Abstract
Background/Objectives
Progression of retinopathy of prematurity (ROP) is associated with increased retinal blood flow velocities. We investigated changes of central retinal arterial and venous blood flow after intravitreal administration of bevacizumab.
Subjects/Methods
Prospective observational study using serial ultrasound Doppler imaging in preterm infants with bevacizumab-treated ROP. Eyes were examined 1 [0–2] days before injection (median [interquartile range]), and at three time points after injection (1 [1–2] days, 6 [3–8] days, and 17 [9–28] days). Preterm infants with ROP stage 2 displaying spontaneous regression served as controls.
Results
In 21 eyes of 12 infants with bevacizumab-treated ROP, peak arterial systolic velocity declined from 13.6 [11.0-16.3] cm/s prior to intravitreal bevacizumab to 11.2 [9.4–13.9] cm/s, 10.6 [9.2–13.3] cm/s and 9.3 [8.2–11.0] cm/s at discharge (p = .002). There was also a decline of the arterial velocity time integral (from 3.1 [2.3–3.9] cm to 2.9 [2.4–3.5], 2.7 [2.3–3.2] cm and 2.2 [2.0–2.7], p = .021) and mean velocity in the central retinal vein (from 4.5 [3.6–5.8] cm/s to 3.7 [2.6–4.1] cm/s, 3.5 [3.0–4.3] cm/s, and 3.2 [2.8–4.6] cm/s, p = .012). Arterial end-diastolic velocity and resistance index remained unchanged. Blood flow velocities in bevacizumab-treated eyes examined before injection were significantly higher than those measured in untreated eyes that ultimately showed spontaneous regression of ROP. Sequential examinations in these controls did not reveal any declines of retinal blood flow velocities.
Conclusion
Increased retinal arterial and venous blood flow velocities in infants with threshold ROP decline following intravitreal bevacizumab injection.
Introduction
Retinopathy of prematurity (ROP) is a disease of developing retinal blood vessels affecting most very preterm infants to varying degrees. To follow the progression and eventual regression of ROP, screening guidelines call for serial dilated fundus exams using binocular indirect ophthalmoscopy with a lid speculum and scleral depression. 1 If advancing ROP threatens to compromise vision, infants are scheduled for treatment with laser photocoagulation or intravitreal injection of agents that neutralize vascular endothelial growth factor (VEGF), such as bevacizumab, 2 ranibizumab, 3 pegaptanib, 4 aflibercept,5,6 or conbercept. 7
Treatment recommendations put forward in national guidelines1,8,9 as well as criteria employed in randomized controlled trials,2,3,10 are based on ophthalmic findings in binocular ophthalmoscopic examinations, comprising zone, stage, and presence of plus disease. Plus disease, as sequentially defined by the International Committee for the Classification of Retinopathy of Prematurity 1984, 11 2005, 12 and 2021, 13 is characterized by venous dilatation and arteriolar tortuosity. However, it is now being recognized that a continuous spectrum of vascular abnormality exists from normal to preplus and plus disease. 13 Progression of plus disease is associated with increasingly larger diameters of arterial and venous vessels of the retina 14 and may eventually lead to iris vascular engorgement, pupillary rigidity, and vitreous haze. 11 These features had been included in the first definition of plus disease but are no longer considered essential for its diagnosis. 13
The exact definition of plus disease has been subject to further changes over time (presence of dilatation and tortuosity in at least two quadrants of the eye in 2005, 12 as opposed to vessels in zone I in 2021 13 ), and there is considerable inter-observer variability in the diagnosis of ROP severity.15–17 In borderline cases for which indicating treatment is a difficult decision, ophthalmologists tend to take neonatological and demographical data into consideration, 18 although these have never been evaluated for that purpose. This suggests that there is an apparent need for further adjunctive measurements to fine-tune ROP diagnosis.
Using color Doppler imaging, we have found previously that blood flow velocities in the central retinal artery (CRA) and central retinal vein (CRV) increase when infants reach stage 2 ROP. 19 Increased blood flow velocities were observed both opposed to baseline and to infants that did not develop ROP. Subsequent plan-wave ultrasound measurements have confirmed that ROP stage correlates with CRA and CRV flow velocities. 20 These data point to increased retinal blood flow as a crucial feature of ROP that is linked to, and may potentially be involved in the progression of the disease.
To further elucidate the association between retinal blood flow and ROP, we asked whether regression of threshold ROP induced by intravitreal injection of bevacizumab was accompanied by a decrease of CRA and CRV blood flow velocities.
Patients and methods
Infants
The study was conducted in preterm infants cared for at three neonatal intensive care units of a regional referral center. Serial fundus exams by indirect ophthalmoscopy followed established guidelines. 1 ROP stage and plus disease was diagnosed as defined by the International Committee for the Classification of Retinopathy of Prematurity, 2005 edition. 12 If a decision was made to schedule an infant for intravitreal bevacizumab, parents or guardians were approached for written informed consent for participation of their infant in the study which entailed one ultrasound measurement before and three ultrasound measurements after the intravitreal injection. Treatment with intravitreal bevacizumab (0.625 mg in 0.025 ml of solution) was done as described.2,21 Furthermore, we recruited infants with ROP stage 2 that did not progress further for serial ultrasound measurements at a similar postmenstrual age as infants treated with bevacizumab.
Measurements
Color Doppler imaging was performed by 3 trained neonatologists (FM, HF and CC) as described previously, 19 using a 5–18 MHz (Phillips L 18–5 spektrum linear array probe) or 13MHz linear array transducer (Vivid S6 9L and Vivid S7 M12L). Pulse wave Doppler frequency was set at 7.1 MHz (EPIQ 7, Philips, Amsterdam, The Netherlands) or 5 MHz (Vivid S6/S7, General Electric, Boston, Ma). The linear probe was placed in a vertical position perpendicular to the eye globe on the closed eyelid using coupling gel with minimal pressure. A fresh 20 g sachet of sterile ultrasound gel (Aquasonic 100 Gel, Parker Laboratories, Fairfield, NJ) was used for each patient and warmed to skin temperature prior to application. All examinations were performed without sedation and kept gentle (ultrasound mechanical index 0.2–0.7, thermal index 0.2–0.6), short (3–15 min), and not stressful for the infants. CRA and CRV blood flow velocities were measured simultaneously by placing the sample volume 2 mm below the sclera in the optic nerve shadow, yielding a pulsatile flow pattern of the CRA combined with a continuous flow of the CRV in the opposite direction. We recorded CRA peak systolic flow velocity, end-diastolic flow velocity, flow velocity time integral (area under the velocity time curve as a measure of arterial blood flow), resistance index (peak systolic flow velocity – end-diastolic flow velocity / peak systolic flow velocity, reflecting the resistance to blood flow by the retinal microvascular bed) and CRV maximum velocity (a measure of total retinal blood return). To minimize variations in blood flow velocity measurements, mean values were taken from uniform blood flow patterns over 3 cardiac cycles with clear signals.
Statistical analysis
Statistical analysis was strictly based on non-parametric tests, using SPSS 25.0 (IBM, Armonk, NY). Numerical data are given as median and interquartile range (IQR). Repeated measures of continuous variables in the same infant were compared by Friedman non-parametric one-way analysis of variance, whereas categorical variables were analyzed by Cochran Q test. Between-group comparisons were assessed by the Mann Whitney U test. P values < .05 were considered significant.
Regulatory approval
The study was approved by the local institutional review board (Ethikkommission der Charité, # EA2/002/16). Prior written informed consent was obtained from all parents or guardians.
Results
A total of 13 preterm infants with severe ROP born between March 2016 and April 2018 underwent intravitreal injection of bevacizumab, of whom 12 infants participated in the study. All bevacizumab-treated eyes infants had ROP in zone II, none had aggressive posterior ROP. One participating infant had bevacizumab injected only in the right eye. Measurements of the left eye and that of further 16 infants with ROP stage ≥ 2 without progress were used as controls (one control infant had only the right eye measured). Characteristics of infants involved in the study are shown in Table 1. Infants treated with bevacizumab had significantly lower gestational age, as well as significantly longer duration of endotracheal ventilation, overall duration of supplemental oxygen, and had received a higher number of red blood cells transfusions, as compared to infants with ROP stage 2 that displayed spontaneous regression.
Characteristics of infants with ROP treated with intravitreal bevacizumab and controls (median, IQR).

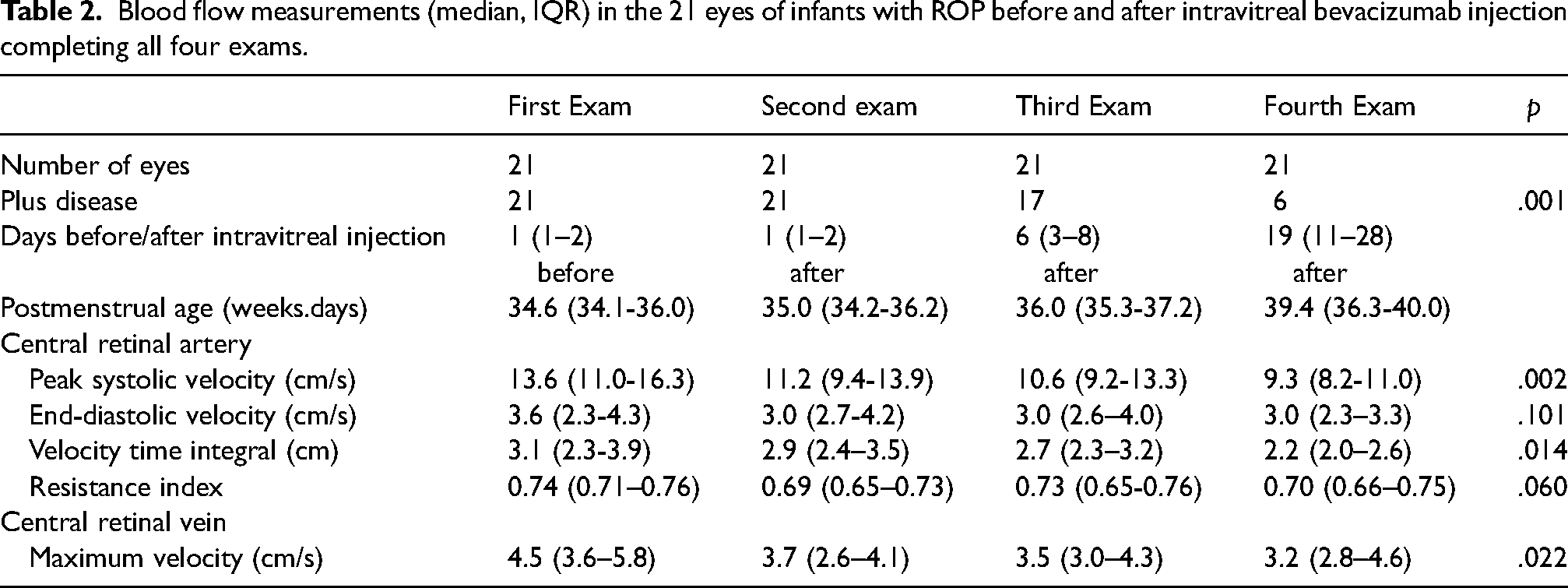
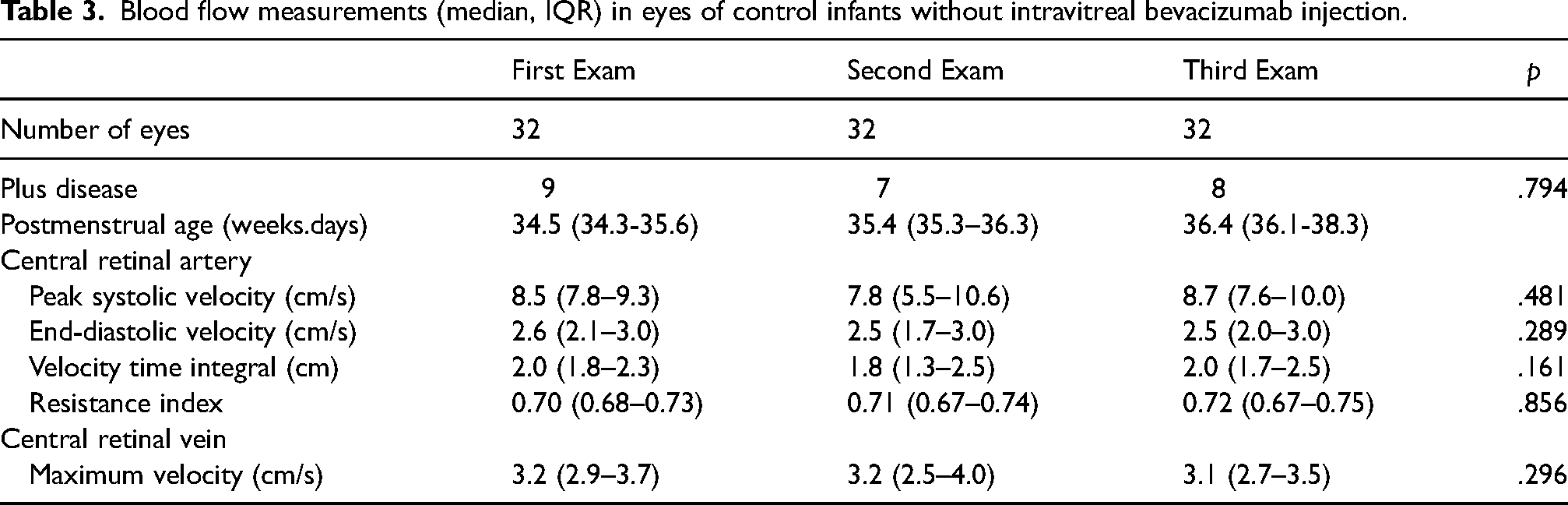
The postmenstrual age at the first, second and third exam did not differ between bevacizumab-treated and control infants (p > .1, respectively). Infants who subsequently received bevacizumab (Table 2) had significantly elevated CRA peak systolic velocity (p < .001), end-diastolic velocity (p = .043), velocity time integral (p = .001), resistance index (p = .005), and CRV maximum velocity (p = .001), as compared to controls (Table 3). Following bevacizumab injection, there was significant decrease of CRA peak systolic velocity and CRA velocity time integral and a trend towards decreased CRA resistance index, while CRA end-diastolic velocity remained unchanged. CRV velocity also decreased significantly (Table 2). In contrast, there were no significant changes in any flow measurements in untreated infants between the first, second, and third exam (Table 3). However, significant differences remained also in the third exam between bevacizumab-treated and control eyes for CRA peak systolic velocity (p = .002), end-diastolic velocity (p = .018) and velocity time integral (p = .002) but not resistance index (p = .412), while the difference for CRV maximum velocity attained borderline significance (p = .051).
Blood flow measurements (median, IQR) in the 21 eyes of infants with ROP before and after intravitreal bevacizumab injection completing all four exams.
Blood flow measurements (median, IQR) in eyes of control infants without intravitreal bevacizumab injection.
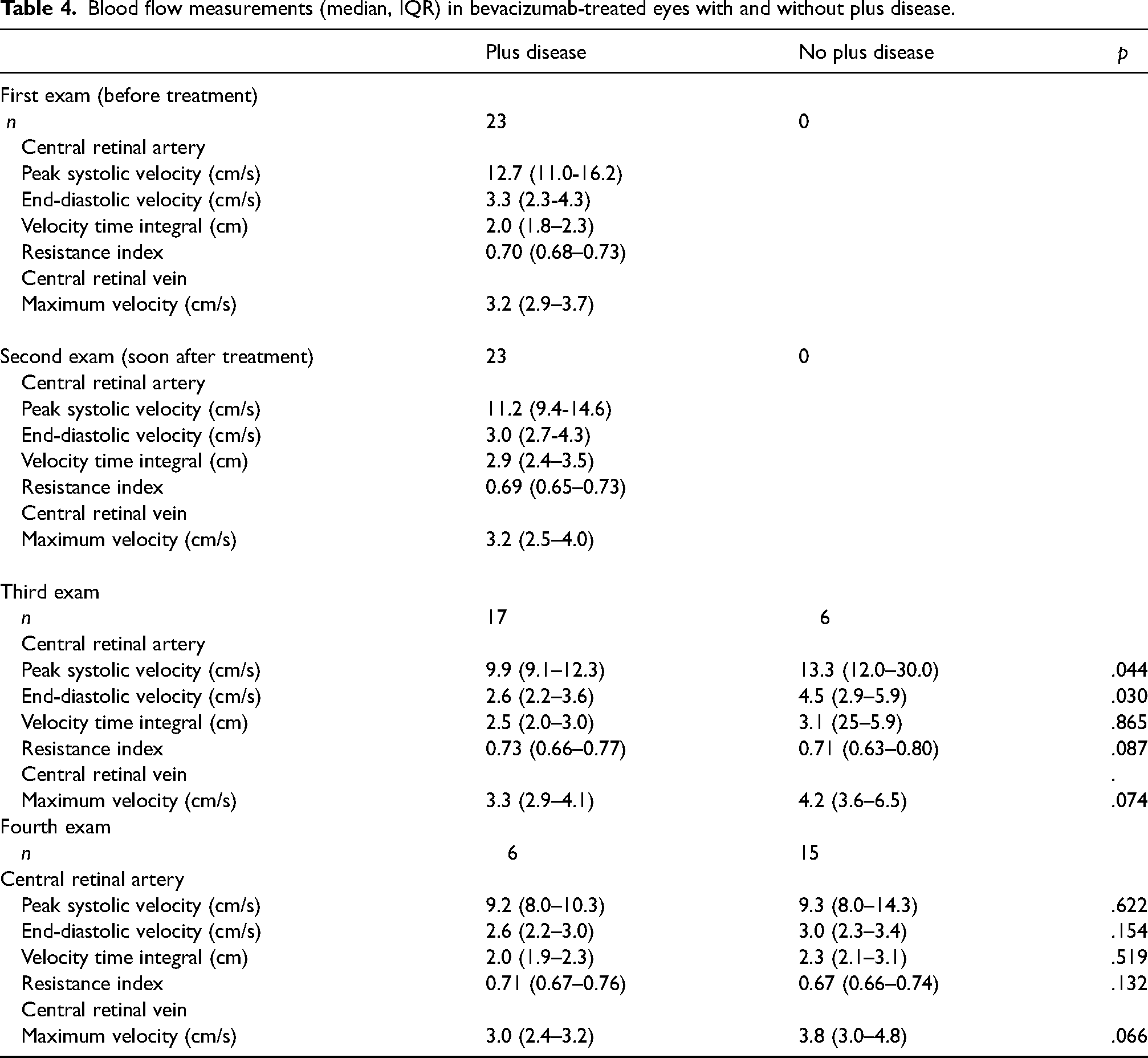
The percentage of eyes with plus disease, as defined by the International Committee for the Classification of Retinopathy of Prematurity 2005 12 criteria, declined significantly after bevacizumab injection while it remained unchanged in controls (Table 2, Table 3). However, plus disease in eyes of the control group, all of which showed spontaneous regression of ROP, was apparently rather mild. In a secondary exploratory analysis, we compared retinal blood flow measurements in eyes with or without plus disease. Eyes reverting from plus to non-plus after bevacizumab featured a transient increase of end-diastolic and systolic velocities in the central retinal arterial vessels, possibly indicating precapillary vasoconstriction (Table 4). At the discharge exam, these differences were no longer visible. In eyes not treated with bevacizumab, there was little relationship between plus disease and arterial retinal blood flow, while venous blood flow tended to be higher in plus versus non-plus eyes (Table 5).
Blood flow measurements (median, IQR) in bevacizumab-treated eyes with and without plus disease.
Blood flow measurements (median, IQR) in control eyes with and without plus disease.

Discussion
Data of this preliminary report indicate that retinal blood flow velocities are increased in infants with severe ROP subsequently treated with bevacizumab when compared to less affected infants measured at a comparable postmenstrual age. After intravitreal bevacizumab injection, CRA peak systolic blood flow velocities and velocity time integrals, as well as CRV velocities, demonstrated a sustained decrease but remained elevated compared to those of less affected infants.
As a limitation, ultrasound examiners were not blinded to the intravitreal bevacizumab injection. The spatial resolution of the 7.5 – 12 MHz probes was not sufficient to estimate retinal vessel diameters and thus to calculate blood flow. Changes in blood flow velocities could reflect changes in vessel diameters or changes in blood flow. As retinal vessels display larger diameters in infants with severe, as opposed to infants with less severe or no ROP,14,22 the increased retinal blood flow velocities observed in infants with ROP stage 3, as opposed to infants with ROP stage 2, point to elevated retinal blood flow as a feature of severe ROP.
Declining arterial retinal blood flow velocities following local anti-VEGF treatment have been observed in three further studies. Reduced peak systolic and mean blood flow velocities in the ophthalmic artery after intravitreal bevacizumab have been reported in 15 infants. 23 In contrast, treatment with intravitreal aflibercept was associated with a slight decline of CRA peak systolic velocity, a marked decline in CRA end-diastolic flow velocity, and an increase in CRA resistance index in 15 preterm infants with severe ROP. 5 Using laser speckle flowgraphy imaging, a technique that requires sedation and pharmacological mydriasis, intravitreal bevacizumab was found to result in decreased venous dilation, arterial tortuosity, and overall ocular blood flow in four preterm infants from Japan. 24 The same group had previously also found reduced ocular blood flow in five infants after laser ablation. 25 Taken together, these data suggest that increased retinal blood flow is a hallmark of ROP with plus disease, which increases during progression of the disease and leads to arterial tortuosity, akin to what can be observed in a free-lying garden hose when the faucet is turned on further. Successful treatment with anti-VEGF agents is associated with a slow decline of this excess blood flow, possibly accompanied by a transient increase of end-diastolic and systolic blood flow velocities associated with some vasoconstriction and the transition from plus to normal.
Fundoscopy performed with a binocular indirect ophthalmoscope is indispensable as the reference examination technique for screening of ROP. It is mostly a rapid procedure lasting only a few minutes but carries some pain and stress. 26 Imaging of all parts of the retina, which is pivotal to correctly diagnose ROP and decide about the need for treatment, requires effective drug-induced mydriasis, and the optical axis must be transparent. Concomitant corneal opacities, cataracts, and other anterior and posterior segment abnormalities may impede direct visualization of retinal vessels. Moreover, pupils may fail to dilate after administration of eye drops in infants with pronounced plus disease featuring iris vascular engorgement, pupillary rigidity, and vitreous haze. 11 Adjunctive diagnostic tools including color Doppler measurements of retinal blood flow might help to estimate disease severity and aid in determining the requirement for treatment in such cases. The measurements can be used at the bedside without sedation of the infant and only require a limited amount of training. However, it remains a topic of future investigations to determine if such ultrasound examinations can be employed to monitor the course of the disease more closely, to improve our understanding of ROP pathophysiology, and to provide ancillary decision aids to treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
De-identified data that underlie the results reported will be supplied to investigators whose proposed use of the data has been approved by a learned intermediary and the institutional review board of the Charité Universitätsmedizin Berlin.
Compliance with Ethical standards
Prior written informed consent was obtained from all parents or guardians.
Ethical approval
The study was conducted according to the tenets of the Declaration of Helsinki and the Ethics Committee of the Charité Universitätsmedizin Berlin (# EA2/002/16).
Animal research
The article does not contain any studies with animals.
Informed consent
Written Informed consent for the treatment protocol and the data collection was obtained from the parents or guardians.
Consent to publish
All authors agreed to publish the article.
