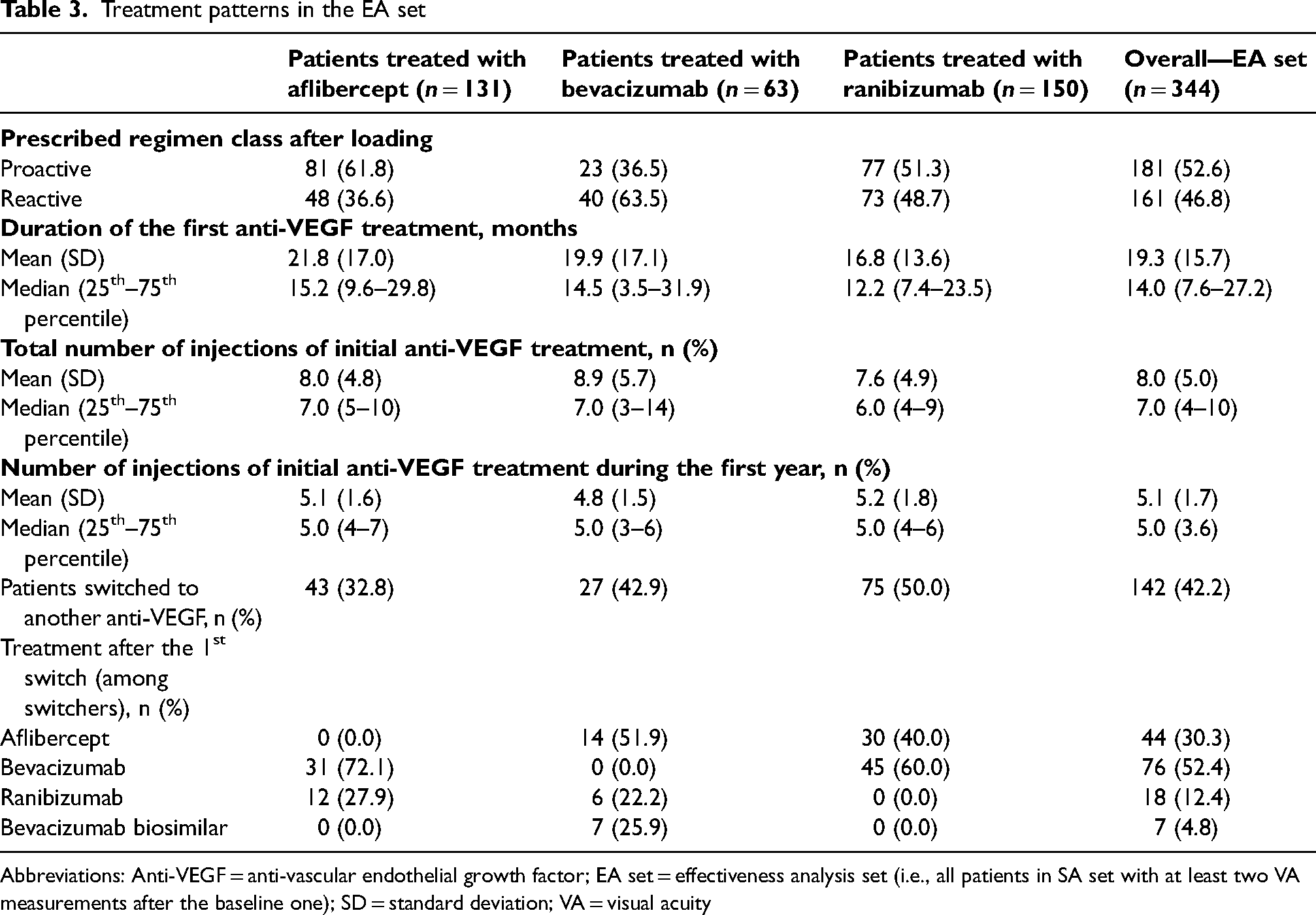
Abbreviations: Anti-VEGF = anti-vascular endothelial growth factor; EA set = effectiveness analysis set (i.e., all patients in SA set with at least two VA measurements after the baseline one); SD = standard deviation; VA = visual acuity
Among the 262 patients who permanently discontinued the first anti-VEGF, 145 switched to another anti-VEGF treatment (83 patients switched to bevacizumab or its biosimilar, 44 to aflibercept, and 18 to ranibizumab). Patients who switched from the initial treatment were 32.8%, 42.9%, and 50% of patients treated with aflibercept, bevacizumab, and ranibizumab, respectively (Table 3 and Figure 3).
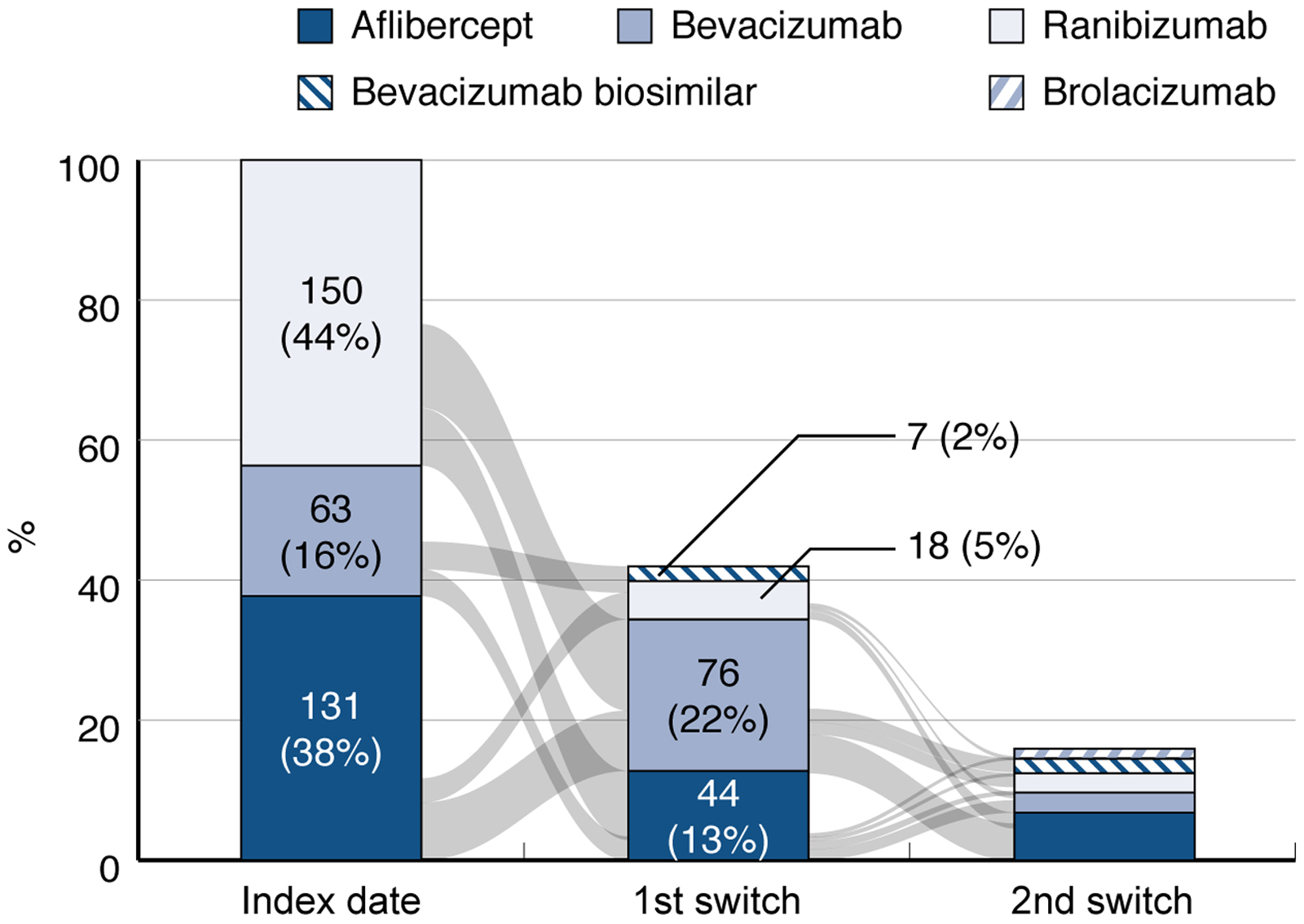
Treatment switch from the index date: Sankey diagram (EA set). Abbreviations: EA: effectiveness analysis set.
CST at baseline was reported in 229 patients included in the EA set; of these, 183 patients also had available information at 52 weeks. The reduction in the median value was −58 μm (25th–75th percentile: −161– −15). No statistically significant difference was observed among the treatment subgroups.
Safety
A total of 110 AE occurred in 81 of 387 (20.9%) patients included in the SA set. Only 1 (0.3%) patient had a serious AE.
Discussion
RADIANCE is a retrospective, observational cohort study conducted on data from 13 Italian centres with the primary aim of describing the real-world treatment patterns of anti-VEGF naïve patients with nAMD treated with intravitreal anti-VEGFs and the associated effectiveness. The study included patients who received the first anti-VEGF injection (namely aflibercept, ranibizumab, and bevacizumab) during the index period (January 2017–November 2018). The characteristics of patients enrolled were in line with the literature17,34,35 and, overall, the study population can be considered representative of the Italian real-world population.
In the subgroup of patients with a VA measurement at week 52 ± 10 (CA set), the overall median and mean VA change at week 52 were +1 letter. Only 109 (41.1%) patients showed a VA improvement ≥ 5 letters, suggesting moderate effectiveness of the anti-VEGF treatments. When stratifying median VA change by anti-VEGF treatment, bevacizumab and ranibizumab showed no gain, whereas aflibercept led to a moderate gain (+3 letters). The difference among treatments was not statistically significant. The overall VA change observed in RADIANCE is lower than that observed in the Italian observational study EAGLE, in which after 1 year of treatment and a mean of 5.6 injections of anti-VEGF, the mean VA increased by 2.45 letters in naïve patients with nAMD. 35 This difference might be due to the inclusion of bevacizumab in the RADIANCE study while it was not included in the EAGLE study. Results in RADIANCE were similar to those observed in a US real-world study that showed a mean gain of 0.95 letters after 1 year of treatment with an anti-VEGF agent (mean injections: 7.3). Results obtained with ranibizumab in RADIANCE are in line with the findings of the multi-country real-world study AURA, which for Italy reported no change in VA to year 1 and a mean of 5.2 injections in 2 years, 36 but differ from those observed in two international real-world studies that showed a mean VA increase of 2.4 and 3.2 letters after a mean of 5 and 5.1 injections, respectively.36,37
More favourable visual outcomes have been reported in clinical trials. Treatment with ranibizumab was associated with a mean VA increase at 1 year that ranged from 11.3 to 7.2 when administered monthly14,38–41 and from 6.8 to 8.2 when administered under a PRN regimen39,41; aflibercept showed a mean VA improvement of 10.9 and 7.6 letters and 8.9 and 7.9 letters when administered monthly and every 2 months, respectively. 13 Finally, the VA gain associated with monthly bevacizumab was 8 letters, while the PRN regimen was associated with +5.9 letters. 41
Nevertheless, although the frequency of anti-VEGF administrations has increased in recent years, in the real-world setting, the mean number of injections was still lower than in randomized clinical trials14,17,38–43 and remains below the pragmatic threshold for appropriate treatment of ≥ 7 injections. 44 Recently, Lanzetta 45 has shown that the frequency of treatment with anti-VEGF in Italy is significantly low and not associated with an improvement in the control of the disease as reported in other countries. This may not be related to the frequency of treatment alone and may be due to organizational and regulatory issues.
Furthermore, in real life, there is still extensive use of the PRN regimen that is associated with non-optimal outcomes due to delays in detecting and treating disease activity.20,46–48 Despite some evidence supporting the association between the T&E regimen and better visual outcomes,24,25 this approach is still not used much, as also observed in RADIANCE, where the most used regimen was PRN (47%) and only 26% of patients were treated with T&E. The recent study of Grassi et al. 43 provides additional real-world evidence from the Bari Intravitreal Injection Registry on the effectiveness of anti-VEGF treatments in nAMD. The study showed that the implementation of a Lean methodology, an approach that minimizes the waste of time and space, and enhances the centre's capacity management, significantly reduced patient wait times and improved treatment adherence within their clinical setting, potentially contributing to enhanced visual outcomes. In fact, over a one-year follow-up the mean number of injections was similar to that observed in the RADIANCE study (6.1), but the mean BCVA change was greater (+6.3 letters), suggesting a role for organizational aspects in patient outcomes.
The correlation between the number of injections and better VA outcomes has been observed in previous real-world studies35–37,48 and was confirmed in the RADIANCE study in which the slight initial VA improvement decreased after the first year as the frequency of injections declined (5.1, 3, and 2.5 injections during the first, second and third years, respectively).35,36,42 The stratification of VA changes based on the number of injections in the first year of treatment revealed a VA improvement of up to 3 letters for patients treated with ≥ 6 injections, whereas no improvement was observed in patients who received 3–5 or 0–2 injections. Aflibercept showed the highest percentage of patients (48.5%) with VA change of at least 5 letters at 52 weeks and the highest percentage of patients (52.5%) treated with ≥ 6 injections, probably because aflibercept was mainly administered following a proactive regimen (i.e., T&E and fixed dosage). On the other hand, when 6 or more injections of bevacizumab or ranibizumab were administered in the first year, the median VA change was 0 and 2 letters, respectively. This may be due to the specific characteristics of the different anti-VEGF agents. Specifically, as shown by the CATT study, bevacizumab should be administered monthly to induce a 1-year change in VA similar to that induced by ranibizumab. 41
The undertreatment observed in the RADIANCE study may jeopardize the overall effectiveness of the treatment and could influence the study's ability to reveal a difference between the anti-VEGF agents. During the study, the number of patients treated with the first anti-VEGF treatment decreased during the time of observation, with a drop of approximately 50% in the second year and a gradual reduction in the following years. In the RADIANCE study, 145 patients (42.2%) switched to another anti-VEGF treatment. Among these, most patients treated with aflibercept and ranibizumab switched to bevacizumab. Because of the lack of VA improvement observed in bevacizumab-treated patients, it is possible that these switches were not strictly driven by efficacy.
This study was subject to the limitations inherent to medical chart reviews, where data were gathered from hospital charts and clinical records. As in all retrospective studies, the risk of having a non-negligible proportion of missing data was taken into account and monitoring visits were performed to minimize this data loss. The retrospective observation window was limited to years 2017–2022, to both limit the risk of informative gaps and avoid generating real-world data not applicable to the current patterns of care. Despite the monitoring and data-cleaning activities, CST measurement was not available in standard clinical practice for one-third of the patients. The RADIANCE study protocol included the presence of a “centre effect” on VA improvements. However, because of the small sample size observed at the centre level and the unequal distribution of centres at the regional and Italian levels, it was decided not to include it in the primary analyses but to conduct a sensitivity analysis of the centres to understand the differences between them. The study showed considerable inter-centre variations in treatment strategies and distribution. This heterogeneity in treatment management may have contributed to the great variability observed in VA improvement.
In conclusion, the RADIANCE study shows that in the real-world setting, there is a suboptimal treatment of naïve Italian patients with nAMD treated with the available anti-VEGF agents, which does not allow for an optimal clinical outcome in terms of VA and may result in worsening long-term visual outcomes. 7 The wide differences in treatment management may have contributed to the significant variability observed in VA changes, with patients who received ≥ 6 injections in the first year showing greater improvements at 52 weeks compared with those receiving fewer injections. These findings highlight the limitations of the current therapeutic alternatives that are effective for the treatment of nAMD, but they might not be optimal because of the high frequency of injections. Some patients are indeed incomplete responders, whereas others might be good responders when anti-VEGF agents are frequently injected leading to a heavy treatment burden for patients, caregivers, and clinicians. In this context, there is a substantial unmet need for more durable treatment options that can reduce the overall treatment burden while maintaining optimal efficacy.
Supplemental Material
sj-docx-1-ejo-10.1177_11206721241310628 - Supplemental material for Real-world evidence of anti-VEGF therapies in patients with neovascular age-related macular degeneration in Italy: The RADIANCE study
Supplemental material, sj-docx-1-ejo-10.1177_11206721241310628 for Real-world evidence of anti-VEGF therapies in patients with neovascular age-related macular degeneration in Italy: The RADIANCE study by Mariacristina Parravano, Francesco Viola, Massimo Nicolò, Stela Vujosevic, Laura Bianchino, Emilia Sicari, Giulia Villa and Paolo Lanzetta in European Journal of Ophthalmology
Supplemental Material
sj-docx-2-ejo-10.1177_11206721241310628 - Supplemental material for Real-world evidence of anti-VEGF therapies in patients with neovascular age-related macular degeneration in Italy: The RADIANCE study
Supplemental material, sj-docx-2-ejo-10.1177_11206721241310628 for Real-world evidence of anti-VEGF therapies in patients with neovascular age-related macular degeneration in Italy: The RADIANCE study by Mariacristina Parravano, Francesco Viola, Massimo Nicolò, Stela Vujosevic, Laura Bianchino, Emilia Sicari, Giulia Villa and Paolo Lanzetta in European Journal of Ophthalmology
Supplemental Material
sj-docx-3-ejo-10.1177_11206721241310628 - Supplemental material for Real-world evidence of anti-VEGF therapies in patients with neovascular age-related macular degeneration in Italy: The RADIANCE study
Supplemental material, sj-docx-3-ejo-10.1177_11206721241310628 for Real-world evidence of anti-VEGF therapies in patients with neovascular age-related macular degeneration in Italy: The RADIANCE study by Mariacristina Parravano, Francesco Viola, Massimo Nicolò, Stela Vujosevic, Laura Bianchino, Emilia Sicari, Giulia Villa and Paolo Lanzetta in European Journal of Ophthalmology
Footnotes
Acknowledgements
Site monitoring, data management, and statistical analysis were performed by MediNeos (Modena, Italy). Writing assistance and journal styling services were provided by Ombretta Bandi from SEEd Medical Publishers and was founded by Roche S.p.A. The authors thank Prof. Giovanni Staurenghi for his contribution as a member of the steering committee of the RADIANCE study.
Author contributions
All authors contributed to conceptualization, writing—review and editing. L.B., E.S., and G.V. contributed to funding acquisition, conceptualization, project administration, resources, supervision, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.P. Abbvie, Novartis, Bayer, Roche, and Zeiss; F.V. Bayer, Novartis, Abbvie, Roche; M.N. Roche; S.V. Abbvie, Alimera, Apellis, Bayer, B&I, Novartis, Roche, and Zeiss; L.B. Roche (employee); E.S. Roche (employee); G.V. Roche (employee); P.L. Aerie, Allergan, Apellis, Bausch & Lomb, Bayer, Biogen, Boehringer Ingelheim, I-Care, Genentech, Novartis, OcularTherapeutix, Outlook Therapeutics, and Roche.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored and financially supported by Roche S.p.A. The research for this paper for the IRCCS-Fondazione Bietti was financially supported by the Italian Ministry of Health and Fondazione Roma, Italy; F. Hoffmann-La Roche.
Research ethics and patient consent
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Fondazione PTV - Policlinico Tor Vergata (protocol code N° 231/21, date of approval 24/11/2021). All living patients signed an informed consent form. Data of deceased and lost to follow-up patients were collected according to local regulations on data privacy.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
