Abstract
Fungal keratitis (FK) is a serious pathogenic disease usually associated with serious ocular complications. The current mainstay of treatment for FK is topical eye drops; however, poor corneal penetration, low bioavailability of the drug and the need to administer high and frequent doses due to the presence of an effective clearance mechanism in the eye result in poor patient compliance. Nanocarriers can extend the duration of drug action through sustained and controlled release of the drug, protect the drug from ocular enzymes and help overcome ocular barriers. In this review, we discussed the mechanisms of action of antifungal drugs, the theoretical basis for the treatment of FK, and recent advances in the clinical treatment of FK. We have summarized the results of research into the most promising nanocarriers for ocular drug delivery and highlight their efficacy and safety in the therapy.
Introduction
Fungal keratitis (FK) is a dangerous eye infection that can swiftly result in blindness. 1 Severe inflammation, corneal ulcers, and the appearance of fungal hyphae in the corneal stroma are all common features of the condition. The filamentous fungi (Fusarium and Aspergillus), as well as yeast-like Candida, are the most prevalent causes of keratopathy that evolves into FK.2–5 Excessive use of broad-spectrum antibiotics and steroids can disturb the equilibrium of microorganisms on the corneal surface, making infections more likely to infiltrate eye tissue. Contamination of eye injury by soil and plant leftovers (typically rich in Fusarium) in farm situations and contact lens wear are supporting and susceptible factor for FK, especially in temperate and tropical climates. 6
The current mainstay of treatment for FK is topical eye drops; however, topical administration has low bioavailability, with typically less than 5% of the drug reaching the target tissue. The main reasons for this are pre-corneal loss factors, including tear dynamics, N absorption, short residence time in the conjunctival tissue and the relative impermeability of the corneal epithelial membrane.7–9 There are two main challenges with topical eye drops: low retention rates and low permeability into the intraocular tissues. So far, attempts have been made to improve the bioavailability of ocular drugs by prolonging the retention time in the conjunctival tissue and improving the permeability of the drug through the cornea.
In recent years, nanotechnology has yielded promising results in the development of ocular drug delivery systems for the treatment of FK.10–12 These nanoscale carrier systems offer several advantages over conventional delivery systems, such as protection of the encapsulated drug from degradation and metabolism, increased residence time in front of the cornea and enhanced corneal permeability. Nanoparticles (NPs), including polymeric NPs, polymeric micelles, nano-hydrogel, and lipid NPs, are considered to be the most promising ocular drug delivery systems for the treatment of FK. This article reviews the current state of research on FK, the characteristics of ocular drug delivery, and the application of nanotechnology in ocular drug delivery.
Research status of Fk
Overview of Fk
FK is a global eye infection that can cause visual impairment and, in more severe cases, blindness. 13 This is most common in tropical and subtropical regions, accounting for an estimated 20–60% of all culture-positive corneal infections in these climates. Infected people are usually young, healthy agricultural or outdoor workers, and contamination of eye damage caused by soil and plant residues (usually rich in Fusarium) in a farm environment is a supporting and risk factor for FK. 14 In temperate regions, ocular surface diseases, such as insufficient tear secretion and eyelid closure defects, as well as contact lens wear, are prone to Candida keratitis. Ocular trauma caused by excessive use of broad-spectrum antibiotics and steroids can disrupt the balance of microorganisms on the corneal surface, thus promoting pathogens to invade eye tissue. 15
Corneal fungal infections are caused by more than 100 different species, although more than 95% are caused by filamentous fungi Fusarium, Aspergillus and yeast. Filamentous fungi are the cause of most fungal infections in tropical and subtropical climates, while yeast is more common in temperate climates. Corneal infections caused by filamentous fungi tend to have a worse prognosis than those caused by yeast.
Subacute eye pain is the most common symptom of FK, which is followed by impaired vision, redness, copious tears or tears, and photophobia. Corneal perforation is a common complication that is five to six times more likely than bacterial keratitis and frequently necessitates corneal removal. Each year, more than one million people may be affected by FK; approximately 10% of eyes will be punctured or removed, and more than 60% of patients will lose vision in one eye (even if treated). 14
Clinical treatment options
Polyenes, azoles, and pyrimidines are three types of antifungal medications that are now used to treat FK 16 (Table 1). The United States Food and Drug Administration (FDA) has approved natamycin (NAT) as the only commercial drug for the treatment of FK. Only local use of NAT suspension is permitted. Amphotericin B (AmB), itraconazole (ITZ), ketoconazole, and fluconazole are examples of medicines that can be delivered systemically in alternative ways. 17
Classification and mechanism of action of antifungals used in FK.
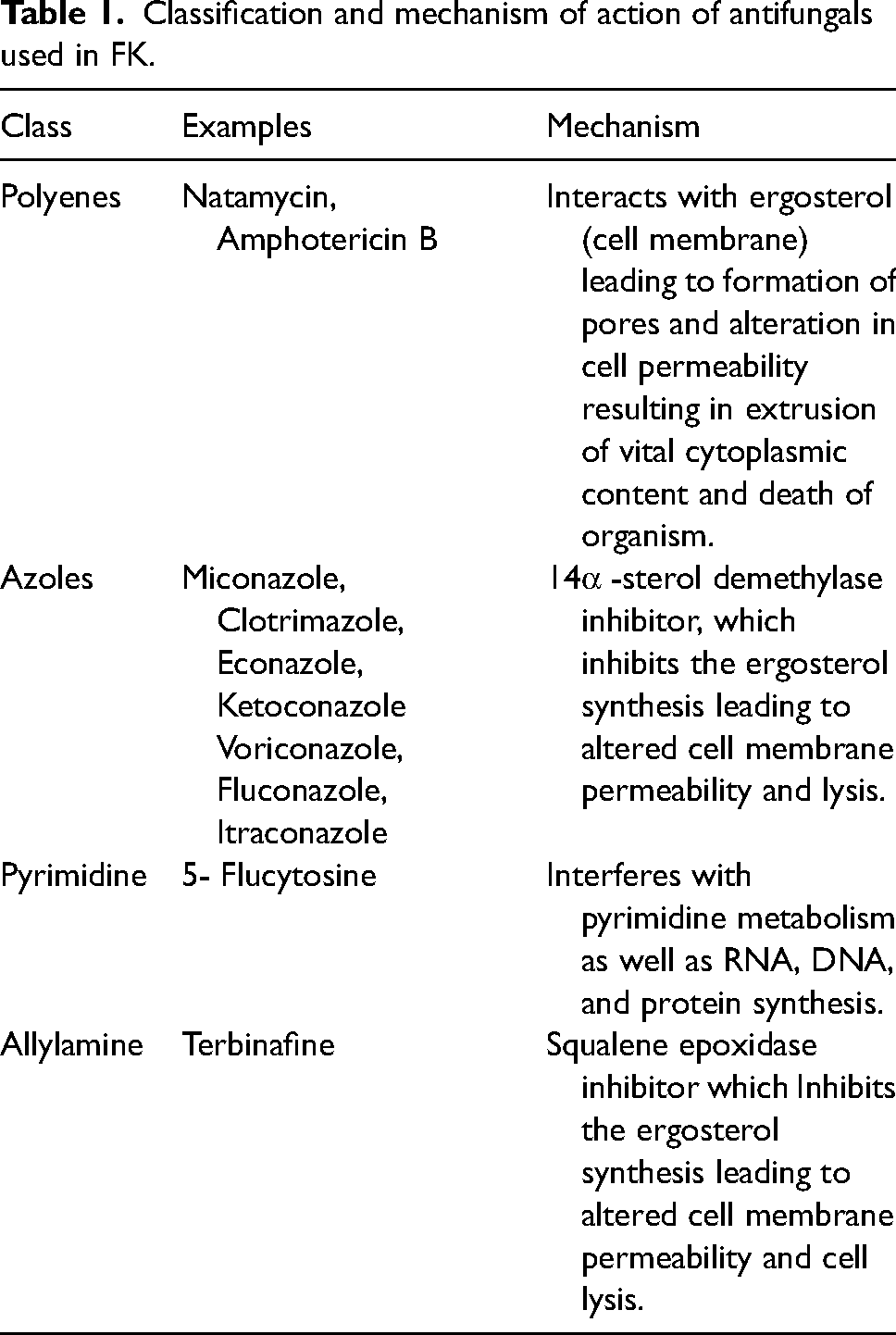
Topical treatment with antifungal medications, including polyenes such as 0.25% AmB eye drops and 5% NAT eye drops, Imidazoles such as 0.5% miconazole eye drops and Pyrimidines such as 1% flucytosine eye drops, 0.15% AmB and 5% NAT eye drops are the first-line antifungal agents. Filamentous fungi should preferably be treated with to 5% NAT, while yeast spp. can be treated with 0.15% AmB, 2% fluconazole, 5% NAT or 1% flucytosine. The more definitive combination regimens are flucytosine + AmB or fluconazole, rifampicin + AmB, etc. Antifungal drugs such as miconazole (5–10 mg) or AmB (0.1 mg) can also be administered systemically, e.g., intravenous miconazole 10–30 mg per kg body weight, divided into three doses per day, usually not exceeding 600 mg per dose. After taking effect, drug treatment needs to be maintained for at least six weeks.
Polyenes antifungals
Polyenes are naturally occurring antifungal compounds produced by the fermentation of actinomycetes, all of which contain a lipophilic macrocyclic lactone ring with an amino sugar attached to the molecule. The structure contains both lipophilic polyene hydrocarbon chains and hydrophilic polyol chains, thus exhibiting amphiphilicity, which also results in poor solubility in water and most organic solvents, and only good solubility in polar organic solvents, such as glacial acetic acid, dimethylformamide and dimethyl sulfoxide.
Eukaryotic cell membranes contain lipids, phospholipid proteins and sterols (ergosterol), which play an important role in the selective action of polyene antifungal drugs. The irreversible binding of polyene antifungals to ergosterol leads to extravasation of intracellular substances such as K+, amino acids and nucleotides by increasing the permeability of the cell membrane, disrupting normal cellular metabolism and thus inhibiting fungal growth.18–20
Azole antifungals
Azole antifungals are also divided into imidazoles and triazoles. Triazoles are discovered on the basis of imidazole research, and the representative drugs of the former are clotrimazole, econazole (ECZ) and ketoconazole, and the latter are fluconazole, ITZ and voriconazole (VCZ). The azoles are used to inhibit the biosynthesis of ergosterol by inhibiting 14-α demethylation. The N atom on the azole can form a complex binding the ferrous ion on the cofactor of fungal CYP450 enzyme, ferric hemoglobin, which inhibits the process of de14-α methylation of CYP450 enzyme, causing ergosterol to lose its normal shape and physical properties, leading to leakage due to membrane permeability change, thus leading to fungal cell death.21,22
Pyrimidine antifungals
The representative drug of pyrimidine antifungal drugs is fluorocytosine. The mechanism of action is that the drug enters the cell through the permease system of the fungal cell and is converted to 5-fluorouracil by the action of cytosine dehydrogenase, which replaces uracil in RNA, or is metabolized to 5-fluorouracil deoxyriboside, which inhibits thymine nucleoside synthase, all with the end result of blocking fungal DNA synthesis. Mammalian cells cannot convert flucytosine to 5-fluorouracil and are therefore unaffected by this drug. 23 Flucytosine is mainly used for infections caused by candidiasis, cryptococcosis and other sensitive fungi. As the fungus is susceptible to resistance when used alone, it needs to be combined with antifungal drugs such as AmB to have a synergistic effect.
Ocular drug delivery barriers
There are many routes of ocular administration, including topical administration, systemic administration, subconjunctival injection, anterior chamber injection, vitreous injection and intrastromal injection. Topical administration is the most suitable route of administration for ophthalmic patients because the ocular surface is the most accessible site for ocular drug delivery, which is more direct and especially suitable for the treatment of anterior segment diseases. Topical administration bypasses the first-pass effect in the liver, avoids the higher doses required for oral administration, and reduces potential adverse effects. However, the presence of multiple ocular barriers (Figure 1), such as tear film, corneal, conjunctival, and dynamic barriers, which allow excessive drug loss, remains a major drawback of topical administration. 24
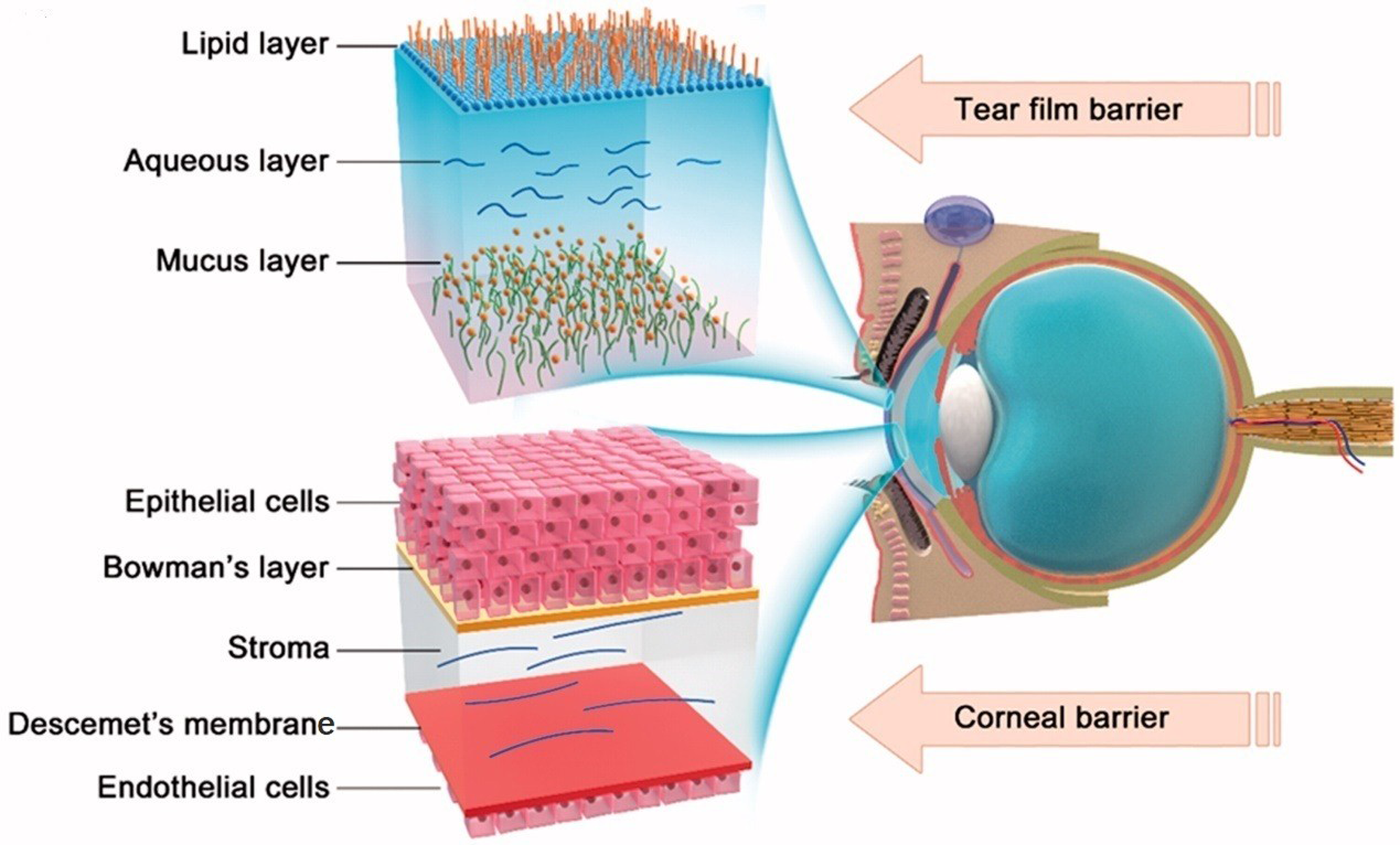
Two major barriers to topical ocular drug delivery. Tear film barrier: a high tear turnover rate and gel-like mucus layer. Corneal barrier: tight junctions in the corneal epithelial cells and alternating polarity five-layer structure. Reprinted from Ref. 25 with permission. Copyright 2018, Taylor & Francis Group.
Tear film barrier
The tear film is the first barrier encountered after topical administration of drugs to the eye. 26 The tear film consists of three layers: a lipophilic outer layer, a hydrophilic middle layer, and a mucus inner layer. Therefore, the hydrophilic/lipophilic nature of the drugs determines whether they can successfully pass through the tear film. In addition, the lacrimal gland moisturizes the cornea and conjunctiva by continuously secreting tear fluid, but the tear renewal rate is only 1 μL/min, and the excess fluid flows into the nasolacrimal duct, which is the main reason for the low bioavailability of drugs. 27 Thus, the physicochemical properties of drug molecules and their carriers, as well as their interaction with the tear fluid, determine whether the drug can cross the tear film barrier and enter the underlying tissues.
Corneal barrier
The cornea consists of the corneal epithelium, the preelastic layer, the stroma, the posterior elastic layer, and the endothelial cell layer. 28 The corneal barrier is composed of mature epithelial cells, and the tight junctions between the cells limit the diffusion of large molecule drugs and water-soluble drugs through the cornea; the rest of the corneal tissue is composed of extracellular matrix, which accounts for about 90% of the corneal thickness and is mainly composed of collagen, keratin sulfate, cartilage sulfate and keratinocytes. 29 The stroma forms a barrier to lipophilic drugs, limiting their penetration; the corneal endothelial cell layer prevents hydrophilic drug molecules from entering the aqueous humor, and this “sandwich-like” structure makes the cornea a unique barrier to most lipophilic and hydrophilic drugs. 30 The corneal surface is generally negatively charged, and drug molecules with a positive surface charge are more likely to bind to and permeate through the cornea than those with a negative charge. Thus, drug penetration in the cornea depends on its molecular weight, charge and hydrophilicity/hydrophobicity.
Conjunctival barrier
The conjunctiva is composed of epithelial cells, cupped cells, and basement membrane. The surface area of the human conjunctiva is several times larger than that of the cornea, and is also much more permeable than the cornea. However, the conjunctival epithelial cells are bound together by tight junctions at the very tip of the epithelium, and the tight junctions between adjacent cells form a barrier. 31
NPs-based ocular drug delivery system
Recent developments in nanotechnology can provide opportunities to overcome the shortcomings and limitations of traditional drug delivery systems, such as low drug bioavailability and low drug penetration into the ocular barrier. Nanocarriers can prolong the action time of drugs through sustained and controlled release, protect drugs from ocular enzymes, and help overcome eye barriers. This can greatly reduce the frequency of administration and increase the tissue concentration of the drug, thereby achieving better pharmacological effects. Researchers have designed multiple nanoformulations for ocular antifungal delivery (Table 2).
A summary of nanocarriers for the treatment of FK studied in recent years.

NAT: natamycin; ECZ: econazole; AmB: amphotericin B; VCZ: voriconazole; TCZ: terconazole; ITZ: Itraconazole; PDG: Poly-D-glucosamine; PCL: Polycaprolactone; CTS: chitosan; α-CD: α-cyclodextrin; HCA: catechol (3,4-dihydroxycinnamic acid); PLGA: poly(lactic-co-glycolic acid; PGMA: poly (glycidyl methacrylate); PPG: poly (propylene glycol); PBA: phenylboronic acid; VE: vitamin E; PEG: polyethylene glycol; DSPE: 1,2-distearoyl-sn-glycero-3-phosphoethanolamine; TMC: Trimethyl chitosan; PEI: polyethylenimine; TPGS: Tocopheryl polyethylene glycol succinate; Na.CMC: Sodium carboxymethylcel-lulose; CIP: Ciprofloxacin.
Polymeric NPs
Nanoparticle polymers commonly used in ophthalmology include poly(lactic-co-glycolic acid) (PLGA),53,54 polyethylene glycol (PEG), 55 polycaprolactone (PCL),56,57 chitosan (CTS),58,59 albumin, 60 and gelatin.61,62
CTS is a naturally occurring cationic polysaccharide with good biocompatibility. In recent years, CTS has been widely used in ocular drug delivery systems, where its positively charged amino groups can adhere to anionic muconic acid residues on the corneal surface, increasing the drug residence time in the eye. 63 In addition, it can open the tight junctions between corneal epithelial cells and improve the penetration of drugs in the cornea.64,65 Bhatta et al. 37 prepared lecithin/CTS NPs by ionic gelation method. Ocular pharmacokinetic results in New Zealand rabbits showed that the NPs exhibited significant mucin adhesion. When compared to commercially available suspensions, the AUC (0-∞) was increased 1.47-fold and the clearance was reduced 7.4-fold. According to PK-PD and pharmacokinetic simulations, the ideal dosing interval for NAT and NPs to maintain approximately two times the MIC90 was 120 and 210 min, respectively. In another study, Chandasana et al. 33 prepared poly-D-glucosamine-functionalized polycaprolactone NPs (PDG-PCL NPs) by nano-precipitation method. NAT could be released for up to 8 h in vitro, according to studies. When compared to the marketed Natamet® (5% w/v), the in vitro antifungal activity was exactly equivalent, the pharmacokinetic parameters were significantly higher, while clearance was significantly lower. To keep NAT concentrations above 10 times the MIC90, one dose every 5 h is the best dosing regimen.
Li et al. 25 synthesized a drug delivery system based on a CTS-conjugated cyclodextrin (CTS-c-CD, abbreviated as CC) polymeric carrier. The results of in vivo and in vitro experiments showed that the prescription loaded with ECZ exhibited significant corneal penetration with good antifungal activity and tolerance after a single ocular surface administration. The intracorneal drug concentration and relative ocular bioavailability were 59 and 29 times higher than those of the control group, respectively. On this basis, catechol (3,4-dihydroxycinnamic acid, HCA) was found to form covalent bonds with thiol and amine groups in the mucus layer via catechol oxidation. 34 Thus, coupling the adherent HCA to the CTS appears to create an upgraded drug delivery platform (CTS-c-(CD + HCA), or CCH for short) loaded with ECZ. This platform appears to enhance drug penetration in the cornea through physical adhesion and strong interactions of covalent bonds with the mucin layer, which prolongs residence time at the ocular surface and opens up tight corneal epithelial junctions. 66
Weifen Zhang's group developed NAT-CTS NPs using an ionic gel method. 35 The study found that the NPs were more bioavailable in the eye, facilitating their penetration in the inner layer. In another study by Weifen Zhang, a co-loaded drug delivery system with an average hydrodynamic particle size of 309–406 nm was prepared using coaxial electrospray (CO-ES) technology for the co-delivery of NAT and clotrimazole (CLZ) with CTS and PLGA using electrostatic spray. 36 In vitro results showed that the combination of CLZ and NAT exhibited a synergistic antifungal effect and ocular irritation studies determined no significant damage or irritation to the cornea. 36
Based on the above research work, we can conclude that CTS-based natural NPs show good results in the ocular delivery of antifungal drugs, mainly due to their adhesive properties. Further functionalization of CTS with a targeting fraction helps to achieve site-specific targeting and reduce toxicity, also showing enhanced antifungal activity.
Polymeric micelles
Polymeric nanomicelles are colloidal dispersion structures with a spherical hydrophobic core-hydrophilic shell formed by self-assembly of amphiphilic carriers in water. This colloidal dispersion structure can encapsulate hydrophobic drugs in the hydrophobic core, improve the solubility of hydrophobic drugs, reduce the adverse effects of drugs, and thus improve the bioavailability of drugs. The hydrophilic shell forms a spatial site barrier that prevents aggregation and ensures solubility of micelles in water or biological environments. In addition, the outer shell can also play the role of drug protection, maintain drug stability, and play the role of slow release. Polymeric nanomicelles, as an ocular drug delivery system, can improve the interaction with the corneal surface and increase the diffusion of drugs in the cornea due to properties such as mucosal adhesion.
PEG-block-poly(glycidyl methacrylate) (PEG-b-PGMA) micelles were synthesized to encapsulate NAT. 37 The epoxy group in PGMA can undergo ring-opening reactions at neutral pH to form hydroxyl groups, which increase the inherent hydrophilicity of the hydrophobic core region of the micelles and provide an additional driving force for drug release, which will encapsulate the hydrophobic drug from the resulting hydrophilic region ‘push’ into the surrounding environment. The shell of the polyethylene glycolized micelles prolongs contact time with the mucus layer of the tear film, acts as a mucus adhesion promoter, and enhances corneal penetration of the drug-laden micelles. The sustained release of NAT from micelles reduced the number of daily doses of NAT from eight to three in rabbits with FK. 67 Han et al. 38 prepared polyhedral oligomeric silsesquioxane (POSS)-modified PEG-PPG copolymers that could self-assemble into micelles for ocular delivery of AmB. POSS is a hydrophobic, highly porous nanomaterial with good bioadhesion. The POSS fragment-mucin binding assay and corneal permeability test proved that the micelle-encapsulated AmB had prolonged residence time on the ocular surface and improved the therapeutic effect. In another report, Sun et al. 39 prepared phenylboronic acid-conjugated chitooligosaccharide-vitamin E copolymer (PBA-CS-VE) for VCZ-adhesive nanomicelles in the treatment of FK. Nanomicelles surface modified with PBA specifically targets and binds to the sialic acid residues in the ocular mucosa. In vitro mucin-binding eyeball adhesion experiments show that the copolymer has strong adhesion.
In general, nanomicelles have received extensive attention as carriers for ocular drug delivery systems. This nanocarrier has the potential to improve ocular bioavailability and patient compliance. Nanomicelle formulations can be used to treat intraocular and ocular surface diseases by topical route of administration.
Lipid-based carriers
Niosomes
Niosomes possess of hydrophilic and hydrophobic micro-regions, which gives them the ability to transport both water-soluble and water-insoluble drugs. At the same time, vesicles have a bilayer membrane structure and good compatibility with biological membranes, making them ideal carriers for ocular delivery of drugs. Compared with liposomes, niosomes have the advantages of better stability, biodegradability, biocompatibility and non-immunogenicity. ElMeshad et al. 45 prepared ITZ-loaded spanlastics with Span 60, Tween 20 and Tween 60 using ethanol injection method. Spanlastics showed a significant increase in the zone of inhibition of Candida albicans cultures compared to ITZ powders. In another report, Ultradeformable bilosomes (UBs) were prepared by ethanol injection method using terconazole, Span 60 and cholesterol as organic phase and bile salt (sodium taurocho-late) and edge activator as aqueous phase. 44 The edge activator was used for the first time to enhance the permeability of bilosomes. In vitro corneal permeability studies showed that the studied UBs significantly enhanced the drug penetration.
The natural polymer CTS and its derivatives have also been used in the modification of niosomes to further enhance corneal permeability. Verma et al. 43 first prepared vesicles containing NAT, Span 60 and cholesterol using a thin film hydration method and subsequently wrapped them with N-trimethyl CTS (TMC) to make them corneal-adhesive. The optimized niosomes had a particle size of 1034 nm, wwith a positive charge, and could be released continuously for 12 h. The TMC-coated niosomes had a higher mucin binding efficiency compared to the uncoated niosomes.
Solid lipid NPs
Solid Lipid NPs (SLNs) are the first generation of lipid NPs that consist of solid natural or synthetic lipids encapsulated in a solid gel drug delivery system. 68 SLNs are derived from O/W emulsions, replacing the liquid lipid of the emulsion with a lipid matrix, which is solid at body temperature and is stabilized by surfactants. Khames et al. 40 prepared NAT-SLN formulations based on thermal emulsification-ultrasonication technique using Precirol ATO 5® as solid lipid, pluronic F68 as surfactant and stearamide as surface charge improver. The optimized NAT-SLNs drug release profile was extended to 10 h with elevated apparent permeability coefficients. Antifungal studies showed that the antifungal effect of NAT-SLNs was significantly improved and the minimum inhibitory concentration was reduced by about 2.5-fold.
Nanostructured lipid carrier
Nanostructured Lipid Carrier (NLC) is formed by adding liquid lipids to solid lipids and mixing the lipids to form a NLC. 69 The liquid lipid destroys the lattice structure of solid lipid, increases the proportion of irregular crystalline shape, and increases the drug loading capacity through space expansion. Adjusting the proportion of liquid lipids ensures that the nanostructured carriers maintain the skeletal structure in vivo, and achieves slow and controlled drug release. Compared with SLN, the advantage of NLCs is to increase the drug loading capacity and achieve slow and controlled drug release. 70 Lipid NPs enhance the drug loading and penetration of lipophilic molecules. 71 Adhesion, stability, and invisibility NPs are obtained by surface modifiers such as PEG, CTS, and lipids containing amine functional groups.
PEG makes the nanoparticle surface hydrophilic and improves ocular bioavailability by interacting with corneal mucus and epithelium. It stabilizes the NPs as well as the drug in the NPs by reducing the contact with the surrounding environment. PEG can be achieved by binding to lipids or electrostatic interactions. The main advantage of conjugated PEG is that NPs do not dissociate from PEG over time in an aqueous environment. In one study, NAT-PEG-NCLs were prepared by thermal homogenization method. 41 Castor oil, Precirol® ATO 5 were used as solid lipid and liquid lipid, respectively. The particle size of NAT-PEG-NLCs was 200–250 nm, and the transcorneal flux of NAT-PEG-NLCs was approximately 2 and 7 times higher than that of NAT-NLCs and Natacyn®in vitro, respectively. This is due to the PEG coating's high hydrophilicity, which allows it to effectively penetrate the mucus layer. Despite the fact that 5% Natacyn® is twice as concentrated in the cornea as NAT-PEG-NLCs (0.3%), NAT-PEG-NLCs can penetrate the intact cornea to reach the iris ciliary body, anterior chamber, and vitreous tissue. 41
In situ hydrogel
The in situ hydrogel can prolong the residence time of the drug on the the corneal surface and improve the permeability of the drug into the cornea to improve the bioavailability of the eye. 72 Therefore, it is more and more widely used in corneal tissue engineering.73,74 Usually, antifungal drugs are prepared as nanoformulations first, and then dispersed in hydrogels to form composite nanoformulations. In situ gel is a kind of preparation which can be transformed from liquid to non-chemical crosslinked semi-solid gel immediately at the drug site by changing physiological conditions (pH, temperature, electrolyte or ionic strength, and light radiation) after administration in solution state. Corneal hydrogels can be divided into synthetic and natural hydrogels according to their origin. Synthetic hydrogels include PEG and cyanoacrylates, among others. Natural hydrogels include collagen, gelatin, CTS, hyaluronic acid, sodium alginate and extracellular matrix hydrogels (decellularized bovine cornea and porcine cornea). Compared with ordinary gels, in situ gels have the advantages of simple preparation, convenient administration and accurate dosage. According to different formation mechanisms, in situ gels can be divided into temperature sensitive in situ gels, ion sensitive in situ gels and pH sensitive in situ gels.
Temperature-sensitive in situ hydrogels
Temperature-sensitive hydrogels use temperature-sensitive polymers, such as poloxamer, CTS, polyn-isopropylacrylamide and other substrates. These polymers exist in liquid form below their low critical dissolution temperature and gelate at or above ambient temperature. Sha et al 46 prepared NAT-loaded tri-block polymer nanoparticles (PLGA-PEI-PEG@NAT) and added them to poloxamer gels (Gel@PLGA-PEI-PEG@NAT). Positively charged PEI could help NPs penetrate the cornea, and PEG modification prolonged circulation of the NPs. Okur et al 50 prepared thermosensitive in situ gels using poloxam 407, poloxam 188, and carboxymethyl cellulose by cold method to increase the residence time and bioavailability of VCZ in the eye mucosa. The concentration of VCZ in the in situ gel preparation was 0.1% (w/w). The gelation temperature of the in situ gel preparation containing VCZ ranged from 29.73 ± 0.21°C to 34.13 ± 0.32 °C. This suggests that the formulations were converted to gel when they dripped onto the surface of the eye. The drug release results showed that the sustained release time of the preparation was 8 h.
Ion sensitive in situ gels
Ion-sensitive in-situ gels mainly react with K+, Na+, Ca2+ and other cations using polymer solutions formed from derivatives of polysaccharides in the body fluid environment to form gels at the site of administration. The commonly used materials are sodium alginate and deacetylated gellan gum. Tear is rich in electrolytes, so ion-sensitive in situ gel systems are rapidly converted to hydrogel in the ocular environment, even at low polymer concentrations. 50 Janga et al 47 prepared NAT bisomes (NB) using the film hydration technique, with a lipid mixture of Span60 and cholesterol selected and sodium taurocholate allowing the bisomes to penetrate the intercellular lipid layer of intact biofilms, and NB was added to nodular gum or xanthan gum dissolved in deionized water to obtain NB in situ gels. The results showed a 6- to 9-fold increase in NB trans-corneal flow. Based on this, this group 48 also prepared a NAT transfer regulator (FN) using phospholipids and low D-alpha tocopherol PEG1000 succinate (TPGS), the addition of gellan gum (0.3% w/v) resulted in immediate in situ gel formation in simulated tear fluid. The formulation also showed higher ocular permeability than the control suspension.
Other in situ gels
Diaz-tome et al. 51 prepared two kinds of ECZ eye hydrogels. ECZ nitrate was first solubilized with α -cyclodextrin and then the inclusion complex of ECZ -α-CD was embedded in a natural polysaccharide ion-sensitive hydrogel (ISH) and hyaluronic acid hydrogel (HAH). The study showed that both hydrogels successfully controlled the release of ECZ nitrate and retained it on the ocular surface for a longer time. NAT biosomes 49 were prepared by reverse-phase evaporation with Span 20, cholesterol, and lipid components with or without dicetylphosphate, and then the optimized NAT biosomes were incorporated into 0.5% Ketorolac tromethamine USP gels with different mucoadhesive polymers Sodium carboxymethylcel-lulose (Na-CMC) and Hydroxypropyl methylcellulose-E4 (HPMC-E4). It was shown that the gels promoted long-term NAT delivery and penetration into corneal tissue.
The gel does not guarantee the physicochemical stability of the drug in the formulation or good targeted drug delivery effect. The bioadhesion of the nano-formulation is similar to that of traditional eye drops, and it cannot adhere to the surface of the eye for a long time, and cannot improve the residence time of the drug on the surface of the eye. Therefore, combining the in-situ gel system with nano-formulations, this new composite system can not only increase the stability of the drug, improve the solubility and targeting effect of the drug through the nano-carrier, but also increases the drug through the in-situ gel drug delivery system. The adhesion of the drug further improves the residence time of the drug on the surface of the cornea. The combination of in situ gel and nano-drug delivery system makes up for the shortcomings of using a single formulation, and the composite nano-formulation has more obvious advantages than a single drug delivery system.
Conclusion
Due to the unique physiology of the eye and the presence of many barriers, drug delivery systems for the treatment of ocular diseases have been a major challenge for pharmaceutical practitioners and also a major problem for pharmaceutical workers. In traditional dosage forms for ophthalmic drug delivery, drug residence time in the eye is short and bioavailability is low, while the use of highly concentrated drugs can cause ocular irritation and in some cases, systemic toxic reactions. With the development of nanotechnology and biomaterials, the delivery and action of drugs to specific tissues based on nanodelivery systems is gradually showing its unique advantages and providing new ideas for the treatment of ocular diseases.
These drug delivery systems can increase the ocular absorption of drugs, prolong the residence time of drugs in the eye, reduce the ocular tissue toxicity of drugs, improve the bioavailability of drugs, and obtain good local therapeutic effects. In recent years, a variety of ocular drug delivery systems have been developed, such as polymeric NPs, polymeric nanomicelles, lipid NPs, and in situ hydrogel. However, ocular drug delivery also faces some challenges, such as some adverse reactions after local administration of certain carriers eyes, and the administration dose and drug release are difficult to control. Therefore, further efforts and exploration are needed to study more effective ocular drug delivery systems.
Footnotes
Acknowledgements
The authors are thankful to the National Natural Science Foundation of China (81973671).
Authors’ contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Availability of data and materials
Not applicable.
Consent for publication
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81973671), the special project of science and technology in 2020 in Qingdao west coast new district (No.2020-45), Shandong Medical and Health Science and Technology Development Plan Project (No.202013020643).
