Abstract
Introduction
The differential diagnosis for serous SRF can involve diseases with widely different pathogenic mechanisms that can range from vascular ocular diseases to ocular tumours and paraneoplastic syndromes. Recently, van Dijk et al. have described in three patients a new entity which they have called serous maculopathy with an absence of retinal pigment epithelium (SMARPE). We hereby describe a case of this infrequent macular disease and report its characteristic findings on multimodal imaging.
Case description
We present the case of a 65-year-old hyperopic woman with a three-year history of visual acuity (VA) loss in her left eye. Prior optical coherence tomography (OCT) had revealed the presence of serous subretinal fluid that had shown no response to treatment with intravitreal injections. On swept source OCT angiography scan, no macular alterations in the retinal vascular plexus structure were noted and there was no evidence of choroidal neovascularization. Ultra-widefield fluorescence angiography of the left eye revealed an early hyperfluorescent macular spot corresponding to the area of absent RPE and late fluorescein pooling. On ultra-widefield indocyanine green angiography there were no central or peripheral abnormalities of choroidal vascularization.
Conclusion
This recently described entity should be considered as a differential diagnosis in persistent serous subretinal fluid. Multimodal imaging helps differentiate SMARPE from its main differential diagnoses
Keywords
Introduction
A great number of diseases and conditions present with serous subretinal fluid (SRF) as the primary finding. The differential diagnosis for serous SRF can involve diseases with widely different pathogenic mechanisms that can range from vascular ocular diseases to ocular tumours and paraneoplastic syndromes,1,2 thus, the importance of identifying the main diagnostic features of each disease.
Recently, van Dijk et al. have described in three patients a new entity which they have called serous maculopathy with an absence of retinal pigment epithelium (SMARPE), characterized by the presence of persistent SRF with a focal area of retinal pigment epithelium (RPE) absence and no RPE detachment or drusen, the patients described (one male and two females) showed a remarkable stability of the SRF with a slight increase in the area of missing RPE over the years; no evidence of choroidal neovascularization (CNV) was found. We hereby describe a case of this infrequent macular disease and report its characteristic findings on multimodal imaging.
Case description
A 65-year-old hyperopic (+4.5D) woman with a three-year history of visual acuity (VA) loss in her left eye. Past examinations had revealed the presence of serous SRF in her left eye, which had been treated with 14 intravitreal anti-VEGF injections (10 ranibizumab, 2 aflibercept and 2 bevacizumab). SRF showed no response to treatment with intravitreal injections, maintaining relative stability. VA had also remained stable for the past 3 years. Given the persistence of SRF, the patient was referred to our centre to asses treatment with photodynamic therapy (PDT).
On the first evaluation at our centre, VA was 80 and 50 letters in her right and left eye using the ETDRS chart, respectively. Fundoscopy revealed a well-delineated atrophic macular area in the left eye. Although a posterior subcapsular cataract made macular assessment difficult, no alterations were noted in the fellow eye. Swept-source optical coherence tomography (SS-OCT) of the left eye showed the presence of serous SRF over a focal oval area of EPR absence, with a horizontal extension of 659μm (Figure 1 a, c-d). The height of SRF and the horizontal diameter of the RPE defect had remained stable since the first exam, three years prior (Figure 1 b). Neither RPE detachments nor drusen or other alterations of the RPE were present. The central choroidal thickness was 312 μm.
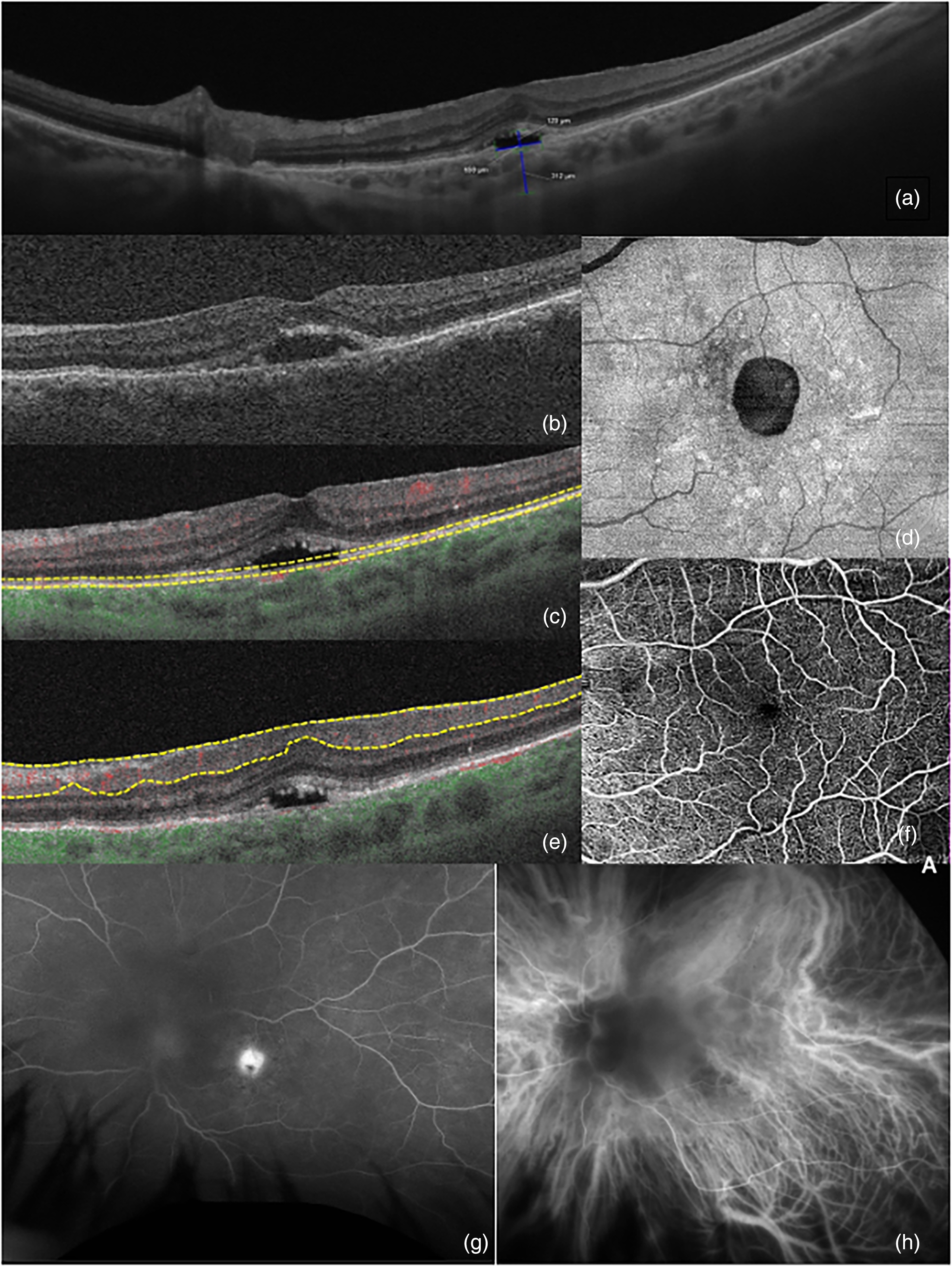
Left eye multimodal imaging of a patient with serous maculopathy with absence of retinal pigment epithelium. (a) Swept-source Optical coherence tomography (SS-OCT) HD scan showing the height of subretinal fluid (SRF) and extension of retinal pigment epithelium (EPR) defect, as well as subfoveal choroidal measurement; (b) OCT B-scan of exam realized three years prior were stability of SRF and EPR defect can be appreciated; (c and d) Enface OCT with segmentation done at EPR level which shows the oval morphology of EPR defect; (e and f) OCT angiography segmented at the superficial retinal vascular plexus without significant alterations; (g) fluorescein angiography which reveals a hyperfluorescent spot with pooling in the area of absent EPR; (h) indocyanine green angiography with no pronounced abnormalities of choroidal vessels.
On SS-OCT angiography (SS-OCTA) scan (Plex Elite 9000 Carl Zeiss Meditec, Dublin CA), no macular alterations in the retinal vascular plexus structure were noted and there was no evidence of CNV beneath or adjacent to the area of SRF (Figure 1 e-f). An en-face image segmented at the RPE level showed a circular area of missing RPE with no evidence of tractions or folds suggesting previous tear (Figure 1 c-d). Ultra-widefield fluorescence angiography (AF) (OPTOS, Dunfermline, Scotland) of the left eye revealed an early hyperfluorescent macular spot corresponding to the area of absent RPE and late fluorescein pooling, no peripheral alterations were noted (Figure 1 g). On ultra-widefield indocyanine green angiography (ICGA) there were no central or peripheral abnormalities of choroidal vascularization (Figure 1 h). Due to the stability of VA, the height of SRF and area of the RPE defect, together with the lack of response to repeated intravitreal injections, and the absence of identifiable complications, observation was the preferred management strategy, with no significant changes observed after 12 months of follow-up.
Conclusion
Multimodal imaging helps differentiate SMARPE from its main differential diagnoses. The absence of RPE detachments and the morphology of the surrounding RPE, evidence of folds, or areas of traction can differentiate this entity from RPE apertures and tears that usually occur in relatively large dome-shaped RPE detachments.1,3,4 The lack of leakage on FA helps differentiate SMARPE from other entities that present CNV.
Given its prevalence and appearance on OCT scans with the presence of serous submacular SRF, central serous chorioretinopathy (CSC) could be considered one of the main differential diagnoses of SMARPE, however, the choroidal alterations associated with CSC such as the presence of pachyvessels and multifocal hyperpermeability on ICGA 5 do not appear to be present on SMARPE.
Regarding SMARPE's pathogenesis, SRF may accumulate due to the lack of the RPE's outer blood-retinal barrier and pumping function.1,6,7 In the cases described until now, the SRF remains remarkably stable over time, the area of RPE absence may slowly increase and VA has shown a slow decline that may be severe in the long term. 8
As for treatment options, in contrast to macular neovascularization present in age-related macular degeneration and CSC, there is no evidence of neovascularization or VEGF drive in SMARPE to support treatment with anti-VEGF injections. Further, CSC is associated with a pachychoroid feature in which hydrostatic pressure from the choroid provides serous detachment of the RPE and the neuroretina, in which treatment of the leakage point may be indicated. 9 Half-dose PDT is a relevant treatment option for CSC, 10 however, this choroidal pathophysiology of CSC is not present in SMARPE, hence, there is no current evidence to support PDT treatment in these cases. Given the relative VA stability, observation seems to be a valid approach.
In conclusion, this recently described entity should be considered as a differential diagnosis for persistent serous SRF. Given the lack of response to intravitreal injections, care should be taken to identify and differentiate it from similar conditions to avoid unnecessary treatment with its possible side effects and complications. More cases and future studies are warranted to better understand the evolution of this disease, as well as its possible complications and management strategies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
