Abstract
Purpose
To investigate the relationship between serum iron status indicators (ferritin) levels and POAG.
Methods
The files of all glaucoma patients who applied to the ophthalmology clinic between January 2018 and January 2022 were reviewed retrospectively. Laboratory data from fasting blood tests, internal medicine outpatient clinic reports, and extensive ophthalmologic examination data, including fundus photographs showing the optic disc, were collected from the files. A control group was formed from individuals with adequate general and eye health, age- and gender-matched individuals who had undergone examination in the ophthalmology clinic within the same date range. Serum iron status indicators and some laboratory data of POAG patients and healthy controls were compared.
Result
Of our participants, consisting of 65 patients with POAG and 72 healthy controls, 84 (61.32%) were female and 53 (38.68%) were male. It was observed that serum ferritin level was significantly higher in POAG patients compared to healthy controls, and the total iron binding capacity was significantly lower (respectively (p = 0.022), (p = 0.002). In logistic regression analysis, it was found that the risk of POAG increased in cases with high serum ferritin levels (OR = 0.982; p = 0.012). In addition, the risk of POAG was found to increase in cases where MCV was lower (OR = 1.121; p = 0.039).
Conclusion
This study shows that higher serum ferritin levels are associated with a higher risk of POAG.
Introduction
Glaucoma is a progressive optic neuropathy that is the leading cause of preventable irreversible blindness worldwide. 1 This disease may remain asymptomatic until the final stages when irreversible, severe visual impairment occurs. 2 High intraocular pressure (IOP) is the most important known risk factor for the development and progression of primary open-angle glaucoma (POAG), the most common type of ocular hypertension and glaucoma. 3 The high complexity and chronic nature of glaucoma as well as the multifactorial pathogenic mechanisms of retinal ganglion cell loss have led to an incomplete understanding of the etiopathogenesis of the disease. 4 Emerging evidence suggests that glaucoma involves several interacting pathogenetic mechanisms, including the mechanical effects of increased IOP, decreased neutrophin support, hypoxia, excitotoxicity, oxidative stress, autoimmune processes, and inflammation. 4
Oxidative stress can be defined as the deterioration of the balance as a result of an increase in the concentration of reactive oxygen species (ROS) and a decrease in antioxidant levels in the cell. Increasing evidence indicates that ROS plays an important role in the pathogenesis of POAG. 5 Previous studies have reported that free iron, a potent oxidant that accumulates with aging, catalyzes the formation of reactive hydroxyl radicals that can cause oxidative damage. 6 In vitro studies have reported that iron regulation plays a role in the pathogenesis of glaucoma, and there are also studies showing that supplemental iron consumption increases the likelihood of self-reported glaucoma.7–10 Serum ferritin level, which is a well-known indicator of body iron stores, has become both an inflammatory marker for various neurodegenerative diseases and recently an important indicator in the evaluation of iron-induced oxidative stress.6,11 The potential role of serum ferritin levels as a marker of inflammation and oxidative stress underlying glaucoma is intriguing, but to our knowledge, studies focusing on this issue are relatively few in the literature.12,13
In the present study, possible relationships between serum iron status indicators and POAG in individuals of Turkish ethnic origin were investigated and the strength of these relationships was measured.
Materials and methods
Patients and methods
In this retrospective case-control study, archival records (outpatient epicrisis, drug prescriptions, and laboratory data) of primary open-angle glaucoma (POAG) patients with at least 2 years of regular follow-up, who were diagnosed with glaucoma according to criteria of the International Society of Geography and Epidemiological Ophthalmology (ISGEO) in Derince Training and Research Hospital Ophthalmology Clinic, were reviewed. This research was conducted in accordance with the Helsinki Declaration and the approval of the Derince Training and Research Hospital institutional review board.
The records of all POAG patients aged 20 years and older, who were examined in ophthalmology and internal medicine outpatient clinics between January 2018 and January 2022, and whose laboratory data, drug prescriptions, and detailed epicrisis were accessed, were independently evaluated by two trained masked ophthalmologists (SAS, EOT). A control group was formed from individuals with adequate general and eye health, age- and gender-matched individuals who had undergone examination in the ophthalmology clinic within the same date range.
The best-corrected visual acuity (BCVA), intraocular pressure (IOP) measurement using a non-contact tonometer (CT 80; Topcon, Tokyo, Japan), slit-lamp biomicroscopy, gonioscopy, pachymetry, all fundus findings, including optic disc evaluation with color fundus photographs taken with the CR-1 Mark II retinal camera (Canon, Melville, NY, USA), and visual field recordings were enrolled. The serum ferritin level, serum iron level, serum total iron-binding capacity (TIBC) level, serum transferrin saturation level, creatine, serum white blood cell (WBC) level, mean corpuscular volume (MCV), serum high-sensitivity C-reactive protein (hs-CRP) level, triglyceride (TG), high-density lipoprotein (HDL), and low-density lipoprotein (LDL) levels were enrolled. Percentage transferrin saturation is calculated by dividing serum iron by TIBC and multiplying the result by 100. 14 The body mass index (BMI) was calculated by dividing the weight in kilograms by the square of the height in meters. All the participants had a BMI of <30 kg/m2.
Examination data of glaucoma patients included in the study
3rd and 4th-degree angle according to Shaffer staging system in gonioscopic angle examination, absence of pseudoexfoliation material in mydriatic anterior segment examination, IOP elevation (>21 mmHg), asymmetry in vertical pit/disc ratios between the two optic nerves (>0.2) or vertical cup-disc ratio in one eye ≥0.7, focal notching and localized nerve fiber defects compatible with glaucoma and glaucomatous changes in the visual field such as arcuate scotoma, nasal step, Siedel scotoma.
Exclusion criteria
Patients with diabetes, anemia, cardiovascular disease, hypertension, gastrointestinal disease, chronic renal failure, or creatinine value above 1.2 mg/dl, those who take iron supplements or take drugs that may affect serum iron levels, those with high serum high-sensitivity C-reactive protein (hs-CRP) and serum white blood cell (WBC) levels that may affect serum ferritin levels, those with refractive error more than ± 3 diopters (D), glaucoma patients caused by branch or central retinal vein occlusion, uveitis, and diabetic retinopathy, conditions such as various retinal abnormalities and stroke that cause visual field defects that can lead to an incorrect diagnosis of glaucomatous disease, those with any ophthalmologic surgery or trauma, and those with a history of alcohol and smoking were excluded from the study.
Statistical analyses
Statistical analysis was conducted using SPSS version 20.0 for Windows. The Kolmogorov-Smirnov test was used to evaluate the normality distribution of each continuous variable. A chi-square test and the Mann–Whitney U test were used to assess the patients’ demographic parameters. The data are presented as mean values, standard deviations, medians, minimums, and maximums. In order to evaluate the significance of the differences between the groups, the Mann-Whitney U test was used for the data that did not fit the normal distribution, and the independent sample t-test was used for the data with a normal distribution. A binary logistic regression analysis was conducted by building a model that included features that affect the presence of POAG. A p-value of less than 0.05 was considered statistically significant.
Results
This study included 137 participants, with 26 (40%) males and 39 (60%) females in the POAG group and 27 (37.5%) males and 45 (62.4%) females in the healthy control group (Table 1). There was no significant difference between the two groups in terms of age and gender (p-values p = 0.127, p = 0.762, respectively). It was observed that the mean serum ferritin levels of POAG patients were significantly higher than healthy controls (Mean ± Std. Dev and p-value, respectively; 63.96 ± 43.11 mg/ng, 45.35 ± 24.68 mg/ng, and p = 0.022). Total iron binding capacity was found to be lower in POAG patients compared to healthy controls (Mean ± Std. Dev and p-value, respectively; 321.85 ± 56.65 mg/ng, 348.42 ± 45.31 mg/dL, 321.85 ± 56.65 mg/dL, and p = 0.002). When the two groups were compared in terms of MCV, it was observed that the POAG group was lower than the healthy controls, but the level of statistical significance was at the limit (Mean ± Std. Dev and p-value, respectively; 90.1 ± 4.39 fl, 491.44 ± 3.42 fl and p = 0.050). Table 1 shows the demographics, serum ferritin, and iron levels, Transferrin saturation, TIBC, HsCRP, WBC, Hb, MCV, TG, LDL, HDL, and creatine of all the participants.
Descriptive data and serum laboratory parameters of different groups.
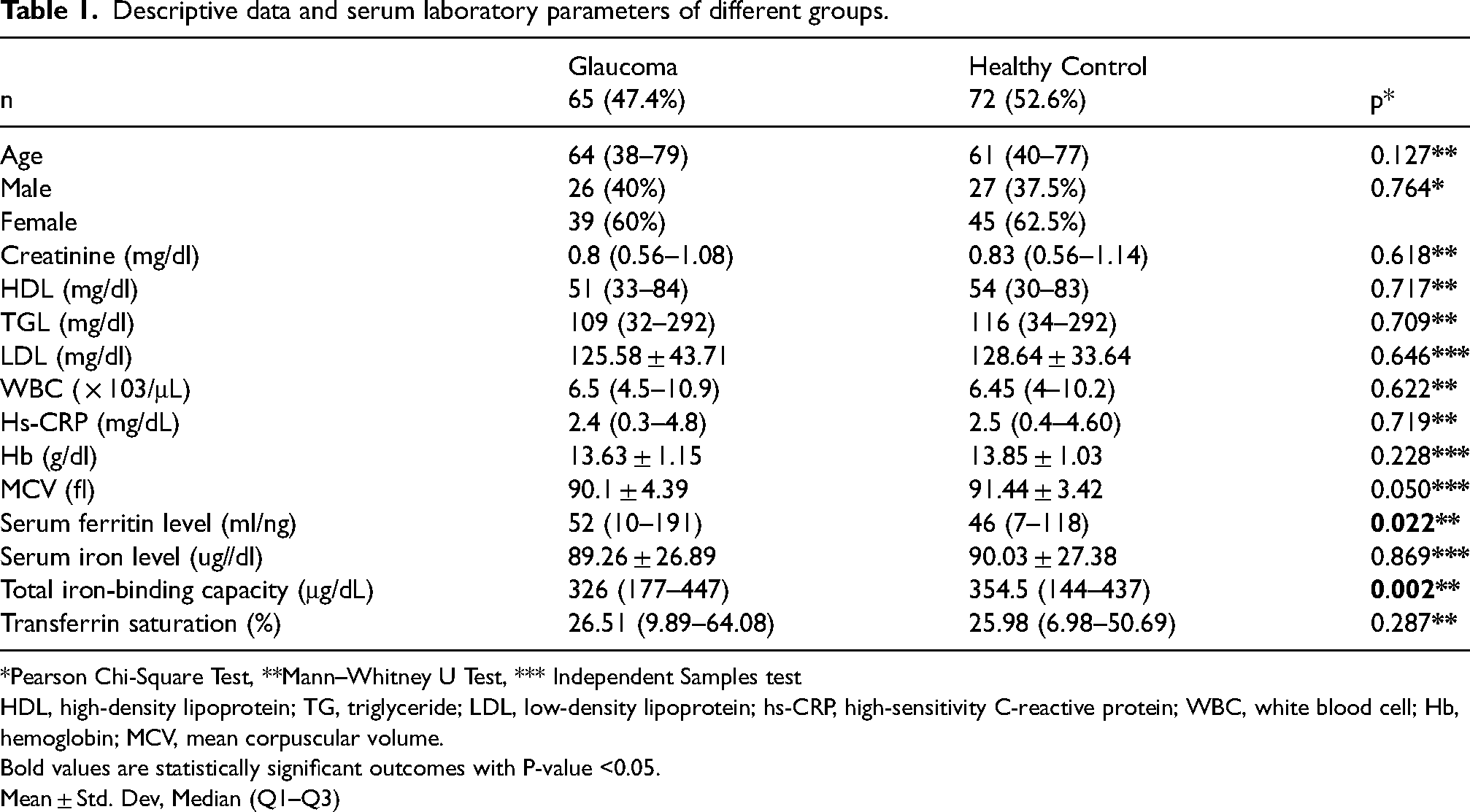
*Pearson Chi-Square Test, **Mann–Whitney U Test, *** Independent Samples test
HDL, high-density lipoprotein; TG, triglyceride; LDL, low-density lipoprotein; hs-CRP, high-sensitivity C-reactive protein; WBC, white blood cell; Hb, hemoglobin; MCV, mean corpuscular volume.
Bold values are statistically significant outcomes with P-value <0.05.
Mean ± Std. Dev, Median (Q1–Q3)
Logistic regression analysis was used to identify risk variables for the presence of POAG. Age, gender, serum ferritin, and iron levels, Transferrin saturation, TIBC, HsCRP, WBC, Hb, MCV, TG, LDL, HDL, and creatine variables were included in the analysis. It was observed that high serum ferritin levels and low MCV levels increased the possibility of POAG development (odds ratio [OR] and p-values, respectively; [OR] = 0.982; p = 0.012, [OR] = 1.121; p = 0.039). When the same logistic regression analysis model was performed separately for men and women, it was observed that high ferritin levels in men increased the probability of developing POAG (odds ratio [OR] and p values, respectively; [OR] = 0.963; p = 0.025), while other variables were not significantly related. In women, there was no significant variation in the probability of developing POAG ([OR] = 0.987; p = 0.175). Logistic regression analysis of POAG and serum laboratory parameters is shown in Table 2.
Logistic regression analysis of POAG and serum laboratory parameters.
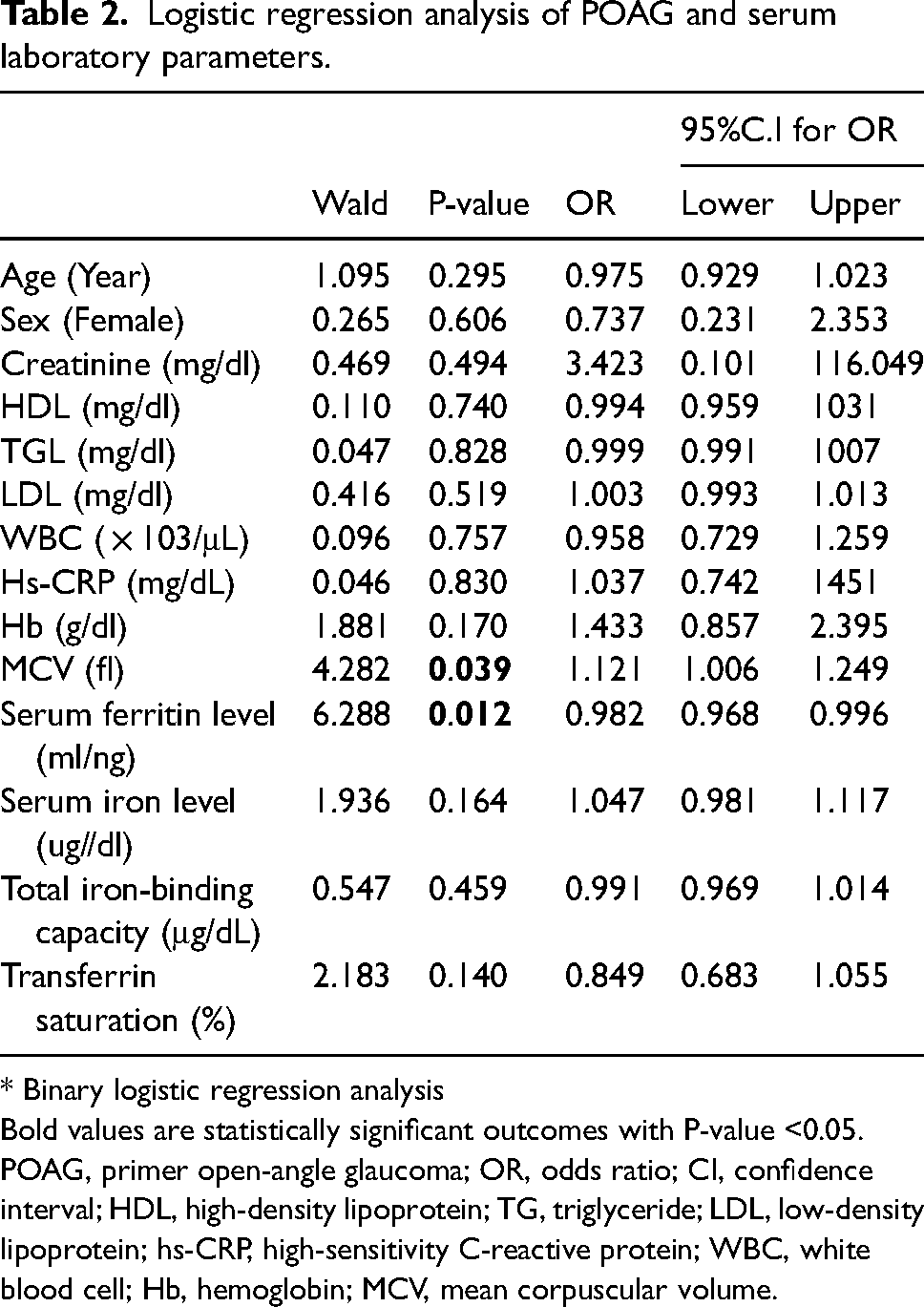
* Binary logistic regression analysis
Bold values are statistically significant outcomes with P-value <0.05.
POAG, primer open-angle glaucoma; OR, odds ratio; CI, confidence interval; HDL, high-density lipoprotein; TG, triglyceride; LDL, low-density lipoprotein; hs-CRP, high-sensitivity C-reactive protein; WBC, white blood cell; Hb, hemoglobin; MCV, mean corpuscular volume.
Discussion
This study presents a comparison of serum iron status indicators (especially ferritin, which is known as both an inflammatory marker and an oxidative stress marker) in POAG and control groups without anemia and without active inflammation (matched in terms of inflammatory factors such as WBC, HsCRP). We observed that the POAG group had higher serum ferritin levels (p = 0.022) and lower TIBC levels (p = 0.002) compared to healthy controls, and a potential association of high serum ferritin ([OR] = 0.982; p = 0.012) and low MCV levels ([OR] = 1.121; p = 0.039) with POAG according to logistic regression analysis.
These results are consistent with one study reporting an association between serum ferritin level greater than 61 ng/mL and glaucoma in a Korean population, 12 and another study in the same population reporting that high serum ferritin level is associated with glaucoma only in males. 13 However, there is a clear difference in mean serum ferritin levels both between the two studies and between them and our study. This may be attributed to the fact that the male and female ratios of the subjects of the 3 studies were different. The mean serum ferritin level reported by Lin et al. 12 and the percentage of males among their subjects were 92.3 ± 6.3 ng/mL and 68.9%, respectively, while the average serum ferritin level reported by Gye et al. 13 and the percentage of males among their subjects were 184.74 ± 151.94 ng/mL and 51.1%, respectively. Our study had a lower percentage of male subjects (40%) than the other two studies, and our mean serum ferritin level was lower (63.96 ± 43.11 mg/ng). It is known that serum ferritin levels are higher in men. 15
Excess tissue iron is known to catalyze the formation of free radicals and poses a significant risk for a wide variety of degenerative diseases, including glaucoma. 16 Previous studies have indicated that iron may play a role in the pathogenesis of glaucoma by affecting aqueous humor outflow and/or retinal ganglion cell survival.7,8 Among the serum iron status indicators, ferritin is the preferred marker for the assessment of iron-induced oxidative stress. 6 Also, Serum ferritin is a well-known acute-phase reactant that reflects the extent of acute and chronic inflammation. 17 Strong positive associations were discovered between serum ferritin and interleukin 6 (IL-6), tissue necrosis factor-alpha (TNF-a), and high-sensitivity C-reactive protein (hsCRP), as well as the presence and/or severity of chronic disease processes. 18 Glaucoma is an extraordinarily complex eye disease in which varying degrees of inflammatory changes may be present along the entire visual pathway from the ocular surface to the posterior segment. 19 Considering the complex effect of ferritin on the mechanisms of inflammation and oxidative stress in the pathogenesis of POAG, the relationship between POAG and other serum iron status indicators, including ferritin, in participants without anemia and with similar laboratory data for inflammation markers is intriguing. To our knowledge, two studies12,13 involving the Korean population reported the association between serum ferritin levels and glaucoma prevalence, while two other studies9,10 suggested the importance of the effect of dietary iron intake on glaucoma prevalence. In this study, similar to the study of Gye et al., 13 in the logistic regression analyses performed to determine the risk variables for POAG in male and female genders, high ferritin levels and increased risk of POAG were observed in men, but no significant relationship was observed in women. Similarly, no significant relationship was observed in women in our study. We can think that the reason for this situation is due to the difference in serum ferritin levels between the sexes. Previous research has reported that men have higher lifetime iron stores than women, especially during the premenopausal period. 15 The finding that men with high serum ferritin levels have a higher risk of glaucoma than those with low serum ferritin levels supports our view. 13
In addition, although there was no statistically significant difference between our findings, MCV levels were higher in the healthy control group, and a significant relationship was observed between glaucoma and low MCV levels. Young erythrocytes are known to have a higher MCV. In a study investigating oxidative damage in rats after an acute strenuous exercise, it was reported that a slight but significant decrease was observed in MCV. 20 Low determination of MCV in POAG may be associated with oxidative damage in the pathogenesis of glaucoma. Although POAG was found to be lower in this study compared to the TIBC control group, no significant association was observed in the regression analysis. In the study of Gye et al., 13 in which they compared TIBC levels according to the presence of glaucoma, lower TIBC was reported in the glaucoma group, but no significant relationship with glaucoma was reported for either gender.
A recent article includes all publications measuring iron indicators in the serum, aqueous humor, and tears of patients with glaucoma and in the retinal tissue layers of primates. 21 In addition to studies reporting increased ferritin and transferrin levels in the retinas of monkeys with glaucoma and humans with glaucoma,22,23 there are several studies reporting increased levels of transferrin in the aqueous humors of people with PAAG, 24 and increased iron levels in the tears of people with glaucoma. 25 Also, when human aqueous humor iron levels were examined, it was determined that they were low in pseudo-exfoliating glaucoma and high in POAG.26,27 Higher transferrin, ferritin, and iron levels and lower ceruloplasmin levels were measured in the serum of patients with glaucoma compared to controls.13,27,28
This study has several limitations, such as a small sample size and retrospective design. The nature of retrospective studies led us to examine the subjects at a particular time. All the laboratory data examined, including the ferritin level of the patients in both groups, were obtained from laboratory analyses requested by the internal medicine department for different reasons. Therefore, there may be a selection bias. In addition, this was a cross-sectional case-control study, which did not offer the possibility to determine causality, but was limited to reporting possible associations between POAG and the few available biomarkers. Despite all the limitations of retrospective cross-sectional studies, we think that this study will lay the groundwork for future large prospective clinical studies.
Conclusions
Although the mechanisms by which ferritin may contribute to the formation of POAG are not clearly understood, in this study, as in the limited number of studies in the literature, a correlation was found between the formation of POAG and high serum ferritin levels. In addition, as far as we know, we observed a relationship between POAG and low MCV as a new finding. More prospective comparative studies with longer follow-ups are needed to confirm these associations.
Footnotes
Author contribution
Sevim Ayça Seyyar: Study design, data acquisition, manuscript; Ecem Onder Tokuç: Manuscript editing, research, data acquisition; Kıvanç Güngör: Manuscript editing; All authors read and approved the final manuscript.
Data availability
The data used to support the findings of this study are included in the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics committee number
Derince Training and Research Hospital ethics committee 2022-69
This study has a retrospective randomized clinical design.
Informed consent not addressed
A study containing data from human subjects. It was carried out with the informed consent of the participants in accordance with the Declaration of Helsinki.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
