Abstract
Myopia is becoming increasingly common in young generations all over the world, and it is predicted to become the most common cause of blindness and visual impairment in later life in the near future. Because myopia can cause serious complications and vision loss, it is critical to create and prescribe effective myopia treatment solutions that can help prevent or delay the onset and progression of myopia. The scientific understanding of myopia's causes, genetic background, environmental conditions, and various management techniques, including therapies to prevent or postpone its development and slow its progression, is rapidly expanding. However, some significant information gaps exist on this subject, making it difficult to develop an effective intervention plan. As with the creation of this present algorithm, a compromise is to work on best practices and reach consensus among a wide number of specialists. The quick rise in information regarding myopia management may be difficult for the busy eye care provider, but it necessitates a continuing need to evaluate new research and implement it into daily practice. To assist eye care providers in developing these strategies, an algorithm has been proposed that covers all aspects of myopia mitigation and management. The algorithm aims to provide practical assistance in choosing and developing an effective myopia management strategy tailored to the individual child. It incorporates the latest research findings and covers a wide range of modalities, from primary, secondary, and tertiary myopia prevention to interventions that reduce the progression of myopia.
Keywords
Introduction
The major aim of this Algorithm Annexe to the “Update and Guidance on Management of Myopia. European Society of Ophthalmology in cooperation with International Myopia Institute” 1 is to give practical advice to eye care providers in choosing and developing an effective myopia management strategy for their patients. The Annexe presents current knowledge in the form of flowcharts and is focused on prevention and delaying onset and then slowing down progression: from environmental variables to avoid myopia onset, through screening and follow-up of pre-myopes to prevent or postpone myopia development, and eventually to myopia management to reduce myopia progression. The rapid increase in knowledge regarding myopia management could be challenging for the busy eye care provider but warrants an ongoing need to monitor new research and incorporate it into personal everyday practice. 2
Limitations
Although extensive literature has been published on myopia management and is freely available, important knowledge gaps have remained, which can lead to difficulties in creating a robust myopia algorithm. In some areas, evidence is difficult to obtain e.g. it is becoming increasingly difficult to conduct randomized controlled trials (RCTs) involving a placebo arm, as this may now be considered unethical,3,4 and because there are individual variations in the progression of myopia, randomisation in itself is challenging. 5 The main aim of this sub-chapter is to explain the limits of a myopia management algorithm and to demonstrate the present gaps in knowledge in order to initiate additional research in these areas.
Why is it not possible to provide simple algorithm(s) or guideline(s), given the present state of the science of myopia control?
- There is a wide variety of differences among myopes currently reported in the literature, including, but not limited to, age of onset, sex, ethnicity, family history of myopia, growth rate, and environmental factors, including computer screen time/near work, outdoor time, work-rest regime. - There are few head-to-head comparisons (“randomized non-inferiority trials”) of the various interventions available.6–8 The majority of studies focused on comparing pharmacological and optical treatments against a non-active control. Results indicated that these interventions could potentially slow down myopic progression and reduce axial elongation, but the results are inconsistent. Limited evidence is available for the effects of these interventions after two or three years, and there is still uncertainty about their long-term effectiveness. There is therefore a need for more rigorous and longer-term studies that compare various myopia control interventions, either used alone or in combination, and for improved methods of monitoring and reporting adverse effects.
9
- There are no well-designed within-patient comparisons (“repeated cross-over trials”) of the various interventions available, so it is not clear whether some interventions are more effective than others in certain groups of patients. - Comparatively few studies have been carried out in non-Asian countries, making it unclear if the efficacy and side effects of interventions are consistent across ethnic groups and geographical regions, especially within Europe. However, recent studies conducted on American and European populations have shown that pharmacological treatments, such as 0.01% atropine, demonstrate reduced progression of axial length and spherical equivalent refraction after treatment.10,11 - Legislative rules differ across countries/regions in Europe; certain treatment regimens, such as topical application of atropine eye drops may have to be used in an “off-licence”, “off-label” or “no-label” mode. Practitioners may understandably be reluctant to use such unlicensed preparations, as this may expose them to the risk of malpractice claims. In addition, atropine eyedrops are generally only available from compounding pharmacies, where variability in production and stability will affect dosage.
12
Open questions yet to be addressed (by RCTs, large independent real-life studies, or other study designs)
- There is a lack of prevalence data obtained using cycloplegia, as studies not using cycloplegia overestimate the frequency of myopia. - Which interventions (other than more time spent outdoors and low-dose atropine13,14) can be used prophylactically to prevent or delay onset of myopia? Could risk factor matrices or scoring systems be useful here to identify when prophylactic use is most indicated?
15
- Which intervention to start and at what age?
- - direct comparisons using – randomised non-inferiority trials examining the efficacy of different treatments (as 1st line therapy). - What factors influence treatment decisions and treatment outcomes?
- - age of myopia onset? rate of previous myopic progression? family history? sex? ethnicity? binocularity? pupil size? lifestyle conditions? social-economic status? etc? and the interactions between these factors? - What is the optimal age to start treatment for different types of myopia (pre-school, primary school, secondary school, and young adulthood)? - Is switching treatment modality effective, and if so, which to which intervention should a patient be switched?
- - repeated cross-over trials to search for scientific evidence of “non-responders” - - right time to switch to another intervention? - - head-to-head comparisons of different switching orders and timings - - what action to take when intervention fails to control myopia progression? - Which combination therapy?
- - direct comparisons of different combination therapies using randomised non-inferiority trials - More data (possibly a systematic review or meta-analysis) is required on the prevalence (and possible characteristics) of patients who fail to respond to the various interventions. - Treatment duration and cessation?
- - comparative data on when and how to stop interventions? Research to test the continuation of efficacy beyond 2–3 years is very limited. Issue of plateau effects/rebound effects.
Further points which are important
- The lack of a centralised data repository for all clinical studies or trials. Although there have been discussions about the creation of a central repository for all data that would allow meta-analyses, no central repository has yet been set up. - The field of myopia control is constantly expanding, and new methods of myopia control are being trialled regularly. This paper includes only the treatment modalities that are currently regulated and widely used for myopia control and prevention.
Solutions used to overcome the aforementioned limitations
The main aim in developing this algorithm was to be as close as possible to the science-based evidence. A challenge exists primarily where no evidence is available. A compromise is to work on “best practices” or “consensus” basis by many experts in the field of myopia. Not doing so leads to a lack of guidance for practitioners to follow, resulting in confusion and poor adoption. The main process was the circulation of the drafts for expert commentary, and where necessary, methods inspired by the Delphi Panel 16 were used to generate consensus in a structured way. The algorithm was developed by members of the board of the European Society of Ophthalmology in cooperation with invited experts and European authors of the International Myopia Institute. The algorithm was endorsed by the SOE executive board. In detail: The first author (BT) wrote the first draft of the algorithm, which was reviewed several times by the last author (JN). The last author wrote the initial draft of the introduction of the manuscript and the limitations of the project following a brainstorming process with four authors (JBJ, JAG, LSG, SR) through email. All authors received the initial draft of the whole text for evaluation, changes, and comments. After receiving all of the authors’ responses, a second draft was created that included all of the suggestions, and a consensus questionnaire was developed on eight particular topics in which the authors’ opinions differed. The consensus questionnaire and the second draft document, which included all of the authors’ recommendations and also the text solutions on consensus subjects, were distributed to all authors. The manuscript and figures were revised based on the responses. This third draft was shared again, and after additional minor changes, it was approved by each author. The article was then delivered to the Board of the European Society of Ophthalmology, who shared it among their Board members and included it on the agenda for the Board meeting. The SOE Myopia Consensus Group was created when 16 Board members (in addition to the six original authors who are SOE Board members: WAA, JTHNDF, ZZN, PGK, IJ, and CNV) provided their views and agreement, and the SOE Executive Board endorsed the paper. The finalised version of the Myopia Algorithm manuscript was sent to all authors for final approval before it was eventually submitted to the journal.
Algorithm
Prevention or delay of myopia onset (primary prevention measures)
1. The main aim is to provide lifestyle advice for any child whereby the clinical evaluation indicates a possibility of reducing the risk of or at least delaying myopia onset. Delaying myopia onset is important because postponing the onset of myopia is expected to reduce the ultimate magnitude of myopia and hence the risk of future high and pathologic myopia.1,17–19 The most important primary prevention measures are listed in Table 1.
Recommendation for primary prevention or delay of myopia onset for school-aged children (Level I is the highest evidence (details see in Elsevier evidence levels) 20 ).

Time spent outdoors is well recognised as a factor preventing the development of myopia (evidence: Level I). 20 It is recommended to spend as much time as possible in natural daylight (with appropriate skin protection from ultraviolet radiation), regardless of age. The timing, brightness and UV light exposure play important role in controlling myopia during outdoor activity.28–30
For school-aged children to have clinically significant protection from myopia inducing stimuli, it is advised that the children participate in outdoor activities for at least 1–2 h every day.1,2,22,31
In parallel with more time spent outdoors, it may be beneficial to promote a healthy visual environment to limit the length of continuous time spent at near work: reading at a short distance or using a close-up screen (Level II and III evidence, Table 1). For any reading or other activity performed at short distances, the distance should be increased to a minimum of 20–30 cm.24–27 In addition, one may discuss whether, except for schoolwork, the time spent with reading or with other activities at short distances should be reduced to no more than 30 to 45 min,24,25 followed by an ideal break of 5–10 min (it may be more effective to take longer breaks, such as five minutes per hour, than the well-known 20-20-20 ocular discomfort rules to lower the risk of myopia).32,33 Data issuing from the COVID-19 pandemic has indicated that using TV displays or projectors for online learning is preferable to using tablets, mobile phones, or PC screens when trying to provide a less myopiagenic environment for children. 34
Natural daylight or exceptionally bright indoor light (over 2500 Lux) are beneficial to use indoors (Level III evidence).26,35–40 The WHO's guidance on physical activity, sedentary behaviour and sleep recommends that children under the age of two years avoid using screens at short distances. For children up to five years old, screen time at short distances should be limited to one hour per day. 41 These recommendations were developed for a wider child-health purpose but may also be helpful in relation to myopia. For children aged five to twelve years, the Erasmus Myopia Research Group has argued that a maximum of two hours per day may be a sensible recommendation. 42
Education campaigns for myopia management and eye care awareness are supported by the MyopiaEd toolkit, developed by the World Health Organization (WHO) and the International Telecommunication Union (ITU). The toolkit provides evidence-based messages for targeted digital communication and implementation guidance. 43
A few studies have recently examined the impact of low dose atropine for the prevention or delaying onset of myopia. Atropine at 0.025% and 0.05% was effective in delaying or preventing the onset of myopia in pre-myopes (0.0D to +1.0D) compared to the control group; however, this finding needs to be confirmed through additional research and it is suggested that the concentration should be as low as possible to minimise adverse effects and encourage compliance.13,14 (See a more detailed discussion of atropine treatment in the tertiary prevention section, in points 10–11).
2. The initial step of the algorithm is a cycloplegic refraction (Figure 1). The use of cycloplegia is recommended to establish the diagnosis of pre-myopia or prevent an overdiagnosis of myopia as well as to provide a robust and more repeatable baseline measurement of myopia with which to compare future measures.44,45 If cycloplegia is not performed, the eye care provider must ensure that the eye is unaccommodated.46,47 Consideration of the uncorrected distance vision will aid in confirming or refuting the presence of myopia where a non-cycloplegic result is utilised.
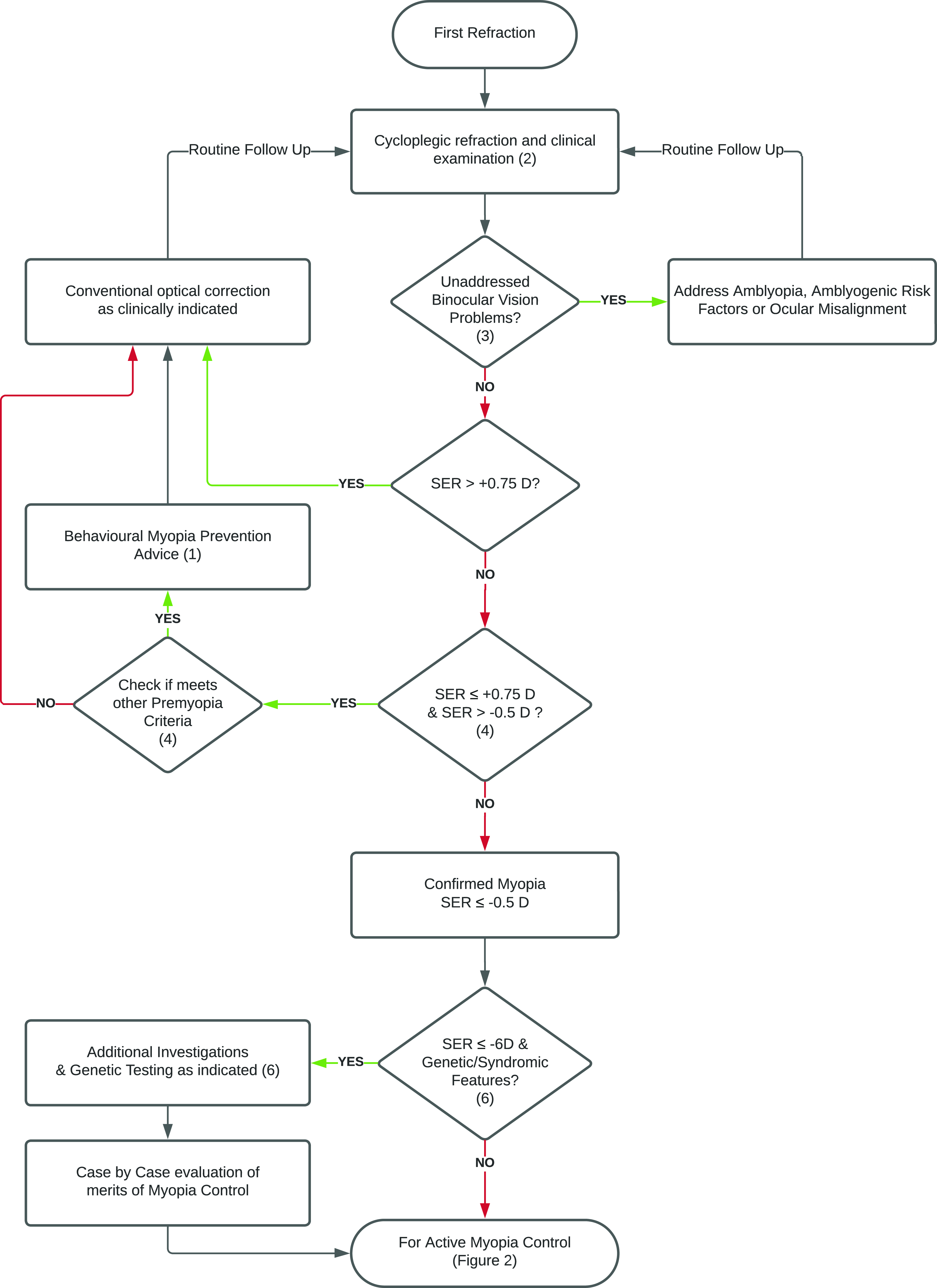
Initial refractive assessment flowchart. Algorithm for the primary and secondary prevention of myopia. Numbers refer to the stages identified in the text. The flowchart needs to be read from top to bottom, however at some decision points, certain responses are going upward and eventually returning to an earlier level of the flowchart, showing the necessity for repeated cycles to monitor and follow-up on myopia development and intervention procedures. (D: diopter, SER: spherical equivalent refraction).
Myopia is not very common (0.2–3.7%) in children under the age of six years, even in Asia,1,48,49 suggesting that a screening examination for myopia may be most effective when conducted around the age of six years. In children with myopia onset at an age of less than 6 years, forms of secondary myopia such as Stickleŕs syndrome and other conditions should be considered/investigated.1,50,51
Screening of pre-myopes (secondary prevention measures)
3. Studies have shown a complex relationship between near-work, ocular accommodation, and myopia development, with no clear causative relationship.
52
Studies have found that the accommodative convergence-to-accommodation (AC/A) ratio is elevated in myopic children and has been documented to be elevated prior to myopia onset, as early as four years prior. However, increased lag of accommodation has not been found to be predictive of myopia onset or progression.
52
Since providing a sharp image on the retina is important, eye care practitioners should still assess the accommodation and convergence systems in all children to ensure normal visual development.
52
4. Pre-myopia is the refractive state of an eye which under cycloplegia has a spherical equivalent between ≤+0.75 D and >−0.5 D. Children aged 6–8 years with pre-myopia have an increased risk of future development of myopia, particularly in conjunction with other quantifiable risk factors such as family history of myopia and ethnicity. 44 Analysis of the risk factors for myopia must be carried out for every child. In addition to quantifiable risk factors such as family history of parental myopia, ethnicity, age, and female sex, lifestyle factors such as less time spent outdoors and more time spent in near work activities should be explored (Table 2).1,19 Monitoring axial length changes is a valuable tool for screening and follow-up of pre-myopes and also for myopia control because the visual consequences of myopia are closely tied to axial elongation.53–55 A growth chart of the eye is a pillar for myopia management. Children who are in the higher percentiles of axial length and those who cross percentiles, indicating more than natural growth, are at increased risk for myopia or even high myopia.56,57
Risk factors associated with prevalence or progression of myopia.

To reduce the risk of myopia, it is recommended to regularly monitor pre-myopes using the axial growth chart percentile curve.56,57 In terms of follow-up, the duration may vary for each individual based on factors such as risk factors and eye length. However, in general, a 6-month follow-up period is recommended.
Reduction of myopia progression (tertiary prevention measures)
5. It is the duty of every eye care provider to proactively discuss myopia control measures with parents and myopic children and offer appropriate interventions (a refractive error of −0.5 D or stronger under cycloplegia) or refer to an appropriate colleague if they do not feel comfortable managing myopia (Figure 2). Although the risk for visual impairment increases to 25% in eyes with an axial length greater than 26 mm and exceeds 90% when the axial length is over 30 mm,
53
it is essential to note that even low and moderate myopia at an early age pose an increased risk.54,55 The need to promote suitable evaluation methods and viable myopia control strategies is essential (environmental, optical, pharmacological). In addition to the optimisation of environmental influences (Tables 1 and 2), the two basic interventions for which there is currently the most evidence to support efficacy are optical and pharmacological.
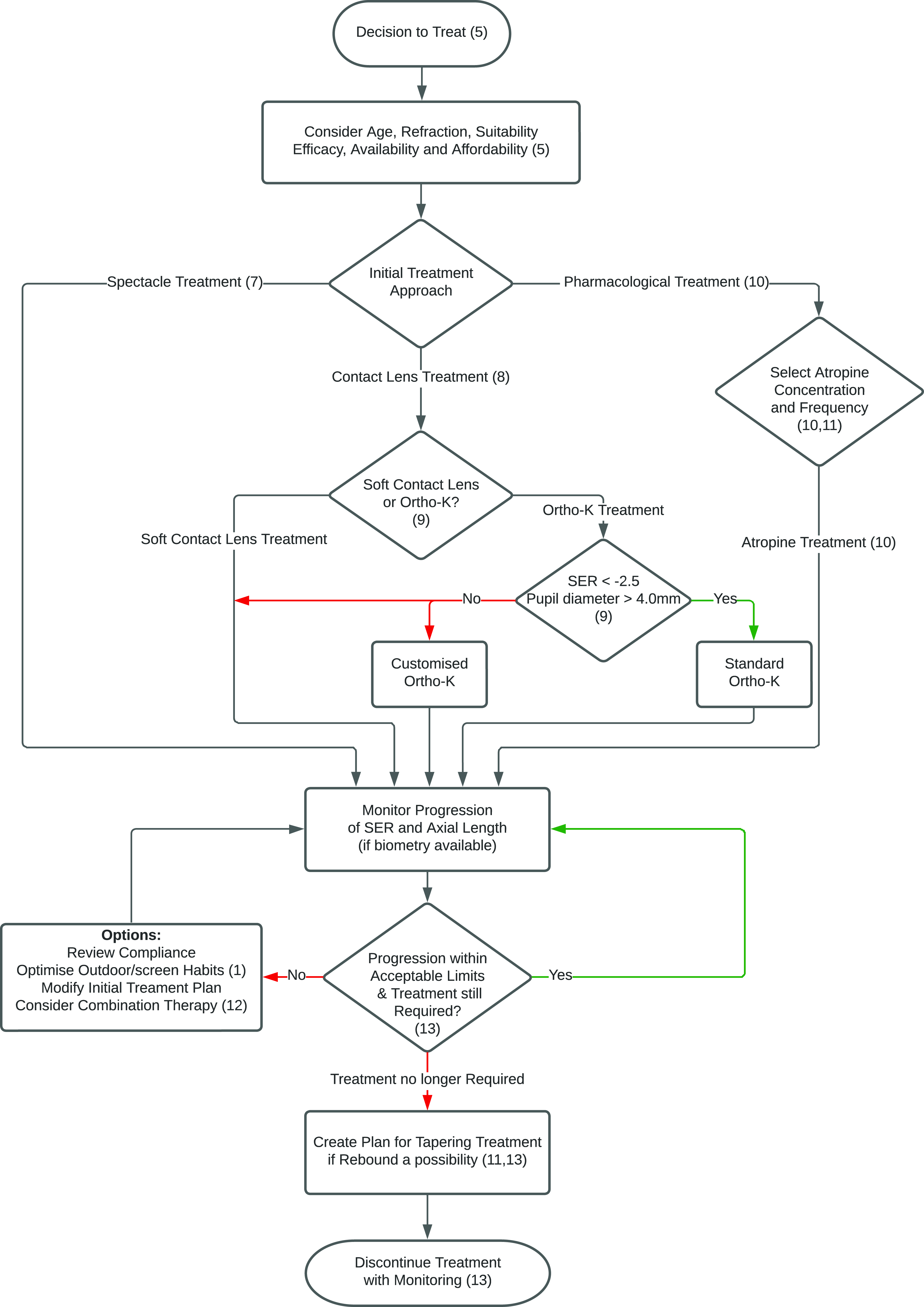
Treatment flowchart. Algorithm for reducing myopia progression. (Ortho-K: orthokeratology, SER: spherical equivalent refraction).
There are several criteria to consider when deciding which option to choose. These include the availability of the option in the patient's country, the practicality of the option given the patient's lifestyle or financial situation, the eye care provider's familiarity with the procedure, and the availability of appropriate instrumentation. If one intervention method fails, another maybe tried, or methods can be combined.1,2,73,74
It should be noted that at present, while these interventions have been shown to be efficacious in many children, children do not all respond equally, and some children do not respond and continue to progress at pace. Eye care practitioners should not over-promise and more research is needed to develop more targeted and personalised approaches to myopia management.
6. After diagnosing high myopia in pre-school children, it's important to determine if there are any associated medical conditions that take priority. A comprehensive clinical history and biometric evaluation are crucial. Further specialised investigations and multidisciplinary evaluations may be necessary, requiring referral to a tertiary care facility following initial diagnosis in primary care. While low-risk interventions can be used for childhood myopia, there's limited evidence of effectiveness for high myopia and syndromic forms. Close monitoring of refraction and axial length is recommended during therapeutic interventions. Conducting randomised clinical trials for myopia interventions in syndromic and monogenic myopia is impractical due to genetic heterogeneity. Pooling outcome data from different clinical sites in disease registries is a viable option for evidence-based myopia management in this complex subtype.
18
7. Do not under-correct patients with myopia1,72,75 (low-certainty evidence, Cochrane-2023)
9
nor over-correct.
76
The blue-light blocking glasses have no effect,
77
and the bifocal or progressive additional eyeglasses have a small effect on myopia control.72,78 8. In some situations, spectacles are not an option. A child may refuse to wear them for various reasons including sport activities during which wearing spectacles is not feasible. Research has demonstrated that the utilisation of contact lenses can enhance the self-perception and engagement of children and teenagers in various activities, resulting in increased satisfaction with the correction of their refractive errors.
46
For spectacle correction, the most recent myopia control spectacle lenses are likely to be a good starting point in younger patients as they have been shown to be effective (with the caveats above) and they are the least invasive option.79–82
When considering myopia management using contact lens corrections the evidence-based options are multifocal soft contact lenses (moderate-certainty evidence, Cochrane-2023),
9
positive spherical aberration contact lenses and night-time orthokeratological lenses (moderate-certainty evidence, Cochrane-2023).
9
Wearing single-vision rigid gas-permeable or soft contact lenses has little to no effect on myopia progression and axial length elongation.72,78
Dealing with higher myopia (−6.0 D or stronger), orthokeratology can be used along with optical overcorrection during the day. 83 However, it is important to note that in this situation, there are other options available, such as using anti-myopia spectacles or daytime soft anti-myopia contact lenses.2,73
In the case of astigmatism, anti-myopia spectacles, regular or customised ortho-K contact lenses may be used. 73 Alternatively, astigmatism could be corrected using spectacle lenses in combination with an anti-myopia contact lens or a soft multifocal toric contact lens could be used, because the uncorrected refractive astigmatism may influence axial elongation. 84
The most serious complication of contact lens wear is microbial keratitis, for which the lifetime risk associated with children using daily disposable soft contact lenses is low (1:431). The risk is higher in orthokeratology (1:67).
85
However, both of these risks are still lower than the risk of developing complications resulting in vision loss associated with high myopia (greater than −6.0 D or AL >26 mm), which has a risk of 1:10, and similar to the risk of developing complications resulting in vision loss associated with lower myopia (less than −3.0 D or AL <26 mm), which ranges from 1:10 to 1:100.
85
9. In conventional orthokeratology lenses, the number of dioptres reduced on the corneal surface aligns with those adjusted in the mid-periphery.
86
This balance plays a crucial role in counteracting the myopic shift in the peripheral retina87–89 and aberrations, particularly spherical aberrations and vertical coma.83,90,91
Pupil size matters. For pupils 4.0 mm or smaller, daytime soft anti-myopic lenses outperform night-time ortho-K lenses.92–97 This is because the ortho-K lens's back optical zone diameter (BOZD), which is responsible for distance visual acuity, is typically 6.0 mm. The slowing effect of soft contact lenses is better in narrow pupils, where this therapeutic zone is within 5.0 mm.
98
The BOZD of an ortho-K contact lens in a case of 6.0 mm relates to a 3.1–3.5 mm treatment zone on the cornea.96,99
The effectiveness of myopia control increases with a greater difference in surface power,93,100,101 as well as with a smaller treatment zone diameter92,94–96,102 (without negatively affecting visual acuity) or a larger pupil size.92,93,95,97
Customised ortho-K lenses can boost the myopic shift effect and offer smaller optical zones for narrow pupils, enhancing their efficacy in slowing progression. However, some authors argue that the diameter of the optic zone has minimal impact on peripheral refraction,
103
and pupil size has no significant influence on myopic progression.
104
Other factors should be considered in addition to their long-term effects on the rate of myopia development.
105
10. Atropine is one of the pillars of myopia control (moderate-certainty evidence, Cochrane-2023).
9
During the past two decades, well-designed clinical studies have reported compelling evidence of its growth inhibiting effect,106–108 but so far, its use has frequently been off-label in many countries. (Further limitations of atropine have been discussed in the introduction.)
12
Atropine inhibits the progression of myopia in a dose-dependent manner, but it also has dose-dependent rebound effects and dose-dependent adverse events.1,73,109
Although in the Low-concentration Atropine for Myopia Progression (LAMP) study, the rebound effect was clinically small at all three concentrations (0.01%, 0.025%, 0.05%), discontinuing treatment at an older age and lower concentrations was associated with a smaller rebound effect.108,109 At a low dosage of 0.01%, atropine has demonstrated conflicting effects on axial elongation. While some studies reported an insufficient effect (statistically not different from the placebo group),107,110,111 several recent studies indicate a notable reduction in axial length growth.10,11,112,113 The frequency of application or dose may be increased when lower doses are proving ineffective.
107
In children who are at risk of developing high myopia in adulthood, it has been recommended by some authors to start with a dose of 0.5% atropine. 114 In order to mitigate any potential adverse reactions to this concentration, the use of photochromic glasses and progressive addition lenses is also recommended. 42
Younger children require a higher (0.05%) concentration of atropine to obtain a similar reduction in myopic progression as compared to older children because younger age is associated with poor treatment response to low concentrations of atropine.
108
According to a recent network meta-analysis, the three concentrations that were most effective for controlling myopia were 1%, 0.5%, and 0.05%. The effects on pupil size and accommodation amplitude were dose-related, and 0.05% was determined to be the most efficient concentration for myopia control as measured by relative risk for total myopia progression.
115
11. After the age of 12 years, children show less rebound after cessation of atropine. This suggests that in children younger than 12 years of age, more consideration should be given before stopping atropine than in children over 12 years of age.116,117 12. In combination therapy, optical interventions are paired with atropine (moderate-certainty evidence, Cochrane-2020).
118
Combination treatment was found to slow myopia progression significantly more than each treatment alone.1,73,119
Evidence based guidelines regarding how to taper doses have not yet been established. Sudden discontinuation of a higher dose of atropine can lead to an increased rate of myopic refractive error progression,
106
although according to some publications, in the case of lower doses, this is not substantiated by accelerated axial length growth.
107
Nevertheless, when children have been treated with higher doses of atropine, a tapering schedule to low doses of atropine is recommended to minimize the risk of a rebound effect.
114
Close axial length monitoring would be useful to monitor for potential rebound during tapering.
Studies demonstrated that combining low-dose atropine concentration with orthokeratology resulted in greater myopia control effect compared to using orthokeratology therapy alone.120–122 The articles describe the combined effect in different ways. The potential mechanism for the combined effect of atropine treatment and orthokeratology lenses is the increased retinal illumination resulting from a larger pupil diameter, which can influence the myopic shift in the peripheral retina and enhance the effect of ortho-K lenses. Moreover, a small but significant increase in pupil diameter leads to higher levels of high-order aberrations, and the elevated aberrations may provide a visual signal that slows eye growth, or the increased choroidal thickness may retard effective eye elongation.
123
There are studies suggesting that Defocus Incorporated Multiple Segments (DIMS) technology combined with 0.01% atropine is more effective than DIMS lenses alone.6,124 However the addition of 0.01% atropine to soft multifocal contact lenses with +2.50-D add powers did not result in a better myopia control than the lenses alone.
125
13. Myopia generally progresses most rapidly during the preteen years (6–12 years).63,64 By the age of 15 years, progression occurs in 52% of cases; by the age of 18 years, it can occur in 23% of cases. At the age of 21–24 years, myopia usually stabilises, except in those with high myopia.64,126,127 Myopia can also occur between the ages of 20 and 30 years, which is a complex process involving a combination of genetic and environmental factors.17,128 Stabilisation is achieved when axial length progression is equal to or less than 0.06 mm per year.128,129 However, it is important to note that axial elongation is age dependent. 57,130
It has been demonstrated that discontinuing the use of ortho-K lenses before the age of 14 years causes axial elongation to accelerate. This indicates that wearing ortho-K should not be discontinued before the age of 14 years or possibly beyond.
131
After cessation of atropine therapy, axial length follow up is needed for an additional 6 to 12 months or longer to ensure there is no rebound effect.
117
Low-dose (0.05%, 0.1%) atropine therapy's effects have been shown to last up to 4.5 years, and the rebound effect may also be less severe in older age after long-term use.
132
Novel methods in myopia management
Current investigations have demonstrated that repeated Low-Level-Red-Light (LLRL) therapy could effectively slow down the progression of myopia.133–139 The therapy utilises desktop laser diode devices that emit long-wavelength light (635–650 nm). The follow-up period in current trials has extended up to 2 years. 136 The treatment effects reported were significant, surpassing those observed with other pharmacologic and optical interventions in terms of magnitude. Furthermore, the LLRL treatment resulted in thickening of the choroid.136,138 Nevertheless, the exact mechanism behind the shrinkage of the axial length remains uncertain and cannot be attributed to changes in choroidal thickness alone. Discontinuation of LLRL therapy showed only modest rebound effects.136,139 To date adverse event monitoring has primarily relied on questionnaires, 136 measures of best-corrected visual acuity, and limited utilisation of optical coherence tomography (OCT) imaging.133,138 This therapy requires further investigation to better understand its effectiveness, underlying mechanisms, and potential long- and short-term adverse events. The latter are particularly important since a recent case report demonstrates an example of possible retinal damage associated with this type of therapy. 140
Conclusions
The role of eye care providers is multi-faceted in ensuring the best available therapy that corresponds to the specific requirements of myopic and pre-myopic children. The proposed algorithm reflects real-life experience that show there is no one method that works for everyone and that myopia management should be tailored to the individual child. The use of axial length growth curve charts and an analysis of family and environmental risk factors can help make well-informed decisions about myopia management and pre-myopic lifestyle advice to delay the onset of myopia. It is important to monitor patients regularly to determine whether the treatment they are receiving is working. If one strategy is not sufficiently effective, a change or combination with another intervention can be considered. By taking these factors into account, we can ensure that we are providing the most effective and personalised management approach for individuals with myopia. The area of myopia management is subject to tremendous research and clinical focus and new evidence, therapies and management options are emerging rapidly. Eye care practitioners have a responsibility to keep up-to-date, respond to the dynamic evidence-base, and ensure their practice continues to provide optimal care for patients.
The SOE myopia consensus group
Angelina Meireles (Sociedade Portuguesa de Oftalmologia, Lisboa, Portugal), Emina Kujundžić Begović (Ophthalmological Society of Bosnia and Herzegovina, Sarajevo, Bosnia and Herzegovina), Zaza D Khotenashvili (Georgia Ophthalmological Society, Tbilisi, Georgia), Camelia Margareta Bogdanici (Romanian Society of Ophthalmology, Iasi, Romania),Steffen Heegaard (Danish Ophthalmological Society, Glostrup, Denmark), Mikheil Omiadze (Georgian Ophthalmology Society, Tbilisi, Georgia), Alexander Stanley Thrane (Norwegian Ophthalmological Society, Oslo, Norway), Iwona Grabska-Liberek (Polish Society of Ophthalmology, Warszawa, Poland), Catherine Creuzot-Garcher (French Society of Ophthalmology, Dijon, France), Hakan Kaymak, (German Ophthalmology Society, Homburg, Germany), Reda Zemaitiene, (Lithuanian Ophthalmological Society, Kaunas, Lithuania), Anat Loewenstein (Israeli Ophthalmological Society, Tel Aviv, Israel), Gunnar Mar Zoega (Icelandic Ophthalmological Society, Reykjavík, Iceland), Benedict Vella Briffa (Malta Association of Ophthalmologists, Msida, Malta), Eugeniu Bendelic (Association of Ophthalmologists of Republic of Moldova, Chisinau, Republic of Moldova), Ilir Arapi (Albanian Ophthalmological Society, Tirana, Albania).
Footnotes
Acknowledgements
The authors would like to thank the International Myopia Institute for the financial support of open access.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JBJ discloses European patent EP 3 271 392, JP 2021-119187, and US 2021 0340237 A1: „Agents for use in the therapeutic or prophylactic treatment of myopia or hyperopia”; AG discloses grants from Alcon, Bausch&Lomb, Zeiss, Teleon, J&J, CooperVision, Hoya, Essilor, Thea, Polpharma, Viatris, consultation: Polpharma, Eyerising, lectures: Thea, Polpharma, Viatris, member of Advisory Boards: Nevakar, GoCheckKids and Thea; JAG discloses editorial board memberships: IOVS, TVST, OPO, grants panel: Fight for Sight UK, consultant for several companies, however the consultancy fee is paid directly by the company to an eye research charity chosen by the company; RCB discloses editorial board memberships: Investigative Ophthalmology and Visual Science (IOVS), Current Eye Research, Scandinavian Journal of Optometry and Visual Science (SJOVS). JRP discloses consultant: Essilor, Hoya, Sightglass, Vyluma, Government funding by ZonMw; JSW discloses consultant: Alcon, Allergan, AOS, Bausch & Lomb, CooperVision, CSIDryEye, DopaVision, M2C Pharmaceuticals, Medmont, Novartis, NuVision, Santen, Scope Ophthalmics, SightGlass, Théa, shares in AstonVision Sciences, Eyoto, Wolffsohn Research Limited, and funding by Alcon, Allergan, Johnson & Johnson Vision, Rayner, M2C Pharmaceuticals, Novartis, NuVision, Scope Ophthalmics, SightGlass, Théa, Topcon, The Eye Doctor; SW is employee of Carl Zeiss Vision International GmbH; SR discloses consultant: BHVI (C), Thea (C); DIF discloses trial support from CooperVision, Topcon and Vyluma Inc., consultant: Coopervision, Thea, Vyluma, EssilorLuxottica, Sightglass, Johnson and Johnson, and commercial interest in Ocumetra Ltd.KJS is in receipt of research funding from Hoya Vision and Vyluma Inc; SJM Is in receipt of research funding from Hoya Vision; HK is consultant for Hoya and Haag-Streit. The other authors declared no potential conflicts of interest with respect to the authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the online publication of this article: The open access fee for this publication is paid by the International Myopia Institute (IMI).
