Abstract
Purpose
To report a recurrence of punctate inner choroidopathy (PIC) with an inflammatory choroidal neovascular membrane (iCNVM) after the Pfizer-BioNTech COVID-19 vaccine.
Methods
Case report.
Results
A 38-year-old female with a history of myopia and previous episodes of PIC and iCNVM presented with distorted vision in her right eye, seven days after receiving the first dose of the Pfizer-BioNTech COVID-19 vaccine. The patient exhibited active PIC lesions with iCNVM confirmed on multimodal imaging. Treatment with a combination of oral corticosteroids and intravitreal anti-VEGF injection led to disease resolution. Subsequent COVID-19 vaccinations, administered while the patient was immunosuppressed, did not lead to disease relapse. However, relapse occurred following the fourth COVID-19 vaccine, when the patient was not immune suppressed.
Conclusion
This case highlights the potential risk of PIC disease relapse following COVID-19 vaccination. Further research is needed to investigate the relationship between COVID-19 vaccination and PIC exacerbation, as well as to determine optimal management strategies in this population, including close observation and consideration of prophylactic immune suppression at the time of COVID-19 vaccine for high-risk individuals.
Keywords
Introduction
Punctate inner choroidopathy (PIC) is an idiopathic inflammatory multifocal choroidopathy, classified as one of the “white dot syndromes”. The inflammatory lesions present with white-yellow colored lesions located at the level of the retinal pigment epithelium and inner choroid. A lack of inflammation within the anterior chamber and vitreous is a hallmark of its presentation. 1 The development of a secondary inflammatory choroidal neovascular membrane (iCNVM) occurs in approximately 63–74% of cases and is the most common cause of vision loss. 2 PIC is an autoimmune inflammatory disease, believed to be often triggered by an environmental stimulus, where antigens target the outer retina and inner choroid. 3
The development of COVID-19 vaccines in response to the global pandemic caused by the coronavirus has significantly changed the trajectory of the crisis, resulting in the saving of millions of lives around the world. Following mass population vaccination there is a growing literature documenting COVID-19 vaccination-related adverse events with new ocular disease and ocular disease reactivation. 4 In particular, COVID-19 vaccination is associated with an increased risk of inflammatory eye disease relapse. 5
Case description
A 38-year-old European female with a history of myopia (refractive error: −4.50 diopters) treated with LASIK, bilateral PIC and previous right inflammatory choroidal neovascular membrane (iCNVM) that was last active at least 1 year earlier. She presented to the acute ophthalmology clinic with a 7-day history of distorted vision in her right eye, 8 days after receiving her first dose of the Pfizer-BioNTech COVID-19 vaccine.
Best-corrected visual acuity (BCVA) was 20/25 and 20/20 in the right and left eyes. Slit-lamp examination revealed a quiet anterior segment with clear media and quiet vitreous. On dilated fundus exam there were multifocal choroidal lesions present in both eyes. Optical coherence tomography (OCT) macular scan of the right eye confirmed activity associated with an existing PIC lesion. OCT-angiography (OCTA) confirmed the presence of an iCNVM within the avascular complex level of the retina [Figure 1]. Screening investigations were non-contributory; a normal serum ACE (38 U/L, normal 20–70), full blood count, QuantiFERON-TB Gold, and treponemal antibody testing. Chest x-ray was normal, showing no evidence of tuberculosis and sarcoidosis.
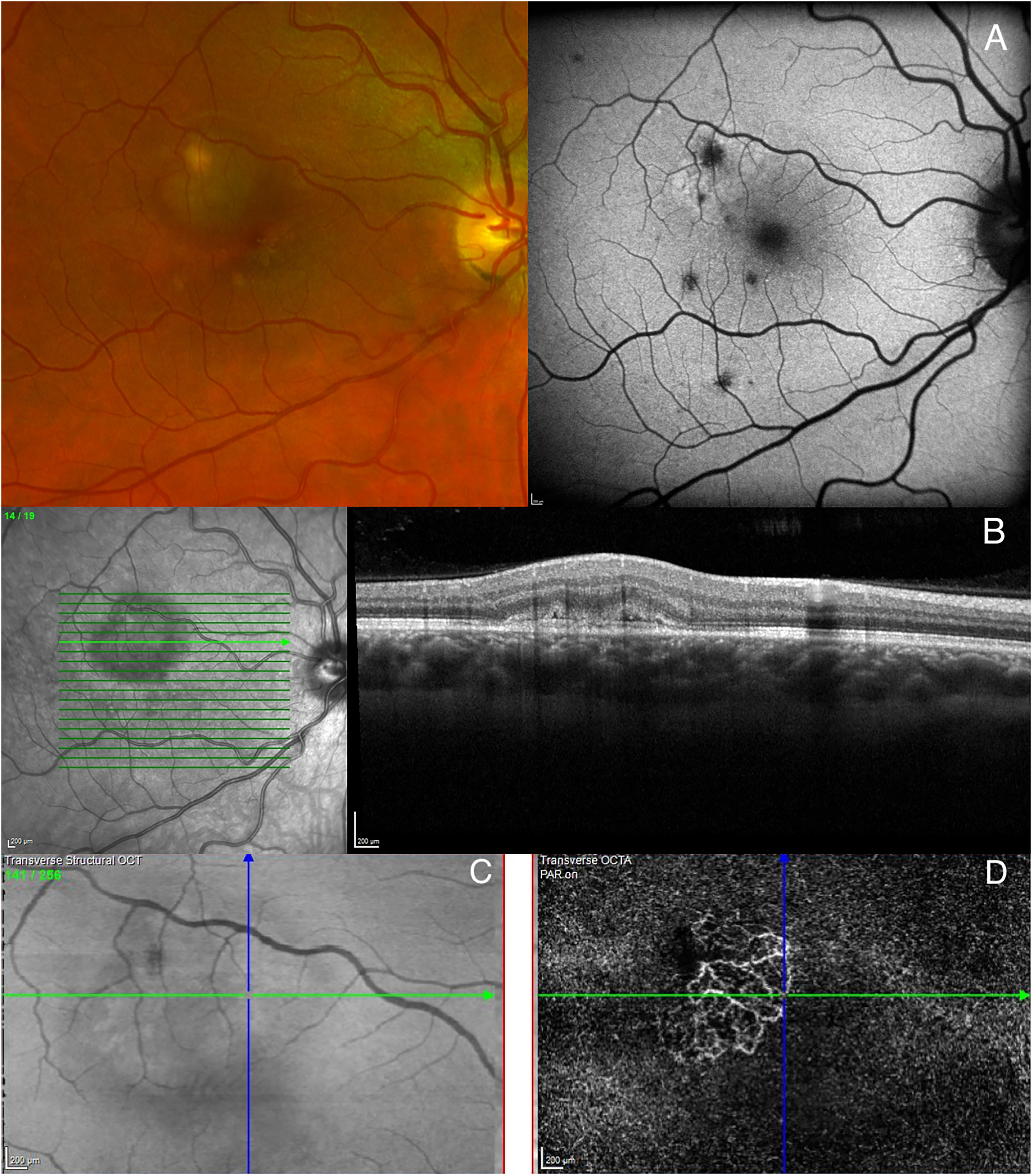
Multimodal imaging in an active phase of PIC recurrence. (A) Right eye color fundus ultrawide field photograph showing small, multifocal, yellow-white spots in the extrafovea. A Fundus Autofluorescence (FAF) image reveals peripapillary retinal pigment epithelium (RPE) atrophy and multiple hypofluorescence spots corresponding to RPE and outer retinal atrophy (B) Horizontal Optical Coherence Tomography (OCT) B-scan through the center of the largest lesion (green line, near-infrared image) illustrates active choroiditis. The PIC lesion demonstrates isoreflective material, splitting the RPE/Bruch's membrane complex. An active inflammatory choroidal neovascular membrane (iCNVM) is supported by the presence of both the pitchfork sign and subretinal fluid as supporting diagnostic elements (C) Structural image (en-face OCT) in comparison with (D) OCT-Angiography (en-face OCTA) highlighting a lacy network of vessels, confirming active iCNVM in the outer retina.
The presentation was consistent with relapsing PIC with active iCNVM and the patient was treated with a combination of oral corticosteroids and intravitreal anti-vascular endothelial growth factor (anti-VEGF) injection. No disease recurrence was observed at the time of the second COVID vaccination, which was given whilst the patient was immunosuppressed on 20 mg of oral prednisone.
Seven months later, there was a mild PIC iCNVM recurrence. Fundus Fluorescein Angiogram (FFA) confirmed iCNVM activity with associated fluorescein leak [Figure 2]. This was treated with a combination of intravitreal anti-VEGF and oral corticosteroids with resolution of activity on OCT. The patient had her third COVID-19 vaccine whilst immunosuppressed with 20 mg of oral prednisone, and there was no further disease recurrence with close monitoring.
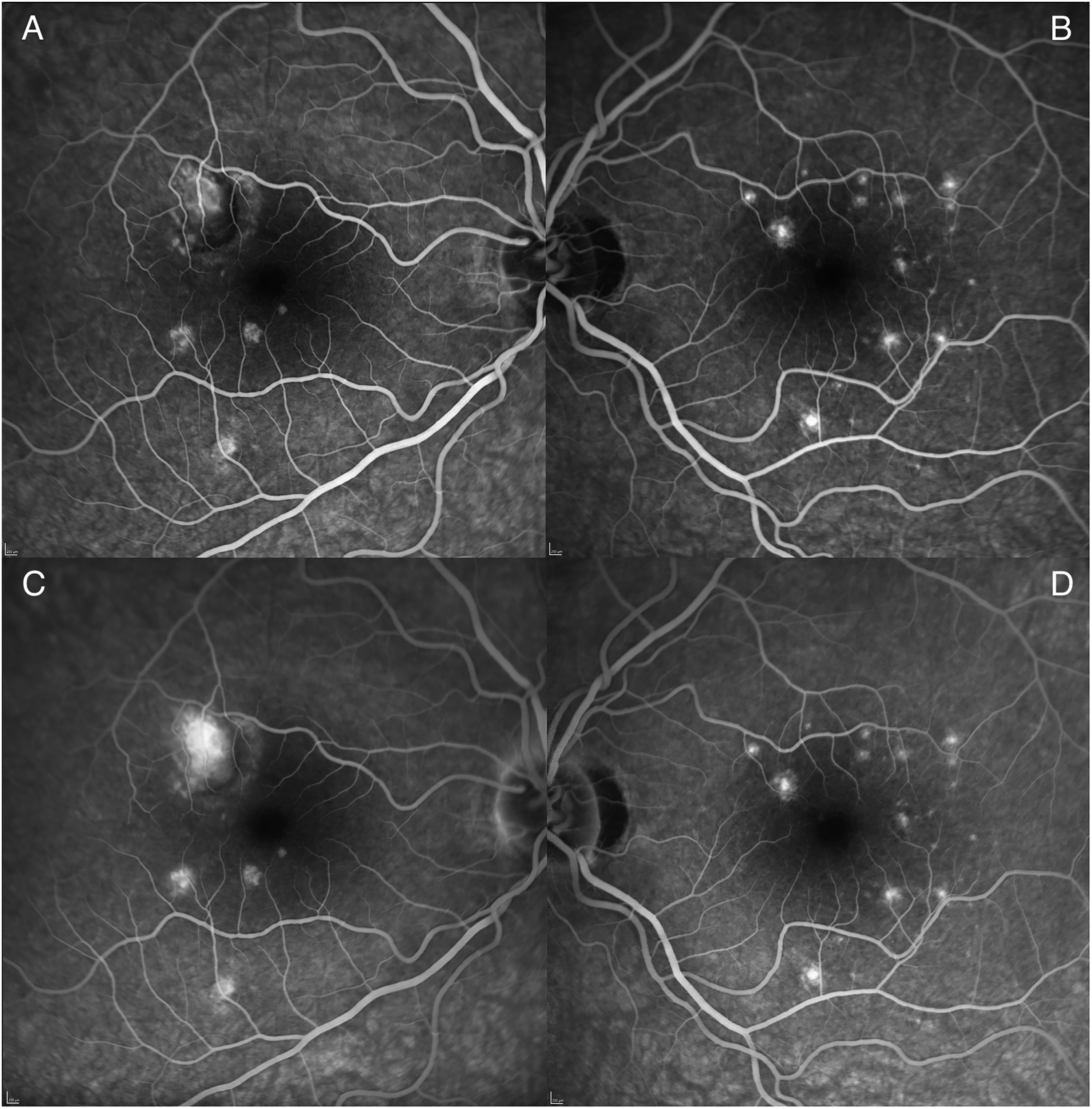
Reactivation of iCNVM demonstrated on Fundus Fluorescein Angiogram (FFA) in the right eye, showing hyperfluroescence in the early phase (A), followed by late leakage (C). The left eye with inactive lesions shows hyperfluorescent in both early (B) and late (D) phases due to RPE window defects.
Nine months later, there was further iCNMV recurrence, treated with intravitreal anti-VEGF injection. The patient had COVID-19 infection 3-months before this episode, and there was no disease reactivation one month after COVID-19, so it is unlikely associated.
The fourth COVID vaccine was given one year after the third COVID vaccination, and 4-months following previous unprovoked relapse. The patient was symptomatic within one month, with new PIC lesions documented on ultrawide field imaging, and subsequent iCNVM reactivation on OCT imaging. Oral corticosteroids had a good response, leading to fading PIC lesions as well as iCNVM resolution [Figure 3]. There was unfortunately a recurrence one month after stopping immunosuppression so a further intravitreal anti-VEGF injection was required. The decision has been made to consider an immunosuppressant steroid-sparing agent.
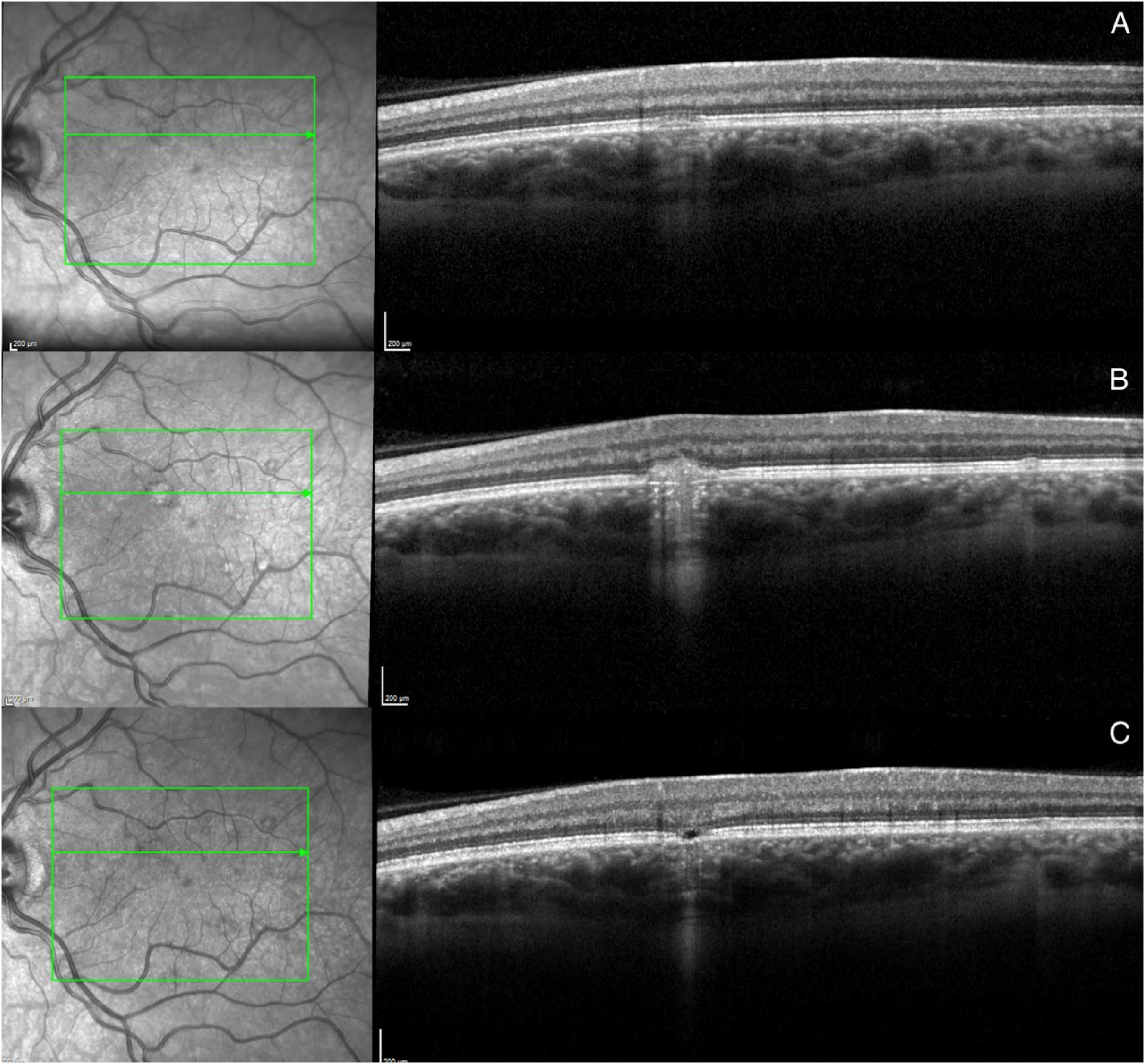
Tracked OCT B-scan images follow reactivation of a left macular PIC lesion. (A) Stage 2 disease, 2 weeks before the vaccine, (B) Stage 3 disease, 1-month post 4th dose of COVID-19 vaccine (C) Stage 4 disease, resolution following treatment with corticosteroids.
Discussion
To the best of our knowledge, this is the first reported case of recurrent PIC disease reactivation associated with Pfizer-BioNTech (BNT162b2 mRNA) COVID-19 vaccination. Over two years, this case demonstrates two disease relapse entities; combined PIC lesions with iCNVM, and new PIC lesions.
The case captures the development and recurrence of both iCNVM and PIC disease with multimodal imaging, which includes time-lapsed scans.6,7 When evaluating an iCNVM, our case highlights the clinical utility of OCTA with detecting choroidal neovascular networks and the presence of late leakage on FFA to confirm recurrence. 6
There is one previous case of PIC disease reactivation three days following the COVID-19 Oxford AstraZeneca vaccine. 8 This case documents disease recurrence, with resolution after treatment with oral prednisolone (including a 6-week tapering period).
The COVID-19 virus, like other viruses (Cytomegalovirus and Epstein-Barr Virus) has also been linked to the development and relapse of autoimmune diseases, and case reports also document the development and reactivation of PIC disease.9,10 Autoimmunity induction is proposed through various mechanisms, which include molecular mimicry, epitope spreading, and bystander activation. 11 Uveitis reactivation following COVID-19 immunization is also suggested by the same theorized processes. 4 Based on the existing evidence, it is advisable not to discourage patients with pre-existing ocular inflammatory conditions from receiving COVID-19 vaccination. The benefits of protection against severe infection will usually significantly outweigh the relatively low risk of triggering a uveitis flare.4,5,12
Our case highlights the potential benefit of prophylactic immunosuppression, given our patient did not experience disease recurrence when immunosuppressed. The role of prophylaxis remains undetermined and should be discussed with patients, including the benefits and risks specific to individual patients. Immunosuppression may be considered in preparation for COVID-19 vaccination in those with a previous history of vaccine-associated flare. Equally, clinicians may also consider closer monitoring given the higher risk of relapse following vaccination, or switching to another vaccine class. 5 Further research is required to better understand the potential and the full range of available options for patients.
There is significant debate as to the role of inflammation in the development of iCNVM and its treatment with immunosuppression. The evidence shows that the use of oral corticosteroids halves the risk of developing iCNVM in the largest cohort of PIC patients. 2 The flare of iCNVM post-vaccine supports the theory that inflammation is likely contributory. In recurrent episodes, steroid-sparing immunosuppression may be required to prevent further disease relapses and secondary chorioretinal atrophy. 6
In conclusion, this case highlights the potential risk of PIC disease relapse following COVID-19 vaccination Further research is required to determine the optimal management strategies in this population, including close observation and considering prophylactic immune suppression at the time of COVID-19 vaccine for high-risk individuals. Ophthalmologists should remain vigilant in monitoring patients with ocular inflammatory conditions after COVID-19 vaccination and promptly treat any relapse to prevent permanent visual impairment.
Consent
Informed consent to publish the details from the affected individual has been obtained.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
