Abstract
Purpose
To report a case of neurogenic dry eye (NDE) that developed after intravitreal ranibizumab injection (IVR).
Case report
A 71-year-old woman had a history of cataract phacoemulsification and intraocular lens (IOL) implantation, Nd:YAG laser posterior capsulotomy and femtosecond laser-assisted laser in situ keratomileusis (FS-LASIK). Aneurysmal dilatation of the inferior temporal retina was found in the left fundus, which was diagnosed as retinal aneurysm, and intravitreal ranibizumab injection was administered. Dry eye was found in the left eye after the first injection. After the second injection, the patient developed severe dry eye with left eye dryness, photophobia, irritation, and blurred vision. The tear film breakup time (TBUT) value was 3 s, the Schirmer test value was 2 mm/5 min, corneal fluorescein staining (CFS) showed diffuse patellar staining of the corneal epithelium with a staining area of >50% and irregular staining at the edge of the corneal flap, and the corneal touch threshold value in the filament length was 1.5 cm in the left eye.
Conclusions
Patients with a history of intraocular lens implantation, Nd:YAG laser posterior capsulotomy, and femtosecond laser-assisted laser in situ keratomileusis surgery may have increased drug permeability due to an increased concentration of anti-VEGF drugs in the aqueous layer and thinning of the stromal layer of the cornea. The corneal subepithelial nerve repair mechanism was destroyed, causing neurogenic dry eye.
Keywords
Introduction
Ranibizumab (Lucentis®, Genentech, South San Francisco, CA), a humanized monoclonal antibody fragment, is an antibody derivative/fragment that can resist vascular endothelial growth factor (VEGF). Intravitreal injection (IVI) can effectively reduce retinal capillary leakage and improve retinal microcirculation.1,2 Anti-VEGF drugs work by binding molecules and blocking and lowering the level of active VEGF in the eye, thereby reducing abnormal angiogenesis and leakage while improving vision. The drugs have a favourable safety profile, with only rare reports of adverse vision-threatening events such as endophthalmitis, vitreous haemorrhage, retinal detachment, and retinal tears, despite their widespread use. 3 At present, ocular surface complications caused by anti-VEGF drugs include corneal epithelial defects, corneal oedema, delayed corneal healing and limbal dysfunction.4–6 To the best of our knowledge, no case of dry eye after intravitreal ranibizumab injection (IVR) has been reported.
Case report
A 71-year-old female patient underwent binocular phacoemulsification and intraocular lens (IOL) in an external hospital in September 2019. In March 2020, she went to the ophthalmology department complaining of obvious visual acuity loss after left eye surgery. Her medical history was unremarkable except for hypertension and hyperlipidaemia. Her uncorrected visual acuity (UCVA) was 20/20 in the right eye and 20/500 in the left eye. The biomedically corrected IOP (bIOP) was measured using a Corvis ST noncontact tonometer (Oculus, Wetzlar, Germany), and the bIOP was 14.8 mmHg in right eye and 15.5 mmHg in left eye. Intraocular lens implantation in the left eye, posterior capsular opacification and unclear fundus (grade II). After Nd:YAG laser posterior capsulotomy of the left eye, visual acuity was improved to 20/50, and fundus examination showed no abnormalities. Visual ghosting was still complained of after Nd:YAG laser posterior capsulotomy. Subjective refraction revealed −1.50DS/-2.75DC*160° in the left eye, and the bIOP was 16.2 mmHg in left eye. After wearing glasses, dizziness and nausea occurred and caused discomfort, so the patient could not tolerate it and refused to wear glasses. The corneal thickness measured by corneal topography was 558 µm, and the number of corneal endothelium was 1850, which ruled out the possibility of keratoconus. With the consent of the patient, topography-guided FS-LASIK was performed in October 2020 (Wavelight ® FS 200 Hz femtosecond laser was used to make corneal flap and Wavelight® EX500 excimer laser cutting). Postoperative examination: her UCVA was 20/20, subjective refraction revealed +0.25DS/+0.50DC*25°, the bIOP was 13.1 mmHg and the corneal thickness was 452 µm in the left eye.
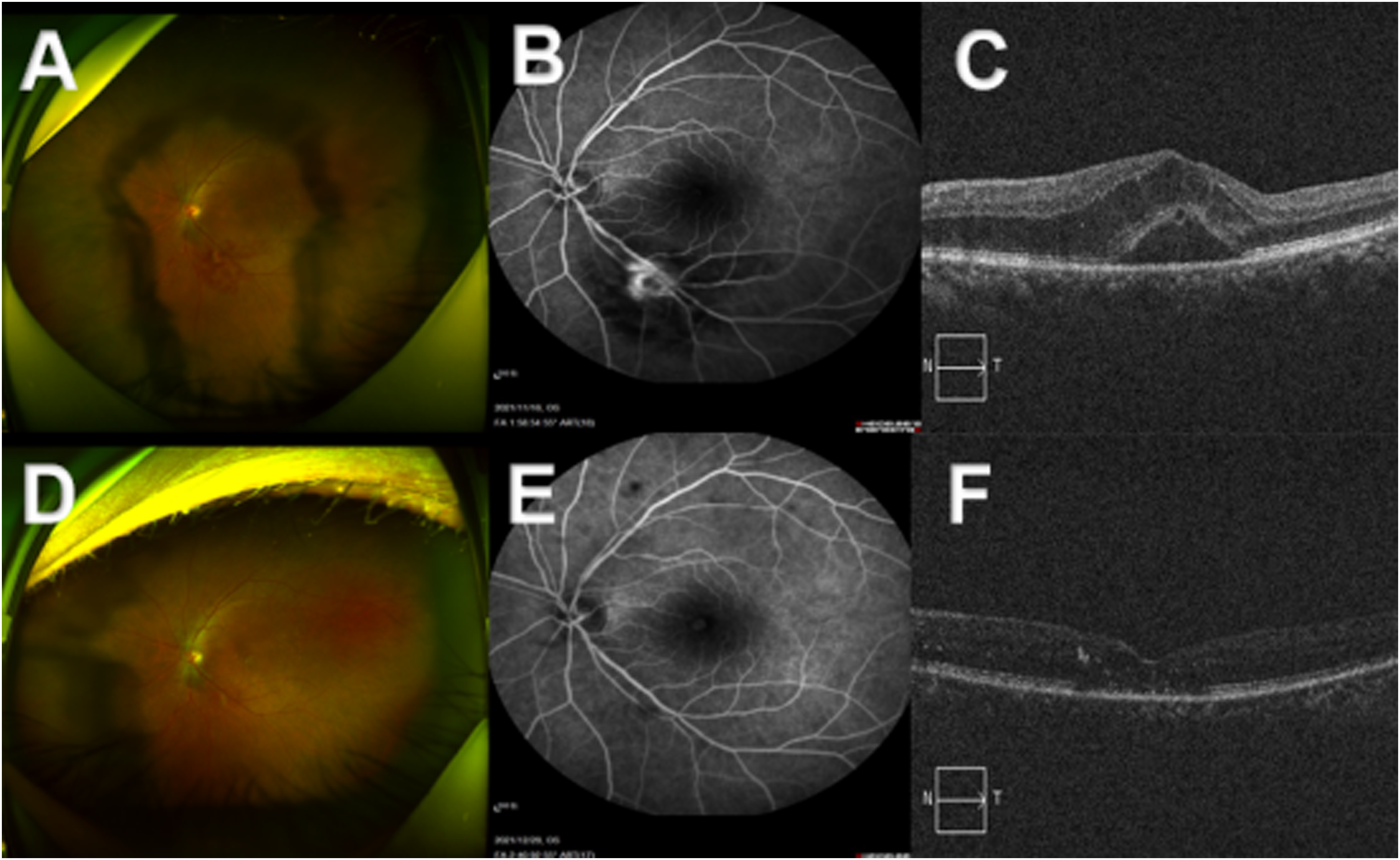
A: The retinal artery is thin with enhanced reflection, and an orange-red round cystic dilatation of the inferior temporal artery, and flake hemorrhage can be seen around it. B: In the early stage, the tumor was quasi-circular and slightly high fluorescence, and the fluorescein leakage in the late stage. C: Cystoid macular edema, Neuroepithelial interlayer edema and exudation. D: The cystic expansion of the inferior temporal branch artery of the left fundus was significantly smaller and the bleeding under the temporal disappeared after the second IVR. E: the aneurysm shrinks and becomes smaller, there is no fluorescent leakage, and the distal artery is filled with defect after the second IVR. F: macular edema subsided, and punctate strong reflex was seen locally in the retina after the second IVR.
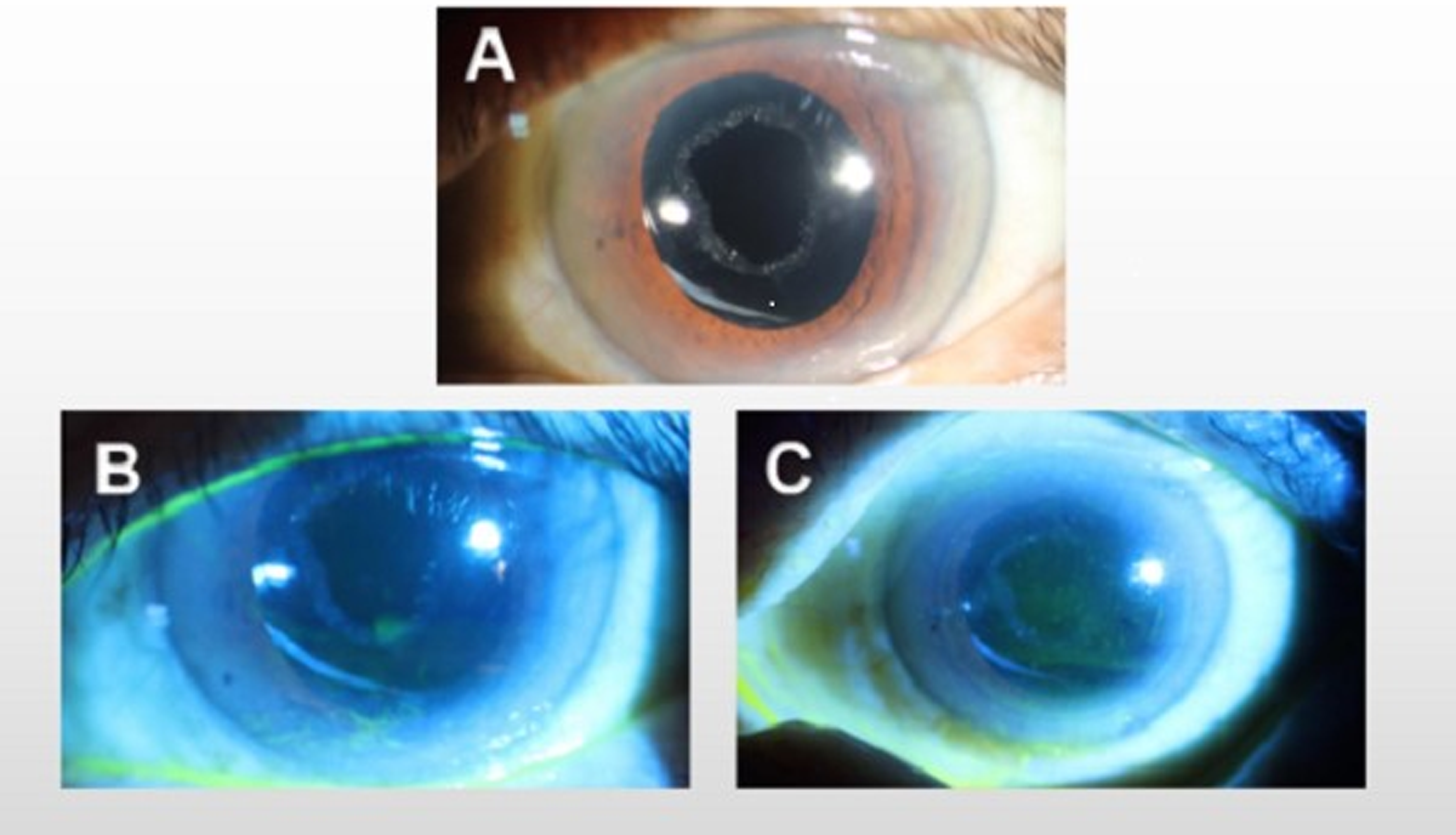
A: Slit-lamp microscopic photographs from the left eye of before IVR. B: Slit-lamp microscopic photographs from the left eye after the first injection, there was significant inferior punctate epitheliopathy and a less severe band of staining inferior to the central ablation zone. C:Slit-lamp microscopic photographs from the left eye after the second IVR,there was diffuse staining of corneal epithelium, staining area >50% and irregular staining at the edge of corneal flap.
In November 2021, the patient went to our department complaining of a sudden decrease in visual acuity in the left eye. The UCVA was 20/20 in the right eye and 20/200 in the left eye, and the bIOP were 15.4 mmHg in right eye and 13.7 mmHg in left eye. The TBUT values were 13 s in the right eye and 9 s in the left eye, and the Schirmer test values were 12 mm/5 min in the right eye and 8 mm/5 min in the left eye. Corneal sensation was assessed with a Cochet–Bonnet corneal anaesthesiometer. The corneal touch threshold values in the filament length were 5.0 cm in the right eye and 4.7 cm in the left eye. Fundus examination showed an orange–red round cystic dilatation of the inferior temporal artery, with patchy haemorrhage and macular oedema around. Fundus fluorescein astronomy (FFA) (Spectralis HRA2, Heidelberg, Germany) showed that in the late stage, there was circular-like high fluorescence with fluorescein leakage under the inferior temporal part. The patient was thus diagnosed with retinal aneurysm and was treated with intravitreal ranibizumab injection (IVR) in the left eye. IVR was performed in our department following standard procedures. These included the administration of one drop of oxybuprocaine hydrochloride 0.5% and one drop of Levofloxacin 0.3% to the eye, followed by injection of 0.05 ml ranibizumab into the vitreous cavity 3.5 mm posterior to the corneal limbus. Levofloxacin 0.3% eye drops were used 4 times a day after surgery.
One week after the operation, the UCVA was 20/32, and the bIOP was 14.1 mmHg in left eye. Fundus examination showed that the bleeding area inferior to the left temporal area was reduced, and macular oedema was significantly reduced. The patient complained of dryness and foreign body sensation in the left eye after injection. The TBUT was 7 s, and the Schirmer test value was 6 mm/5 min. Corneal fluorescein staining (CFS) showed significant inferior punctate epitheliopathy and a less severe band of staining inferior to the central ablation zone. The corneal touch threshold value in the filament length was 3.0 cm in the left eye. The patient was treated with preservative-free artificial tears 6 times a day.
In December 2021, the second IVR was performed. One week after the operation, the UCVA was 20/25, and the bIOP was 13.5 mmHg in left eye. The patient complained of dryness, photophobia, irritation and blurred vision in the left eye since the injection, which was more serious than that after the first injection. Fundus examination revealed that the cystic expansion of the inferior temporal branch artery was significantly smaller than before, the bleeding disappeared, the macular oedema subsided, and FFA showed that the tumour body atrophied and decreased. The TBUT was 3 s, and the Schirmer test was 2 mm/5 min. The CFL showed diffuse staining of the corneal epithelium and irregular staining at the edge of the corneal flap. The corneal touch threshold value in the filament length was 1.5 cm in the left eye. Ranibizumab concentrations in aqueous humour samples were quantified by concentrations in aqueous humour samples by enzyme-linked immunosorbent assay (ELISA). Approximately 0.15 ml aqueous humour samples were obtained via corneal paracentesis anterior to the limbus. The concentration of unbound ranibizumab in the aqueous humour was 20.3 µg/ml. The patient was treated with 3% diquafosol tetrasodium (DQS) 6 times a day, 0.02% fluorometholone eye drops (Santen) 4 times a day and recombinant bovine basic fibroblast growth factor eye gel 4 times a day. The TBUT value was 8 s, the Schirmer test value was 5 mm/5 min, and the corneal touch threshold value in the filament length was 2.6 cm in the left eye. Figure 1 shows the changes of the patient's fundus before and after IVR. Figure 2 shows the changes in the anterior segment of the patient's eye before and after IVR. Informed consent was obtained from the patient for the purpose of publication.
Discussion
Ralizumab is an anti-VEGF drug widely used in exudative age-related macular degeneration (AMD), diabetic macular oedema (DME), macular oedema secondary to central or branch retinal vein occlusion (RVO) and other vitreoretinal diseases.7–11 Retinal aneurysm is an acquired local dilation of the retinal artery, and VEGF plays a key role in its development. 12 With the development and application of anti-VEGF targeted drugs, anti-VEGF combined with laser photocoagulation has become the main treatment method for retinal aneurysm. Complications of intravitreal anti-VEGF injection can be attributed to injection procedures or drug effects, and no dry eye case has been reported after intravitreal anti-VEGF administration.
Dry eye is a multifactorial ocular surface disease characterized by tear film instability and tear hyperosmolarity and accompanied by ocular discomfort symptoms. The destruction of tear film stability is the core mechanism of dry eye. 13 Corneal nerves play an important role in protecting the cornea from irritants and helping to maintain a healthy ocular surface.14,15 Damage to the corneal nerve leads to the destruction of tear film integrity and function, reduced corneal surface perception, and dry eye. 16 The corneal nerve originates from the ocular branch of the first branch of the trigeminal nerve (TV). The corneal nerve is emitted from the ciliary nerve, first passes through the limbus into the stromal layer of the cornea to form the radial distribution of the corneal stromal nerve plexus, and then branches out between the corneal epithelium. 17 During femtosecond laser-assisted laser in situ keratomileusis (FS-LASIK), all the nerves along the entire circumference (except the hinge) of the flap are transected, leading to decreased corneal surface perception, reduced blink frequency, and reduced tear secretion. 18 Corneal nerve fibres were damaged after LASIK surgery, which inevitably decreased corneal sensitivity. 19 Considering corneal sensitivity, proper corneal nerve fibres are essential for normal sensitivity. 20 Moptom et al. 21 showed that although neural parameters improved over time, the subbasal plexus did not fully recover preoperatively at least 10 years after LASIK. Both the main nerve density and nerve branch density continued to be significantly lower, although neural parameters improved over time. Other studies 22 also found that the recovery rate of nerve density at 6 months postoperatively was less than 25%, the nerve density at 2 years was only 64% of the preoperative values, and the mean nerve density 5 years after the surgery was 24% less than preoperatively.
The regeneration of corneal nerves is a complex process. Nerve growth factor (NGF) plays an important role in the self-recovery of corneal nerve injury. 23 NGF is primarily expressed by epithelial and stromal cells, and it is now believed to be a crucial factor for facilitating corneal nerve regeneration and maintaining normal corneal sensation and an intact corneal epithelium. 24 NGF has been reported to promote the repair of corneal nerve injury after LASIK, increase corneal stroma nerve branches and promote axonal lengthening. 18 Gong et al. 25 found that the corneal sensitivity of rabbit eyes treated with NGF after LASIK was significantly improved and that the stability of the tear film was improved. VEGF is a nutritional factor of the corneal nerve and plays a role in mediating corneal nerve repair. It is possible the decoy mechanisms, such as soluble VEGFR1 receptor, that affect angiogenesis help to regulate and modulate the effect of VEGF on corneal nerves. In the healthy cornea, these mechanisms may quench excessive nerve growth; however, in the injured cornea, downregulation of VEGF quenching is possible, thus promoting neurogenesis in the healing cornea. One possibility would be the existence of two different pathways for signalling by VEGF, one for vessel growth and permeability and another for mediating the neurotrophic effects. 26 VEGF and its receptors are expressed by neurons, which stimulate the proliferation of cortical neurons, protect central and peripheral neurons from hypoxia and death, and promote the growth of peripheral neuron axons. They are important participants in nerve growth. 27 Eyes with a history of anti-VEGF injection showed significantly lower corneal nerve density, total length, fibres, trunks, bifurcations, branches, and beads than eyes without a history of anti-VEGF injection. 28 After bevacizumab eye drops were used in rats, downregulation of NGF caused by VEGF inhibition delayed corneal wound healing, suggesting that NGF and VEGF have mutually promoting effects on neurodegeneration. 29
After IVI, the drug concentration in the aqueous humour is affected by three factors: the state of the vitreous body, lens and posterior capsule. Previous cataract surgery may affect the pharmacokinetics of intravitreal injection drugs, with replacement of the native lens with an artificial implant of less volume, the volume of the vitreous, and therefore the volume of distribution of drug increases.30,31 The posterior capsule-ciliary band system implanted in the IOL is an important diffusion barrier, providing stability and integrity of the anterior and posterior segments. Ohrloff et al. 32 reported that laser posterior capsulotomy with diameters of 3 mm to 5 mm resulted in a loss of protective barrier and showed accelerated diffusion of fluorescein in the anteroatrial direction. The highest drug concentration of approximately 36.9-66.1 μg/mL was detected in aqueous fluid on the first day after 0.5 mg of intravitreal injection of ranibizumab in patients without vitrectomy, and the elimination half-life (T1/2) was 7.19 days. 33 In this case, aqueous humour was extracted for 7 days, and the concentration of ralizumab was 20.3 μg/mL, which was significantly higher than that of the patient with vitreous injection under natural lens conditions, indicating that after IOL implantation, the concentration of vitreous drug diffused to the anterior chamber after laser posterior capsulotomy.
Corneal drug permeability is known to be affected by drug properties, such as lipophilicity and molecular size. 34 Corneal epithelial and endothelial cells are lipid soluble, while the stromal layer of the cornea is water soluble, forming a lipid-water-lipid sandwich layer. 35 Corneal epithelial cells have tight connections in the shallowest cells, which hinders the penetration of paracellular drugs from tear film to epithelial cell space, while corneal endothelial cells have loose connections, which is conducive to the passage of drugs in the anterior chamber (such as ralizumab) between the aqueous humour and stromal layer. 36 The high concentration of ralizumab in the patient's aqueous fluid (20.3 μg/ml) further increased drug permeability to the thin matrix (383 mm) that had been laser-cut after FS-LASIK.
Ranibizumab is a small monoclonal antibody Fab fragment (48 kDa) that can easily penetrate into the corneal subepithelium from the aqueous environment through the corneal endothelium to the significantly thinned matrix. The expression of VEGF was downregulated by ranibizumab, and the expression of NGF, which is significantly related to VEGF, was also decreased. Corneal nerve injury during LASIK is a pathogenic factor of neurogenic dry eye (NDE). One year after surgery, the subbasal corneal nerve plexus could not recover its preoperative pattern, and the density of major nerves and nerve branches decreased significantly. The decreased expression of VEGF and NGF destroys the corneal defence and repair mechanism, decreases corneal sensitivity, and reduces reflex tear secretion and blink regulated by corneal nerve reflex, resulting in tear film instability and NDE.
As intravitreal injection of anti-VEGF drugs has been widely used in the treatment of vitreoretinal and macular diseases, its potential adverse reactions, such as endophthalmitis, lens injury, increased intraocular pressure, and vitreous haemorrhage, have attracted attention, but ocular surface complications should also be of great concern. In this case, we report the first case of retinal aneurysm resulting in NDE after IVI. For such patients, the use of artificial tears at the same time to reduce the occurrence of dry eyes during anti-VEGF treatment; If the patient's dry eye worsens after anti-VEGF treatment, local immunosuppressive agents, Intense pulsed light and lacrimal duct embolism can be used to relieve the symptoms of the patient. The history of intraocular lens implantation, Nd:YAG laser posterior capsulotomy, and FS-LASIK may be a risk factor for NDE induced by IVI, which requires further verification by more clinical cases.
Footnotes
Acknowledgements
The authors thank the patient for agreeing to have his case published
Author contributions
Conceptualization: YJ L.
Writing—original draft: SHL,YHF
Writing—review & editing:YJ L,SHL, FLJ
Consent for publication
Written consent for the publication of identifying clinical/personal information and images was obtained from the patient.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
