Abstract
Purpose
To determine the total alpha-synuclein (αSyn) reflex tears and its association with retinal layers thickness in Parkinson's disease (PD).
Methods
Fifty-two eyes of 26 PD subjects and 52 eyes of age-and sex-matched healthy controls were included. Total αSyn in reflex tears was quantified using a human total αSyn enzyme-linked immunosorbent assay (ELISA) kit. The retinal thickness was evaluated with spectral-domain optical coherence tomography. The Movement Disorder Society-Unified Parkinsońs Disease Rating Scale (MDS-UPDRS), Non-Motor Symptoms Scale (NMSS), and Montreal Cognitive Assessment (MoCA) were used to assess motor, non-motor, and cognition.
Results
In PD, total αSyn levels were increased compared to control subjects [1.76pg/mL (IQR 1.74–1.80) vs 1.73pg/mL (IQR 1.70–1.77), p < 0.004]. The nerve fiber layer, ganglion cell layer, internal plexiform layer, inner nuclear layer, and outer nuclear layer were thinner in PD in comparison with controls (p < 0.05). The outer plexiform layer and retinal pigment epithelium were thicker in PD (p < 0.05). The total αSyn levels positively correlated with the central volume of the inner nuclear layer (r = 0.357, p = 0.009).
Conclusion
Total αSyn reflex tear levels were increased in subjects with PD compared to controls. PD patients showed significant thinning of the inner retinal layers and thickening of outer retinal layers in comparison with controls. Total αSyn levels positively correlate with the central volume of the inner nuclear layer in PD. The combination of these biomarkers might have a possible role as a diagnostic tool in PD subjects.
Keywords
Introduction
Parkinson's disease (PD) is the second most common neurodegenerative disorder, affecting 1% of people over 60 and 4% older than 80 years. 1 It is characterized by motor and non-motor symptoms. 2 The pathological hallmark is alpha-synuclein (αSyn) abnormal accumulation and aggregation in the brain and dopaminergic neuronal loss in substantia nigra. Interestingly, αSyn has been identified in other organs, including the retina, skin, heart, liver, appendix, and colon. As well as body fluids like cerebrospinal fluid, blood, saliva and tears.3–5
Visual symptoms are common in PD, 6 including worsening visual acuity, low sensitivity to contrast, and alterations in color vision.7,8 These symptoms are present because of central visual processing deficits and retina or macula dysfunction. 9 The mechanisms that lead to vision impairment in PD are still unclear but are partially due to retinal dopamine deficiency.10,11 The retina is considered an extension of the central nervous system and contains dopaminergic amacrine cells in the inner nuclear retinal layer. 11 These cells help to integrate visual information presented to the ganglion cell. 12 PD subjects showed thinning of the inner retinal layers; similar changes are found in glaucoma and other neurodegenerative diseases. 13 These findings suggest that retinal degeneration could be associated with neurodegenerative diseases. As a consequence, other body fluids are necessary to determine the usefulness of the retina in PD.
The lacrimal gland is the principal source of tear proteins and is innervated by parasympathetic and sympathetic neurons degenerated in PD.14,15 Given the importance of αSyn accumulation and spreading from the brain to the periphery, along with the broad spectrum of ophthalmological manifestations in PD, this study quantified total αSyn in reflex tears and their association with retinal layers thickness among patients with PD. These results may improve our understanding of PD and aid in the search for accessible and non-invasive diagnostic biomarkers in PD.
Methods
A cross-sectional study including subjects with PD diagnosis and age-and sex-matched healthy controls (HC) was carried out. PD subjects were diagnosed according to the MDS clinical diagnostic criteria for Parkinson's disease. 16 They attended the Movement Disorder outpatient clinic at the National Institute of Neurology and Neurosurgery in Mexico City and were enrolled. HC were volunteers recruited from an ophthalmological hospital.
Exclusion criteria for the study's purposes included: systemic diseases, including inflammatory, autoimmune, hematologic, neoplasia, diabetes mellitus, thyroid disease, alcohol addiction, infection, hepatic or renal failure, and use of ophthalmic drops within the last month. In addition, subjects with an ocular disease, including a history of high intraocular pressure, glaucoma, age-related macular degeneration, or other macular diseases that could affect the retinal structure or surgery in the last three months, were also excluded. HC with a family history of PD, parkinsonism, tremor, and dementia was also excluded. All HC underwent neurological examinations.
Patients were evaluated by a neurologist with expertise in movement disorders using the Spanish version of the Movement Disorders Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS), 17 the Non-Motor Symptoms Scale (NMSS), 18 and the Montreal Cognitive Assessment (MoCA). 19 All PD subjects were in the “ON” state during the examination. Clinical and demographic data were collected, including age, gender, age at disease onset, side of symptoms onset, and current antiparkinsonian treatment. Levodopa (L-dopa) equivalent daily dose (LEDD) was calculated. 20 Severity of disease was defined using the Hoehn and Yahr (HY) stage. 21 In HC only the MoCA was used. After the neurologic evaluation, all subjects underwent an ophthalmologic examination of best-corrected visual acuity, autorefractometry with a cutoff for exclusion based on refraction (6D), slit-lamp examination, Goldman applanation tonometry and funduscopy.
All subjects accepted to participate in the study and signed the informed consent form. The study was approved by the local Ethics Committee (protocol 162/19) and achieved in accordance with the Declaration of Helsinki.
Tear sample collection and αSyn quantification
All reflex tears were collected in the morning within a one-hour timeframe around 10 am and 11 am with a Schirmer strip without topical anesthesia. 22 A Schirmer paper strip was placed over the lateral one-third of the lower lid margin in both eyes. The subjects’ eyes were closed, and after 5 min, the strips were removed and stored at −80°C until analysis. Tear proteins were eluted by adding Phosphate-buffer saline with protease inhibitors with consequent centrifugation at 10,000 g for 10 s. Total αSyn was quantified using the Human αSyn ELISA kit (Product KHB0061, Invitrogen, Frederick, MD) according to the manufacturer's instructions. All measurements were achieved in duplicate, averaged, and normalized to total tear protein concentration for comparison between samples. 5
Optical coherence tomography
SD-OCT imaging was performed using the Heidelberg Spectral Domain OCT (Heidelberg Engineering, Heidelberg, Germany) in a dark room by qualified staff. Scans were quality controlled (>15 dB quality) and each retinal layer was segmented (RNFL = retinal nerve fiber layer, GCL = ganglion cell layer, IPL = inner plexiform layer, INL = inner nuclear layer, OPL = outer plexiform layer, ONL = outer nuclear layer, RPE = retinal pigment epithelium, IRL = inner retinal layers and ORL = outer retinal layers). Each retinal layer thickness was divided into circles of 1 mm, 3 mm, and 6 mm, according to the Early Treatment Diabetic Retinopathy Study grid. The inner ring was defined as the central thickness, and the inner and outer rings were divided into four zones designated as the superior, nasal, inferior, and temporal zones. Scans of the mean thickness of each of the nine zones, the total macular volume, and the foveal thickness scans were included in the analysis. 23
Statistical analyses
Statistical evaluations were performed using SPSS software (version 17.0; SPSS Inc., Chicago, IL, USA). The Shapiro-Wilk test was used to examine if the continuous variables were normally distributed. The Wilcoxon Signed Rank Test, the Mann-Whitney-U-Test, and the Spearman rank correlation coefficient were used for analysis since distribution did not meet the criteria for normality. The right and left eyes were analyzed independently. Statistical differences were considered significant if p < 0.05.
Results
26 subjects with PD (52 eyes) and 26 age-and sex-matched healthy controls were enrolled for analysis. The mean age was 63.69 ± 8.91 years old, 18 males (69.2%) and 8 females (30.8%). Twelve (46.2%) among the 26 PD subjects presented motor symptom predominance on the right side, though the remaining 14 patients (53.8%) presented predominance on the left side. All subjects with PD were receiving antiparkinsonian treatment: 20 (76.9%) were on L-dopa, 16 (61.5%) were on dopaminergic agonists, and 6 (23.1%) were on monoamine oxidase inhibitors. Concerning the severity of disease, 2 (8%) were classified with HY stage 1, 15 (58%) had HY stage 2, 8 (31%) had HY stage 3, and 1 (4%) had severe disease (HY stage 4). The mean NMSS was 58.17 ± 52.28 points. Visual acuity was better in the control group in comparison with PD patients. There were no differences in spherical equivalent and axial length between the subjects with PD and controls. The MoCA score was lower in the PD group than in HC (22.54 ± 4.98 vs 23.81 ± 3.33, p = 0.005). Table 1 summarizes the demographics and clinical characteristics.
Demographics and clinical characteristics of Parkinson's disease and healthy controls.
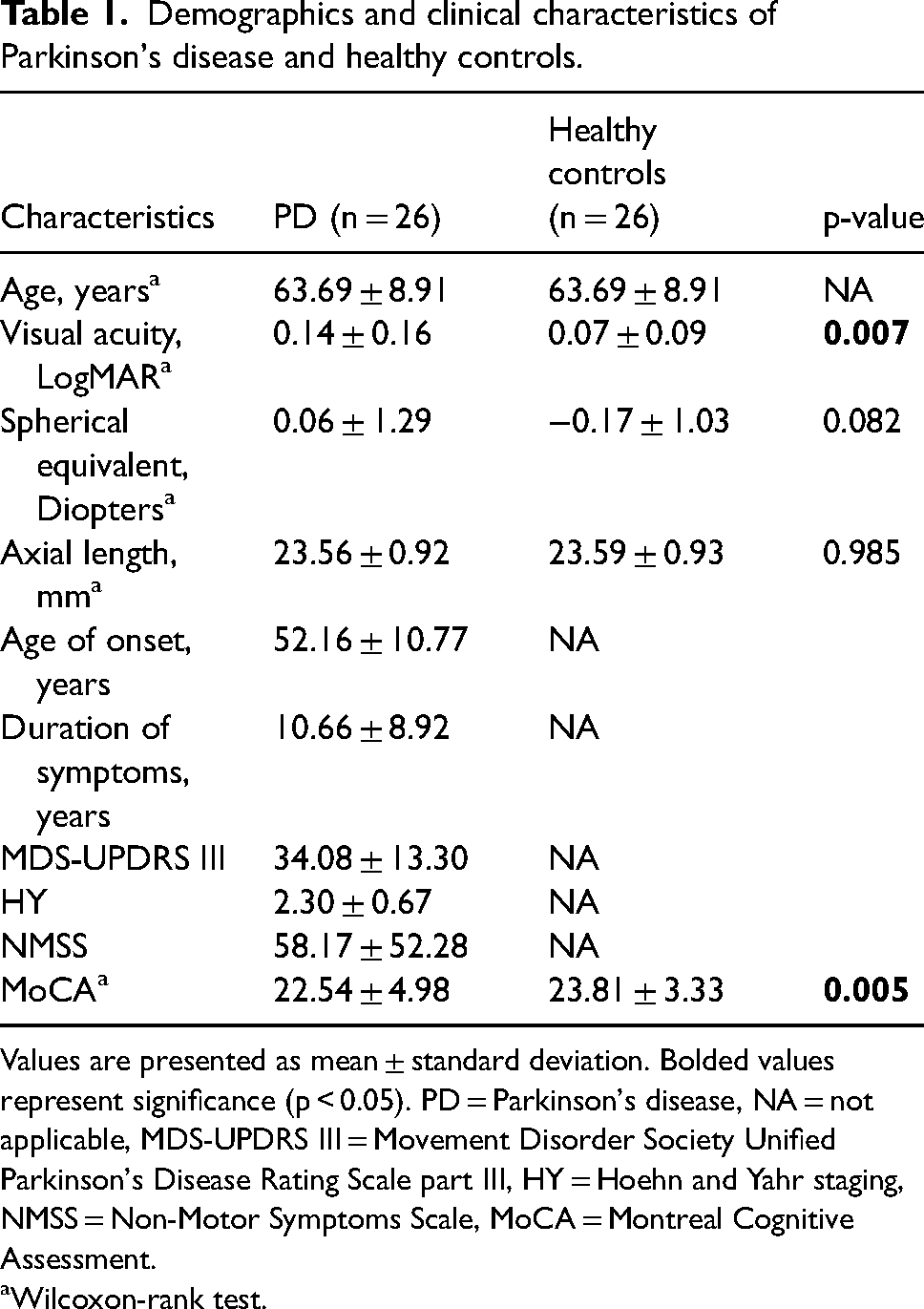
Values are presented as mean ± standard deviation. Bolded values represent significance (p < 0.05). PD = Parkinson's disease, NA = not applicable, MDS-UPDRS III = Movement Disorder Society Unified Parkinson's Disease Rating Scale part III, HY = Hoehn and Yahr staging, NMSS = Non-Motor Symptoms Scale, MoCA = Montreal Cognitive Assessment.
Wilcoxon-rank test.
Comparison of total αSyn values between PD subjects and controls
The median of total αSyn in PD subjects was higher when compared with controls [1.76pg/mL (IQR 1.74–1.80) vs 1.73pg/mL (IQR 1.70–1.77), p = 0.004].
Comparison of OCT thickness parameters between PD subjects and controls
The median total macular retinal volume in PD subjects was 0.31mm3 thinner than controls, p < 0.001 (Figure 1). Fovea thickness in the nasal inner was 14μm, nasal outer 10.5μm, superior inner 11μm, superior outer 13μm, temporal inner 8.5μm, temporal outer 9μm, inferior inner 12μm, and inferior outer 12.5μm thinner in PD than controls, p < 0.001. In NFL, the segment nasal outer was 3μm, the superior outer 1.5μm, and the inferior outer 4.5μm thinner in PD than in controls. The median total volume of GCL was 0.05 mm3 thinner in PD in comparison with controls, p = 0.003. The nasal outer was 3μm, superior inner 0.5μm, superior outer 3μm, temporal inner 2μm, temporal outer 4μm, inferior inner 2μm, and inferior outer 3μm thinner in GCL in subjects with PD than controls, p < 0.05. In PD, the IPL median total volume was 0.1 mm3 thinner than controls, p = 0.0001. In addition, in IPL, the nasal inner was 3μm, nasal outer and nasal superior inner 2μm, superior outer, superior inner and temporal outer 1.5μm, and the inferior inner and inferior outer 2μm thinner than in controls, p < 0.05. The segment nasal inner of OPL was 2μm thicker in PD than HC, p = 0.039. ONL median total volume was 0.1 mm3 in PD thinner than controls, p = 0.006. The central segment was 10μm, the nasal inner 11μm, the nasal outer 5μm, the superior outer 7μm, the temporal inner 6.5μm, the temporal outer 3.5μm, and the inferior outer 2.5μm thinner in PD than controls, p < 0.05. The median total volume of RPE was 0.03 mm3 in PD thicker than HC, p = 0.004. The central segment was 1μm, the nasal outer 1μm, the superior inner 0.5μm, the superior outer 1μm, the temporal inner 2μm, temporal outer 1μm, and the inferior outer 1μm thicker in PD than HC, p < 0.05.
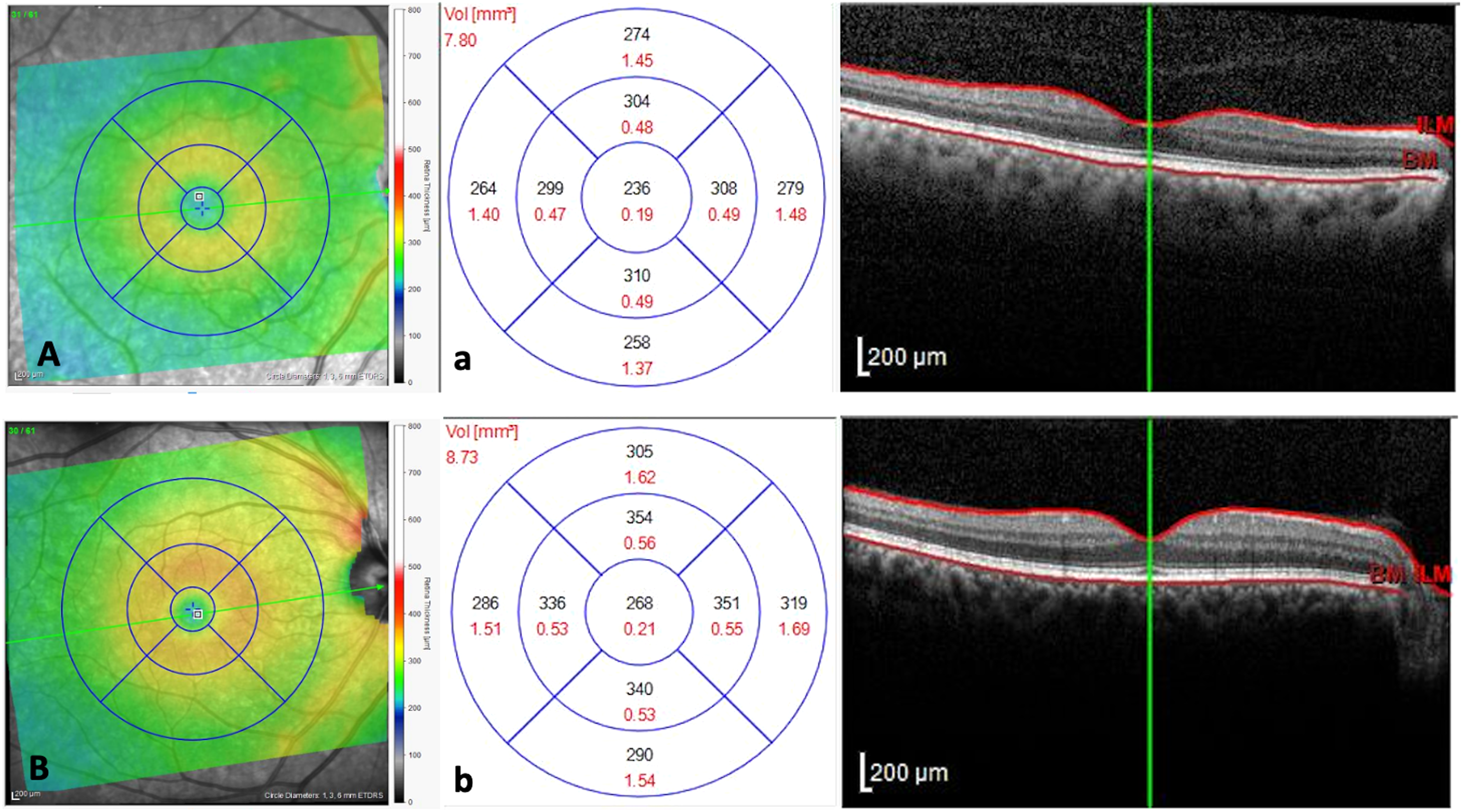
Parkinson's disease patient (A) and control (B) showing the acquisition of central retinal thickness using spectral-domain optical coherence tomography (SD-OCT) (a, b) Measurement of the central macular thickness as well as the subfield thickness.
When we segmented the IRL, the median total volume was 0.37 mm3 in PD thinner than HC, p < 0.001. The nasal inner was 14.5μm, nasal outer 12μm, superior inner 15μm, superior outer 13.5μm, temporal inner 13μm, temporal outer 10.5μm, inferior inner 16μm, and inferior outer 12.5μm thinner in PD than HC. When we compared the ORL segments between PD and HC, the central segment was 1.5μm, the nasal outer, superior inner, superior outer, temporal outer, inferior inner, and inferior outer 1μm, and the temporal inner 2μm thicker in PD than HC.
Supplementary material (S1 Table) includes the comparison of retinal layer thickness between PD subjects and HC.
Association between total αSyn in reflex tears and OCT thickness parameters in PD subjects
Central thickness and the macular volume segments were not significantly correlated with total αSyn in reflex tears. In contrast, the INL central volume was positively correlated with αSyn in reflex tears (r = 0.357, p = 0.009). The ONL had the most segments significantly correlated with total αSyn values in reflex tears all of which were positive including ONL nasal outer segment (r = 0.281, p = 0.043), nasal outer volume (r = 0.296, p = 0.033), superior outer (r = 0.330, p = 0.017) and superior outer volume (r = 0.334, p = 0.015). Also, the ORL temporal inner was positively correlated with total αSyn in reflex tears (r = 0.276, p = 0.047).
Association between total αSyn in reflex tears and clinical parameters in PD subjects
Clinical parameters except for dopaminergic agonist treatment were not significantly correlated with total αSyn in reflex tears of PD subjects. Patients treated with dopaminergic agonists had lower levels of total αSyn in reflex tears than subjects without treatment [1.75pg/mL (IQR 1.74–1.77) vs 1.79pg/mL (IQR 1.76–1.84), p = 0.002].
Discussion
The data presented in this report shows that total αSyn in reflex tears is increased in PD subjects compared with controls. The tear fluid is composed of proteins, and their principal source is the lacrimal gland. 24 Tearing secretion could be basal or reflex. 25 In the present study, we studied reflex tears because has not been impacted by aging.26–28 In line with our findings, two previous studies using an ultrasensitive single-molecule array system to measure total soluble αSyn in tears found a significantly increased total soluble αSyn in PD subjects compared to controls. 3 In contrast, two studies using an enzyme-linked immunosorbent assay reported increased oligomeric and decreased total αSyn in basal and reflex tears in PD subjects compared with controls.4,5 This variance in the results could be explained by the methods used to quantify the total αSyn in tear fluid.
Levels of αSyn in tears can be a potential biomarker for PD. Levels of αSyn in blood and cerebrospinal fluid (CSF) have been studied, showing discrepant results. 29 Some studies reported increased levels of αSyn in plasma and serum in PD,30–34 while others showed decreased levels compared to age-matched healthy controls.35,36 These differences in results could be explained by technical reasons, such as contamination by αSyn from erythrocytes or by the use of diverse assay techniques. 29 In relation to αSyn in CSF, some analyses showed a reduced mean total αSyn in PD compared with age-matched controls or other neurological controls. Nevertheless, other studies did not detect any differences between the PD and control groups.37–39 In terms of clinical feasibility, measuring αSyn levels in tears is less invasive and less expensive compared to measuring αSyn levels in blood or CSF. Tears can be collected non-invasively and easily obtained during routine eye exams. In contrast, obtaining blood and CSF samples requires more invasive procedures, such as a lumbar puncture or blood draw. However, further research is needed to determine the sensitivity and specificity of measuring αSyn levels in tears as a biomarker for PD compared to measuring αSyn levels in blood or CSF. Additionally, it is possible that a combination of biomarkers, including αSyn levels in tears, blood, and CSF, may provide a more comprehensive and accurate assessment of PD.
According to the majority of reports, PD patients typically have thinner macular thickness. 40 Our results described a reduced macular thickness in all quadrants and a sparing of the foveal region in PD patients compared with controls. Similar results have been reported by Sengupta et al., who compared the macular volume between 34 PD subjects and 50 healthy age-matched controls using the Spectralis OCT. 41 They described reduced inner and outer macular regions in all sectors and preserved foveal volume. They additionally propose that central macular sparing is justifiable because the fovea only contains photoreceptor cones and lacks ganglion cells and their axons. As a result, in PD, there is an absence of ganglion cells without impact on the photoreceptors, contrasting to other neurodegenerative diseases where the fovea is spared. Aaker et al. detected a thinning of the inner inferior, outer superior, and outer nasal macular quadrants of 18 eyes of nine patients with PD. 42 Furthermore, Ahn et al. reported a thinning appearance in inferior and temporal inner segments. They suggest that nigral dopaminergic degeneration could be the cause of retinal thinning. A recent study by Abd Hamid et al. related that the mean macula thickness average, central, outer superior, outer inferior, and outer nasal was lower in PD than in controls. 6
Additionally, our findings indicated that GCL, IPL, INL, and ONL were thinner, although OPL and RPE were thicker in PD. Wang et al. found similar results from a more extensive study of PD. OPL was thicker in PD patients, while RNFL, GCL, IPL, INL, and ONL were thinner. HY staging scores made these results stand out more. In addition, the RNFL and INL thinning and the thickening of OPL occur in HY 2 and HY 3 stages. 43 These outcomes are consistent with our population, which primarily included HY 2 stages.
The thinning of the INL but not the OPL and RPE may be due to the fact that the INL is metabolically more active than the OPL and RPE, which makes it more susceptible to damage and degeneration. The INL contains a higher density of cells and requires more oxygen and nutrients to function properly.44,45 Therefore, any condition affecting oxygen and nutrient supply to the retina, such as ischemia or inflammation, may lead to the thinning of the INL. In addition, the macular retinal layer contains dopaminergic cells.43,46,47 Dopamine is a retina neurotransmitter that regulates neural activity and integrates visual information.48,49 The nuclei of dopaminergic cells are situated predominantly in the inner layers close to ganglion cells. Excessive glutamate synthesis, neuronal death, and cellular dysregulation may result from a dopamine deficit. 50 Previous studies have shown that PD patients have low levels of dopamine and an accumulation of αSyn in the retina.51,52 Another factor for the reduction of INL thickness may be the dopamine depletion observed in PD, leading to an impaired interaction between dopaminergic cells and ganglion cells. 51 This observation was corroborated in a mouse model overexpressing αSyn. Marroco et al. 53 demonstrated that overexpression of αSyn in the mouse retina leads to neurodegeneration of dopaminergic amacrine cells (which precedes ganglion cell degeneration) causing a decrease of light-adapted responses and visual acuity. Besides, a reduction of the dopaminergic amacrine cell number and the number of their synapsis contact in the retina of PD patients has been reported. 52 These findings support the idea that the eye constitutes an easily accessible model for studying Parkinson's disease
Our study's most interesting and novel finding was a positive correlation between total αSyn levels in tears and the central volume of the inner nuclear layer (r = 0.357, p = 0.009). The lacrimal gland has dopamine and dopamine receptors.54,55 The parasympathetic and sympathetic nervous systems and stimulation of postsynaptic D1-like receptors regulate lacrimal fluid protein secretion. 56 Similar to photoreceptors, amacrine, bipolar and horizontal retinal cells have dopamine receptors.57,58 The αSyn is primarily found in presynaptic terminals and can have an impact on dopamine levels, including those in the retina. 59 αSyn is found in the axons of photoreceptors, bipolar and amacrine cells of the retina and is present in the presynaptic terminals of neurons in the external and internal plexiform layers. 60
The limitations of our study are mainly related to the relatively small sample size. Second, we did not include patients in the early or premotor stage of the disease; advanced-stage PD patients were also underrepresented, which limited our analysis of the correlation of total αSyn in tears with changes in the retinal structure with the disease severity of PD. A longitudinal study using repeated SD-OCT and quantification of αSyn in tears on the same patients will give better information about how the disease is progressing. Also, we quantified total αSyn, but oligomeric αSyn should be considered in future studies because it is considered the primary cause of neurotoxicity in PD. Finally, the diagnosis of PD was established on clinical diagnostic criteria and lacks of neuropathology confirmation.
In summary, our study provides corroborative evidence of a significant difference in total αSyn reflex tear levels when comparing PD subjects to HC. Tears are an easily accessible biological fluid, and sampling is less invasive than blood or cerebrospinal fluid liquid. Tear fluid is an excellent source to measure specific biomarkers and allow a prompt diagnosis. Furthermore, we observed thinner inner retinal layers and thickener outer retinal layers in PD subjects compared to controls. The positive correlation between total αSyn levels and changes in the inner nuclear layer point to a potential diagnostic tool for PD patients.
Supplemental Material
sj-docx-1-ejo-10.1177_11206721231173725 - Supplemental material for Is there any correlation between alpha-synuclein levels in tears and retinal layer thickness in Parkinson's disease?
Supplemental material, sj-docx-1-ejo-10.1177_11206721231173725 for Is there any correlation between alpha-synuclein levels in tears and retinal layer thickness in Parkinson's disease? by Azyadeh Camacho-Ordonez, Amin Cervantes-Arriaga, Mayela Rodríguez-Violante, Ana Jimena Hernandez-Medrano, Selma Alin Somilleda-Ventura, Hector J. Pérez-Cano, Ángel Nava-Castañeda and Oscar Guerrero-Berger in European Journal of Ophthalmology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Fundacion Hospital Nuestra Señora de la Luz, IAP.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
