Abstract
Purpose
To examine the radiological differences between bacterial orbital cellulitis (OC) and diffuse non-specific orbital inflammation (DNSOI) on magnetic resonance imaging (MRI).
Methods
Retrospective study of patients with OC and DNSOI with an MRI orbital scan. Localised orbital inflammation (e.g., idiopathic dacryoadenitis and myositis), quiescent orbital inflammation and pre-septal cellulitis were excluded.
Results
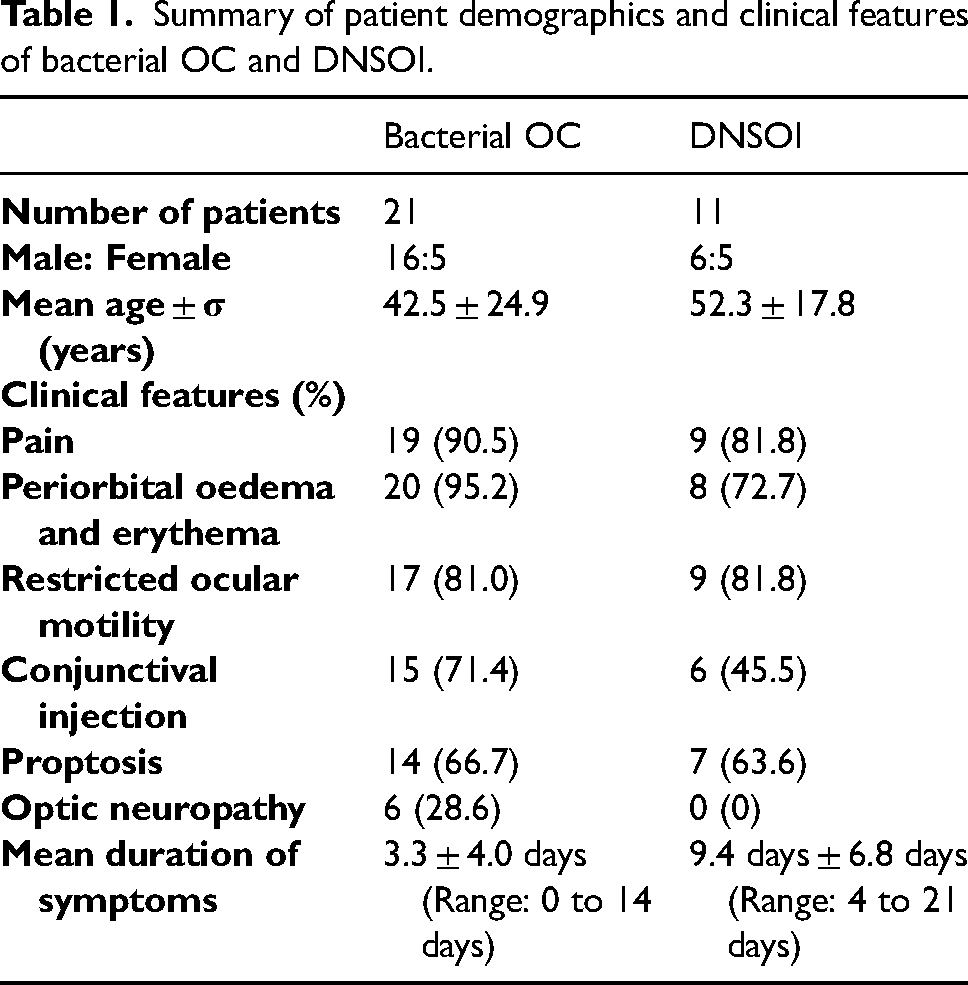
Thirty-two patients presenting between 2008 and 2023, including twenty-one OC patients (mean age: 42.5 ± 24.9 years old, male: 6), and eleven DNSOI patients (mean age: 52.3 ± 17.8 years old, male: 16). Both OC and DNSOI demonstrate orbital fat contrast-enhancement. However, whilst OC demonstrated a hyperintense T2 signal (P < 0.001), variable signal was observed in DNSOI, with a hypointense T2 signal more suggestive of DNSOI (P = 0.012). When the lacrimal glands were involved, indistinct margins were more likely in OC (P < 0.001), whilst gross enlargement and contrast-enhancement was observed in DNSOI (P = 0.032 and 0.017, respectively). Peripheral contrast-enhancement of the extraocular muscle (EOM) (P = 0.002) was more common in OC, whilst DNSOI demonstrated variable contrast-enhancement throughout the affected EOM (P < 0.001). The presence of contralateral abnormalities, such as lacrimal gland enlargement and EOM involvement, are more suggestive of DNSOI.
Conclusion
Several MRI features, beyond overt sinogenic disease, may help to differentiate OC from DNSOI, including the orbital fat signal intensity, EOM and/or lacrimal gland involvement, and contralateral orbital abnormalities. However, these features may not be specific, and thus highlights the ongoing radiological dilemma clinicians are faced when tasked with differentiating between infectious and non-infectious orbital inflammatory disease.
Keywords
Introduction
The differentiation between diffuse non-specific orbital inflammation (DNSOI) and bacterial orbital cellulitis (OC) in the acute setting may be difficult. Both conditions can present with acute-onset periorbital oedema and erythema, proptosis, restricted or painful extraocular movements and diplopia.1–3 Although systemic inflammatory markers may be useful tools in differentiating between these two entities, radiological evaluation with orbital computed tomography (CT) and magnetic resonance imaging (MRI) remains pivotal.4–7 DNSOI and OC are entities with different pathogenesis and expedient differentiation facilitates early appropriate management, with immunosuppressive corticosteroid therapy for DNSOI, and targeted antimicrobials and surgical intervention (if indicated) for OC. Additionally, the ability to provide a clinico-radiological diagnosis may help guide clinician decision making in proceeding to histopathological diagnosis of non-infective inflammatory orbital condition. DNSOI remains a diagnosis of exclusion, whereby evidence of an underlying specific inflammatory condition (e.g., IgG4-related orbital disease) is not demonstrated. CT is often the first-line imaging modality for orbital disease, however, MRI can further characterise orbital structures and pathology. 8
It has been well known that key radiological markers such as the presence of orbital collections or adjacent paranasal sinus disease favour a diagnosis of OC over DNSOI. However, in the absence of overt sinogenic disease or preceding trauma, it can still present as a diagnostic dilemma. Delays in diagnosis prevent initiation of appropriate treatment, and the course of management for DNSOI and OC are vastly different. The aim of this study was to evaluate and characterise additional radiological patterns on MRI which may help to differentiate between OC and DNSOI.
Methods
Subjects
This was a retrospective study of patients with a clinico-radiological diagnosis of bacterial OC and DNSOI who had MRI orbital scans. Exclusion criteria were patients with localised orbital inflammation (including isolated dacryoadenitis and myositis). OC was diagnosed by the clinico-radiological presentation, microbiological results, and clinical response to antimicrobial therapy and/or surgical intervention. Sinogenic forms of OC were not excluded from this study, as additional radiological features beyond sinusitis may help to differentiate between OC and DNSOI. Meanwhile, a diagnosis of DNSOI was supported by the clinico-radiological presentation, relevant histopathology (if conducted), and response to appropriate immunosuppressive therapy (I.e., systemic corticosteroid therapy). In all patients with DNSOI, there was exclusion of specific causes of OID via autoimmune serology, histopathological analysis (if conducted) and serial follow-up.
Patients were identified from a tertiary Oculoplastic Unit in South Australia. Data recorded included patient demographics (age at presentation, gender, relevant past medical history and medications), clinical presentation, laboratory investigations (white-cell count [WCC], C-reactive protein [CRP], relevant microbiological analysis), MRI features, course of management and clinical outcomes. All research was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board (CALHN HREC: 18536) with a waiver of consent obtained.
Image acquisition and analysis
MRI orbital scans in all patients were conducted on either 1.5 Tesla or 3.0 Tesla scanners, and sequences included a combination of axial and coronal T1- and T2-weighted imaging (T1 and T2), fat-supressed contrast-enhanced T1 (T1 FS CE) and fat-supressed T2 (T2 FS). MRI orbital scans were reviewed by an experienced oculoplastic surgeon (JT) and neuroradiologist (SP). Radiological features such as morphology (e.g., extent of involvement, distinct margins and gross enlargement), contrast-enhancement pattern, and signal abnormalities were analysed. Morphology of orbital structures were determined qualitatively, supported by indistinct margins, asymmetry to the contralateral side and/or gross enlargement. Additionally, signal abnormalities were described relative to normal extraocular muscle (EOM) (i.e., hyperintense, isointense or hypointense to EOM). 9
Statistical analysis
Where applicable, results are expressed as means ± standard deviation (σ) and presented in relevant tables. Statistical analysis was performed with SPSS (IBM corporation, New York). Where applicable, results are expressed as means ± standard deviation (σ) and presented in relevant tables. Differences in means were analysed by the Independent Sample's t-test with P < 0.05 deemed statistically significant. Fisher's exact test was used to analyse differences in radiological features between OC and DNSOI.
Results
Patient demographics and clinical features
This study included thirty-two patients presenting between 2008 and 2023, of which twenty-one had OC and eleven had DNSOI. Table 1 summarises the demographics and common clinical features of OC and DNSOI. Mean age was similar and there was no gender predilection between DNSOI and OC (P = 0.258 and 0.210, respectively). Six cases of DNSOI underwent a biopsy demonstrating non-specific inflammatory changes. Meanwhile, in the remaining five patients, a diagnosis of DNSOI was supported by clinic-radiological findings, exclusion of specific causes of OID (via autoimmune serology and radiology), response to steroids and/or unresponsive to antibiotics.
Summary of patient demographics and clinical features of bacterial OC and DNSOI.
Radiological features
Thirty-two MRI orbital scans were reviewed from thrity-two patients. Table 2 summarises the radiological features of all cases. Of the twenty-one cases of OC, sixteen (76.2%) had adjacent acute paranasal sinusitis, and nine (42.9%) had intracranial extension.
Comparison of orbital and extra-orbital radiological features of OC and DNSOI.
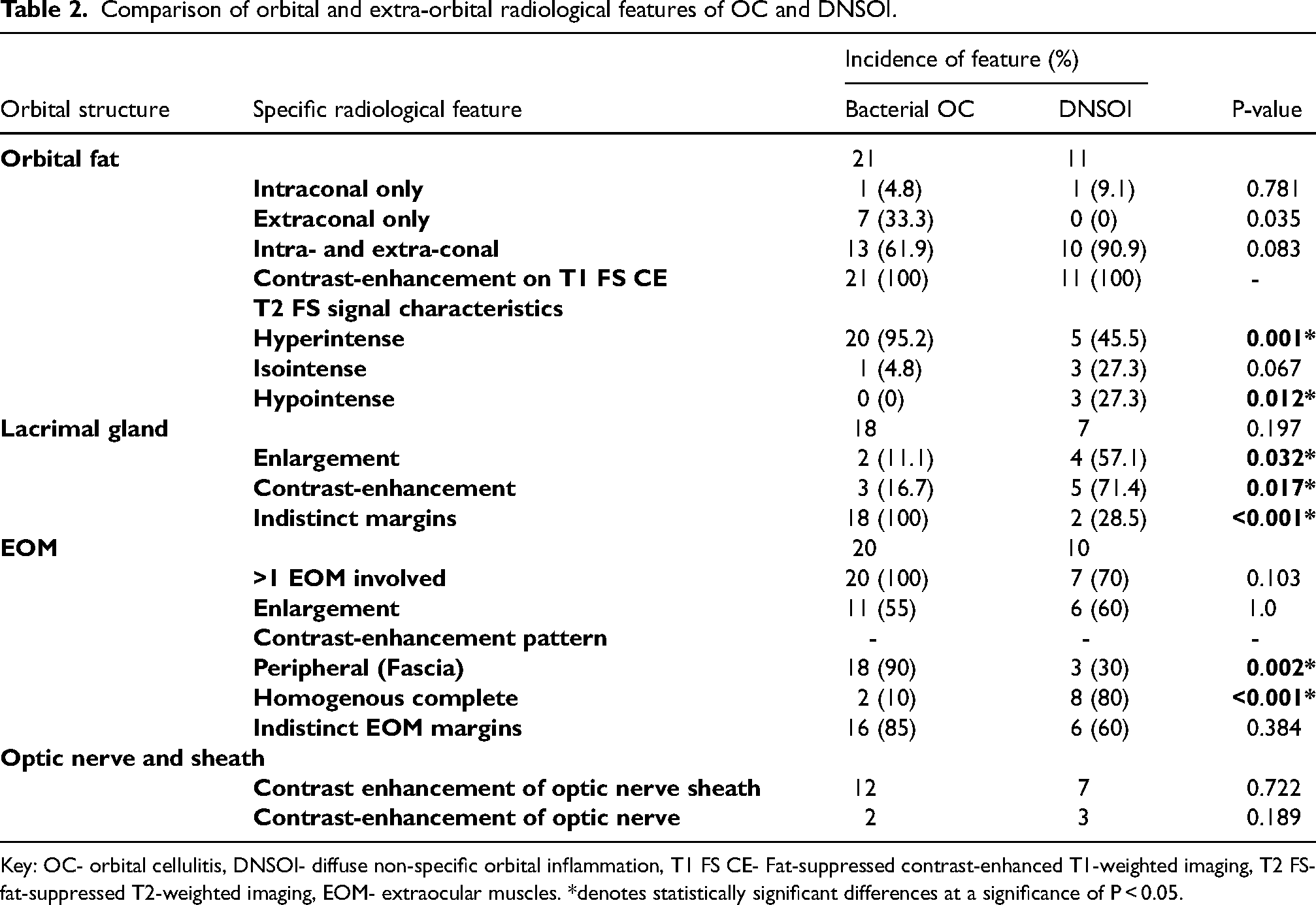
Key: OC- orbital cellulitis, DNSOI- diffuse non-specific orbital inflammation, T1 FS CE- Fat-suppressed contrast-enhanced T1-weighted imaging, T2 FS- fat-suppressed T2-weighted imaging, EOM- extraocular muscles. *denotes statistically significant differences at a significance of P < 0.05.
Orbital fat involvement in both OC and DNSOI demonstrated contrast-enhancement on T1 FS CE (Figures 1C-F and 2B). On T2 FS, DNSOI demonstrated variable T2 signal (ranging from hypointense to hyperintense signal), whilst OC had hyperintense T2 signal of involved structures (P = 0.001). A hypointense T2 signal was more suggestive of DNSOI (P = 0.012). Lacrimal gland involvement in OC was most commonly demonstrated by loss of distinct margins (18/18, 100%) (P < 0.001), whilst contrast-enhancement (5/7, 71.4%) and enlargement (4/7, 57.1%) were more commonly observed in DNSOI (Figure 1E) (P = 0.017 and 0.032, respectively).
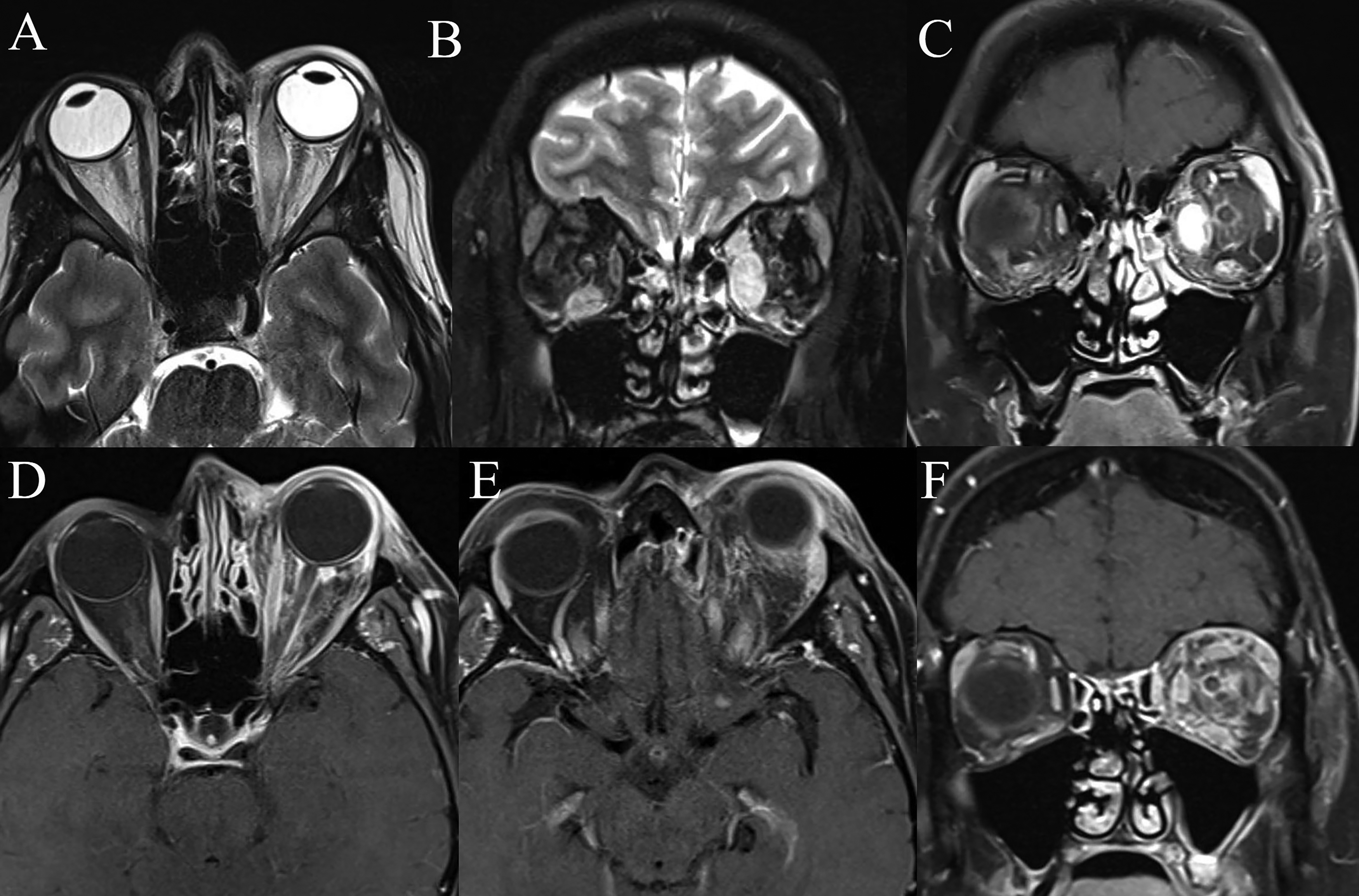
MRI orbital scans of DNSOI with predominant involvement of the medial rectus and lacrimal gland.
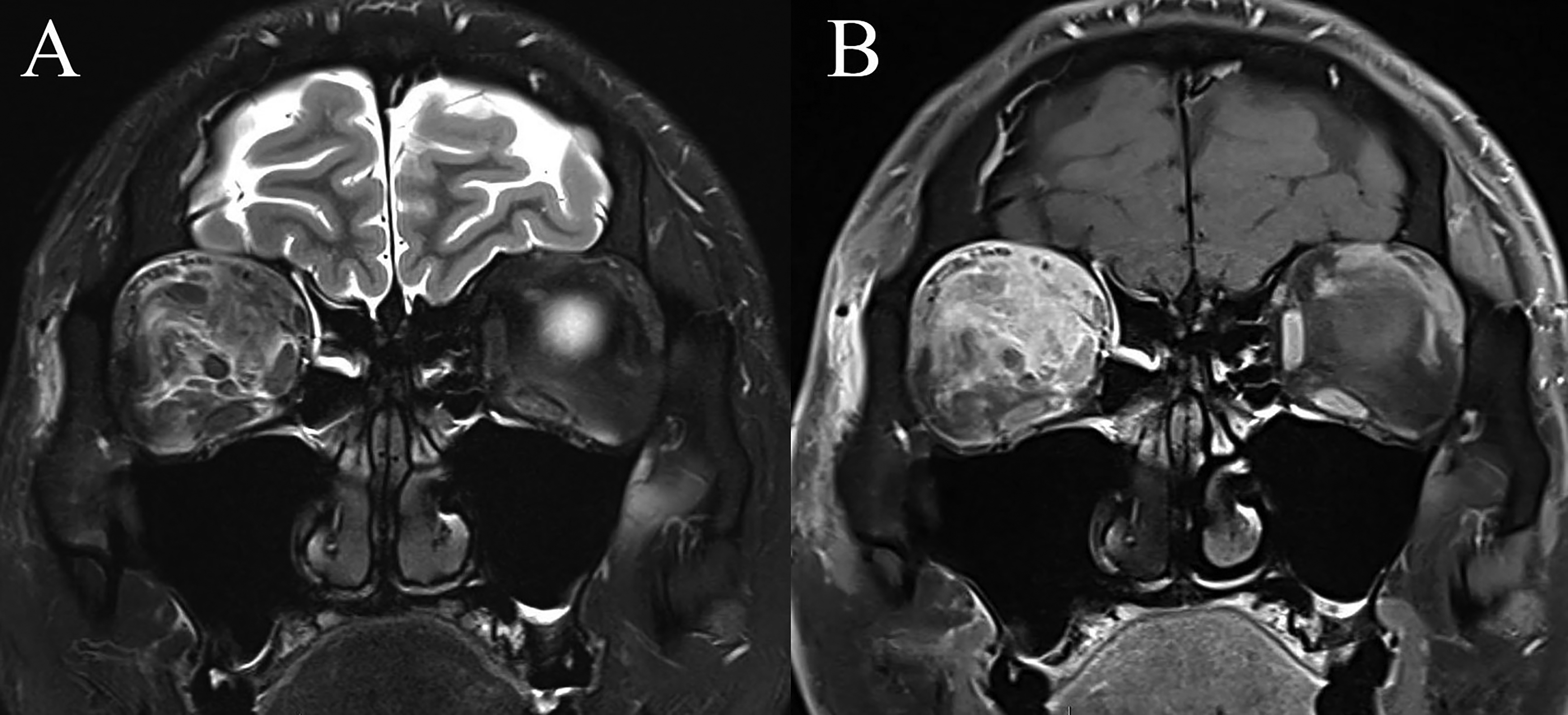
Coronal MRI orbital scans of a 30-year-old male with right DNSOI.
Of the twenty cases of OC with EOM involvement, all had at least 1 EOM affected. EOM involvement in OC was typically observed as loss of EOM margins in areas contiguous with orbital fat involvement, with peripheral (fascial) contrast-enhancement (Figure 3) (P = 0.002). Diffuse or fusiform EOM enlargement in OC occurred to a less significant degree compared to DNSOI. Of the ten DNSOI cases with EOM involvement, three (30%) had only a single EOM involved, whilst seven (70%) had at least 1 EOM affected. Contrast-enhancement of the EOM in DNSOI was variable but was predominantly homogenous and diffuse throughout the EOM (8/10, 80%) (Figures 1C, 1D and 2) (P < 0.001).
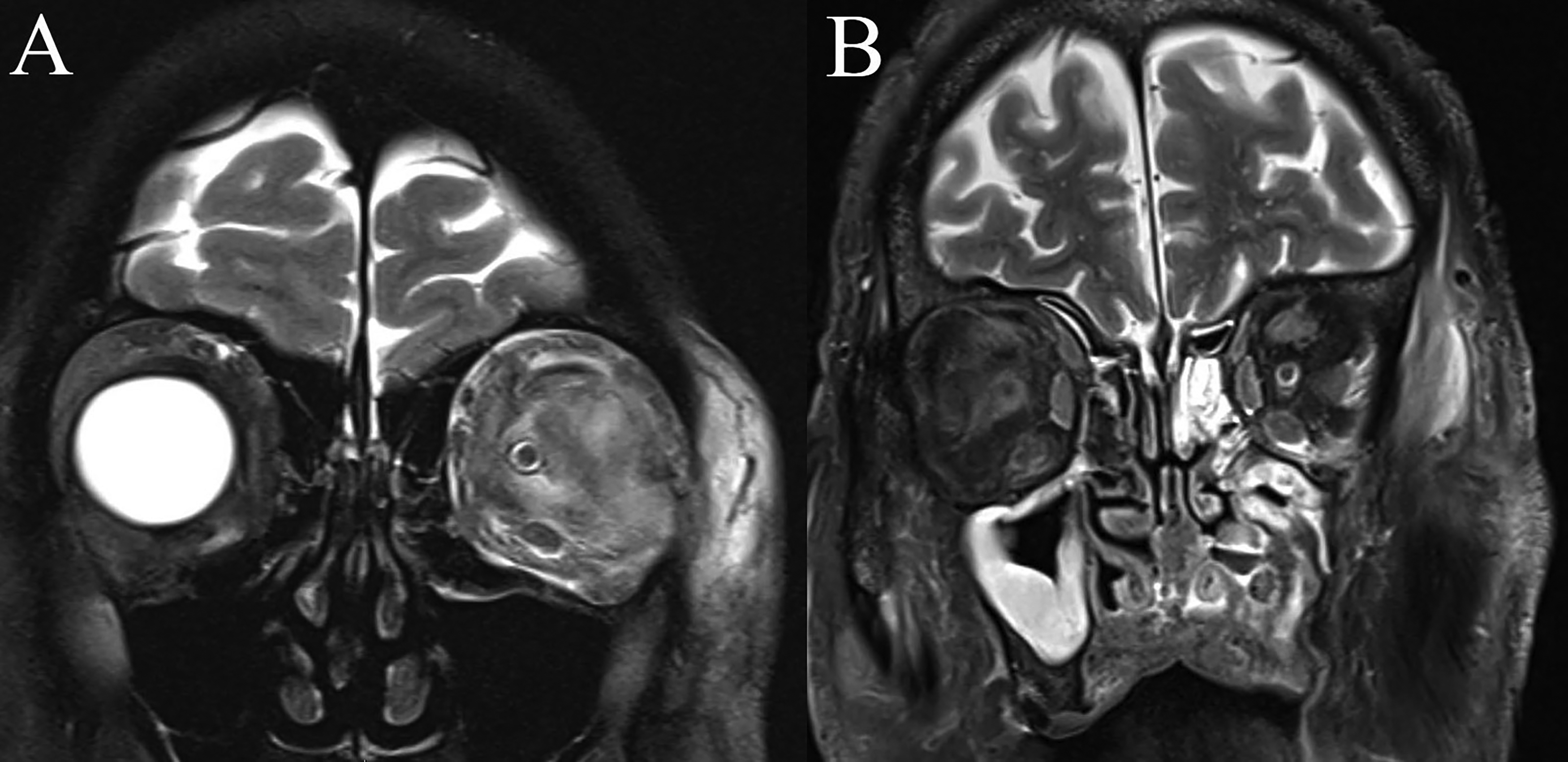
Coronal MRI orbital scans of a 49-year-old female with left non-sinogenic OC (
Four (36.4%) DNSOI cases had additional radiological changes within the contralateral orbit. These features ranged from lacrimal gland enlargement and/or contrast-enhancement, and EOM enlargement and/or contrast-enhancement. Meanwhile, no cases of OC demonstrated radiological abnormalities within the contralateral orbit. Table 3 summarises key differences for the radiological presentation of OC and DNSOI.
Summary of key radiological differences between OC and DNSOI.

Key: OC- orbital cellulitis, DNSOI- diffuse non-specific orbital inflammation, T1 FS CE- fat-suppressed contrast-enhanced T1-weighted imaging, T2 FS- fat-suppressed T2-weighted imaging, EOM- extraocular muscle.
Discussion
OC and DNSOI share a similar clinico-radiological presentation, but differentiating the two entities is crucial to determine the best course of management, which can be rather divergent. Radiological features such as adjacent sinus disease (unilateral or contralateral) or presence of a subperiosteal abscess may be a clear indication of a diagnosis of OC. 1 In the absence of these radiological features, DNSOI may be difficult to distinguish from OC and additional radiological changes within the orbit may be identified. Additionally, infective collections in early OC may occasionally resemble the inflammatory lesion seen in DNSOI.10,11 DNSOI is an entity which can be a significant diagnostic challenge, as differentials remains broad and encompass a range of both infective and specific inflammatory aetiologies. Delineating between OC and DNSOI may help guide decision making to proceed with obtaining a histopathological diagnosis via an orbital biopsy.
The pertinent findings from this study are that differences exist between OC and DNSOI for multiple qualitative MRI parameters, pertaining to orbital fat T2 signal, lacrimal gland and EOM involvement, and contralateral orbital changes (Table 3). These radiological changes are additional to other overt signs of OC, such as adjacent sinus disease or intracranial extension. Whilst orbital fat demonstrated contrast-enhancement in both OC and DNSOI, there was a difference in the signal on FS CE T2 sequence, where OC cases had a hyperintense T2 signal of orbital fat (95.2%), while there was a variable T2 signal in DNSOI (i.e Hyperintense T2 in 45.5%) (P = 0.001). A hypointense T2 signal of orbital fat was suggestive of DNSOI (P = 0.012). Although a greater proportion of OC patients had a hyperintense T2 signal in OC, DNSOI can also demonstrate hyperintense T2 signal in active inflammation and is thus not specific. Additionally, isointense T2 signal was non-differentiating (P = 0.067). Furthermore, OC may demonstrate loss of distinct margins of the lacrimal gland when involved (P < 0.001), whilst gross lacrimal gland enlargement and contrast-enhancement may be more common in DNSOI (P = 0.032 and 0.017, respectively). Diffuse multiple EOM involvement is more common in OC compared to DNSOI, albeit not at a statistically significant level (P = 0.103). In OC, there was peripheral contrast-enhancement and indistinct EOM margins in areas of contiguous orbital fat inflammation (P = 0.002). Contrastingly, DNSOI cases tended to demonstrate variable contrast-enhancement pattern throughout the affected EOM (P < 0.001). Finally, the presence of contralateral radiological changes, such as lacrimal gland enlargement and EOM involvement, was more suggestive of DNSOI. Ultimately, this study demonstrates that various radiological features, beyond overt sinogenic disease, may be non-differentiating between OC and DNSOI. The authors acknowledge however, that some of these radiological features are not specific, and further highlights the radiological dilemma clinician may be faced when differentiating between DNSOI and orbital infections.
NSOI represents a wide and heterogenous spectrum of clinico-radiological presentations. On MRI, T1- and T2-WI may show hypointensity and contrast-enhancement due to cellular infiltration and fibrosis.12–14 Consistent with our study, inflammation of the orbital fat in DNSOI on FS CE T2 may demonstrate variable signal intensity, depending on the tissue composition, however in active disease, hyperintensity of the orbital fat may be expected.15,16 It is known that in idiopathic orbital myositis (IOM), a subgroup within the spectrum of entities classified under NSOI, EOM may appear isointense on T1 and hyperintense on T2, with different patterns of distribution described.14,17,18 Similar patterns of T2-signal and contrast-enhancement were observed in several cases of DNSOI within this study. Additionally, it has been reported that in OC, unilateral orbital involvement and indistinct margins of the EOM are more commonly observed than in inflammatory disease on CT scans, and this was consistent with MR orbital scans in this study. Similarly, lacrimal gland involvement in DNSOI was characterised by both enlargement and contrast-enhancement. Meanwhile, in OC loss of distinct lacrimal gland margins was more commonly observed. In DNSOI, radiological changes observed in the EOM and/or lacrimal gland, may be a result of contiguous orbital fat inflammation or may arise from the primary inflammatory process occurring within the specific orbital structure itself. 19 Additionally, contralateral orbital changes were more common in DNSOI compared to OC, however, other specific localised and systemic inflammatory conditions such as IgG4-related disease and thyroid eye disease may also demonstrate bilateral changes. 20
There are several limitations to this study including the retrospective nature and relatively small sample size of OC and DNSOI. Following initial CT orbital imaging, MRI may be indicated in situations of diagnostic uncertainty or for further evaluation of orbital/intracranial complications. Thus, the study inclusion criteria may depict cases of greater disease severity. 21 Furthermore, future research may be directed at quantitative analysis of differentiating between OC and DNSOI, including parameters such as DWI and ADC values, and measurements of orbital structures involved. Literature surrounding normative measurements; the qualitative and quantitative analysis of various orbital disease (e.g.,: OC); and selective analysis of diffusion weighted imaging (DWI) and apparent diffusion coefficient (ADC) values between OC, NSOI and orbital lymphoproliferative disorders exist but remain limited.22–24
In conclusion, differentiating between OC and DNSOI remains a complex diagnostic dilemma as clinico-radiological presentation may be very similar. This study has demonstrated that several radiological features on MRI, beyond overt sinogenic disease, may help to differentiate OC from DNSOI, including the pattern of orbital fat signal intensity, EOM and/or lacrimal gland involvement, along with radiological abnormalities in the contralateral orbit. However, these radiological features may not be specific, and thus further highlights the radiological dilemma which clinicians are faced when tasked with differentiating between infectious and non-infectious orbital inflammatory disease.
Footnotes
Acknowledgments
None.
Data availability statement
The data that support the findings of this study are available from the corresponding author, [TA], upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
