Abstract
Purpose
In Descemet Membrane Endothelial Keratoplasty (DMEK) a gas bubble is usually used to attach the graft to the host cornea. In this study, we observed the bubble size and bubble-graft coverage at different gaze angles following DMEK.
Methods
This observational prospective study analyzed 465 images of patients who underwent an uneventful DMEK. Intraoperatively, the anterior chamber was filled up to 90% of its volume with a 20% Sulfur Hexafluoride (SF6) gas-air mixture. Postoperatively, the bubble was photographed daily in different gaze angles ranging from a supine position (0°) to an upright position (90°) and a slightly inclined position (105°). The primary outcomes were bubble-graft coverage and bubble diameters depending on the gaze angle and time after DMEK.
Results
The highest bubble-graft coverage was achieved at a 0° gaze angle at all times of measurement. In the first 48 h after DMEK, the mean bubble-graft coverage was over 85% at a gaze angle between 0° and 45°. Starting 72 h after DMEK, the graft coverage declined at all gaze angles. The graft coverage at a 0° gaze angle was 88.61 ± 10.90% after 96 postoperative hours, while the graft coverage was below 85% at all other gaze angles.
Conclusion
Our clinical results provide novel insight into variation in bubble-graft coverage as a function of gaze angle and may be used to aid in patient counselling for appropriate body positioning following DMEK to prevent early graft detachment. Maintaining supine positioning seems to be most advantageous starting 48 h after DMEK.
Keywords
Introduction
Descemet Membrane Endothelial Keratoplasty (DMEK) is one of the standard procedures for treatment of endothelial diseases such as Fuchs’ endothelial corneal dystrophy (FECD).1,2 Compared to other lamellar keratoplasties such as Ultrathin Descemet Stripping Automated Endothelial Keratoplasty, DMEK results in a better visual outcome after one year.3–5 However, DMEK has a higher risk for complications such as intraocular pressure rise3,6 or graft detachment and subsequent need for re-bubbling.3,4 Furthermore, re-bubbling is associated with a higher decrease in endothelial cell density,7–9 possibly increasing the risk for long-term graft failure. Hence, the intra- and postoperative care needs to be optimized to reduce the graft detachment rate after DMEK.
In DMEK, an air or gas bubble is injected into the anterior chamber to help attach the corneal graft to the host cornea. While the postoperative intraocular pressure does not have an effect on graft detachment rates, 10 two studies found that a smaller bubble size is an independent risk factor for postoperative graft detachment.11,12 A smaller bubble possibly offers a smaller “bubble-graft coverage”, thereby providing insufficient support for graft attachment. Thus, to maximize the postoperative bubble-graft coverage and lower the risk of graft detachment, patients are usually instructed to keep a strict supine positioning immediately after surgery.
Two mathematical studies have shown that with increasing anterior chamber depth, an appropriate patient positioning tends to be more important than the amount of gas fill regarding the graft coverage.13,14 However, no published data exists on actual clinical data regarding bubble-graft coverage after DMEK. Additionally, only one retrospective case series reported the time until complete resorption of an air bubble in the anterior chamber, but the time-dependent decrease in size of the bubble was not elaborately reported. 15
Herein, the purpose of this study was to clinically analyze the gas bubble size and bubble-graft coverage in the postoperative period after DMEK depending on the gaze angle.
Materials and methods
In total, ten patients with FECD who underwent uneventful DMEK were included into this monocentric, prospective, observational case series. Inclusion criteria were presence of FECD, pseudophakia and a planned DMEK to treat FECD. Exclusion criteria were complications during or after surgery, phakic DMEKs, or presence of other ocular diseases. Preoperatively, we performed an anterior segment optical coherence tomography (AS-OCT, Anterion, Heidelberg Engineering GmbH, Heidelberg, Germany) to obtain the White-to-White distance.
This study was approved by the Ethics Committee of the Medical Faculty of University Heidelberg, Germany (ID: S-565/2023), and was performed in accordance with the tenets of the Declaration of Helsinki. Written informed consent for this study was obtained from all patients.
Surgical procedure
Before surgery, an iridotomy with a Nd:YAG laser was performed at the 6 and 12 o’clock positions to minimize the risk of pupillary blockage.
All surgeries were performed by the same experienced surgeon (V.A.A.) under general anesthesia. Overall, four clear corneal incisions were made. The incisions at 3 and 9 o’clock were made with a 0.9 mm sized keratome, one incision at 10 o’clock was made with a 1.5 mm sized keratome and the largest incision at 12 o’clock was made with a 2.7 mm keratome to implant the corneal graft.
After a 9 mm descemetorhexis, the 8 mm corneal graft was inserted into the anterior chamber using a Viscoject-Bio 2.2 cartridge (Medicel AG, Altenrhein, Switzerland) with an injector. The graft was unfolded using corneal tapping maneuvers. After successful unfolding and central positioning of the corneal graft, 100% air tamponade was performed for one minute. Then, the anterior chamber was filled with a 20%
Gaze angle
Starting three hours after DMEK, we photographed the treated eye daily in different gaze angles using a digital camera with a resolution of 12 Megapixel. As visualized in Figure 1, the examined gaze angles ranged from the recommended supine position, which is described as a 0° gaze angle, in 15° intervals up to a slightly inclined gaze angle (105°). The respective body positions were upheld using an electric bed with automated electric body positioning functions and a goniometer (Würth GmbH & Co. KG, Kuenzelsau-Gaisbach, Germany). The subsequent gaze angle photo series were obtained in 24-h intervals after surgery.
Visualization of the analyzed gaze angles in lateral view ranging from 0° (supine position) over 90° (upright position) to 105° (slightly inclined position).
Image analysis
The ImageJ software (Version 1.51, U. S. National Institutes of Health, Bethesda, Maryland, USA) was used for image analysis. Each image was calibrated using the individual White-to-White distance obtained in the AS-OCT measurement. The horizontal and vertical diameter of the gas bubble were measured in every image (Figure 2). To measure the graft coverage of a graft with an 8 mm diameter, an overlay was automatically created using a newly programmed macro, which calculates a perfect circle with an 8 mm diameter using the individual White-to-White-calibration of each image. The overlay was positioned centrally as shown in Figure 2. Then, the pixel amount of the simulated graft covered by the gas bubble was measured and divided by the pixel amount of the whole simulated graft to obtain the bubble-graft coverage.
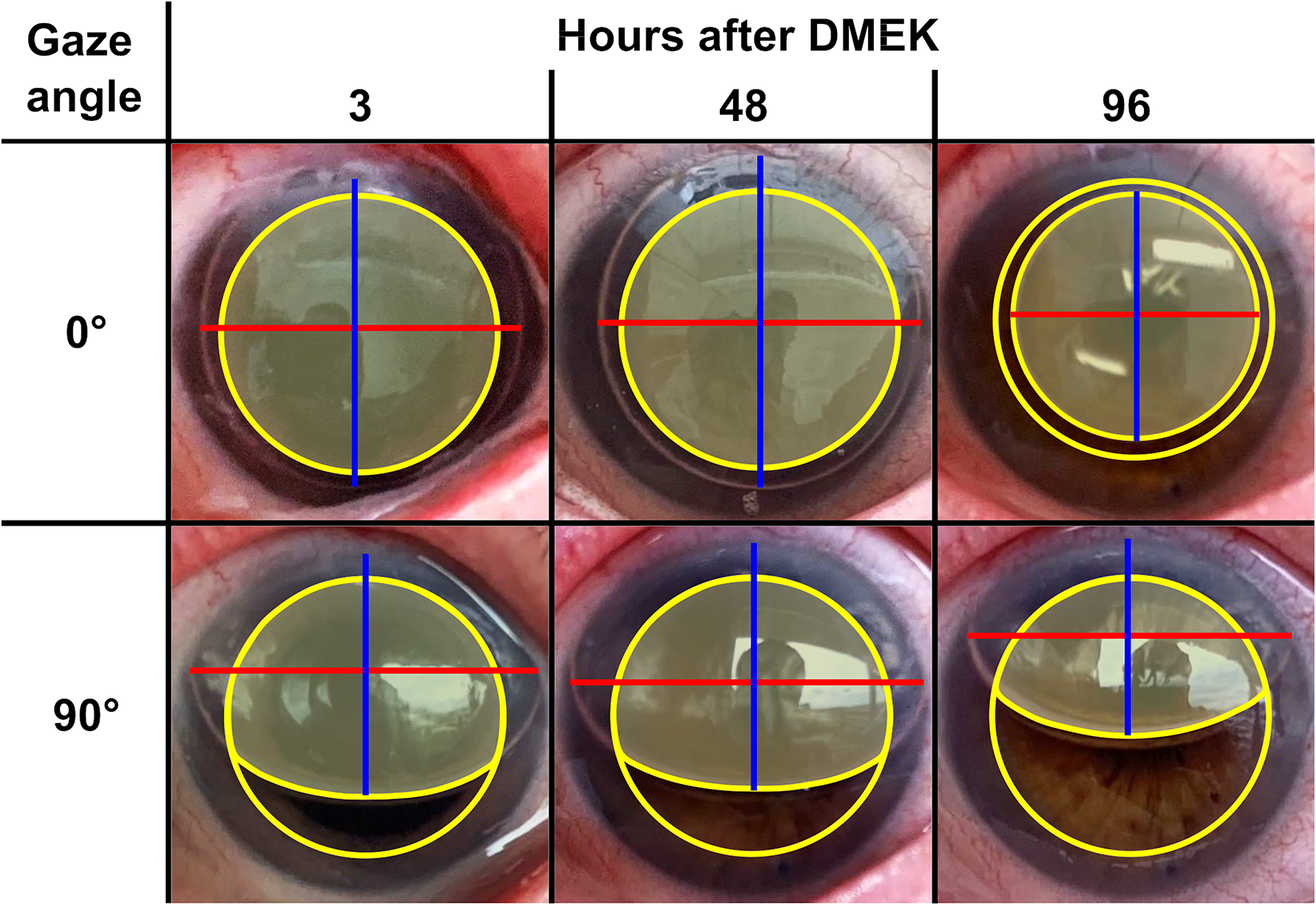
Example of the performed measurements shown it the images of one patient at a gaze angle of 0° (supine position) and 90° (upright position) 3, 48 and 96 h after Descemet membrane endothelial keratoplasty (DMEK). Red = Horizontal bubble diameter. Blue = Vertical bubble diameter. Yellow circle = 8 mm diameter graft. Yellow area = Graft area covered by the gas bubble.
Statistical analysis
The main outcome measures were bubble-graft coverage depending on gaze angle and time after DMEK, and horizontal and vertical bubble diameter. The statistical analysis was performed with SPSS for Windows (Version 29, IBM, Armonk, New York, USA). To describe the data, we reported the mean value, standard deviation and/or range.
Results
In total, 465 postoperative images obtained from ten eyes of ten patients were analyzed. The patients’ age ranged from 65 to 79 years. The mean White-to-White distance measured by the AS-OCT was 11.87 mm ± 0.43 mm. The images of each patient were calibrated with their respective White-to-White distance.
Bubble-graft coverage
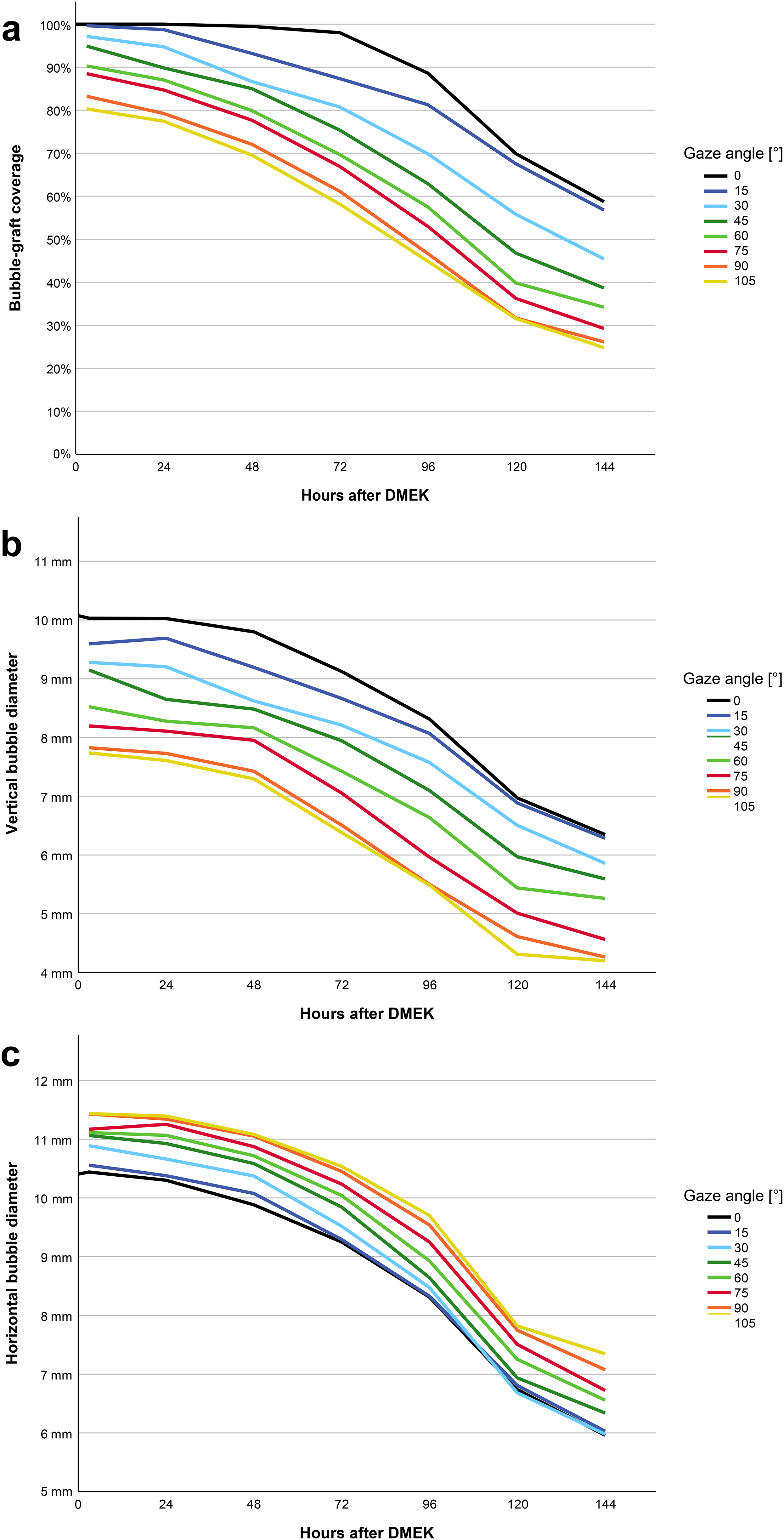
The bubble-graft coverage decreased over time and was constantly lower in more upright gaze angles (as shown in the Table 1). In the first 24 h after DMEK, a gaze angle of up to 60° provided a bubble-graft coverage over 85%. At 48 h after DMEK, the mean bubble-graft coverage was over 85% at all gaze angles up to 45°. At 72 h after DMEK, gaze angles of 0° and 15° provided a bubble-graft coverage over 85%. At 96 h after DMEK, a mean bubble-graft coverage of more than 85% was only observed at a 0° gaze angle. Afterwards, the mean bubble-graft coverage decreased rapidly with a maximum bubble-graft coverage at 120 and 144 postoperative hours of 69.88% and 58.75%, respectively (Figure 3a).
Overview of the mean bubble-graft coverage depending on the gaze angle up to 144 h after Descemet membrane endothelial keratoplasty. SD = Standard deviation, N/A = not applicable.
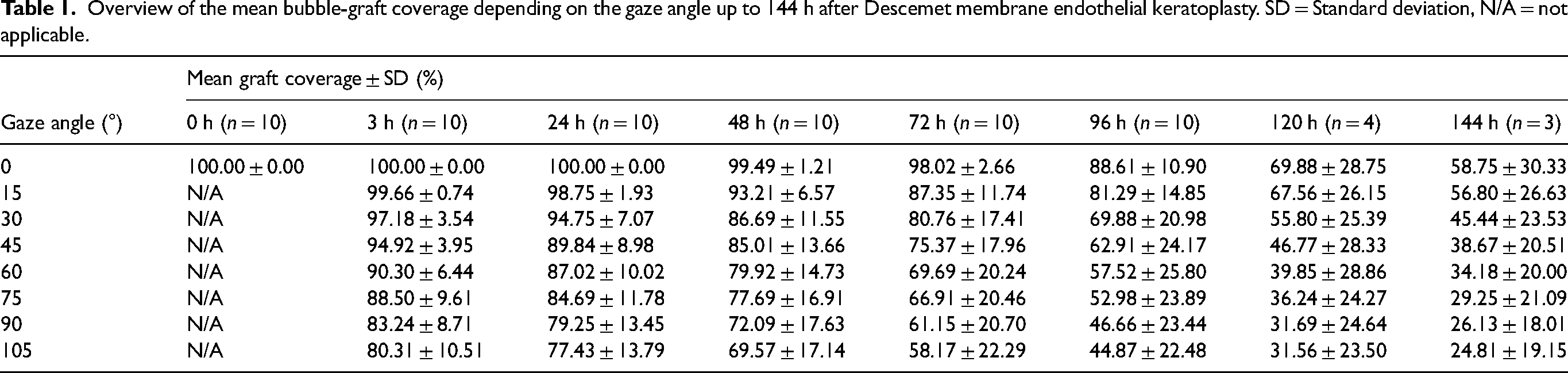
The maximum bubble-graft coverage was achieved in a supine position (0° gaze angle) immediately until 24 h after DMEK with a complete bubble-graft coverage (100%). The minimum bubble-graft coverage was observed at 144 postoperative hours at a gaze angle of 105° (slightly inclined position) with a mean coverage of 24.81%.
The difference in mean bubble-graft coverage between the positions with the worst and best coverage grew over time until 120 h after DMEK. After 3 h, the difference was 19.69%, which grew to 55.32% 120 h after DMEK (24 h: 22.57%; 48 h: 29.92%; 72 h: 39.85%; 96 h: 43.74%). At the measurement 144 h after DMEK, this difference reduced to 33.94%, indicating a lower gaze angle dependence.
The standard deviation of mean bubble-graft coverage was low especially in the early examinations after DMEK and in a more supine gaze angle. In later examinations and higher gaze angles the bubble-graft coverage showed a higher variation in our study group.
Vertical bubble diameter
The vertical bubble diameter corresponded to the gaze angle, with a higher gaze angle resulting in a smaller vertical bubble diameter. This relationship was consistent at all times of measurement. At the first measurement series 3 h after DMEK, the vertical bubble diameter was the largest at a gaze angle of 0° with a vertical diameter of 10.03 ± 0.51 mm (Figure 3b). In the first 48 h after DMEK, a slow decrease in vertical diameter was observed, with an accelerated decrease afterwards (Figure 3b). At the final measurement 144 h after DMEK, the mean vertical bubble diameter ranged between 4.20 mm and 6.35 mm at both gaze angles of 105° and 0°.
Horizontal bubble diameter
At the first measurement series 3 h after DMEK, the horizontal bubble diameter was the largest at a gaze angle of 105° with a horizontal diameter of 11.43 ± 0.50 mm (Figure 3c). In the first 48 h after DMEK, the horizontal bubble diameter only decreased slowly with a mean diameter over 10 mm in all gaze angles except for the 0° gaze angle. Afterwards, the reduction rate in horizontal diameter increased. A larger gaze angle resulted in a higher horizontal bubble diameter at every time of measurement. The gas volume formed a circular bubble in lower gas angles whereas the gas volume accumulated in the superior chamber angle and had a more lenticular shape in higher gaze angles.
Discussion
In this clinical study, we analyzed the gas bubble behavior after DMEK and showed that the bubble-graft coverage is over 85% in the first 48 h after surgery in a gaze angle of up to 45°. Afterwards, a steady decline in bubble-graft coverage was observed with a gaze angle of 0° resulting in the highest bubble-graft coverage at all times of measurement. The difference in bubble-graft coverage between the positions with the highest and lowest graft coverage increased steadily until 120 h after DMEK indicating a higher gaze angle dependence in the later postoperative period.
To date, only a few studies have examined the graft coverage of the gas bubble in the anterior chamber. Ćirković et al. stated that an 80% anterior chamber volume air bubble will cover 100% of a centered 8 mm graft in a supine position and 90% in an upright position three hours after DMEK. 12 This is congruent to our results 3 h after DMEK. Later examinations of the bubble-graft coverage were not analyzed in their study. Two mathematical models found that the bubble-graft coverage in shallow anterior chambers is more sensitive to the gas fill than the patient's positioning whereas in deep anterior chambers the patient's positioning is more important.13,14 This relationship is especially important for pseudophakic patients since they tend to have a deeper anterior chamber. However, both studies only mathematically modeled the behavior of the bubble in the anterior chamber with various settings but did not clinically analyze the bubble behavior and decrease over time.
Interestingly, we observed that the horizontal bubble diameter is the lowest at a 0° gaze angle compared to all other examined gaze angles. In a supine position the bubble is nearly circular in en-face images and therefore has the lowest diameter. In higher gaze angles, the gas accumulates in the top of the anterior chamber and is more lenticular-shaped, thus increasing the horizontal diameter while decreasing the vertical diameter. This bubble behavior may be one of the causes of the decrease in bubble-graft coverage in higher gaze angles next to the sole displacement of the bubble.
A clinical study showed that after a graft detachment in the first eye of a patient, the subsequent eye of the same patient has an increased risk for graft detachment. 16 While this correlation could be due to pathophysiological or genetic factors, the patient's compliance for supine positioning may also influence the bilateral risk increase. The incompliance of patients could be reduced if the supine positioning is only needed for a short time. Due to the lower bubble-graft coverage in all gaze angles after 48 h following DMEK, a strict supine positioning of the patients may be most advantageous to prevent graft detachment starting 48 h after DMEK.
Of note, the presented results only apply to the usage of a 20% SF6-gas-air-mixture. While the bubble formation and constitution do not significantly differ between an air or gas bubble, 13 the time until resorption may differ. Thus, the postoperative bubble behavior including bubble diameter and bubble-graft coverage is most likely dependent on the gas mixture used. A previous study found that there is no significant difference in graft detachment rate when comparing a strict supine positioning for 48 h after DMEK (with an air bubble installation) to an uninstructed postoperative care. 17 However, due to the usage of air to ensure graft attachment, which is usually resorbed after 48 to 72 h, 15 the bubble-graft coverage may be low in the initial phase after surgery even in a supine position. Therefore, a large area of the graft may not be covered by the air bubble sufficiently in the early postoperative period independent of gaze angle, which could result in early graft detachment. Von Marchtaler et al. found that the use of a SF6-gas bubble after DMEK results in a better graft adhesion than the use of an air bubble with a comparable risk of pupillary blockage. 18 As shown in our study, when using a SF6 bubble the bubble-graft coverage was still over 69% in the first 48 h after DMEK in all examined gaze angles. Therefore, the difference in graft adhesion could be explained by a prolonged presence of a SF6-gas bubble compared to an air bubble with a subsequent higher and longer bubble-graft coverage. Future studies evaluating the benefit of postoperative positioning should consider the difference in resorption time depending on the type of gas used.
A limitation of this study is the small sample size. However, this is the first clinical study reporting the bubble-graft coverage after DMEK. Furthermore, we believe the rigorous and extensive measurements performed for each patient provide sufficient data to demonstrate the tendency of bubble-graft coverage behavior during the early postoperative period following DMEK. Another limitation of this study is the two-dimensional measurement of the graft coverage. Due to the curvature of the cornea, the area in contact with the gas bubble may be higher than stated in this article. However, we consider this effect to be small and clinically negligible.
In conclusion, our analysis suggests that a strict supine positioning may not be imperative in the first 48 h following DMEK given the large bubble-graft coverage during this time window at all gaze angles. However,
Footnotes
Credit statement
M.F.: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Visualization, Roles/Writing - original draft; H.-S.S.: Data curation, Formal analysis, Investigation, Writing - review & editing; R.C.B.: Data curation, Formal analysis, Investigation, Writing - review & editing; C.M.M.: Data curation, Investigation, Writing - review & editing; T.M.Y.: Data curation, Investigation, Writing - review & editing; R.K.: Funding acquisition, Project administration, Resources, Supervision, Writing - review & editing; G.U.A.: Funding acquisition, Project administration, Resources, Supervision, Writing - review & editing; V.A.A.: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Visualization, Writing - review & editing
Data availability statement
The datasets generated and analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Declaration of conflicting interests
T. M. Yildirim reports lecture fees from Alcon Laboratories and Hoya Corp. and nonfinancial support from Johnson & Johnson Vision.
R. Khoramnia report grants, personal fees, and nonfinancial support from Johnson & Johnson Vision, Alcon Laboratories, Hoya Corp., Kowa Co., Ltd., Teleon, Physiol S.A., Sifi Medtech Srl, and Rayner Intraocular Lenses Ltd.; grants and personal fees from Oculus Optikgeräte GmbH, Carl Zeiss Meditec AG, Heidelberg Engineering and Ophtec BV; and grants from Acufocus, Inc., outside the submitted work.
G.U. Auffarth reports lecture fees and research grants from Johnson & Johnson Vision, Alcon Laboratories, Carl Zeiss Meditec AG, Hoya Corp., Kowa Co., Ltd., Oculentis GmbH/Teleon, Rayner Intraocular Lenses Ltd., Santen, Sifi Medtech Srl, and Biotech.
V.A. Augustin reports research grant from Kowa Co., Ltd.
For the remaining authors, no financial disclosures were declared.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Klaus-Tschira-Foundation (Heidelberg, Germany).
