Abstract
Introduction
A detailed understanding of the anatomical and structural changes occurring in the retina following intravitreal fluocinolone acetonide implantation may help improve the management and prognosis of persistent or recurrent diabetic macular edema (DME).
Methods
Overall, 45 eyes (from 35 patients) with refractory center-involved DME received an intravitreal fluocinolone acetonide implant. They were monitored at baseline and at 6, 12, 24, and 36 months for best-corrected visual acuity (BCVA), central foveal thickness (CFT), and the seven retinal parameters used in the classification of diabetic maculopathy recently developed at the European School for Advanced Studies in Ophthalmology (ESASO).
Results
Within 6 months of implantation, significant improvements were evident in BCVA, CFT, maculopathy stage, and the percentage of eyes with: intraretinal cysts; CFT > 30% above the upper normal value; and disrupted or absent ellipsoid zone (EZ) and/or external limiting membrane (ELM). Significant improvements were still maintained at 36 months post-implantation. At month 36, early treatment with the implant (i.e., after < 6 previous intravitreal injections for DME) trended toward being more effective than later treatment in improving BCVA, CFT, maculopathy stage, and the percentage of eyes with CFT > 30% above the upper normal value. However, statistical significance was not achieved.
Conclusion
In persistent or recurrent DME, fluocinolone acetonide implantation can be effective in improving maculopathy stage and reducing the percentage of eyes with: intraretinal cysts; CFT > 30% above the upper normal value; and disrupted or absent EZ and/or ELM. It can also increase BCVA and reduce CFT.
Keywords
Introduction
Macular edema is the major cause of vision loss in patients with diabetes mellitus. 1 In a healthy retina, fluid balance is controlled by an intact blood-retinal barrier (BRB) and by the normal functioning of Müller cells and the retinal pigment epithelium (RPE).2,3 However, in diabetic macular edema (DME), breakdown of the inner BRB and dysfunction of the Müller cells and RPE can lead to intracellular and extracellular edema. 2 Breakdown of the BRB is initially reversible but can lead to irreversible damage if the edema overcomes the stretching capacity of the retina, damaging bipolar axons and disrupting the transmission of visual signals. These structural changes mean that later attempts to resolve the edema may not lead to proportional recovery of vision. 4 Thus, to optimize the chances of improving visual acuity, early resolution of edema is paramount before structural changes become irreversible. Early resolution of edema is likely also important in preventing visual outcomes from worsening as a result of fluctuating central subfield thickness. 5
Despite central macular edema/retinal thickness often being used to determine the need for intravitreal treatment of DME, it is a relatively poor surrogate for retinal function as it does not correlate closely with visual acuity, and it is not predictive of final outcomes.4,6 Although reducing extracellular fluid may improve visual acuity up to a point, further drying may not necessarily improve vision any further 4 as neurons and glial cells may be lost in the process. The location of accumulated fluid can also be important — a greater height of intraretinal cystoid fluid is associated with poorer visual acuity whereas the presence of subretinal fluid can enhance visual acuity gain during anti-vascular endothelial growth factor (anti-VEGF) treatment. 7 Such enhancement is believed to be attributable to the integrity of the ellipsoid layer (which is important for visual acuity) being maintained in the presence of subretinal fluid. 4 (An intact photoreceptor layer at baseline is also known to be associated with better functional responses to intravitreal corticosteroid treatment. 8 )
A greater understanding of retinal characteristics that reflect the severity of diabetic retinopathy may help the management of DME and the design of clinical trials. 9 With the advent of high resolution retinal imaging using spectral domain optical coherence tomography (SD-OCT), DME can now be characterized not just in terms of retinal thickness but also in many other ways, including: the integrity of the ellipsoid zone (EZ) and external limiting membrane (ELM); the size of intraretinal cysts; the number of hyperreflective foci; and the presence or absence of disorganized retinal inner layers (DRIL), subretinal fluid, and vitreoretinal interface abnormalities. Some of these may be useful predictors of DME severity, progression, or prognosis. 10
An international panel of experts recently proposed a classification system that stages diabetic maculopathy simply and objectively so it can be used in the evaluation and prognosis of DME by both non-retinal experts and retinal experts, and in both everyday clinical practice and clinical studies. 11 Developed at the European School for Advanced Studies in Ophthalmology (ESASO), this system evaluates SD-OCT images by grading seven retinal parameters: retinal thickening; cysts; EZ and/or ELM status; DRIL; hyperreflective foci; subretinal fluid; and vitreoretinal relationship. The first four of these parameters are used to identify the stage of maculopathy along the continuum of DME progression (as early DME, advanced DME, severe DME, or atrophic maculopathy). These four stages reflect the progressive severity of the condition, which can be reversible in the early stages but irreversible in the later stages. Early DME commonly corresponds to good visual acuity and a shorter duration of hyperglycemia, whereas atrophic maculopathy is generally secondary to long-standing macular edema. Identifying a patient's stage of maculopathy is critical for setting expectations and optimizing treatment management.
This study sought to use the ESASO criteria to characterize the response of eyes with persistent or recurrent DME to an intravitreal fluocinolone acetonide implant (ILUVIEN, Alimera Sciences Ltd., Aldershot, UK) to obtain a more detailed understanding of the effects of such treatment on the retina. In Europe, this implant is indicated for the treatment of vision impairment associated with chronic DME considered insufficiently responsive to available therapies (such as anti-VEGF — which is considered the first-line therapy for the majority of eyes with DME — and short-term corticosteroid intravitreal injections). 12 The implant contains 190 μg fluocinolone acetonide, which is designed to be released continuously at a microdose of ∼0.2 μg/day for up to 3 years — thereby offering the potential for sustained and long-lasting effectiveness and a reduction in treatment burden relative to multiple intravitreal injections. In both clinical trials 13 and real-world studies, 14 a single implant has been shown to offer sustained structural and functional improvements for up to 3 years in eyes with persistent or recurrent DME. Furthermore, it appears similarly effective in vitrectomized and non-vitrectomized eyes. 15
Methods
Study design and objective
This study is a retrospective cohort study enrolling eyes that had received a single intravitreal fluocinolone acetonide implant (ILUVIEN) to treat persistent or recurrent DME. The primary objective was to determine the effect of this treatment on the seven retinal parameters of the ESASO classification system, and on the stage of maculopathy, at 6, 12, 24, and 36 months after implantation. The study was approved by the Ethics Committee of Centro Hospitalar Universitário do Porto (2017.093 [084-DEFI/082-CES]) and conducted according to the Declaration of Helsinki (2013 amendment). All patients signed informed consent.
Patients
Clinical records from consecutive adults being treated by an ophthalmologist at a university hospital (Centro Hospitalar Universitário de Santo António, Porto, Portugal) were eligible for evaluation if the eye had received, at least 36 months previously, its first fluocinolone acetonide implant (ILUVIEN) for center-involved DME (central foveal thickness [CFT] > 315 μm in the presence of intraretinal fluid). The DME was required to have been present for at least 24 months at the time of the implantation and to be refractory to intravitreal anti-VEGF or short-term corticosteroid treatment (refractoriness being defined as persistent intraretinal fluid and/or subretinal fluid on SD-OCT, regardless of visual acuity, after ≥ 3 monthly anti-VEGF injections or at least 6 months after injection of dexamethasone or triamcinolone acetonide). Exclusion criteria included: any other concomitant ocular disease, except cataract, that could cause macular edema or compromise visual acuity; a history of ocular trauma or intraocular surgery (except uncomplicated cataract extraction or vitrectomy at least 3 or 6 months, respectively, before fluocinolone acetonide implantation); and short-acting intraocular corticosteroid treatment in the 6 months before the fluocinolone acetonide implantation.
A subgroup analysis was performed to compare eyes that had previously received < 6 versus ≥ 6 intravitreal anti-VEGF and/or corticosteroid injections for DME before fluocinolone acetonide implantation. These were termed the early switch and late switch subgroups.
Clinical outcomes
Patient demographic information was evaluated at baseline (prior to fluocinolone acetonide implantation) and best-corrected visual acuity (BCVA) data and SD-OCT scans were evaluated at baseline and months 6, 12, 24, and 36. BCVA was assessed using Early Treatment Diabetic Retinopathy Study (ETDRS) letters. SD-OCT scans were obtained using a Heidelberg Spectralis® HRA + OCT imaging platform, version 1.10.2.0 (Heidelberg Engineering, Heidelberg, Germany) using a macular scan with high resolution mode spanning a 20 × 20° frame. Scans were evaluated by two highly trained medical doctors (JL and CC) with disagreements resolved by a senior medical retinal specialist (BP). The evaluators were not masked to clinical information or timepoints.
The SD-OCT scans were evaluated for the percentage of eyes having each of the ESASO criteria:
– CFT > 30% above the upper limit of normal using Spectralis SD-OCT (315 μm), so > 410 μm – Subretinal fluid – Intraretinal cysts – > 30 hyperreflective foci with < 30 μm diameter, reflectivity similar to the nerve fiber layer, and no backshadowing – DRIL – Disrupted or absent EZ and/or ELM – Vitreomacular interface abnormalities (any visible adhesion or traction between the vitreous cortex and the retina).
These retinal parameters are associated with macular edema/inflammation (CFT, subretinal fluid, and intraretinal cysts) or with structural changes in the retina (hyperreflective foci, DRIL, EZ/ELM, and vitreomacular abnormalities). Subretinal fluid, intraretinal cysts, hyperreflective foci, DRIL, and EZ/ELM status were evaluated within the central 1 mm of five line scans (the foveal scan and the two above and two below it).
We staged the diabetic maculopathy according to the ESASO classification. 11 Thus, if the EZ/ELM was intact, and CFT was more than 10% but less than 30% higher than the upper normal value of 315 μm, 16 maculopathy was classified as early. 11 If the EZ/ELM was intact and CFT was > 30% higher than the upper normal value, or if the EZ/ELM was disrupted, maculopathy was classified as advanced. If the EZ/ELM was absent, maculopathy was classified as either severe (if CFT was > 10% higher than the upper normal value) or atrophic (if CFT was < 10% higher than the upper normal value).
Any supplementary therapies administered during the follow-up period were noted.
Statistical analyses
All statistical analyses were performed using Stata® software, release 14 (StataCorp LLC, College Station, Texas, USA). Analyses excluded missing data and p values of < 0.05 were considered statistically significant. Paired sample means and paired sample medians of BCVA and CFT were compared with baseline using Student's t test and the Wilcoxon test, respectively. The percentage of eyes with each retinal parameter, and the overall distribution of maculopathy stage, were compared with baseline using the McNemar test.
Across the early switch and late switch subgroups, continuous variables were compared using Student's t-test with Welch correction for inequality of variance, and categorical variables were compared using Fisher's exact Chi-test.
Results
Overall, 45 eyes were enrolled in the study from 35 patients, with fluocinolone acetonide implantations performed between 2 April 2015 and 5 June 2017. At baseline, mean BCVA was 42 ETDRS letters and mean CFT was 546 μm. Maculopathy stage was classified as advanced in 73% of eyes and early in 27% of eyes. Baseline demographics show no significant differences between the early and late switch subgroups except the late switch group had a relatively greater mean duration of DME and a greater number of previous anti-VEGF and corticosteroid injections for DME (Table 1).
Baseline demographics.
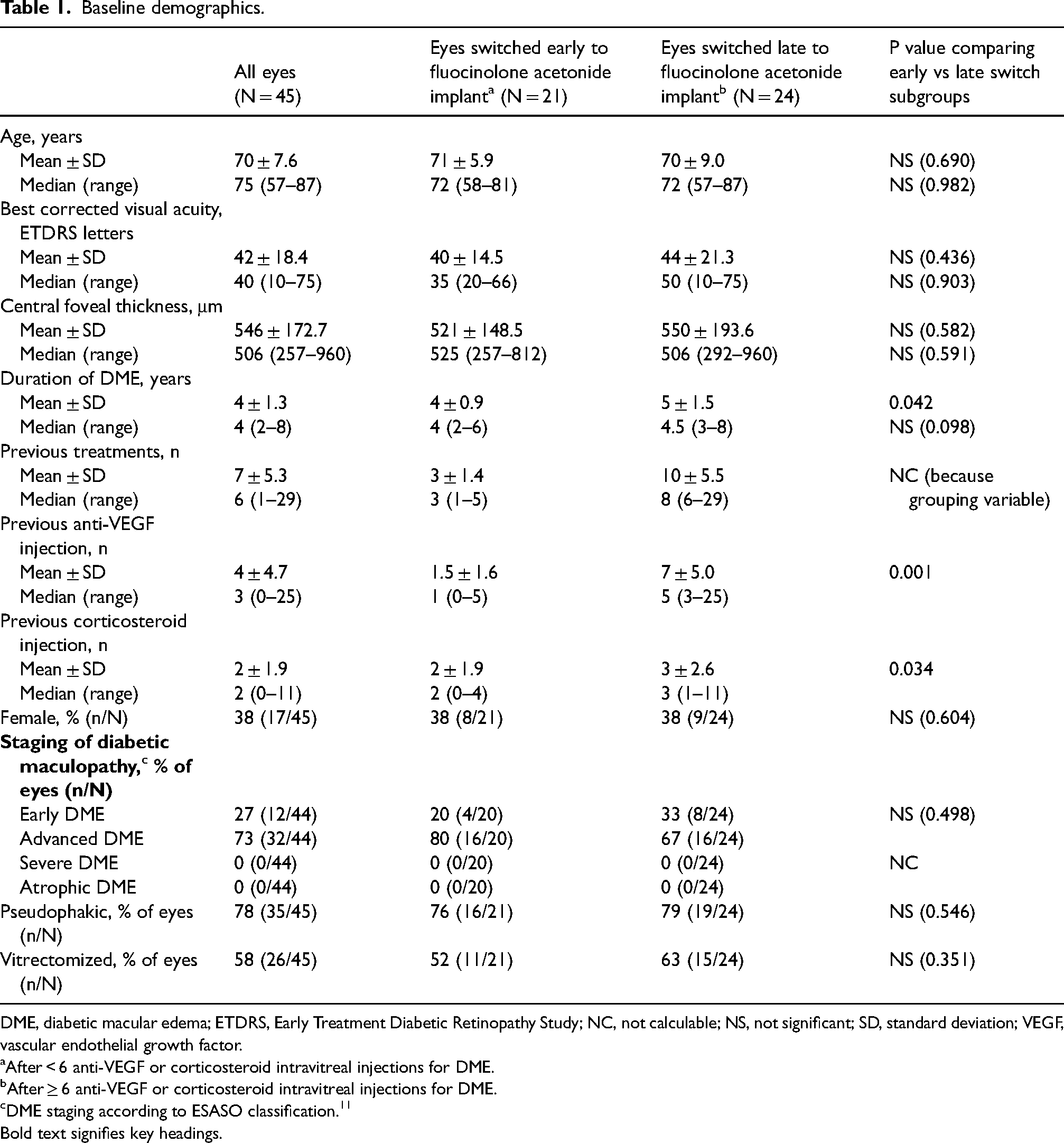
DME, diabetic macular edema; ETDRS, Early Treatment Diabetic Retinopathy Study; NC, not calculable; NS, not significant; SD, standard deviation; VEGF, vascular endothelial growth factor.
After < 6 anti-VEGF or corticosteroid intravitreal injections for DME.
After ≥ 6 anti-VEGF or corticosteroid intravitreal injections for DME.
DME staging according to ESASO classification. 11
Bold text signifies key headings.
BCVA and CFT
Fluocinolone acetonide implantation was associated with a significant increase in BCVA and a significant decline in CFT (p < 0.001 from baseline at all timepoints; Figure 1 and Table 2). At month 36, the increase from baseline in mean BCVA was numerically larger in the early switch group than in the late switch group (19 versus 9 ETDRS letters) (Figure 1 and Table 3). Similarly, the decline from baseline in median CFT was numerically larger in the early switch group than in the late switch group (268 versus 178 μm) (Figure 1 and Table 3).
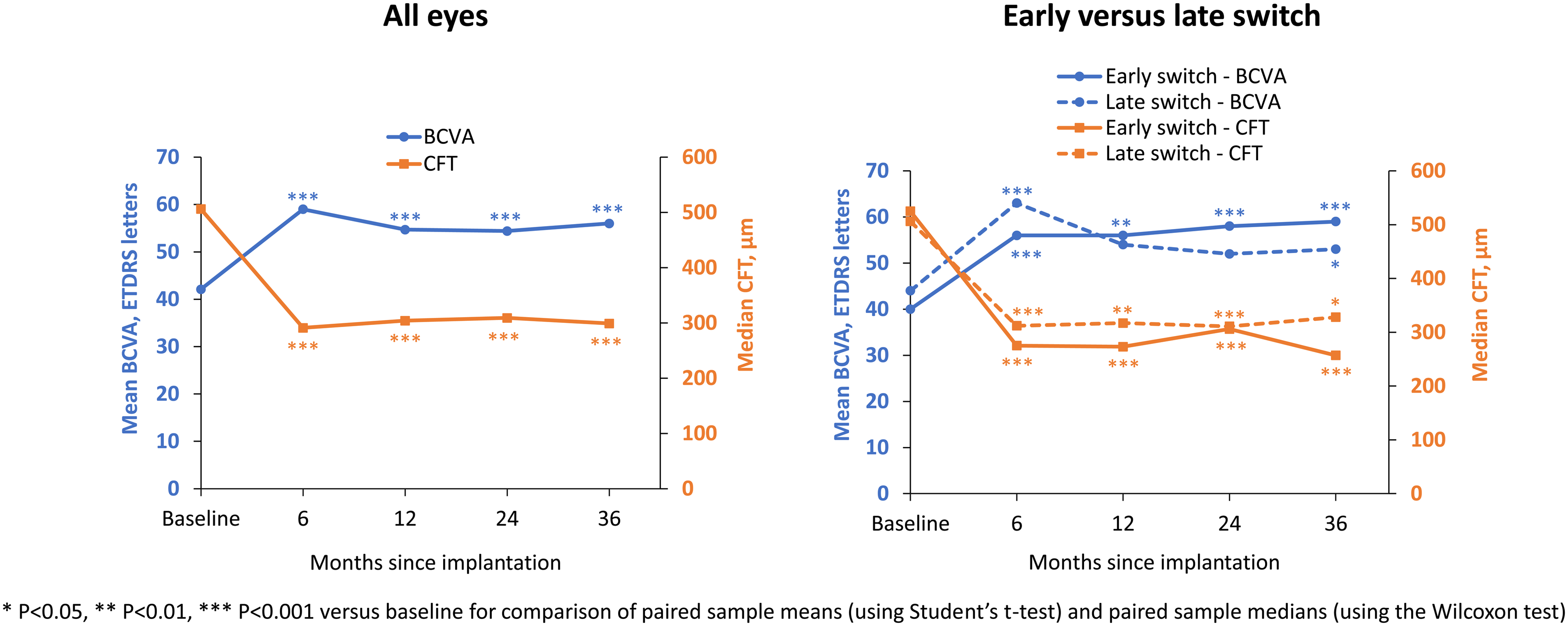
Improvement in best-corrected visual acuity (BCVA) and central foveal thickness (CFT) after fluocinolone acetonide implantation. Left graph, all eyes (N = 45). Right graph, subgroups of eyes switched to corticosteroid either early (n = 21) or late (n = 24). Early switch was after < 6 intravitreal injections for diabetic macular edema (DME), late switch was after ≥ 6 intravitreal injections for DME.
Outcomes across all patients.
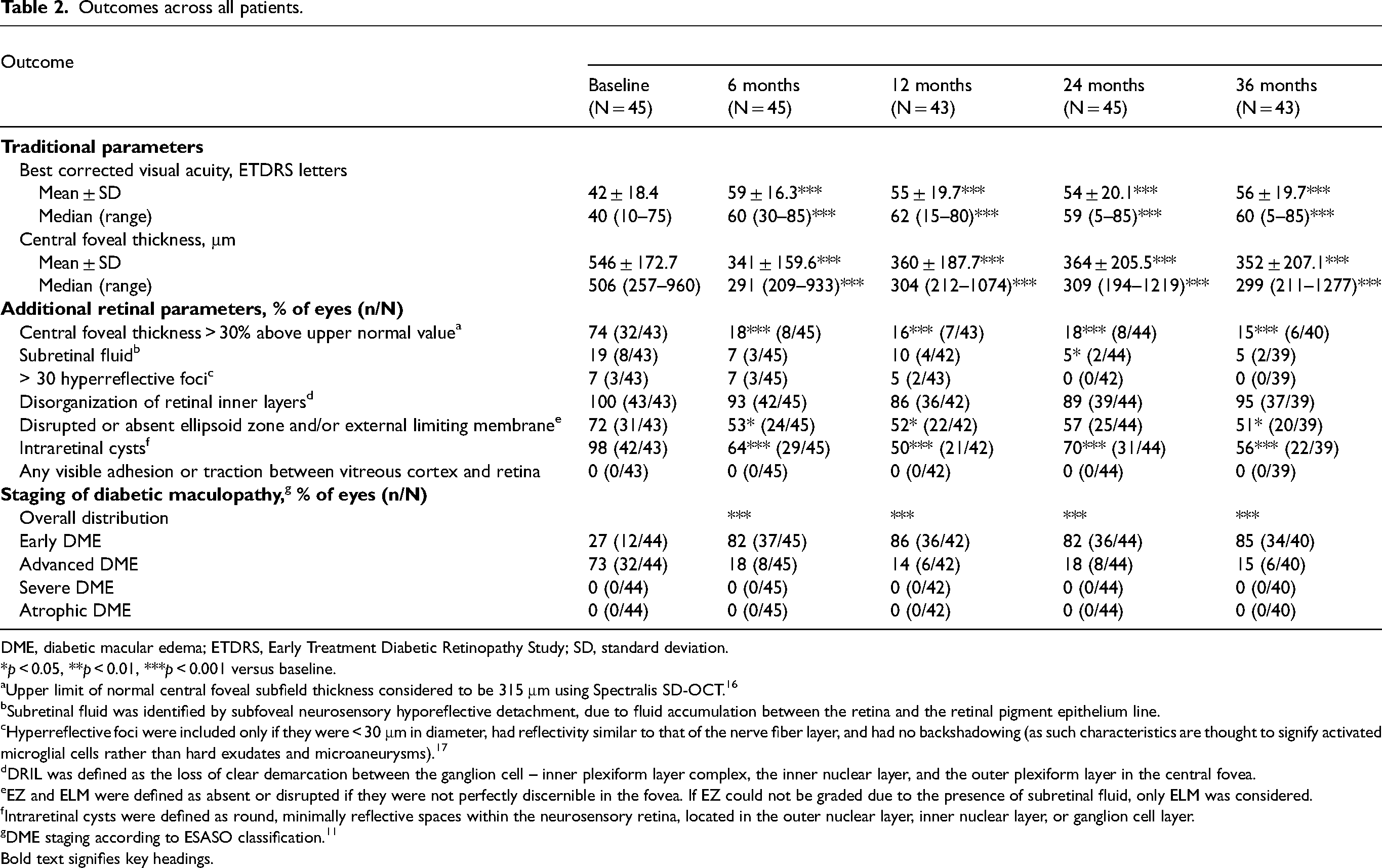
DME, diabetic macular edema; ETDRS, Early Treatment Diabetic Retinopathy Study; SD, standard deviation.
*p < 0.05, **p < 0.01, ***p < 0.001 versus baseline.
Upper limit of normal central foveal subfield thickness considered to be 315 μm using Spectralis SD-OCT. 16
Subretinal fluid was identified by subfoveal neurosensory hyporeflective detachment, due to fluid accumulation between the retina and the retinal pigment epithelium line.
Hyperreflective foci were included only if they were < 30 μm in diameter, had reflectivity similar to that of the nerve fiber layer, and had no backshadowing (as such characteristics are thought to signify activated microglial cells rather than hard exudates and microaneurysms). 17
DRIL was defined as the loss of clear demarcation between the ganglion cell – inner plexiform layer complex, the inner nuclear layer, and the outer plexiform layer in the central fovea.
EZ and ELM were defined as absent or disrupted if they were not perfectly discernible in the fovea. If EZ could not be graded due to the presence of subretinal fluid, only ELM was considered.
Intraretinal cysts were defined as round, minimally reflective spaces within the neurosensory retina, located in the outer nuclear layer, inner nuclear layer, or ganglion cell layer.
DME staging according to ESASO classification. 11
Bold text signifies key headings.
Outcomes among early switch and late switch subgroups.
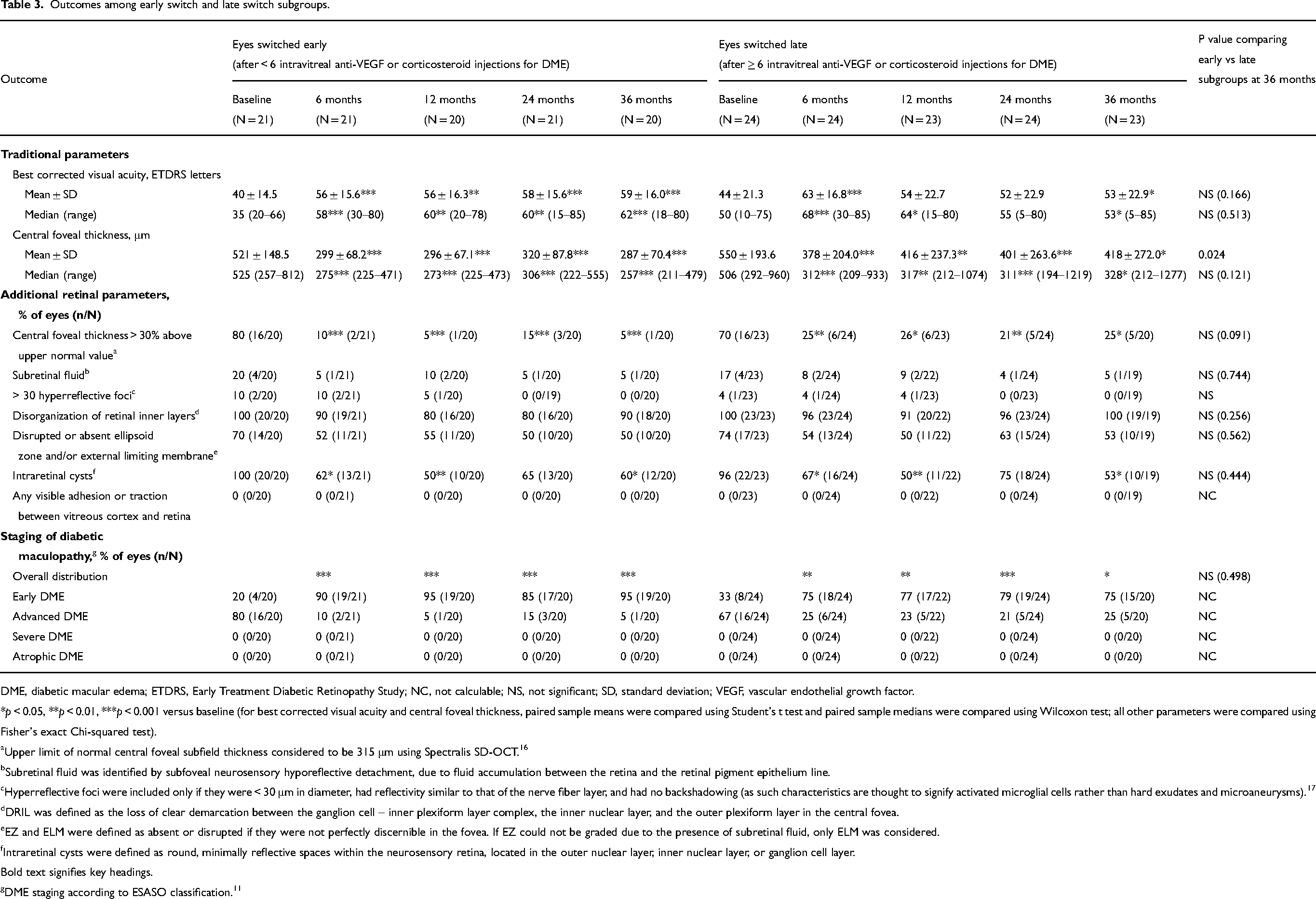
DME, diabetic macular edema; ETDRS, Early Treatment Diabetic Retinopathy Study; NC, not calculable; NS, not significant; SD, standard deviation; VEGF, vascular endothelial growth factor.
*p < 0.05, **p < 0.01, ***p < 0.001 versus baseline (for best corrected visual acuity and central foveal thickness, paired sample means were compared using Student's t test and paired sample medians were compared using Wilcoxon test; all other parameters were compared using Fisher's exact Chi-squared test).
Upper limit of normal central foveal subfield thickness considered to be 315 μm using Spectralis SD-OCT. 16
Subretinal fluid was identified by subfoveal neurosensory hyporeflective detachment, due to fluid accumulation between the retina and the retinal pigment epithelium line.
Hyperreflective foci were included only if they were < 30 μm in diameter, had reflectivity similar to that of the nerve fiber layer, and had no backshadowing (as such characteristics are thought to signify activated microglial cells rather than hard exudates and microaneurysms). 17
DRIL was defined as the loss of clear demarcation between the ganglion cell – inner plexiform layer complex, the inner nuclear layer, and the outer plexiform layer in the central fovea.
EZ and ELM were defined as absent or disrupted if they were not perfectly discernible in the fovea. If EZ could not be graded due to the presence of subretinal fluid, only ELM was considered.
Intraretinal cysts were defined as round, minimally reflective spaces within the neurosensory retina, located in the outer nuclear layer, inner nuclear layer, or ganglion cell layer.
Bold text signifies key headings.
DME staging according to ESASO classification. 11
Retinal parameters
Between baseline and month 36, the percentage of eyes with the following retinal parameters declined (i.e., improved) from: 74% to 15% for CFT > 30% above the upper normal value (p < 0.001); 98% to 56% for intraretinal cysts (p < 0.001); 72% to 51% for disrupted or absent EZ/ELM (p < 0.05); 19% to 5% for subretinal fluid (NS); 7% to 0% for > 30 hyperreflective foci (NS); and 100% to 95% for DRIL (NS) (Table 2 and Figure 2).
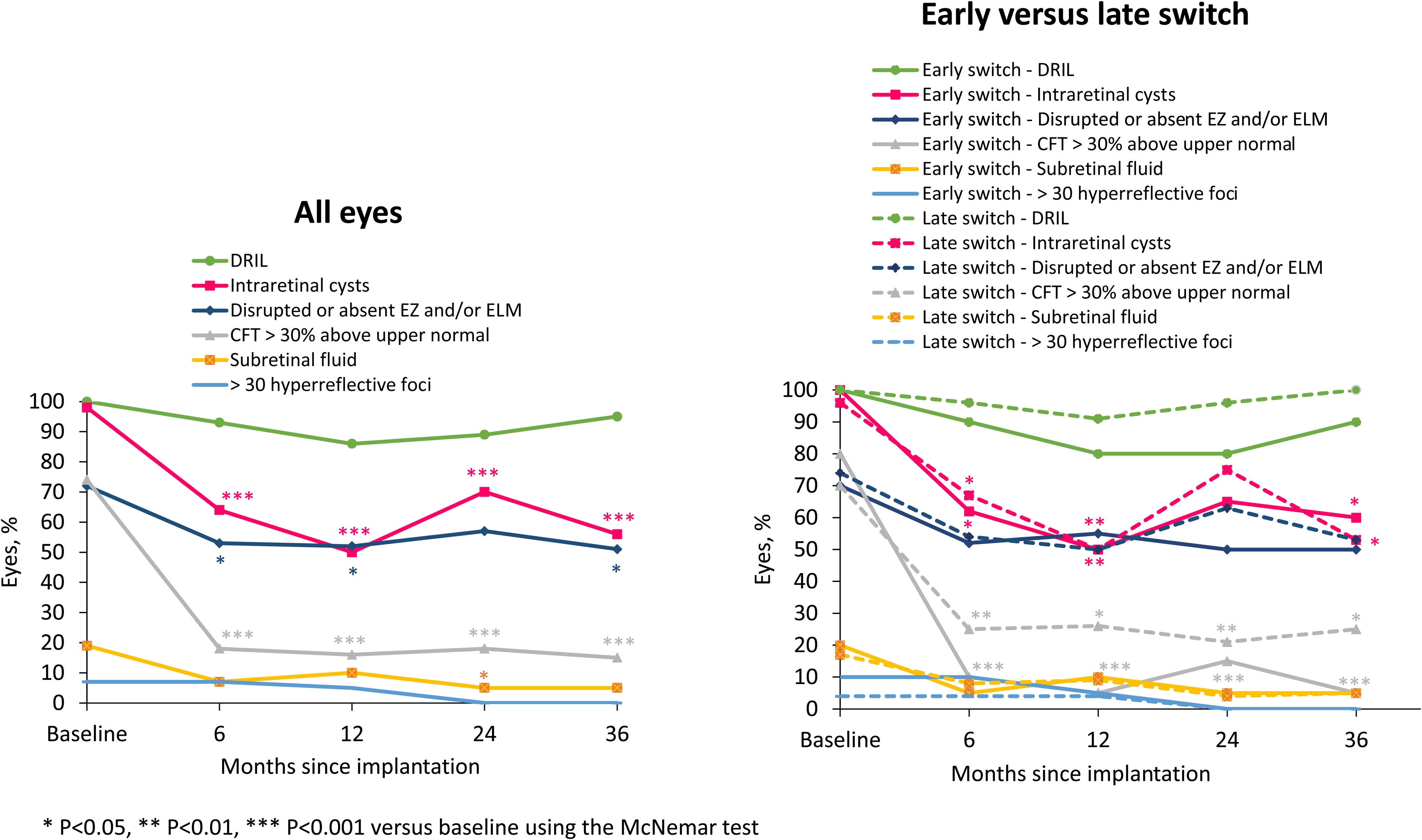
Change in retinal parameters after fluocinolone acetonide implantation. Left graph, all eyes (N = 45). Right graph, subgroups of eyes switched to corticosteroid either early (n = 21) or late (n = 24). Early switch was after < 6 intravitreal injections for diabetic macular edema (DME), late switch was after ≥ 6 intravitreal injections for DME. CFT, central foveal thickness; DRIL, disorganized retinal inner layers; ELM, external limiting membrane; EZ, ellipsoid zone.
The reduction in the percentage of eyes with each retinal parameter was consistently greater in the early switch group than the late switch group for CFT > 30% above upper normal value (75% reduction from baseline at month 36 in the early switch group versus 45% in the late switch group [80%‐5% and 70%‐25%, respectively]) and for DRIL (10% versus 0% at month 36 [100%‐90% and 100%‐100%, respectively]) (Figure 2 and Table 3). For the other retinal parameters, the percentage of eyes affected appeared comparable between the early and late switch groups. No vitreomacular interface abnormalities were observed at baseline or any later timepoint.
Maculopathy stage
Fluocinolone acetonide implantation was associated with a significant change in the distribution of maculopathy stages at all timepoints (p < 0.001 from baseline) — from baseline to month 36, the percentage of eyes with early maculopathy increased from 27% to 85% and the percentage of eyes with advanced maculopathy declined from 73% to 15% (Table 2 and Figure 3). The changes were greater in the early switch group than in the late switch group (the proportion of eyes with early maculopathy increasing from 20% to 95% in the early switch group compared with 33% to 75% in the late switch group, and the proportion of eyes with advanced maculopathy declining from 80% to 5% in the early switch group compared with 67% to 25% in the late switch group (Table 3 and Figure 3)).
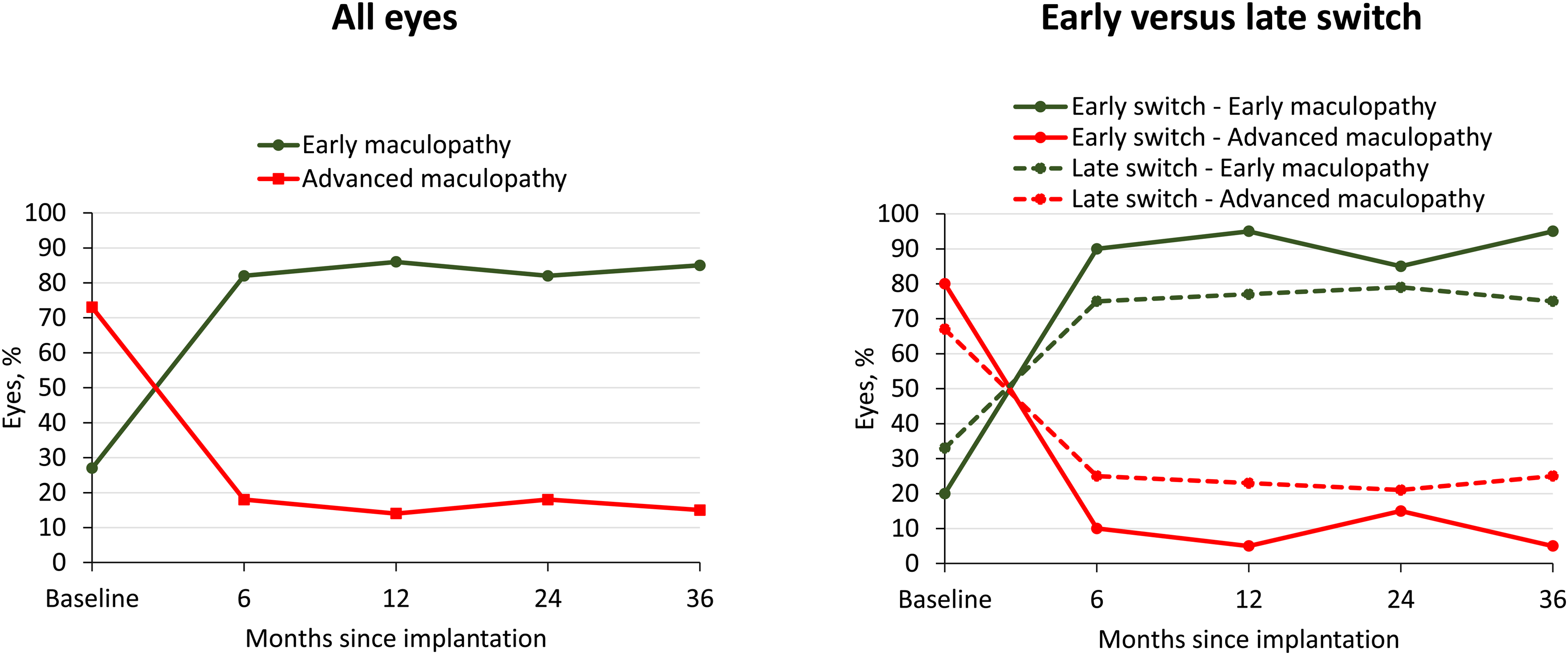
Change in maculopathy stage after fluocinolone acetonide implantation. Left graph, all eyes (N = 45). Right graph, subgroups of eyes switched to corticosteroid either early (n = 21) or late (n = 24). Early switch was after < 6 intravitreal injections for diabetic macular edema (DME), late switch was after ≥ 6 intravitreal injections for DME. The overall distribution of maculopathy, within its categories, was significantly changed from baseline at all timepoints (p < 0.001 for all eyes and early switch group and p < 0.05 for late switch group using the McNemar test).
Supplemental therapy
During the 36-month follow-up period, 22 eyes (49%) received additional anti-VEGF or corticosteroid injection(s), 11 (24%) received laser treatment, 6 (13%) had glaucoma surgery, and 4 (9%) were re-treated with another fluocinolone acetonide implant. All 10 phakic eyes required cataract surgery. At baseline, 24% of eyes were already receiving IOP-lowering drops and an additional 43% of eyes started such therapy after fluocinolone acetonide implant treatment (thus a total of 67% of eyes were receiving drops at the end of follow-up).
Discussion
Classically, central macular thickness has been used to determine both the need for intravitreal treatment and the response to treatment. However, retinal structure and function are highly complex and additional evaluations related to the composition and integrity of different retinal layers would provide a better characterization of DME and could be beneficial in determining the need for treatment. The ESASO classification of DME was developed to facilitate this.
The ESASO classification system provides a simple means of classifying the stage of maculopathy and, for example, distinguishing between advanced and severe maculopathy. This is important because advanced and severe maculopathy (which differ in terms of EZ/ELM status) may differ greatly in terms of treatment response and visual outcome. For example, whereas the successful treatment of DME could lead to visual improvement in advanced maculopathy it may lead to little or no functional improvement in severe maculopathy. It is therefore critical that maculopathy stage is considered both in research protocols and everyday clinical practice. In the clinic, using the ESASO criteria to determine the maculopathy stage could influence treatment choices. For example, when the maculopathy stage suggests there is only a small chance of clinical benefit (as might be the case if the EZ/ELM is absent), patients and clinicians might decide against pursuing options with a high treatment burden. Conversely, when the maculopathy stage suggests a more favorable prognosis, reluctant or non-adherent patients might be encouraged to continue with their recommended treatment when otherwise they might not have been receptive to this.
To our knowledge, this is the first study demonstrating a real-world use of ESASO classification in the follow-up of DME after fluocinolone acetonide implantation. Our results characterize the effects of fluocinolone acetonide implantation on a range of key retinal parameters and support the effectiveness of the fluocinolone acetonide implant in the treatment of persistent or recurrent DME. This treatment was associated with significant improvements in BCVA, CFT, and maculopathy stage. Treatment was also associated with a significant reduction in the proportion of eyes having each of the following at multiple timepoints: CFT > 30% above the upper normal value; intraretinal cysts; and disrupted or absent EZ and/or ELM. Additionally, treatment was associated with a significant reduction in the proportion of eyes with subretinal fluid at month 24. Among all the retinal parameters evaluated, the magnitude of the reductions was greatest for (a) CFT > 30% above the upper normal value and (b) intraretinal cysts, suggesting that these parameters offer the most sensitive reflection of the effect of treatment. The improvements in the retinal parameters were accompanied by a corresponding regression in maculopathy stage (from advanced maculopathy to early maculopathy) within 6 months in the majority of eyes.
The data show a trend suggesting that switching to a corticosteroid early (after < 6 previous injections for DME) rather than late (after ≥ 6 previous injections for DME) lowers the likelihood of eyes having (a) CFT > 30% above the upper normal value or (b) DRIL. Although these differences were not statistically significant in these relatively small subgroups, it remains possible that these retinal parameters could reflect changes that occur relatively early in DME progression and could be more readily reversible in earlier DME than later DME. The regression of diabetic maculopathy was also more pronounced in the early switch group than the late switch group; this might be at least partly attributable to the significantly lower mean duration of DME in the early switch group at baseline (4 years, versus 5 years in the later switch group; P < 0.05), as this could lower the chance that DME progression had become irreversible.
Retinal parameters such as those used in the ESASO system have previously been evaluated as potential biomarkers for DME by several other authors. However, we believe the results detailed here are the largest dataset specifically evaluating the effects of fluocinolone acetonide implantation on these criteria. A smaller study has evaluated a variety of retinal parameters for 12 months post-implantation 18 and another study has evaluated the effect of retinal characteristics at baseline on functional response post-implantation. 8
Other authors have previously outlined key information about each of the retinal parameters studied. Thus, DRIL signifies poor definition of the boundaries of the inner retinal layers9,19,20 and may result from the snapping of bipolar axons when edema pushes them beyond their limit of elasticity.20,21 It is associated with poor visual acuity 9 and appears to be a negative predictor of visual acuity in both current and resolved DME. 22 DRIL is also strongly associated with disruption of the EZ and ELM. 9 Disruption of the EZ signifies damage to the macular photoreceptors 23 and carries a greater risk than DRIL of poor visual recovery. 24 Disruption of the ELM may follow disruption of the EZ 24 and may allow fluid in the outer retina to move more easily into the subretinal space.25,26 Dexamethasone implantation has been reported to be associated with a significant reduction in DRIL and a small (though non-significant) reduction in EZ/ELM disruption.27,28 And, consistent with our findings here, fluocinolone acetonide implantation has been reported to be associated with a significant reduction in the prevalence of subretinal fluid and a non-significant reduction in the prevalence of DRIL. 18
Regarding intraretinal cysts, it has previously been reported that the percentage of eyes with cysts significantly declines after fluocinolone acetonide implantation 18 (which is also consistent with our findings), and that the size of cysts significantly declines after dexamethasone implantation. 29 The prognostic significance of intraretinal cystoid spaces depends on their size, location, and association with hyperreflective material. 20
Regarding hyperreflective foci, these are associated with a poor visual prognosis in DME.30,31 They may be active inflammatory cells and a reflection of damaged retinal tissue. 32 The number of hyperreflective spots has previously been reported to decline significantly after dexamethasone implantation, 29 which is consistent with our findings of a (non-significant) decline in the percentage of eyes with > 30 hyperreflective foci.
The percentage of eyes given supplementary intravitreal therapy in the present analyses was higher than in results reported in a recent meta-analysis of real-world evaluations of fluocinolone acetonide implant treatment in DME (49% versus 39%). 33 However, this has to be viewed in the context of our follow-up being relatively longer (3 years versus 1–2 years in several studies in the meta-analysis). Although this incidence of supplemental therapy is not surprising, it does suggest that the sustained reduction in CFT at month 36 might not be entirely due to the implant but could be partly attributable to supplemental therapy. Nevertheless, any such contribution appears to be relatively small as results from other real-life studies with a smaller percentage of eyes receiving supplemental intravitreal therapy for DME after fluocinolone acetonide implantation (0% in a subgroup, 34 19%, 35 and 32% 36 ) still demonstrate sustained reductions in central macular thickness for up to 1, 34 2, 35 and 3 years, 36 respectively.
Limitations of this study include its retrospective nature, lack of a control group, lack of masking during scan evaluations, limited number of eyes evaluated, and inclusion of 10/45 eyes that were contralateral to eyes already in the study. In addition, 58% of eyes enrolled in the study were vitrectomized. Although this could have an influence on ESASO OCT grading parameters, especially if peeling of the epiretinal membrane ± internal limiting membrane occurred, results from previous real-world evaluations of vitrectomized eyes show statistically significant improvements in functional and anatomical outcomes after fluocinolone acetonide implant treatment of DME 37 and suggest such treatment may be at least as effective in vitrectomized eyes as in non-vitrectomized eyes.15,18,38,39 In future studies, the inclusion of eyes with shorter DME durations may further help our understanding of retinal changes occurring during earlier stages of DME progression.
In summary, although the evaluation of BCVA and retinal thickness are traditionally the key parameters involved in the assessment of DME and its treatment, they are not well correlated and do not describe DME thoroughly enough. Retinal thickening should not be the only structural parameter included in assessments of retinal damage because the same degree of retinal thickening can occur in different stages of maculopathy and the integrity of the EZ/ELM is pivotal in defining the stage of maculopathy. 11 There is a need to further classify DME to include parameters that characterize its: severity (as this cannot be gauged from the duration of DME alone); anticipated response to treatment; and prognosis. 11 In addition to the EZ/ELM status, the presence of DRIL, intraretinal cysts, subretinal fluid, and hyperreflective foci are useful in characterizing diabetic maculopathy and may prove useful in determining prognosis and guiding optimal treatment. We hope the retinal evaluations detailed here will become standard in both clinical research and everyday clinical practice and that they will lead to a better understanding of DME and, ultimately, help optimize the management and prognosis of DME.
Conclusion
Our evaluation of ESASO criteria in eyes with persistent or recurrent DME show that fluocinolone acetonide implantation can be effective in improving maculopathy stage and reducing the incidence of eyes with: intraretinal cysts; CFT > 30% above the upper normal value; and disrupted or absent EZ and/or ELM. An increase in BCVA and reduction in CFT were also evident but the changes in the ESASO parameters provide a more detailed insight into the nature of the retinal changes occurring alongside these more commonly reported parameters.
Our data also show a trend suggesting that early corticosteroid implantation (before 6 intravitreal injections of anti-VEGF and/or corticosteroid have been administered) may help achieve greater clinical improvements than later implantation. Although larger studies are needed to provide the necessary statistical power to confirm this, early implantation may be an important consideration in treatment management and can be adopted now.
Footnotes
Acknowledgments
Writing assistance was funded by Alimera Sciences Ltd. and provided by Gill Shears PhD (Write on Target Ltd., Leighton Buzzard, UK) who wrote the first draft manuscript. All authors reviewed and critiqued the initial and final drafts and authorized Gill Shears to submit the final manuscript, financial disclosures, and other declarations to the journal on their behalf.
Authors’ contributions
All authors made significant contributions towards the conception and design of this study. João Leite and Catarina Castro collected the data. João Leite, André Ferreira, and Bernardete Pessoa analyzed and interpreted the data and summarized the study findings. All authors revised or critically reviewed the article; gave approval of the version to be published; agreed on the choice of journal for submission; and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
The authors declare that there is no conflict of interest other than writing assistance being funded by Alimera Sciences Ltd.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The authors received no financial support for the research, authorship, and publication of this article other than medical writing services and article processing charges funded directly by Alimera Sciences Ltd.
