Abstract
Purpose
To report the use of intravitreal triamcinolone to treat macular edema associated with isolated perifoveal exudative vascular anomalous complex (PVAC) and resembling lesion (PVAC-RL).
Methods
In this case series, three diabetic patients (3 eyes) with PVAC-RLs and one healthy patient (1 eye) with PVAC lesion associated with cystic spaces underwent three intravitreal injections of aflibercept before switching to one intravitreal triamcinolone injection in each patient.
Results
Macular edema improved from 297.5 ± 8.10 μm, at baseline, to 269.2 ± 8.89 μm, after triamcinolone
Conclusion
PVAC and PVAC-RL are rare and often misdiagnosed lesions that may be associated with decreased vision. Our outcomes suggest that intravitreal injection of triamcinolone may be an effective and affordable treatment for PVAC and PVAC-RL with intraretinal fluid.
Keywords
Introduction
Perifoveal exudative vascular anomalous complex (PEVAC) was first described by Querques et al. in 2011, and so far, it has appeared in a few case reports.1–3 More recently, in 2020, Sacconi et al. renamed PEVAC into ePVAC and nePVAC based on the exudative or non-exudative (subclinical) stage of the disease. 4 On fundus examination, it is characterized by a large, perifoveal, aneurysmal abnormality that may be associated with hemorrhages and/or hard exudates, and on spectral domain optical coherence tomography (SD-OCT), it appears as a round or oval hyperreflective lesion with hyporeflective lumen, which may be surrounded by intraretinal cysts.5,6 Patients with PVAC are often healthy, without diabetes or uncontrolled hypertension, or other retinal or choroidal vasculopathy.1–3,5 However, this lesion has been reported in subjects with dry age-related macular degeneration (AMD) and myopic macular degeneration, and in patients with diabetes or hypertension (PVAC-resembling lesions). 7 We present here a case series involving 4 patients (4 eyes), where one patient had isolated PVAC lesion, and the other three patients had PVAC-resembling lesion with macular edema. To date, there are few cases reporting the findings, course, outcomes, and treatment of this condition. The aim of this case series is to report the use of intravitreal triamcinolone to treat isolated PVAC and PVAC-resembling lesions accompanied by intraretinal cystic spaces.
Case report
Among 1000 patients with macular diseases seen at the Hospital Naval Marcilio Dias from 2020 to 2022, we observed this aneurysmal lesion in 5 cases, although 1 of 5 cases did not present macular edema in a follow -up of more than 4 years.
The first patient was a 63-year-old white female complaining of decreased visual acuity in the right eye (RE). The patient had controlled hypertension and dry AMD. On examination at baseline, the best-corrected visual acuity (BCVA) in Snellen chart was 20/30 in her RE and 20/20 in her left eye (LE). Anterior segment examination and intraocular pressure (IOP) were normal in both eyes. Fundus examination (Carl Zeiss Meditec, Jena, Germany), of the RE showed 2 large perifoveal aneurysmal lesions, and both eyes had scattered cuticular drusen in the posterior pole and narrowing of the arterioles (Figure 1A, and Figure 1E). In fundus autofluorescence photography (FAF), the lesions were seen as perifoveal hypoautofluorescent lesions (Figure 1B). Fluorescein angiography (FA) (Carl Zeiss Meditec, Jena, Germany) revealed 2 well-defined hyperfluorescent perifoveal aneurysmal dilations without leakage, and a rarefaction of retinal capillaries in the perilesional areas (Figure 1H). On OCT angiography (Topcon, Tokyo, Japan), 2 aneurysmal lesions with rarefaction of the retinal capillaries in the perilesional areas were present in the superficial vascular complex (Figure 1C) and deep vascular complex (Figure 1D). These PVACs were detected as oval hyperreflective lesions surrounding a dark lumen with reflective material, sited from the ganglion cell layer (GCL) to the outer nuclear layer (ONL), associated with intraretinal cystic space on SD-OCT– DRI -OCT Triton Swept Source (Topcon, Tokyo, Japan) of the RE (Figure 1F and 1H). The patient was followed up with no treatment, and after 6 months, the RE showed an increase in intraretinal cystic spaces on SD-OCT and a decrease in BCVA to 20/40 (Figure 1G). She received 3 doses of aflibercept without success, so we switched to intravitreal triamcinolone. After 2 weeks of treatment with triamcinolone, BCVA and SD-OCT images improved to 20/25 and no intraretinal fluid, respectively, without recurrence for almost 12 months (Figure 1G*).
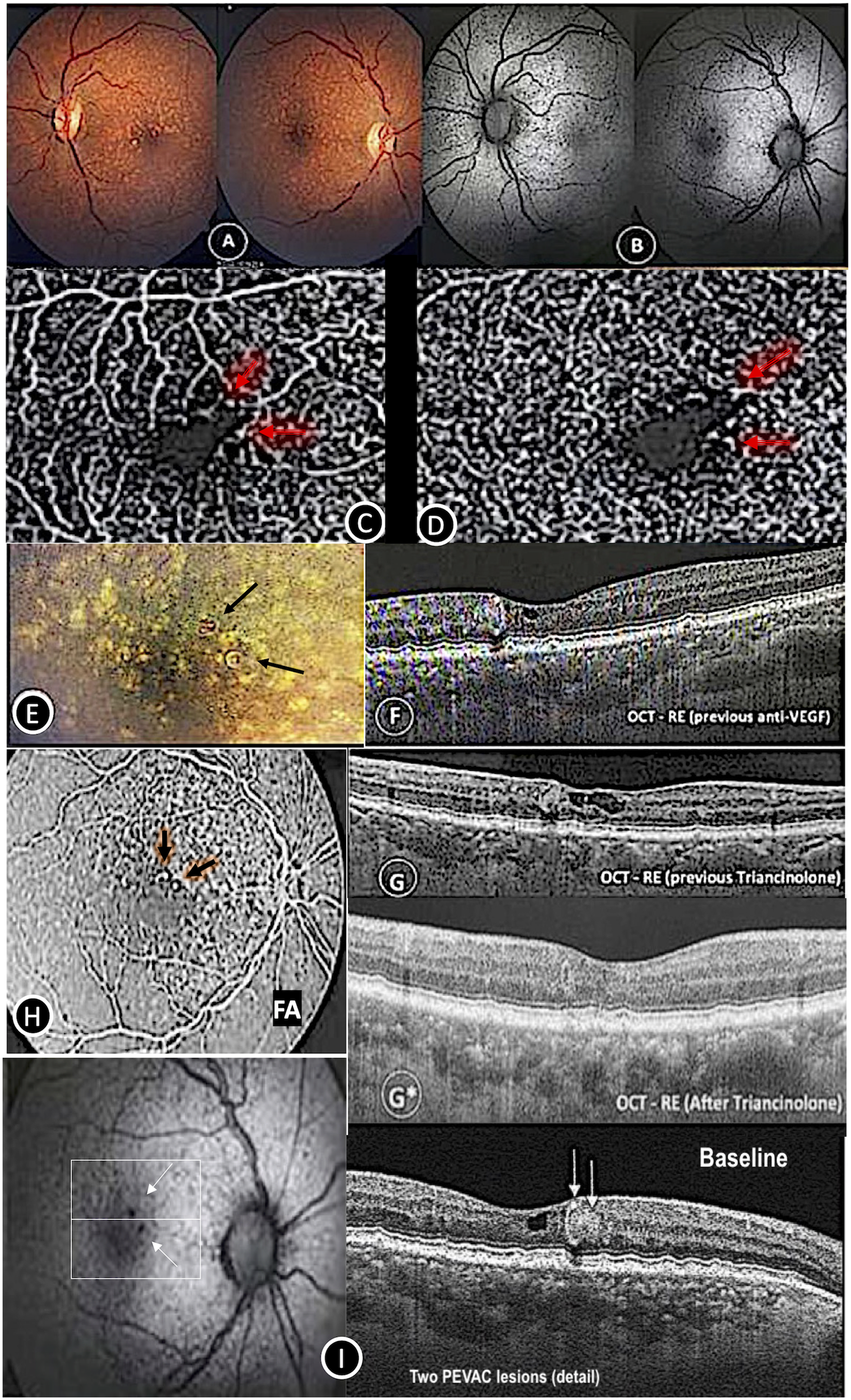
Case 1 – PVAC and macular cuticular drusen. A: Color fundus LE and RE PVAC, B: Autofluorescence – RE PVAC detail, C: OCTA – PEVAC superficial capillary plexus, D: OCTA – deep foveal capillary plexus; E: color fundus RE PVAC vascular detail; F, G, G*: SD-OCT – Effect of anti-VEGF and triamcinolone therapy on PEVAC and cystoid macular edema, H: fluorescein angiography RE PVAC detail, and I: 2 PVAC lesions.
The second patient was a 64-year-old white man with type 2 diabetes mellitus, macular edema (DME) in his LE, glycated hemoglobin of 6.8% and creatinine of 0.99 mg/dl, controlled glaucoma with timolol, and he had no relevant surgical history. At baseline, BCVA was 20/40 in his LE, and fundus examination showed a large perifoveal aneurysmal lesion associated with hard exudate (Figure 2A). Additionally, OCT-A identified flow in the superficial and deep capillary plexus (Figure 2B and 2B*); and intraretinal fluid and typical oval hyperreflective PVAC- resembling lesion were present on SD-OCT (Figure 2C). He received 3 doses of intravitreal injection of aflibercept, which reduced the intraretinal fluid but without total resolution, and BCVA remained unchanged (Figure 2D). After 3 months of follow-up, we switched the treatment with aflibercept to intravitreal triamcinolone, and BCVA improved to 20/30 and intraretinal fluid disappeared for at least 10 months of follow-up (Figure 2E).
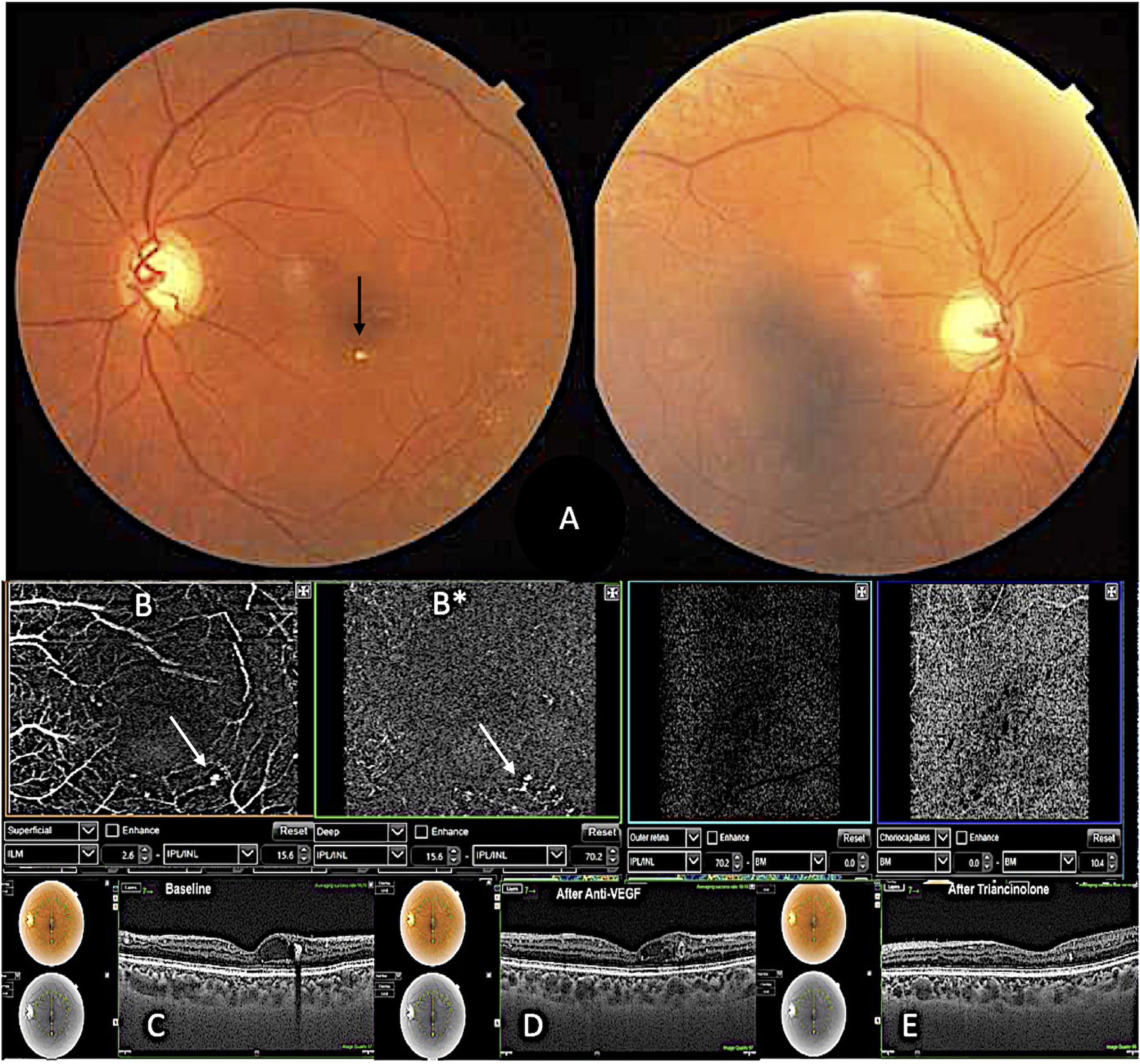
Case 2 – macular PVAC A: color fundus le and RE PVAC vascular detail, B: OCTA B* – PVAC superficial foveal capillary plexus, B*: OCTA – deep foveal capillary plexus; C, D, E – effect of anti-VEGF and triamcinolone therapy on PVAC and cystoid macular edema.
The third case was a 78-year-old white man with type 2 diabetes mellitus who had macular edema in his RE, glycated hemoglobin of 7.8% and creatinine of 0.88 mg/dl, and he was phakic in both eyes. His baseline BCVA was 20/50 in the RE. The anterior segment and IOP were unremarkable in both eyes, whereas fundus examination showed perifoveal aneurysmal lesion in his RE. OCTA showed flow in the deep capillary plexus (Figure 3C and 3D). SD-OCT showed intraretinal fluid and oval hyperreflective PVAC-resembling lesion (Figure 3A). Patient underwent 3 intravitreal injections of aflibercept without any improvement (Figure 3A*). We then changed the treatment to intravitreal triamcinolone. After 1 month of follow-up, BCVA increased to 20/25 and the intraretinal fluid improved (Figure 3B).
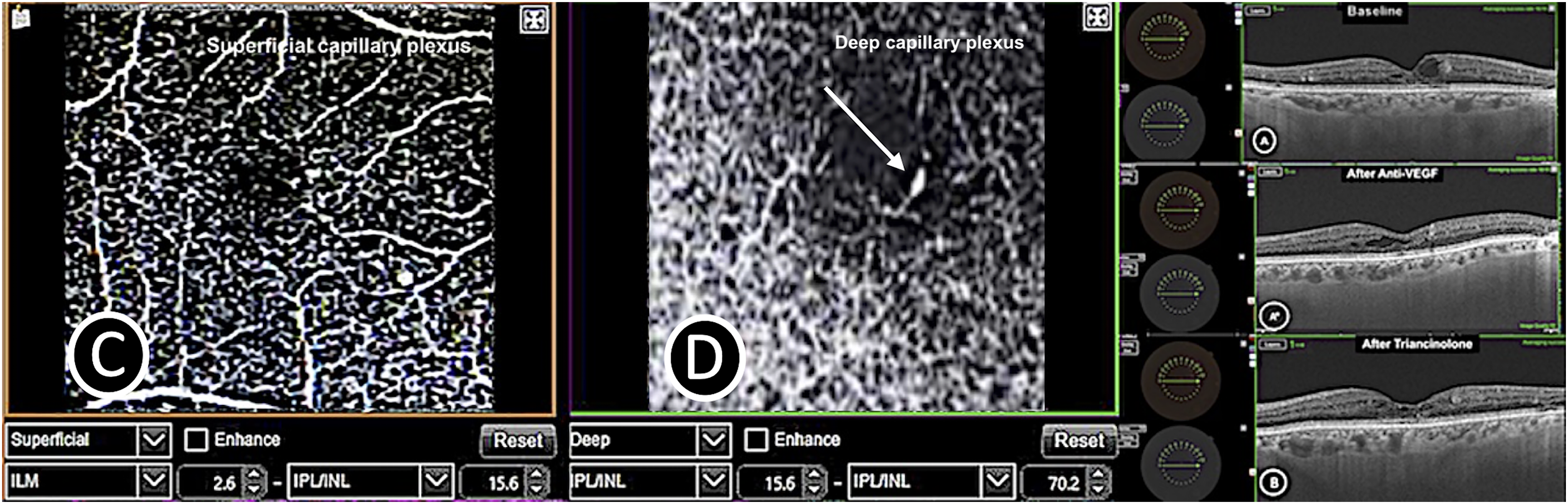
Case 3 – macular PVAC A, A*, B: effect of anti-VEGF, and triamcinolone therapy on PVAC and cystoid macular edema. C: OCTA – superficial capillary plexus, D: OCTA – PVAC in the deep foveal capillary plexus.
The last patient was a 78-year-old white male, pseudophakic, diabetic under treatment with insulin, with glycated hemoglobin of 7.0% and creatinine of 0.88 mg/dl. He was followed up for exudative AMD in the RE and dry AMD in the LE. In his LE, BCVA was 20/30; we observed cuticular drusen and a large perifoveal aneurysmal lesion associated with cystic spaces on SD-OCT (Figure 4A). OCTA showed flow in the deep plexus (Figure 4C and D). Baseline characteristics remained unchanged after 3 intravitreal injections of aflibercept. BCVA improved to 20/25, and the intraretinal fluid and PVAC-resembling lesion disappeared after one intravitreal injection of triamcinolone (Figure 4B).
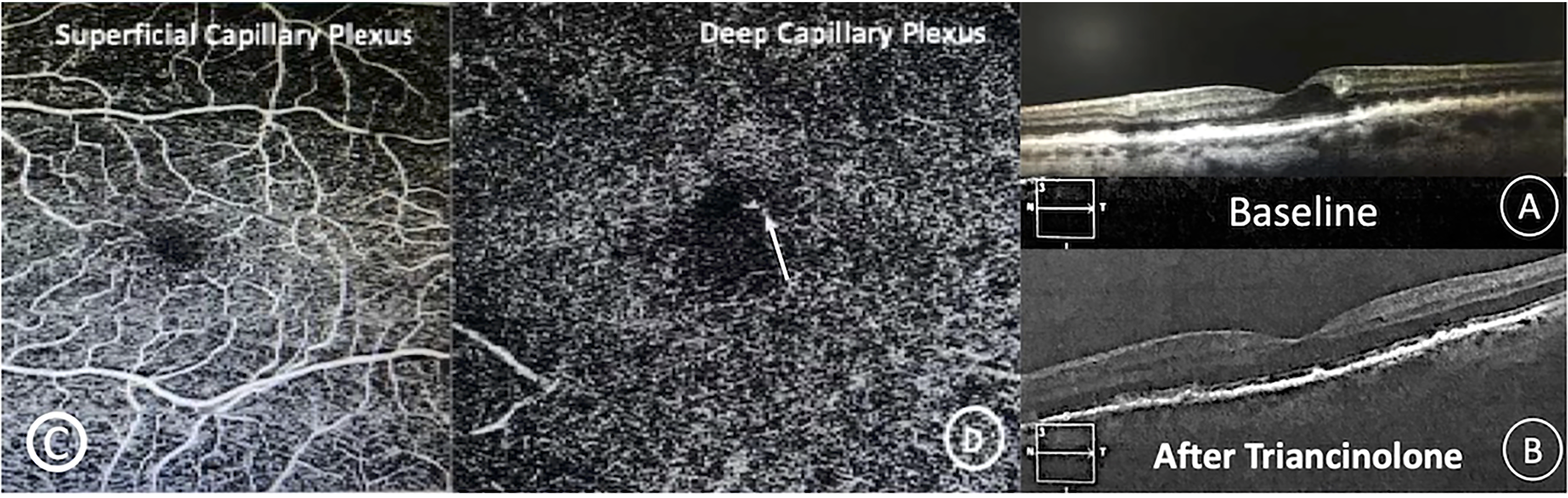
Case four – macular PVAC A: effect of before and after triamcinolone therapy on PVAC and cystoid macular edema. C: OCTA – superficial foveal capillary plexus, D: OCTA – PVAC (Arrow) deep foveal capillary plexus.
All patients were treated with 3 intravitreal injections of aflibercept without success, before switching to 1 intravitreal injection of triamcinolone, which proved to be effective. Therefore, ophthalmologists should be aware that macular edema associated with PVAC may not respond to anti-vascular endothelial growth factor (VEGF).1,4 In all cases we observed that BCVA and intraretinal cystic spaces improved. Macular edema improved from 297.5 ± 8.10 μm, at baseline, to 269.2 ± 8.89 μm, after triamcinolone
Discussion
The pathogenesis of PVAC and its treatment are not yet established.1–3,6,8 It was first identified in a healthy patient without any other vascular or inflammatory disease. However, in 2019, Venkatsh et al. and Zhang et al. identified PVAC in a diabetic patient who was unresponsive to anti-VEGF injections.9,10 Moreover, in 2021, Smid and associates referred to a lesion as PVAC-resembling when it occurred in patients affected by other vascular or inflammatory abnormalities. 7 Despite the similar clinical, morphological, and vascular features between PVAC and PVAC-resembling lesions, PVAC lesion is considered a microangiopathy of unknown origin as it occurs in healthy patients. However, Smid et al. observed ischemic attack or stroke in 40% of patients with PVAC and controlled hypertension in 70% of patients, supposing that it may be related to a general cardiovascular disorder. Moreover, they postulate that ageing may play a role in the development of PVAC lesions. Of note, the underlying vascular disease may stimulate the formation of PVAC-RL in patients. 7
Some authors have been advocating the use of thermal laser photocoagulation but this should be considered with caution when applied close to the fovea due to the risk of paracentral scotoma. 1 Still, the micropulse laser (MPL) is a minimally damaging treatment used for direct block of leaking microangiomas and also may diminish local inflammation by deactivating retinal microglia. Fu and colleagues believed that a photoinduced biological effect on neuroretina tissues and cells may play a role in PVAC treatment with MPL. 11 We think that the use of intravitreal triamcinolone may be an effective and affordable alternative for the treatment of PVAC associated with macular edema. Our results imply an inflammatory mechanism associated with PVAC accompanied by macular edema. In contrast, Mrejen and coworkers believed that PVAC stands apart from local inflammation. 1 To the best of our knowledge, this is the first time that PVAC associated with macular edema was treated with intravitreal triamcinolone.
Another belief is that PVAC may be a progressive degeneration of retinal endothelial cells which explains the ineffectiveness of intravitreal anti-VEGF injections. 3 Thus, Querques et al., Sacconi et al., Kim et al., and Zhang et al. described cases with intraretinal fluid secondary to PVAC lesions that were unresponsive to intravitreal anti-VEGF.1,3,5,10 Indeed, Spaide et al. reported five PVAC cases, one case with PVAC-resembling lesion associated with stable diabetic retinopathy and four cases with PVAC. All patients presented with secondary macular edema and were treated with anti-VEGF, resulting in incomplete response in 1 patient and no response in 4 patients. Yellow laser photocoagulation was then applied in unresponsive patients, and macular edema was resolved, leading to involution of these lesions. These authors supposed that the leakage from these capillaries was not mediated by VEGF. Instead, an increase expression of matrix metalloproteinase-9, which breaks-down basement membrane proteins, would lead to a decrease in pericytes. In addition, an imbalance between a reduced wall strength and an increased wall tension would trigger the aneurysm formation. Notably, another hypothesis to be considered is a Muller cell-driven process, as these cells play a role in maintaining the inner blood-retina barrier and regulating the extracellular space volume. 12 This could explain why PVAC lesion appeared concomitantly with lamellar hole-associated epiretinal membrane in a case. 13
In 2020, Mrejen and colleagues evaluated a series of 8 patients with PVAC imaged with OCTA. Among these patients, 6 patients were observed, and 2 patients were treated with anti-VEGF. One patient responded to the therapy, despite the need for 13 intravitreal injections of anti-VEGF to achieve stability, and maintain a BCVA of 20/32 for at least 8 months of follow-up. The other subject was unresponsive to 3 intravitreal anti-VEGF but was successfully switched to thermal laser photocoagulation. These authors speculated that many intravitreal injections of anti-VEGF were required to treat macular edema in capillary aneurysms. 2 Therefore, it is possible that Sacconi et al. and Verhoekx et al. did not use a sufficient number of anti-VEGF injections to achieve an effective response with this therapy.3,6 Another point that cannot be ruled out is the natural history of these lesions. 1 Sacconi et al., Kim et al., and Verhoekx et al. identified a total of 10 patients with spontaneous resolution of the cystic spaces, and in 3 patients the PVAC lesion completely vanished.3,5,6 PVAC size may be correlated with the presence of intraretinal fluid as observed in the study of Verhoekx et al. 6 In addition, we also identified one patient with a small PVAC lesion that did not present with secondary macular edema for more than 4 years of follow-up. In 2022, Tombolini and associates reported the use of topical diclofenac, a non-steroidal anti-inflammatory drug (NSAID) to treat a patient diagnosed with isolated PVAC lesion. This was based on the NSAID property of inhibiting the expression of cyclooxygenase enzyme isoforms and their products, called prostaglandins and thromboxanes which are involved in the inflammatory response. 13 Similarly, we also believe that the use of intravitreal steroidal anti-inflammatory drug therapy benefited our patients with macular edema secondary to PVAC. Judging from the few cases that have been published relating isolated PVAC and resembling lesions, it is still uncertain whether PVAC spontaneously remits over time or responds to any type of treatment. More studies are needed to better explain this.
Footnotes
Acknowledgements
Not relevant
Authors’ contributions
The first author collected the data, performed the analysis, the co-authors participated in analyzing the data and writing the paper. All authors read and approved the final manuscript.
Availability of data and material
The authors confirm that all the data supporting the findings of this study are available within the article.
Consent for publication
Not relevant
Competing interests
There is no Competing interest for any of the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The current case series was approved by the Research and Ethics Committee of the Universidade Federal Fluminense, UFF and adhered to the Guidelines and Norms of the Declaration of Helsinki to Human Health Research. All participants signed an informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
