Abstract
Purpose
To analyze the prescribing trends over a 7-years period, between 2013 and 2020, in a tertiary hospital (Hospital Clinico San Carlos, Madrid, Spain) and its health area.
Material and methods
A retrospective study on the data collected from the information systems, “farm@web” and “Farmadrid”, of glaucoma prescriptions in the framework of a public health system (Spanish National Health System) during the last seven years.
Results
Prostaglandin analogues were the most commonly used drugs in monotherapy during the study period (range: 36.82% − 47.07%). Fixed combinations of topical hypotensives had an upward trend since 2013 (range: 39.99% − 54.21%), becoming the most dispensed drugs in 2020 (48.99%). Preservative-free eye drops (lacking benzalkonium chloride, BAK) have displaced preservative containing topical treatments in all pharmacological groups. In 2013, BAK-preserved eye drops accounted for 91.1% of the total prescriptions, however in 2020 they only accounted for 34.2% of total prescriptions.
Conclusions
The results of the present study highlight the current trend to avoid BAK-preserved eye drops for the treatment of glaucoma.
Keywords
Introduction
Glaucoma is a highly prevalent disease responsible for 12-14% of cases of blindness.1–4 In recent decades, this percentage has decreased thanks to currently available treatments.
The most common first line of treatment is topical drugs used in monotherapy or combined. The reduction in the number of packs, eased by the use of fixed-combinations (FCs), increases compliance, eliminates possible washout effects, and has the hypotensive efficacy equivalent to that obtained by separate instillation of their components. 5 The adverse effects are similar to when drugs are administered individually. However, the prevalence and severity of local adverse effects, primarily dry eye symptoms, worsen with the higher the number of eye drops and the longer the treatment. The FCs currently marketed in Spain are listed in Supplemental file 1.
Preservatives are often used to prevent contamination of the packaging. These have been hypothesized to aggravate the symptoms caused by dry eye disease and create a predisposition to failure of glaucoma surgeries. 6 Benzalkonium chloride (BAK), used in approximately 70% of ophthalmic formulations, 7 has been identified as an independent risk factor for the coexistence of ocular surface disorders in patients with glaucoma. 8 BAK destabilizes goblet cells and induces squamous metaplasia of the conjunctival epithelium, reduces the number of epithelial cells making the lipid layer of the tear film unstable, favoring excessive evaporation of the aqueous fraction of the tear film. In addition, it has been reported that after 5-10 years of treatment with BAK-containing eye drops, BAK accumulates in the trabecular meshwork and may cause worsening of glaucoma. 7
Given the adverse effects of BAK and the current legislation on antimicrobial preservatives (Real Farmacopea Española. 5th ed. Madrid: Ministerio de Sanidad y Consumo; 2007), 9 other classes of preservatives have been developed: polyquaternium-1 (Polyquad), Stabilised Oxychlorine Complex (SOC) (PURITE™), sodium perborate (GenAqua), the SofZia ionic-buffered solution and new preservative-free (PF) dosage systems such as COMOD and ABAK. Given the above, the prescribing trends may have changed in recent years, as the therapeutic indication should no longer only depend on the drug to be prescribed to the patient but also on other factors such as the formulation of the eye drops.
In Spain, the estimated prevalence of glaucoma is 2.1%. 10 The Spanish National Health System (NHS) is based on a public model (General Health Law 14/1986) that provides universal coverage for Spanish citizens. The NHS finances a percentage of the cost of drugs ranging from 40% to 60%, depending on the patient's income. For retired people, coverage is extended to 90%.
In Spain, each state is further divided into health areas, corresponding to each hospital. The information and registration systems, “farm@web” and “Farmadrid”, available in Madrid, have been operational since 2013, recording the medicines dispensed at our health area.
This paper, through the analysis of the data collected, aims to highlight the changes in prescribing trends in the health area of the HCSC over the last seven years through the analysis of prescription data and to propose possible explanations for these changes.
Material and methods
Data on topical hypotensive drugs for glaucoma prescribed between 2013 and 2020 were collected from October 2020 to April 2021. Given the characteristics of this study in accordance with current legislation in Spain, approval by an ethics committee was not required.
The Hospital Clinico San Carlos (HCSC) in Madrid (Spain) is a public hospital, and the population of its health area was 374,369 inhabitants in 2019 (last recorded data). 11 In the Glaucoma Unit of the HCSC, there were 18,138 appointments in 2017, which corresponded to approximately 20.2% of the total activity of the Ophthalmology Department at the HCSC. The workload per doctor was about 2,591 appointments per doctor per year, which is the average for third-level hospitals.
Data on the prescribing patterns of ocular antiglaucomatous drugs in the public care in our health area were obtained from the information systems “farm@web” and “Farmadrid”. Data between 2013 and 2020 were included in the present study.
The farm@web platform records prescriptions by active ingredient or trade name (generic or brand name medicines), providing numerical data on how many times the drugs have been prescribed. Farmadrid collects prescriptions by active ingredient and also offers further information on sex and age group ([0-14 years], [15-44 years], [45-64 years], [65-74 years] and [>75 years]). Neither farm@web nor Farmadrid link prescription information to individual patients but to more general population groups.
The data collected between 2013 and 2018 came from the register of prescription coupons sent from drugstores to the Madrid College of Pharmacists. Therefore, they reflected not the medicines prescribed but only those dispensed. The data from 2018 onwards, after the implementation of the e-Prescription in Madrid through the so-called Single Prescription Module (SPM)e, corresponded to the prescriptions given out. However, the disadvantages of this data collection system are that it is not possible to know whether the patient actually bought the medicine and that in case each patient's e-Prescription is not kept fully up to date, the total number of prescriptions may be artificially inflated.
The data analysis was performed using Microsoft Office Excel v. 2008. In the trend analysis, as the absolute number of prescriptions recorded was higher from 2018 onwards, the results were expressed as a percentage of the total to account for the different data collection methods. Prescribing trends were studied by pharmacological groups, active ingredients, formulations with and without preservatives, age groups and sex. In addition, a systematic search of MEDLINE (Pubmed) was conducted to discuss possible causes of the observed trend changes.
Results
Prescribing trends by pharmacological groups are depicted in
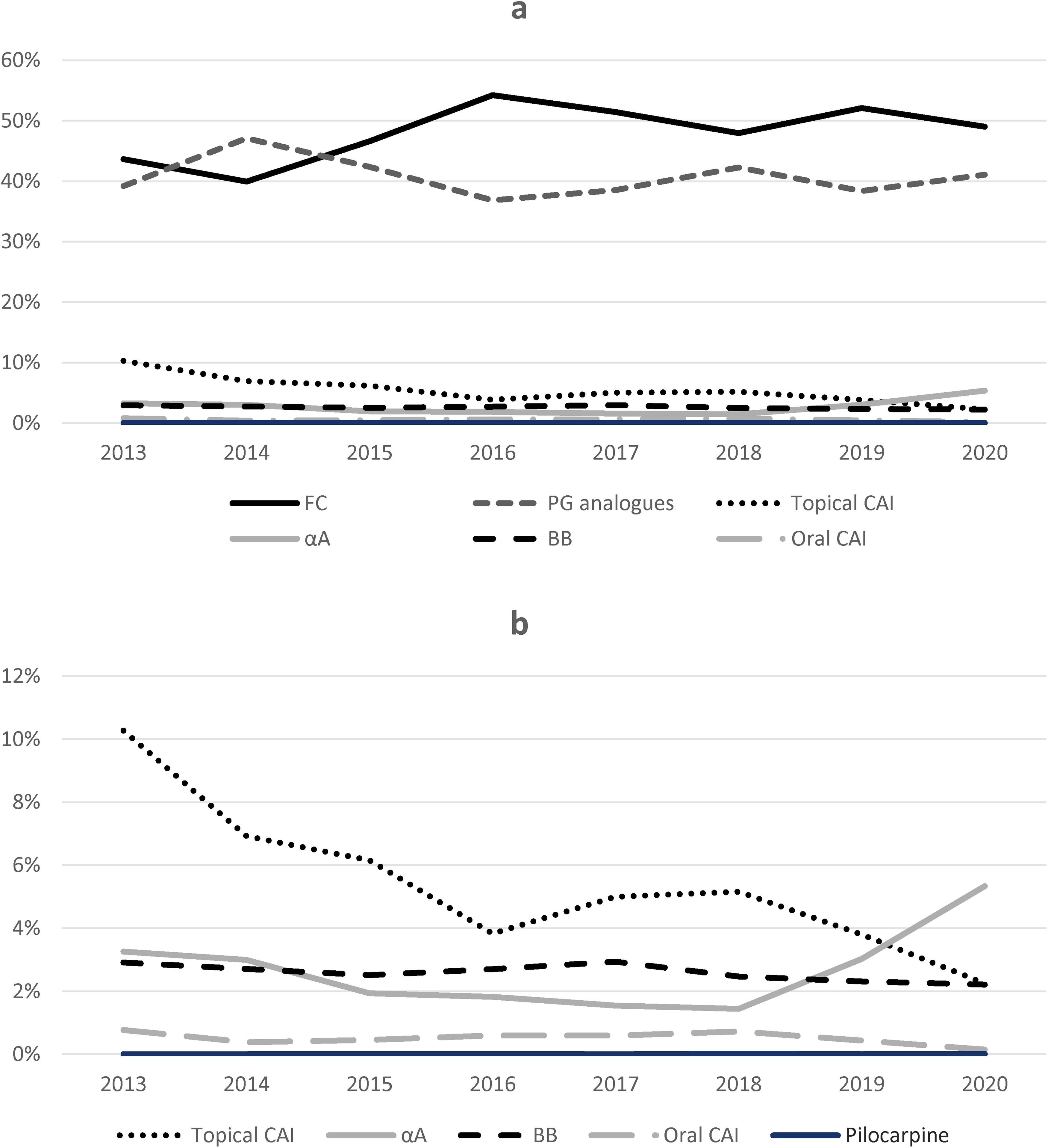
1a. Use of topical antihypertensives in the health area of the Hospital Clinico San Carlos, from 2013 to 2020. 1b. Detail of topical antiglaucomatous less commonly used in the health area of the Hospital Clinico San Carlos, from 2013 to 2020. Note the differences in scale.
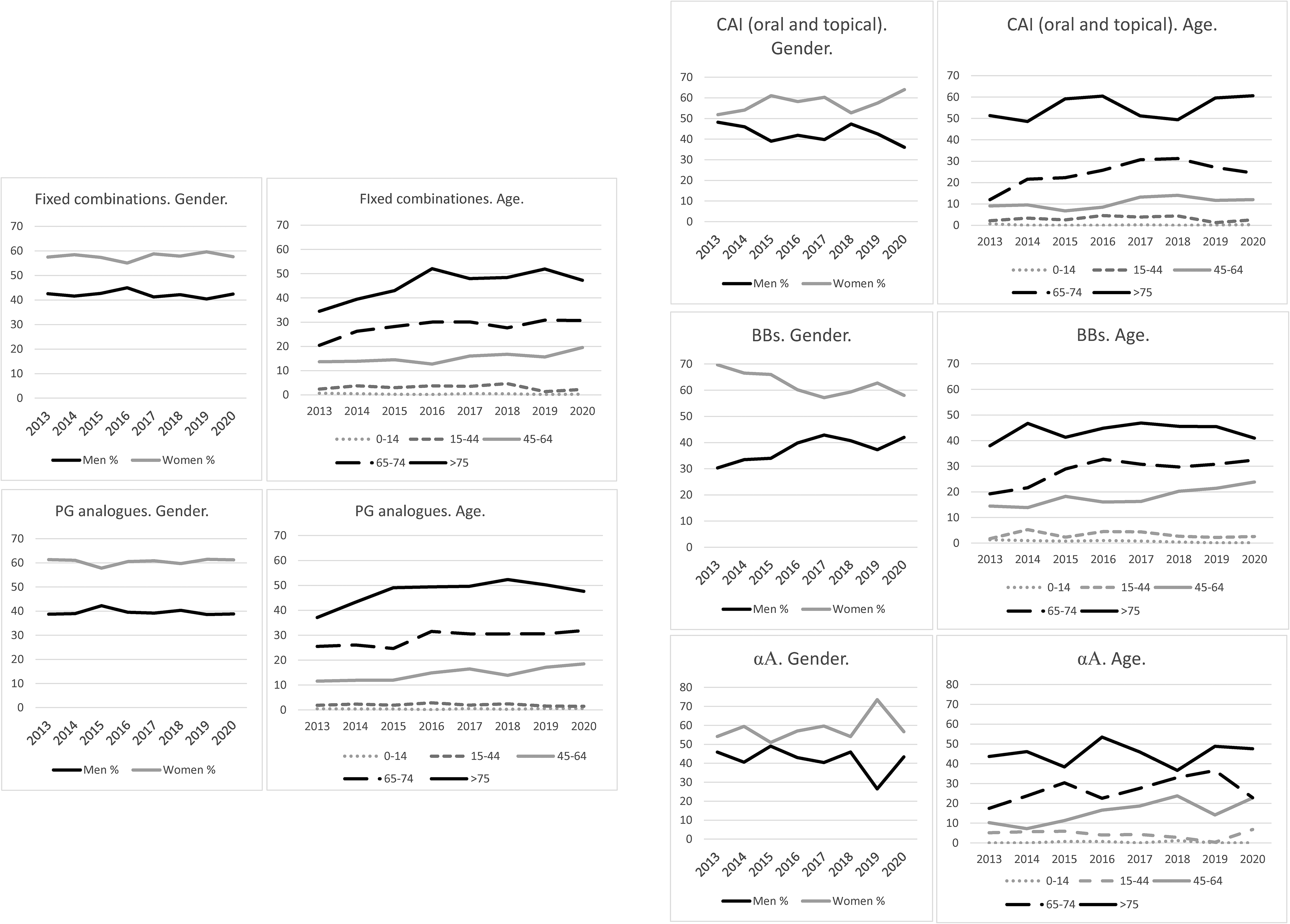
Use of ocular antiglaucomatous drugs stratified by sex and age, for the different pharmacological groups in the health area of Hospital Clinico San Carlos, from 2013 to 2020. Note the differences in scale on the y-axis.
Regarding the least prescribed pharmacological groups (
PG analogues’ prescription trend showed the greatest variation compared to other pharmacological groups (
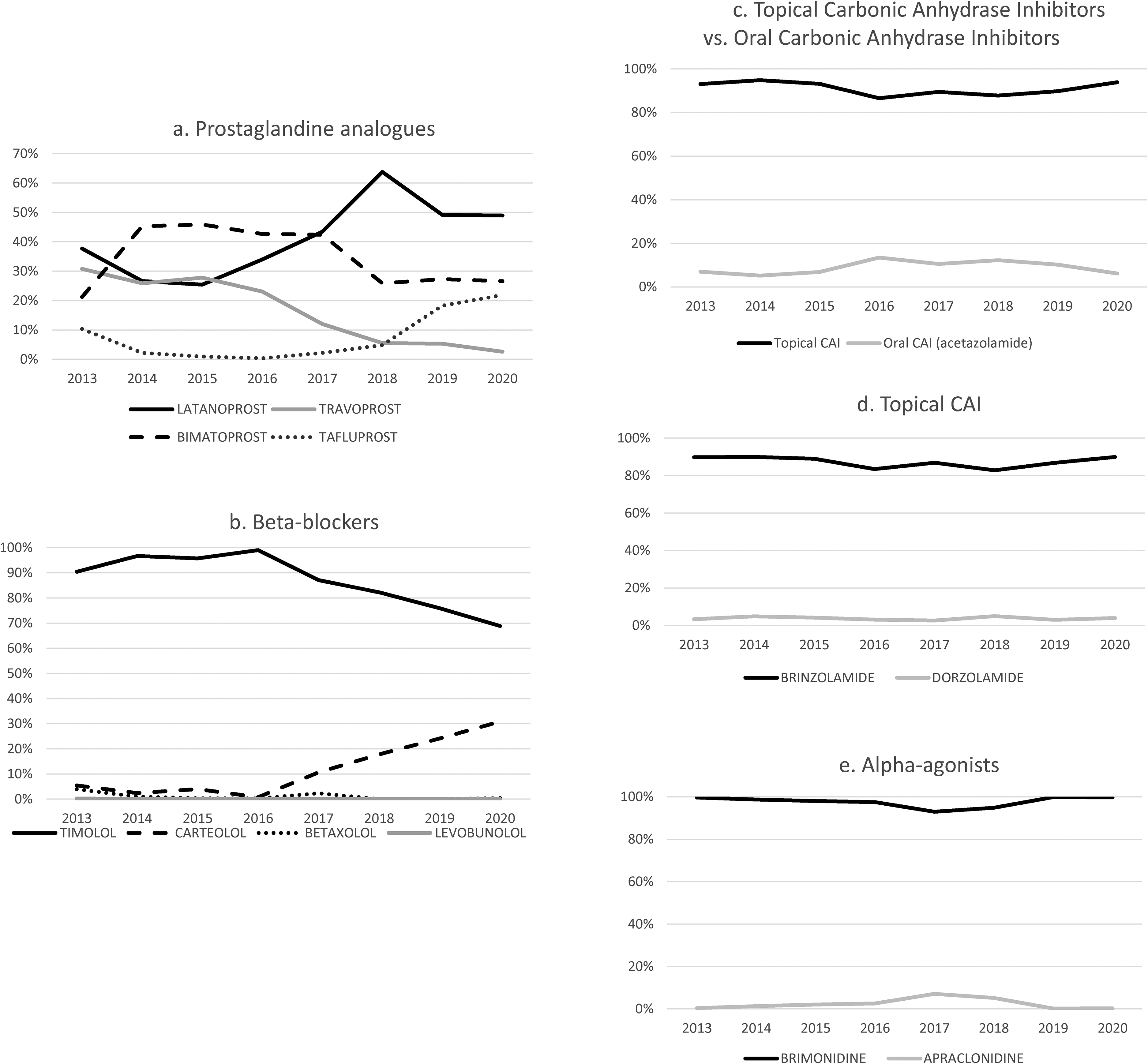
Comparative use of the different active ingredients by therapeutical groups: 2a. Prostaglandin analogues, 2b. Beta-blockers, 2c. Topical versus oral carbonic anhydrase inhibitors, 2d. Topical carbonic anhydrase inhibitors, 2e. Selective alpha2-agonists in the health area of the Hospital Clinico San Carlos, from 2013 to 2020. Note the differences in scale on the y-axis.
Regarding Beta-Blockers (BBs), (
During the period under review, topical CAIs accounted for more than 86.5% of the CAIs prescribed
The comparison of the FCs used in the HCSC health area grouped by therapeutic groups and by combinations of active ingredients is summarized in
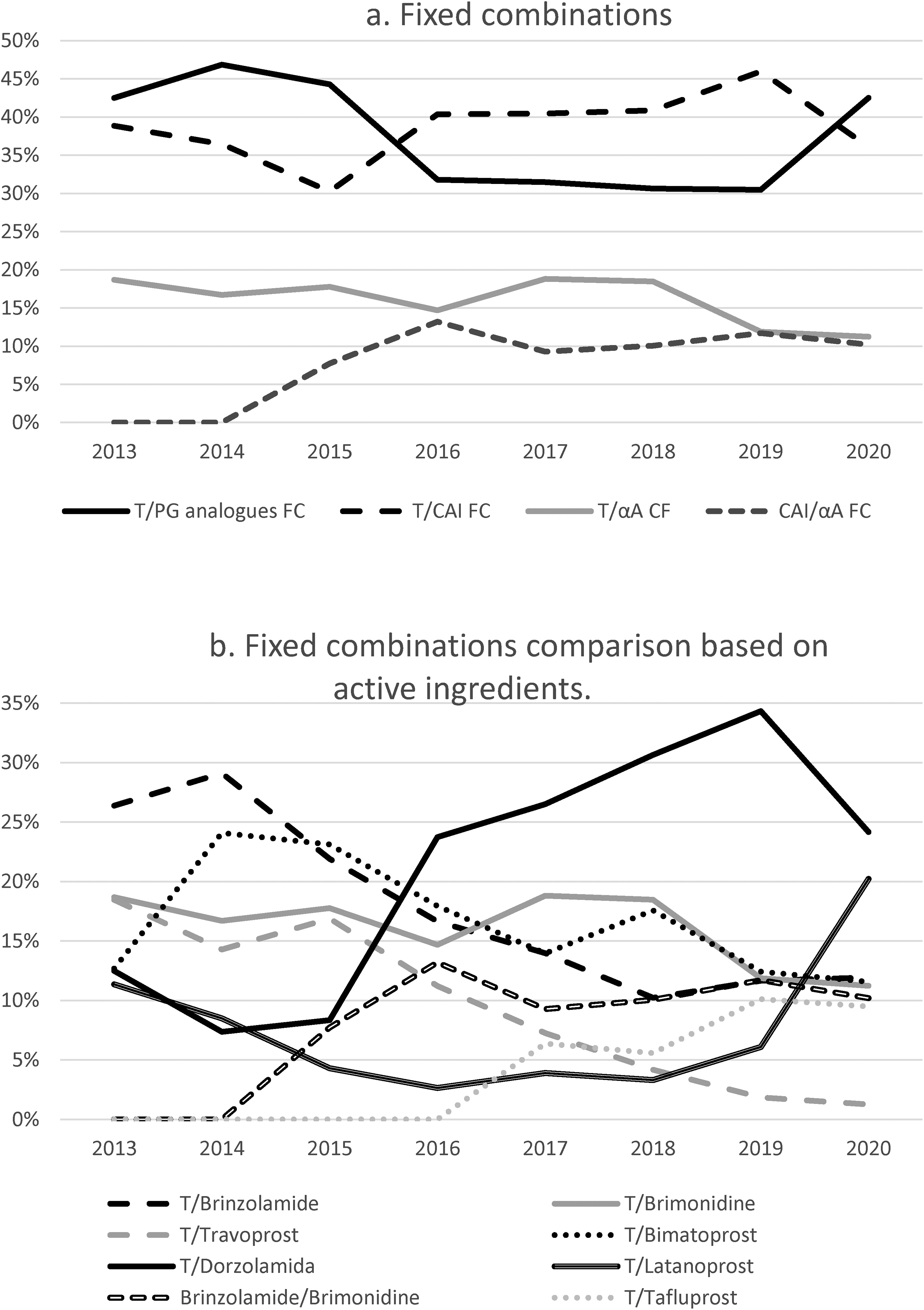
Comparison of the use of fixed combinations by pharmacological groups (3a) and by active ingredients (3b) in the health area of Hospital Clinico San Carlos, from 2013 to 2020. Note the differences in scale on the y-axis.
Among Timolol/PG analogues FCs (
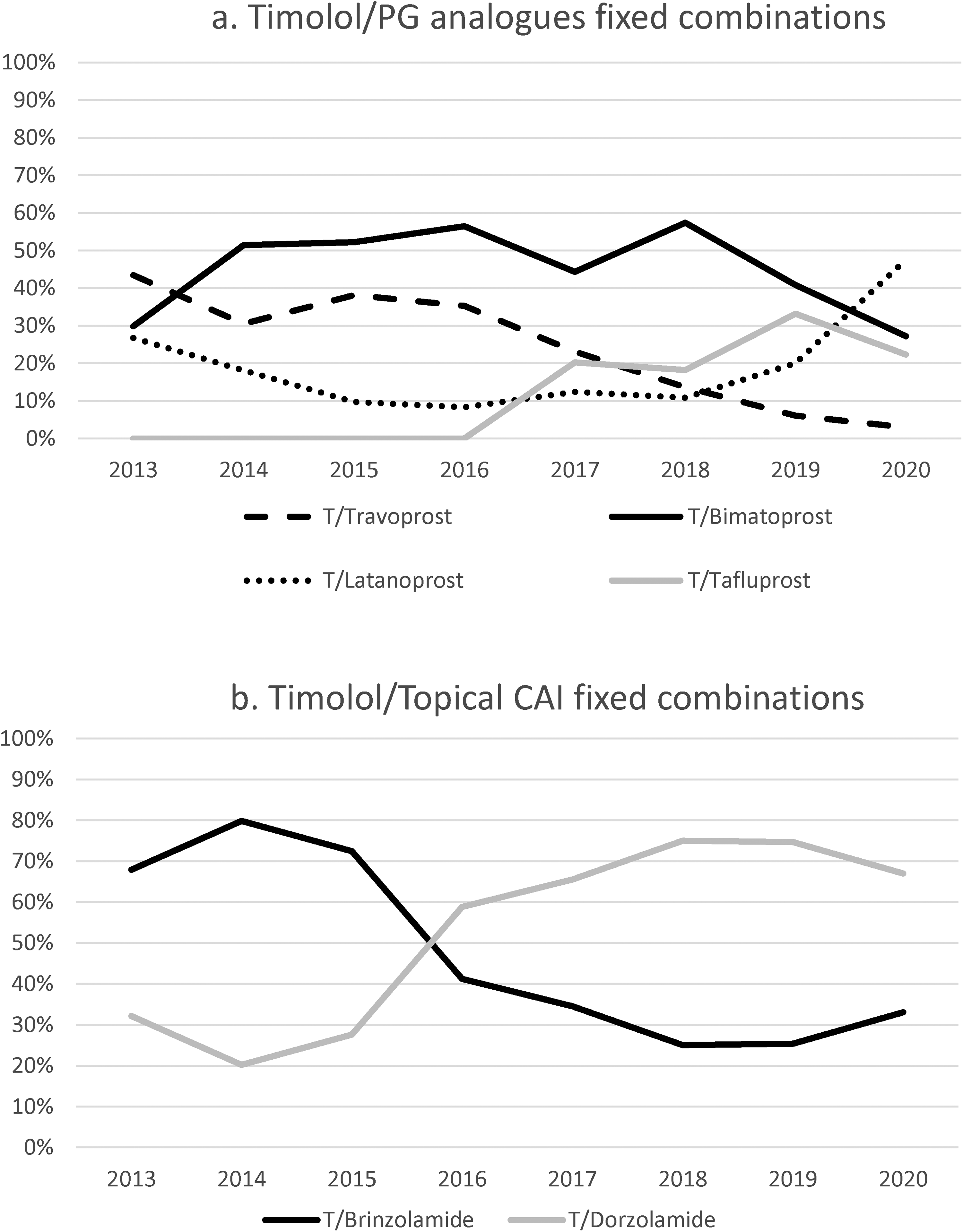
Comparative use of fixed combinations of Timolol/prostaglandin analogues (4A) and Timolol/CAI (4B) in the health area of Hospital Clinico San Carlos, from 2013 to 2020. Note the differences in scale on the y-axis. Since there is only available a fixed-combination of Timolol/alpha-agonist and of Brinzolamide/Brimonidine, these were excluded.
In
Regarding the use of preservatives, initially, more than 90% of prescriptions for all pharmacological groups contained BAK in 2013 (
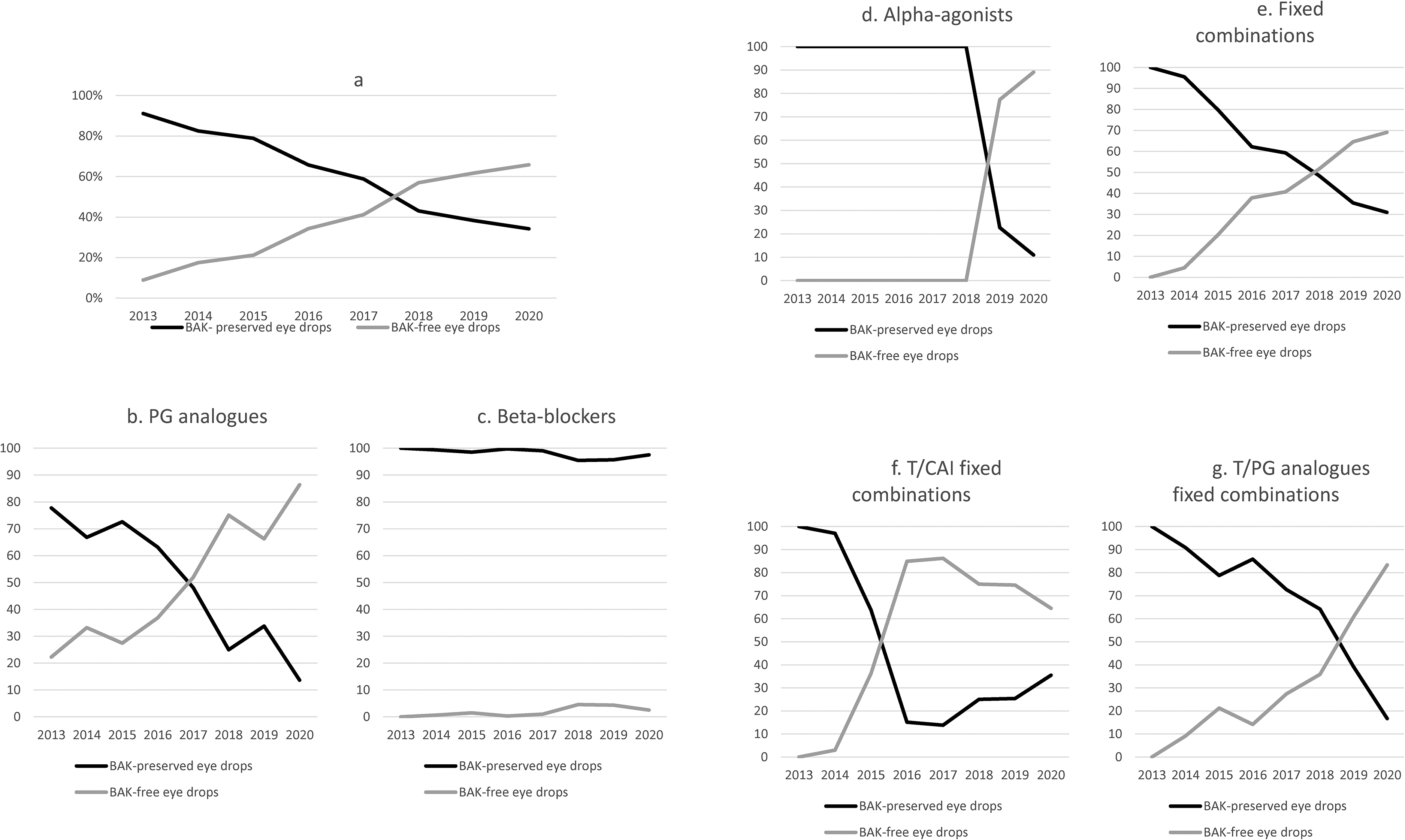
Use of ocular antiglaucomatous drugs with and without preservatives in the health area of Hospital Clinico San Carlos, from 2013 to 2020 (6a). Use by pharmacological groups of prostaglandin analogues (6b), beta-blockers (6c), alpha2-agonists (6d) and fixed combinations (6e-g) with and without preservatives in the health area of the Hospital Clinico San Carlos, from 2013 to 2020. Note the differences in scale on the y-axis.
The trends for preservative and PF drugs divided by drug families are shown in
Discussion
This is the first study to date on the evolution of prescribing patterns for glaucoma medications in Spain. The health area considered corresponds to the population of a tertiary hospital (HCSC, Madrid, Spain) between 2013 and 2020.
Current clinical practice guidelines1,12,13 establish PG analogues as the first therapeutic step for both POAG and ocular hypertension (OHT) due to their greater hypotensive effect 14 and fewer systemic adverse effects. 15 Correspondingly, in our study, PG analogues were the most commonly prescribed drugs in monotherapy (41.06% of prescriptions in 2020).
Forty-nine percent of the patients with OHT are said to require at least two topical agents within 5 years from diagnosis, 6 therefore, the different therapeutic agents should be combined in cases of insufficient intraocular pressure (IOP) control or disease progression.1,12,13 The current recommendation is, whenever possible, to use FCs given the greater convenience of dosing, instillation and the lower dose of preservatives. 15 This recommendation made FC the most frequently prescribed medicines during the seven years under study (48.99% of prescriptions in 2020).
In the present study, the percentage of drugs dispensed as separated agents has decreased, probably, secondarily to the emergence of new FCs. Same results have been reported by Hogg et al. in England. 16
This is the case of topical CAIs, whose fall is explained by the widespread use of Timolol/CAI FCs. On the other hand, BBs and α2A have maintained the numbers given that they retain other indications (non-responders to PG analogues, IOP peaks, etc.). However, this does not reflect a change in the prescribing pattern from one pharmacological group to another, but a preference for prescribing FCs when multiple medications are required instead of combining several drops.
The first studies on the toxicity of preservatives, published in the 1970s, 17 were carried out in animal models and reported alterations in the tear film, damage to the cornea, conjunctiva, trabecular meshwork and lens induced by BAK. 18 Since 2007, an increasing number of studies in humans have shown that if exposed long-term, BAK accumulates in the trabecular meshwork, altering the cytoskeleton, intercellular adhesion and the mechanosensitive and contractile properties of the trabecular meshwork. 19 With the advent of the first PF drugs, many studies emerged demonstrating significant differences in quality of life, symptomatology, success of filtering surgery and adherence to therapy, 18 with no differences in terms of efficacy.20,21
The latest recommendations emphasize the usefulness of PF eye drops in patients under three or more drugs, high likelihood of future glaucoma surgery or previous ocular surface disease. 22 However, the use of PF drugs is not universally recommended, especially in other healthcare systems where the patient bears the cost of medication.
In 2017, the recommendation from the NICE guideline was that generic topical drugs should be used as first line, as the cost related to the used of brand medicines is not always justified by their benefits. Hogg et al. 16 reported the use of generic bimatoprost in 2017 could have made the United Kingdom to save £31.4 million, which represented 34.7% of all 2017 glaucoma prescribing cost. 16 This analysis has not been possible to be made in our article; however, as English healthcare system is also public, these data could be comparable.
In contrast, Nguyen et al. 23 evaluated the prescription of branded and generic PG analogues in the United States. They concluded the interaction between prescriptors and the industry could be responsible for more indications of branded drugs, even when no difference in efficacy is demonstrated. 23 Bearing in mind in this healthcare system, the patient must pay for the drug, they alert about the risks this may have on treatment adherence. In Spain, as the stated pays part of the price, this is not frequently a limitation and we may be less sensitive to this fact.
Some recent studies have reported no differences in terms of ocular surface symptoms in patients under BAK-containing and PF eye drops,24–26 concluding that in the absence of randomized studies demonstrating the superiority of PF eye drops, PF drugs should be indicated only for patients with polytherapy or previous ocular surface disease.
Despite the literature is controversial in some points, there is consensus on the cumulative effects of BAK and its possible deleterious effects. According to Thygesen et al., 26 although PF medications should be indicated according to the risk of ocular surface disease, collectively, a significantly large proportion of glaucoma patients would have such risk factors. Therefore, it is appropriate to recommend PF eyedrops in the treatment of glaucoma. It is also essential to consider that single-dose eye drops are currently the most common PF packaging, which implies higher cost, risk of corneal injury and greater difficulty in instilling the drug in elderly patients. 27
Our study confirms the current trend to avoid preservative-containing drugs. In 2013, 91.1% of the expenditure was for BAK-containing formulations, while at the time of this study's completion in 2020, 65.8% of prescriptions were for PF eye drops. Weather conditions in Madrid, such as variable temperature and low rates of humidity, tend to worsen dry eye symptoms, and this could make Madrid professionals more likely to prescribe PF drugs to glaucoma patients.
The change in PG analogues prescription is worth noting, being 20% PF in 2013 and 85% in 2020 (
There was little change in BBs prescription. As shown in
It is important to note in terms of α2A, only two formulations for Brimonidine are currently available. With the appearance in 2018 of the first PF α2A (Brimvera®, Lab. Esteve Pharmaceuticals), the α2A used until then (Alphagan®, Lab. Allergan) was relegated to only 10.95% of the prescriptions. The most prescribed CAI is Brinzolamide (89.70% in 2013, 89.90% in 2020). However, there is currently no PF CAI.
Another example of the change in prescribing trends is FCs, 100% preservative-based in 2013, and 69.06% PF drugs in 2020 (
Currently, there are only three PF Timolol/PG analogues FCs on the market: Fixaprost® (Lab. Théa, 2018) (Timolol/Latanoprost), Taptiqom® (Lab. Santen, 2015) (Timolol/Tafluprost) and Ganfort® (Lab. Allergan), which is only BAK-free in the single-dose format. Although the IOP-lowering effect of Bimatoprost is slightly superior to PG analogues,5,14 the use of this drug, both in monotherapy and as part of FC with BBs, has decreased. Vizibim® (Lab. Bausch&Lomb, 2017), is the only drug that contains isolated Bimatoprost and does not have preservatives, while the FC with Timolol, Ganfort® (Lab. Allergan), has been surpassed by Timolol/Latanoprost FC.
There have also been changes in Timolol/CAIs FCs prescriptions. In 2013, the Timolol/Brinzolamide FC accounted for 70% of the prescriptions, while in 2020 the Timolol/Dorzolamide FC represented 65%. Currently, three drugs with the PF combination Timolol/Dorzolamide are available, Cosopt PF® (Lab. Santen), Duokopt® (Lab. Théa) and Cosduo® (Lab. Santen); while the only Timolol/Brinzolamide FC, Azarga® (Lab. Novartis), does contain BAK.
Our study also analyzed prescribing trends by sex, showing a higher percentage of drugs dispensed among women than men for all pharmacological groups. This could be due to a higher prevalence of POAG in females (RR 0.71) 28 or to the greater life expectancy of the female sex, 4 given that no significant differences have been reported in terms of therapeutic adherence.29,30
Regarding the age-stratified analysis, the results of our study are consistent with the premise that the risk of glaucoma increases with each decade of life (OR 1.73), 31 since for all therapeutic groups the main age group under treatment corresponded to patients over 75 years of age.
The present study is not without limitations, which need to be recognized to interpret the results. Firstly, this is a descriptive study in which the prescription data were not linked to individual patient records. This made it impossible to know the actual treatment of the patients, compliance, prescription patterns (monotherapy, bitherapy in non-fixed combinations, triple therapy), indications (POAG vs. OHT; chronical vs. ad hoc treatment) and treatment efficacy. Also, data associated with ethnicity or race are not known. Secondly, as mentioned above, given that the data came from prescription registers in the public health sector, it may lack information on drugs prescribed and dispensed that are not financed by the NHS, such as those prescribed by private institutions. Imprecision of medical and pharmacy claims must be considered, since these could be responsible for inaccuracies. Finally, regarding the characteristics of the health system in Spain, in which prescriptions do not need to be signed by the NHS, professionals might be more prone to indicate PF drugs, despite their higher cost. Moreover, as the patients pay only partially for the cost of the drugs, the price does not condition the prescription as much as it could in other countries. The loss of patents in the following years could condition the drug prescription trends to go back to what it was. Future studies will be necessary to confirm this.
In conclusion, the results of our study tease out the current trend to avoid eye drops containing BAK, although the appearance of PF eye drops for glaucoma treatment has not significantly changed the most prescribed active ingredients, which are the main drugs in monotherapy, PG analogues. The availability of PF formulations may be influencing on the active ingredients indicated for glaucoma patients. Prospective, individual-based studies are needed to analyze the direct and indirect costs of the treatments used, and pharmacoeconomic studies on the cost-utility and cost-benefit of these treatments.
Supplemental Material
sj-docx-1-ejo-10.1177_11206721231170034 - Supplemental material for Prescription trends for preservative free glaucoma medication in a public health system
Supplemental material, sj-docx-1-ejo-10.1177_11206721231170034 for Prescription trends for preservative free glaucoma medication in a public health system by Pilar Pérez-García, Bárbara Burgos-Blasco, Laura Morales-Fernández, Ana Fernández-Ruiz-Morón, Verónica Gómez-Calleja, Carlos Oribio-Quinto, Isabel Collado-Vincueria, Julian Garcia-Feijoo and Jose María Martinez-de-la-Casa in European Journal of Ophthalmology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
